Services on Demand
Journal
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Dental Journal
On-line version ISSN 0375-1562Print version ISSN 0011-8516
S. Afr. dent. j. vol.77 n.8 Johannesburg Sep. 2022
https://doi.org/10.17159/2519-0105/2022/v77no8a2
RESEARCH
The search for a healthy sugar substitute in aid to lower the incidence of Early Childhood Caries: a comparison of sucrose, xylitol, erythritol and stevia
N MoelichI; N PotgieterII; FS BothaIII; J Wesley-SmithIV; C van WykV
IDivision of Paedodontics, Department of Odontology, School of Dentistry, Faculty of Health Sciences, University of Pretoria, Gauteng, South Africa
IIDepartment of Paediatric Dentistry, Faculty of Dentistry, Unversity of the Western Cape, South Africa
IIIFaculty of Veterinary Sciences, Paraclinical Sciences, University of Pretoria, Gauteng, South Africa
IVElectron Microscope Unit, Sefako Makgatho Heatlh Sciences University, Gauteng, South Africa
VDepartment of Community Dentistry, School of Dentistry, Faculty of Health Sciences, University of Pretoria, Gauteng, South Africa
ABSTRACT
AIM: A pursuit to And a healthy alternative to sucrose with less cariogenic potential, which can potentially lower the incidence of Early Childhood Caries (ECC), by means of comparison.
METHODS: Primary tooth enamel blocks (n=32) were randomly divided into four groups and exposed to 5% concentrations of the respective test groups (sucrose, xylitol, erythritol and stevia). All samples were inoculated with S. mutans standard strain (ATCC 25175) at room temperature. Analysis of Colony Forming Units (CFUs), acidity measurements (pH) and Scanning Electron Microscopy (SEM) observations were done after 6, 12, 18 and 24 h and compared.
RESULTS: After 6 h, the marginal mean CFU count indicated equal S. mutans growth in all groups. Stevia showed lower CFU counts compared to other groups at 12, 18 and 24 h. The pH levels for all non-fermentable sugar substitutes (NSS) initially decreased but never below the critical pH=5.5 and stabilized from 12 to 18 h. The pH levels of sucrose dropped and remained below pH=5.5 at all time intervals. The SEM analysis of S. mutans supported the CFU results indicating growth in the presence of sucrose and reduction in the presence of the NSS.
CONCLUSIONS: Compared to sucrose, xylitol, erythritol and stevia have less cariogenic potential with reduced growth of S. mutans and subsequent acidity levels. Stevia had the least cariogenic potential of all the NSS tested, followed by erythritol and then xylitol.
Keywords: Cariogenic potential, erythritol, stevia, Streptococcus mutans, xylitol, CFU, pH
INTRODUCTION
Early Childhood Caries (ECC) is one of the most prevalent diseases in children and a serious concern in public health care systems worldwide.1 Streptococcus mutans (S. mutans), in the presence of sucrose, is responsible for the onset, presence and development of dental decay in most instances.2 Studies suggest that the consumption of non-fermentable sugar substitutes (NSS) can affect the metabolism of microorganisms, with subsequent reduction of acidity in the mouth.3-6 A pH level in the mouth of 5.5 is known as the critical level, as this level of acidity and below leads to disintegration of the organic compound of the enamel and dentine, leading to demineralization and subsequent cavity formation.7 Limited research regarding the effect of NSS on the metabolism of cariogenic microorganisms and the resulting pH values inspired this study. The effect of sucrose and different NSS (namely xylitol, erythritol and stevia- hereafter refered to as sweeteners) upon the colony forming unit (CFU) of S. mutans, pH of the incubation medium and morphological appearance of bacterial cells, were investigated at various intervals over a 24 h period. Findings from this study generated new evidence regarding the use of healthy and safe sugar substitutes to lower the incidence of ECC.
MATERIALS AND METHODS
The research was conducted as a randomized controlled, cross-sectional, observational in vitro study of which the methodology is summarized in Figure 1. The aim of the study was to compare the cariogenic potential of sucrose and sweeteners xylitol, erythritol and stevia

Obtaining enamel blocks:
For the results to be applicable to ECC, primary teeth that were extracted as part of a comprehensive treatment plan irrelevant to this study were used. Enamel blocks (2 mm x 2 mm) were produced from the sound buccal surfaces of extracted primary molar teeth, to create a surface for biofllm formation. Blocks were prepared by using a diamond wafering blade in an Isomet 11-1180 low speed saw (Buehler Ltd, Lake Bluff, Illinois, USA). Blocks were sterilised in an autoclave (Already Enterprise Inc., Taipei, Taiwan) at 121°C for 15 minutes (min). The 32 enamel blocks were grouped in pairs and then divided into four well plates, representative of the four time intervals each containing the respective sweeteners and sucrose as control (2x4x4=32), and placed facing upward in individual tissue wells (Figure 2).

Preparation of sweetener solutions:
The technique described by Pfaffmann et al.8 was modified and used to prepare the 5% concentration level for the four sweeteners respectively. A preparation was based on weight by volume,8 and made up by precisely weighing 5 g of a sweetener and dissolving it in 20 mL sterile water. The sweetener preparation was filter sterilized (0.22 μΜ Millipore filters, Merck SA) and added to Peptone-Casein-Soy-Basal medium (PCSBM) to constitute the final sweetener solutions. The respective sweetener solutions were then added to two wells per well plate respectively (2 wells x 4 sweeteners x 4 well plate = 32). A laboratory assistant prepared the sweetener solutions and added the solutions to the well plates (test groups marked as A, B, C, D) as the lead researcher was blinded to which well plates contained which sweetener.
Inoculation of sweetener solutions:
Solutions in all well plates were inoculated with S. mutans standard strain (ATCC 25175), adjusted to a 1% McFarland standard one solution (3 x 108 CFU/mL) at room temperature (25±3°C). Microorganisms were tested for purity by means of Gram-staining.9
CFU determination:
For the purpose of determining Colony Forming Units (CFU), serial dilution and the spread plate technique (adapted from Bauman et al.9) were performed as illustrated in Figure 3. At each time interval (viz. 6, 12, 18 and 24 h) two samples were collected from the well plates harbouring the enamel blocks and sweetener solutions and diluted up to factor 10-8. Each dilution was plated in triplicate to eliminate bias, resulting in 48 plates per group, per time interval (2 samples x 8 dilutions x 3 plates=48 plates available for CFU analysis). All samples, dilutions and well plates were clearly marked.
Petri dishes (90 mm x 15 mm) (Sigma Chemical Co., St. Louis, MO, USA) containing prepared Agar were used for plating. The Agar was prepared by dissolving 5 g Glucose, 23 g Mueler-Hinton Agar and 15 g Bacteriological Agar in 1000 mL distilled, sterilized water. After plating, Petri dishes were placed in a Labotec incubator (Labotec (Pty) Ltd., Midrand, RSA) at 37°C for 24 h and CFU were assessed and compared to estimate the growth of viable bacterial cells in each sample. The most countable consecutive three dilution ranges were identified within each group, amounting to eighteen plates per group that were assessed for CFU at each time interval. The CFU were counted, by means of the standard plate count method, whereby the CFU were counted and multiplied with the dilution factor. These were then converted to log values.
Determining of pH-values:
At each time interval, the pH value of the media containing the respective sweeteners, was determined in triplicate using a microelectrode pH meter (Oakton pH700 pH/mV/ °C /F Bench Meter).
At each time interval, the two enamel blocks from each group were collected from the well plates for scanning electron microscopy (SEM) analysis to determine the extent of biofilm formation. Samples were placed in marked, sterile well plates and prepared for SEM according to standard methods for biological materials.10 Enamel slabs were kept in a 96% alcohol solution (Sigma Chemical Co., St. Louis, MO, USA) prior to final preparation. Samples were then placed on spotting plates, covered with 100% alcohol (Sigma Chemical Co., St. Louis, MO, USA) and left for 10 min. The 100% alcohol was gradually substituted with Hexamethyldisilazane (HMDS) (Fluka, Castle Hill, Australia), using disposable Pasteur pipettes and left to evaporate. Hexamethyldisilazane lowers surface tension around the sample and therefore eliminates the likelihood of distortion of the cells during the final drying stages before SEM examination. The enamel slabs were mounted on stubs, sputter coated with chromium and analysed at 1500x, 3000x and 8000x using a Zeiss Supra 55 VP Field Emission variable pressure Scanning Electron Microscope (FE-SEM) (Zeiss, Germany) at Sefako Makgatho Health Sciences University, South Africa.
Statistical analysis
The data was statistically analysed using a factorial study design with factors sweetener (sucrose, xylitol, erythritol and stevia) and time (6, 12, 18 and 24 h). The data for CFU counts and pH were quantitatively analysed using a two-way analysis of variance (ANOVA), with main factors sweetener and time, inclusive of interaction between factors. The level of significance was set at p < 0.05. Descriptive analysis was used for biofilm formation using SEM.
Ethical clearance
Ethical clearance was obtained from the Ethics Committee, Faculty of Health Sciences, University of Pretoria, South Africa (ethical clearance number 86/2019). Ethical and safety guidelines, for the handling and disposal of human teeth and laboratory research were strictly followed.
RESULTS
Comparisons of the CFU between the sucrose and NSS groups at the different time intervals are reported in Table I and illustrated graphically in Figure 4. Significant differences between the CFU count in the presence of sucrose and the respective NSS were noted after the 6 h time interval.
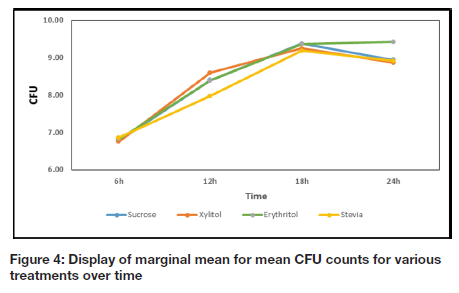
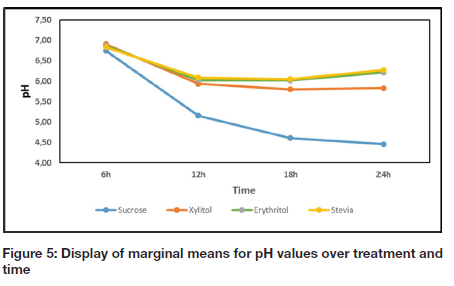
A comparison of the pH values for each group (at different time intervals) are reported in Table II and depicted as a graph in Figure 5. The results in the current study show a direct correlation between the increase in CFU and the decrease in pH value for all groups with the exception of erythritol (Table I, Figure 4, Table II, and Figure 5). When the CFU stabilized or decreased, the pH stabilized or increased accordingly, for sucrose, xylitol and stevia (Table I, Figure 4, Table II, and Figure 5).
The SEM images of the current study show clusters of S. mutans in the presence of sucrose at 12 h and 18 h (Figure 6). However, stressed S. mutans cells could be observed in the presence of erythritol at 18 h and 24 h (Figure 6). Scanning Electron Microscopy also revealed stressed bacterial cells with cob-web-like structure in the presence of stevia at 18 h and 24 h (Figure 6). Bacterial cells were only found in sheltered areas in the presence of xylitol at 12 h (Figure 6).
DISCUSSION
Sucrose
It was observed that sucrose showed statistically significant higher CFU counts than the NSS tested, with the exception of erythritol, at all the time intervals and xylitol at 12 h. Sucrose serves as substrate for S. mutans, enabling it to multiply and grow.11-12 Frequent and excessive intake of sucrose, therefore result in elevated levels of S. mutans in saliva.11-12 The SEM analysis also confirmed positive growth of S. mutans at all time intervals in the presence of sucrose (Figure 6a, b). Sucrose is known to cause a drop in oral pH because of bacterial fermentation of ingested sucrose, glucose, fructose, refined carbohydrates and cooked starches.11-13 After 12 h, the pH value for sucrose was statistically lower than the pH values of xylitol, erythritol and stevia for all time intervals (Table II, Figure 5).
Xylitol
The significant difference between the CFU count for xylitol and sucrose at the 6 h interval can be attributed to the fact that xylitol has a similar structure to sucrose and is therefore initially recognized by the microbial cell as a potential nutrient. Xylitol enters the bacterial cell and is expelled from the microbial cell as xylitol again, thus not utilized by the bacteria as a source of energy.14-15 Miyasawa et al.16 found that in the presence of xylitol, the lactic acid end-product of the metabolic pathway of bacteria is decreased, but that formic and acetic acids are increased which may support the initial drop in pH reported in this study.16
The SEM analysis produced corroborating evidence about the attachment of bacterial cells to the enamel surface. Although the CFU count in the presence of xylitol was higher than that of the other NSS at 12 h, there were not many bacterial cells visible on the enamel surface (Figure 6g). This is in line with the findings of Badet et al.17 where the clear inhibitory effect of biofilm formation in the presense of xylitol was demonstrated. This is also substantiated by the findings of Jacques et al.18 in an earlier study, stating that xylitol leads to a reduction in lipoteichoic acid, which is essential for adhesion to the enamel surface. Although the pH value measured for xylitol was statistically lower than those of erythritol and stevia after 24 h, it stabilized and never dropped below the critical level of 5.5 (Table 2, Figure 5).
Erythritol
The CFU for erythritol was statistically higher than the CFU for all the other sweeteners at 24 h (Table 1, Figure 4), and is supported by is similar to an in vivo study whereby the growth of S. mutans in the presence of erythritol was inhibited only during later growth phases.19 Certain bacterial cells still seemed turgid and healthy while others seemed to have died with cob-weblike attachments anchoring them to the surface of the enamel at 18 and 24 h (Figure 6c, d). The elevation in the pH value for erythritol from 18 h to 24 h may be attributed to an altered metabolic cycle of S. mutans, which is in correlation with a longitudinal study conducted by Runnel et al.20 that reported that bacteria in erythritol produce less acid compared to other polyols over time.
Stevia
The increase in CFU count for stevia after 6 h may be due to S. mutans that recognised the NSS as potential source of nutrition and the presence of carbohydrate bulking agents.21 At 12 h, the CFU for stevia was statistically lower (Table I, Figure 4) compared to all the other sweeteners and also evident in the SEM analysis with very few cells visible on the enamel structure (Figure 6e). Although studies have confirmed stevia's inhibitory effect on growth of bacteria, research is still lacking on the exact mechanism.22-23 It is however postulated that stevia inhibits the formation of polysaccharide, necessary for bacterial cell adhesion and dental biofilm formation.7 The lower CFU count for stevia at 12 h may also be attributed to the exhaustion of carbohydrate bulking agents and the incapacity of S. mutans to metabolize stevia with subsequent inhibition of growth of the microorganisms.14-15 The CFU for stevia dropped significantly after 18 h (Table I, Figure 4), which was again confirmed with SEM where only a few bacterial cells were observed and dead cells, forming cob-weblike structures seen on the enamel surface (Figure 6f). Although there was an initial drop in the pH value in the presence of stevia, the value stabilized after 12 h and was elevated after 18 h (Figure 5). This elevation of pH is similar to the findings of Giacaman et al.7 whereby stevia had significant lower acidogenicity compared to other commercial sweeteners.
Overall
The significance of the pH level of the media containing sucrose dropping below pH=5.5 and remaining below this critical level, should be highlighted. The CFU and SEM observations indicate positive bacterial growth in the presence of sucrose, with growth retardation of NSS confirmed with SEM. Although the CFU counts do not reflect this throughout the experiment, it can be attributed to the solid tooth structure vs. liquid medium not providing equivalent conditions in providing bacterial attachment. However, S. mutans activity and metabolism is a causative factor of dental caries due to acid production.24 The results of the study therefore confirmed that the NSS, xylitol, erythritol and stevia, have a lower cariogenic potential compared to sucrose, in vitro, since none of the NSS tested produced a pH lower than 5.5.
Limitations
The limitations of this in vitro study are that it did not fully mimic the in vivo environment that is influenced by buffering of saliva, variable salivary flow rates, different quantities of bacterial material on teeth and the variety of bacteria capable of producing acid or alkaline substances.
CONCLUSION
This study yielded justifiable evidence that NSS (xylitol, erythritol and stevia) have less cariogenic potential when compared to sucrose. Considering CFU counts, pH values and SEM analysis over 24 h, stevia showed the least cariogenic potential of all the NSS tested, followed by erythritol and then xylitol. The safe and effective way to incorporate NSS, as sugar substitutes in the daily diet of children, which can ultimately contribute to lowering the incidence of ECC, should however be further researched.
Conflict of interest
There was no conflict of interest.
REFERENCES
1. Pitts NB, Baez RJ, Diaz-Guillory C, Donly KJ, Alberto Feldens C, McGrath C, Phantumvanit C, Seow P, Kim W, Nikolai S, Yupin S, Norman T, Svante T. Early childhood caries: IAPD bangkok declaration. Journal of Dentistry for Children. 2019; 86(2):72-86. [ Links ]
2. Patel M. Dental caries vaccine: Are we there yet? Lett Appl microbiol. 2020; 70(1):2-12. [ Links ]
3. Mäkinen KK, Isotupa KP, Mäkinen P-L, Söderling E, Song KB, Nam SH, Jeong SH. Six-month polyol chewing-gum programme in kindergarten-age children: A feasibility study focusing on mutans streptococci and dental plaque. International dental journal. 2005; 55(2):81-8. [ Links ]
4. Baer A. Significance and promotion of sugar substitution for the prevention of dental-caries. Lebenson Wiss Technol. 1989; 22(2):46-53. [ Links ]
5. Imfeld T. Efficacy of sweeteners and sugar substitutes in caries prevention. Caries Res 1993; 27(Suppl. 1):50-5. [ Links ]
6. O'Donnell K, Kearsley M. Sweeteners and sugar alternatives in food technology: John Wiley & Sons; 2012. [ Links ]
7. Giacaman RA, Campos P, Munoz-Sandoval C, Castro RJ. Cariogenic potential of commercial sweeteners in an experimental biofilm caries model on enamel. Arch Oral Biol. 2013; 58(9):1116-22. [ Links ]
8. Pfaffmann C, Young P, Dethier V, Richter C, Stellar E. The preparation of solutions for research in chemoreception and food acceptance. J. Comp. Physiol Psychol. 1954; 47(1):93. [ Links ]
9. Bauman RW, Machunis-Masuoka E, Cosby CD. Microbiology: With diseases by body system. 3rd ed: Benjamin Cummings San Francisco; 2012. [ Links ]
10. Glauert AM. Fixation, dehydration and embedding of biological specimens. Volume 3, North- Holland; 1975. [ Links ]
11. Marshall TA. Low intake of sugars may reduce risk of dental caries. J Evid Based Dent Prac. 2014; 14(2):56-8. [ Links ]
12. Chan A, Ellepola K, Truong T, Balan P, Koo H, Seneviratne CJ. Inhibitory effects of xylitol and sorbitol on streptococcus mutans and candida albicans biofilms are repressed by the presence of sucrose. Arch Oral Biol. 2020; 119:104886. [ Links ]
13. Stephan R. Intra-oral hydrogen-ion concentrations associated with dental caries activity. J Dent Res. 1944; 23(4):257-66. [ Links ]
14. Ghezelbash GR, Nahvi I, Rabbani M. Comparative inhibitory effect of xylitol and erythritol on the growth and biofilm formation of oral streptococci. Afr J Microbiol Res. 2012; 6(20):4404-8. [ Links ]
15. 15. Takahashi N. Oral microbiome metabolism: From "who are they?" to "what are they doing?". J Dent Res. 2015; 94(12):1628-37. [ Links ]
16. Miyasawa-Hori H, Aizawa S, Takahashi N. Difference in the xylitol sensitivity of acid production among streptococcus mutans strains and the biochemical mechanism. Mol Oral Microbiol 2006; 21(4):201-5. [ Links ]
17. Badet C, Furiga A, Thébaud N. Effect of xylitol on an in vitro model of oral biofilm. Oral health & preventive dentistry. 2008; 6(4) [ Links ]
18. Jacques N, Hardy L, Campbell L, Knox K, Evans J, Wicken A. Effect of carbohydrate source and growth conditions on the production of lipoteichoic acid by streptococcus mutans ingbritt. Infection and Immunity. 1979; 26(3):1079-87. [ Links ]
19. de Cock P, Mäkinen K, Honkala E, Saag M, Kennepohl E, Eapen A. Erythritol is more effective than xylitol and sorbitol in managing oral health endpoints. Int J Dent. 2016; 6:1-15. [ Links ]
20. Runnel R, Mäkinen KK, Honkala S, Olak J, Mäkinen P-L, Nõmmela R, Vahlberg T, Honkala E, Saag M. Effect of three-year consumption of erythritol, xylitol and sorbitol candies on various plaque and salivary caries-related variables. J Dent. 2013; 41(12):1236-44. [ Links ]
21. Peldyak J, Makinen KK. Xylitol for caries prevention. J Dent Hyg. 2002; 76(4):276-85. [ Links ]
22. Gamboa F, Chaves M. Antimicrobial potential of extracts from stevia rebaudiana leaves against bacteria of importance in dental caries. Acta Odontol Latinoam 2012; 25(2):171-5. [ Links ]
23. Gardana C, Simonetti P, Canzi E, Zanchi R, Pietta P. Metabolism of stevioside and rebaudioside a from stevia rebaudiana extracts by human microflora. J Agric Food Chem. 2003; 51(22):6618-22. [ Links ]
24. Stephan RM. Intra-oral hydrogen-ion concentrations associated with dental caries activity. Journal of dental research. 1944; 23(4):257-66. [ Links ]
 Correspondence:
Correspondence:
Prof. C van Wyk
Department of Community Dentistry, School of Dentistry
Faculty of Health Sciences, University of Pretoria
Gauteng, South Africa, PO Box 1266
Pretoria, 0001, South Africa
E-mail: candice.vanwyk@up.ac.za
Author contributions:
1 . Dr Nadine Moelich 30%
2 . Dr Nicoline Potgieter 20%
3 . Dr Francien S. Botha 15%
4 . Dr James Wesley-Smith 10%
5 . Prof Candice van Wyk 25%













