Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Journal of the Southern African Institute of Mining and Metallurgy
On-line version ISSN 2411-9717
Print version ISSN 2225-6253
J. S. Afr. Inst. Min. Metall. vol.121 n.3 Johannesburg Mar. 2021
http://dx.doi.org/10.17159/2411-9717/1383/2021
PROFESSIONAL TECHNICAL AND SCIENTIFIC PAPERS
Copper slag as a potential source of critical elements - A case study from Tsumeb, Namibia
S. LohmeierI; B.G. LottermoserI; T. SchirmerII; D. GallhoferIII
IInstitute of Mineral Resources Engineering, RWTH Aachen University, Wüllnerstraße 2, 52062 Aachen
IIInstitute of Disposal Research, Department of Mineralogy, Geochemistry, Salt deposits, Clausthal University of Technology, Adolph-Roemer Straße 2A, 38678 Clausthal-Zellerfeld
IIIInstitute of Earth Sciences, University of Graz, Universitätsplatz 2/II, 8010 Graz, Austria
SYNOPSIS
At a time of resource consumption, it is important to study the chemical composition of mining and metallurgical wastes to prevent the dissipative loss of metals and metalloids from the mining value chain. In particular, the recovery of critical elements from wastes is an option to increase the resources of such materials that are economically significant and have an overall supply risk. In this paper we report on the chemical composition, in particular the critical element content, of granulated slag originating from historical smelting activities in the Tsumeb area, Namibia. Laboratory-based inductively coupled plasma-mass spectrometry (ICP-MS) and X-ray fluorescence (XRF) analyses as well as portable X-ray fluorescence (pXRF) demonstrate that the slags are on average enriched in base metals (Cu 0.7 wt%, Pb 2.7 wt%, Zn 4.7 wt%), trace metals and metalloids (Cd approx. 50 mg/kg, Mo approx. 910 mg/kg) as well as critical elements (As approx. 6300 mg/kg, Bi approx. 3 mg/kg, Co approx. 200 mg/kg, Ga approx. 100 mg/kg, In approx. 9 mg/kg, Sb approx. 470 mg/kg). While metals and metalloids such as As, Mo and Pb can be determined reliably using pXRF instruments, the technique has inherent limitations in evaluating the contents of certain critical elements (Ga, Sb). However, there are positive correlations between the As, Mo, and Pb contents determined by pXRF and the Ga and Sb contents obtained through ICP-MS and XRF. Thus, quantitative pXRF analysis for As, Mo, and Pb allows calculation of Ga and Sb abundances in the slags. This work demonstrates that pXRF analysers are a valuable tool to screen smelting slags for their chemical composition and to predict the likely contents of critical elements.
Keywords: base metal slag, portable XRF, critical elements, secondary resource.
Introduction
The consumption of critical elements for high-technology applications is increasing exponentially, and scenarios predict that future demand will exceed current annual worldwide production (European Commission 2010, 2017). Recent improvements in extraction and recovery technologies have the potential to render secondary materials such as copper slags important sources of critical elements (e.g. PGE, In, Bi) (Binnemans et al., 2015).
Nonferrous slags are known to be highly heterogeneus and mineralogically diverse materials (Piatak, Parsons, and Seal II, 2015). They comprise variable amounts of glass and crystallized phases and may also contain relic ore, gangue minerals, and fluxes. The composition of slags depends on the metallurgical processes and the composition of the processed ore(s) and the fluxes used. Consequently, slags display a wide range in bulk chemical composition, with major concentrations (wt% range) of Al2O3, SiO2, and Fe2O3, and lesser amounts of TiO2, MnO, CaO, MgO, Na2O, K2O, P2O5, and S (Piatak, Parsons and Seal II, 2015). In addition, historical nonferrous slags are commonly characterized by elevated metal and metalloid contents as a result of inefficient metal recovery technologies, with wt% levels of Cu, Pb, and Zn (Ettler, Cervinka, and Johan, 2009; Lottermoser, 2002; Vítková et αι., 2010). The metals are contained within the silicate glass and various slag phases, as well as within primary and weathering-induced secondary minerals, including native elements, sulphides, oxides, hydroxides, chlorides, carbonates, sulphates, arsenates, and silicates (Ettler et al., 2001; Piatak, Parsons, and Seal II, 2015). In these studies, laboratory-based methods have commonly been applied for the determination of major (>1 wt%) and trace elements (<0.01 wt%) (Ettler et al., 2009). In particular, inductively coupled plasma-mass spectrometry (ICP-MS) and atomic absorption or emission spectrometry (AAS/AES) have proven their merits for establishing the bulk chemical composition of these slags (Potysz et al., 2015). However, the concentrations of critical elements in slags and the application of field-portable instruments like portable X-ray fluorescence (pXRF) for such targeted analysis remain largely unexplored.
The purpose of this study is to appraise the use of combined ICP-MS, AAS, XRF, and pXRF data to establish the bulk chemistry and the presence of critical elements in granulated copper slags from the Tsumeb smelter, Namibia. The pXRF method is compared with ICP-MS and XRF techniques and the application of pXRF analysis for quantitative analysis of slag samples is validated. Hence, this case study contributes to our understanding of critical elements in slags and demonstrates that pXRF is a useful addition for the chemical characterization of pyrometallurgical wastes.
Background
Smelting site
The Tsumeb smelting complex, currently operated by Dundee Precious Metals Ltd., is located approximately 430 km north of Windhoek, the capital city of Namibia, on the northern slopes of the Otavi Mountains (Figure 1). Smelting in the area goes back to 1907, when the German Otavi Minen-und EisenbahnGesellschaft constructed two lead-copper blast furnaces to smelt local dolomitic ore (Mapani et al., 2014). The present smelter complex was constructed in the early 1960s by Tsumeb Corporation Limited to process largely sulphidic Cu-Pb ore from the Tsumeb mine, and later also ores from Kombat (Kramer and Hultman, 1973) and Khusib Springs (Melcher, 2003). The site originally featured an integrated copper and lead section (with refinery) and smaller plants that intermittently produced cadmium metal, arsenic trioxide, and germanium (Acid Plant Database, 2020; Dundee Precious Metals Tsumeb, 2020; Mapani et al., 2014, and references therein). During the early 1980s, a slag mill was built to reprocess old copper reverbatory slags, which were milled and subsequently treated by flotation. The resulting concentrate was then processed in the smelters to extract Cu and Pb. In 1986, sodium antimonite was produced (Mapani et al., 2014, and references therein). Between 1980 and 1996, the reverbatory furnaces used fuel oil, and the smelting charge was pelletized with pulverized coal, as well as quartz, chert, and lime as fluxes (Ettler et al., 2009; Mapani et al., 2014). Since the flooding of the De Wet Shaft and the subsequent closure of the Tsumeb mine in 1996 (Bowell and Mocke, 2018), copper ores from the Democratic Republic of Congo, Zambia, Mauritania, Botswana, Greece, Russia, and in particular from Bulgaria and South America have been processed in the refurbished Ausmelt furnace, with various fluxes and fuelled with locally produced charcoal and heavy furnace oil (Jarosítková et al., 2017; Kabbash and Smith, 2016; Mapani et al., 2014). The granulated slag is processed in the slag milling plant and the by-product sulphuric acid is sold to industrial consumers in Namibia (Dundee Precious Metals, 2020). Tsumeb blister copper production benefits distinctly from the considerable tolerance of the Tsumeb smelter towards arsenic and lead in copper concentrates feed (Mapani et al., 2014). Resulting from the long processing history, three different types of slags can be distinguished (Ettler et al., 2009). Granulated slag from the reprocessing of older slags (slag type III; Ettler et al., 2009) is the subject of this study.
Materials and methods
Sampling
A large slag dump (approx. 40 000 m2) is located adjacent to the smelting complex and contains approximately 1.5 Mt of slag. At present, the reprocessed slags comprise granulated fragments ranging in grain size from a few millimetres in diameter to predominantly powder-size due to granulation and/ or milling (Jarosítková et al., 2017; Figures 1, 2, 3). During sampling in 2018, the slag heap was divided into 20 equal sectors (approximately 1 000 m2). Each sector was covered by a number of parallel traverses to obtain a representative 5 kg sample of powdered to millimetre-sized slag pieces from each sector (sample numbers TSS1-TSS20). An additional slag hand specimen was taken during a field sampling campaign in 2019 (sample number TSSF).

Sample processing and laboratory-based analysis
Slag samples were air-dried and homogenized. A representative aliquot was milled to analytical fineness using a tungsten-carbide swing mill in the Department of Mineral Processing at RWTH Aachen University. Milled powders were sent to SGS Bulgaria, Bor Laboratory/Serbia for conventional ICP-MS analysis for Al, As, Ba, Be, Bi, Ca, Cd, Ce, Co, Cr, Cs, Cu, Fe, Ga, Hf, In, K, La, Li, Lu, Mg, Mn, Mo, Na, Nb, Ni, P, Rb, Sb, Sc, Se, Sn, Sr, Ta, Tb, Te, Ti, Th, Tl, U, V, W, Y, Yb, and Zr after HNO3-HF-HClO4 and HCl digestion. Pb and Zn were analysed by AAS. Samples with high Cu values were re-analysed by AAS. In addition, milled powders were submitted to Australian Laboratory Services (ALS, Loughrea, Ireland) for conventional XRF analysis for major elements (Al, Ca, Fe, K, Mg, Mn, Na, P, S, Si, Ti), and minor/trace elements (As, Ba, Cl, Cr, Cu, Mo, Pb, Sb, Zn). LOI was determined by sintering at 1000°C (Table I).
XRD analyses were performed on pulps (<2 pm), using an X'Pert Pro (PANalytical) instrument with data collector and an X'Pert HighScore system equipped with a Cu-LFF (Empyrian) tube and an ADS tool at the Institute of Disposal Research (IDR) at Clausthal University of Technology (TUC). Qualitative evaluation was done using the X'Pert HighScore software from PANalytics (Figure 4).
Electron microprobe (EMP) mapping of As, Cu, Ni, Pb, and Sb was done on a slag hand-specimen mounted in epoxy resin at the IDR, TUC (Figure 3). An In-As alloy was used for As (Lαi), chalcopyrite for Cu (Kα), pentlandite for Ni (Kα), crocoite for Pb (Mα), and stibnite for Sb (Lα) calibration. EMP analyses were performed using an acceleration voltage of 15 kV and a 20 nA beam current.
Portable X-ray fluorescence spectroscopy
Chemplex sample cups were filled with milled sample powders. ProleneTM thin films were used to guarantee simple, and at the same time comparable, analytical settings. All sample cups were backfilled with stuffing fibre. Analysis was carried out using a Niton XL3t 900 hand-held XRF instrument connected to a radiation protection chamber at the IDR (TUC). The analyser is equipped with a 50 kV Ag target X-ray tube. Analyses were done in 'environmental mode - minerals with Cu/Zn', with a total measurement time of 100 seconds (Table II). Analyses were repeated five times. To test reproducibility and homogeneity two sample cups were prepared for each pulp; the results were almost identical. The instrument was calibrated using the following certified reference materials (CRMs): (i) OREAS 24b, 24c, 36, 37, 112, 131b, 132b, 133b, 134b, 160, 623, 932 of the OREAS pXRF Zn-Pb-Ag sulphide kit; (ii) RTS-3a and MP-1b CRMs of the CANMET Mining and Mineral Sciences Laboratories Canada, and (iii) SRM 2780 of the National Institute of Standards and Technology. Calibration factors, slope, and intercept, were obtained using the provided CorrectCalc software program of the Niton device. To monitor the drift of the instrument and to assure quality control, a set of six CRMs (OREAS 36, 131b, 132b, 133b, 134b, 623) was analysed three times at the start and at the end of every measuring day plus once after every sample (plus duplicate).
For calibration of the pXRF device, regression analysis was done using the aforementioned CRMs. The quality of the linear relationship between a CRM and a pXRF value is expressed by the coefficient of determination (R2), which is at the best 1.00. Very good R2 values were obtained for TiO2, MnO, CaO, K2O, As, Ba, Cd, Cu, Mo, Pb, Rb, Sn and Zn (R2 = 0.99) and for SiO2, Fe2O3, S, Sb, Sr and Zr (R2 = 0.96-0.98), when filtered for outliers. However, R2 is low for Al2O3 (R2 = 0.53), and only acceptable for MgO (R2 = 0.86). Root mean square errors (RMSEs) are largely below unity, with the exception of SiO2, Al2O3, and Fe2O3 (Table III).
Precision (i.e. the degree to which repeated measurements under unchanged conditions show the same result) and accuracy ( i.e. proximity of measurement results to the true value) are important parameters to evaluate the quality of chemical analyses. Precision of pXRF values can be assessed via the percentage relative standard deviation (RSD), and accuracy via the relative difference (%RD), using the criteria after Jenner (1996) and Piercy and Devine (2014). Most major elements, metals, and metalloids show on average excellent (RSD 0-3%) to good (RSD 7-10%) precision, with the exception of MgO, Cd, and Rb (RSD >10%). Considering average values, As, Ba, Cu, Mo, Pb, Sn, and Zn show excellent precision (RSD 0-3%) and Sb very good precision (RSD 7-10%).
The average accuracy of pXRF data, compared to ICP-MS data and XRF data, is variable. Elements/oxides like SiO2, TiO2, Fe2O3, Cd, Cu, Mo, Pb, Sr, and Zn show an acceptable accuracy (%RD < 20) compared to the ICP-MS and XRF values. An acceptable accuracy is also evident for MnO, CaO, Ba, and Sb compared to the XRF data, but accuracy is only within the 23-36%RD range compared to ICP-MS results. The average accuracy of As is close to 20%RD. Accuracy for Al2O3 is poor, as well as for MgO, Rb, and in particular Sn.
Results and discussion
Slag mineralogy
Granulated slags from Tsumeb are largely composed of X-ray amorphous substances, in particular vitreous materials, which cannot be distinguished by XRD. This feature is clearly reflected in all XRD patterns by the lack of clearly defined X-ray peaks (Figure 4). Only (synthetic) augite and quartz could be identified in most slag samples. In addition, an uncommon spinel with Cu-Zn-Al compounds is present, as well as unknown Mg-Al oxide, Mg-Fe oxide, and Pb oxide phases. The 'quartz' proportion can probably be ascribed to the high-temperature, low-pressure SiO2 compound tridymite and/or cristobalite (Figure 5). No sulphide minerals, intermetallic compounds, pure metals, or arsenates (cf. Ettler et al., 2009) could be detected in the sampled materials.
Element mapping of As, Cu, Ni, Pb, and Sb (sample TSSF) reveals a largely very heterogeneous distribution of these elements within granulated slag powder (Figure 3) as well as within slag particles. Moreover, imaging gives clear evidence for predominance of glassy fragments in quenched milled slag. Sulphides and oxides are rare. There is a certain match between element distribution maps of As and Sb, as well as of Cu and Pb, but the distinctly greater abundance of As over Sb and Pb over Cu is obvious.
Slag geochemistry
The chemical composition of the slag samples is reported in Table I. The slags are largely composed of SiO2 (23.42-39.11 wt%; av. 34.34 wt%), Fe2O3TOT (28.76-39.88 wt%; av. 33.86 wt%), and CaO (8.88-17.50 wt%; av. 12.60 wt%), as well as minor Al2O3 (3.03-4.85 wt%; av. 3.91 wt%) and MgO (3.01-5.52 wt%; av. 4.22 wt%) as is typical for base metal slags (Figure 5). P2O5 and S contents are comparatively low at 0.19-0.45 wt% (av. 0.30 wt%) and 0.12-1.13 wt % (0 0.48), respectively. However, the slags are significantly enriched in metals and metalloids: Pb (1.55-7.40 wt%; av. 2.68 wt%), Zn (2.11-9.25 wt%; av. 4.70 wt%), Cu (4 986-15 400 mg/kg; av. 7 290 mg/kg), As (2 094-11 200 mg/kg; av. 5 607 mg/kg), Ba (1 3736 359 mg/kg; av. 3 692 mg/kg), and Mo (478-1 465 mg/kg; av. 913 mg/kg). Likewise, high contents of >100 mg/kg are shown by Cr (152-447 mg/kg; av. 331 mg/kg), Co (117255 mg/kg; av. 203 mg/kg), Ga (47-194 mg/kg; av. 101 mg/kg), Sb (215-822 mg/kg; av. 470 mg/kg), Sn (39-123 mg/kg; av. 73 mg/kg), Sr (224-459 mg/kg; av. 331 mg/kg), and V (81-231 mg/kg; av. 128 mg/kg). Several of these elements (As, Cr, Co, Ga, Sb, V) are considered as critical elements according to the USGS (2018). The bulk chemical composition of the granulated slags generally agrees with that documented by Ettler et al. (2009) and Jarosítková et al. (2017). Lower As and S as well as high Cd and Sb values detected in this study are likely due to higher sample numbers (Ettler et al., 2009: n = 2; this study: n = 20).
Compared to average crustal abundances, the granulated slag is highly enriched in As [approx. 2 530 x], Pb, [approx. 2 450 x], Sb [approx. 2 350 x], and Mo [approx. 1 140 x]. Zn [approx. 650 x], Cd [approx. 635 x], Cu [270 x], Te [approx. 180 x], In [approx. 170 x], and W [approx. 115 x] also are distinctly enriched; however, W values have to be interpreted with caution as a tungsten-carbide mill was used for milling. Moreover, a notable enrichment is shown by Se [64 x], Sn [43 x], U [24 x], Bi [17 x], S [12 x], Ba [12 x], Co [8 x], and Ga [6 x (Figure 6). The major elements Fe, Mn [4 x], Ca and P [3 x] are slightly enriched, as well as Cr, Th, Ta, Ni, Ce, and Tl [2 x]. La, Y, Li, Tb, Yb, Lu, Sr, Mg, V, Nb, Hf, Zr, and Si have the same abundance as in average bulk continental crust. Be, Ti, Cs, Rb, Na, K, Al, and Sc are slightly depleted. The important critical elements As, Sb, Te, and In are among the most enriched elements in Tsumeb granulated slag. In addition, although less enriched, are the four critical elements Sn, U, Bi, Ga, and Co.
Prediction of geochemical composition from pXRF data
XRF and ICP-MS analysis are well-established techniques for determining the composition of geological materials and metallurgical wastes (e.g. Potysz et al., 2015). Such instruments used for elemental analysis must be operated in a controlled laboratory environment. Progress in XRF instrumentation has opened the way to measurement of materials in the field by means of portable XRF analysis. Portable XRF can screen for multiple elements simultaneously in a variety of materials, using one device with minimal sample preparation. However, many critical elements are only poorly determined in materials by the energy-dispersive pXRF technology (Gallhofer and Lottermoser 2018). Comparison of laboratory-generated XRF and ICP-MS data with pXRF data reveals specific element trends and may allow predictions of likely chemical compositions (Figure 7).
The chemical composition of slag samples analysed by pXRF is reported in Table II. To compare pXRF with ICP-MS/AAS and XRF data, linear regression functions were calculated for pXRF -ICP-MS/AAS and pXRF - XRF data-sets, which reveal very good correlations (R2 > 0.90) for As, Ba, Cd, Mo, P, Pb, Si, Sn, Sr, and Zn (Figure 7). Good correlations (R2 > 0.80) are shown by Ca and Mn. Cu shows distinctly better correlations for the pXRF - ICP-MS/AAS values than for the pXRF - XRF data-sets (RrcP-MS/AAS-pXRF = 0.94; RXRF-pXRF = 0.81), but for Sb the opposite is the case (Ricp-Ms/AAs-pxRF = 0.73; RxRF-pxRF = 0.92; Figure 7). Correlation of ICP-MS and XRF data is good for most elements (R2 > 0.80), and for major elements and Sb within an acceptable range (R2 > 0.70).
Analysis of critical elements using pXRF is notoriously difficult. In the studied slags, pXRF provides precise and accurate data for only As. Other critical elements (e.g. Ga, Sb, Sn) can be detected; however, accuracy is either poor or concentrations are close to the detection limit. Cobalt is difficult to analyse by pXRF, in particular if Fe is present (Sieber and Pella, 1986), due to X-ray peak overlap (Gallhofer and Lottermoser, 2018), although concentrations are in the 100-200 mg/kg range.
Calculation of correlation coefficients for element pairs from the ICP-MS and pXRF data-sets reveals clear positive correlations of Ga, Sb, and Sn (analysed by ICP-MS) with As (analysed by pXRF), as well as a less certain correlation of Mo and As. Current commercial pXRF detectors are not capable of analysing Ga routinely. Only a few detectors can analyse for Ga if concentrations are well above 100 mg/kg (Williams-Thorpe, 2008), and if the sample matrix allows Ga detection (Lemière, 2018). For the Tsumeb slags, considering that Ga concentrations correlate with the As and Mo concentrations, Ga contents can be indirectly determined by pXRF analysis of As and Mo. pXRF analyses of both As and Mo are of excellent precision, and accuracy is acceptable for As (RSD = 1.79; %RDICP-MS = 22; %RDXRF = 25; averages) for both pXRF - ICP-MS and pXRF -XRF data-sets and is good for Mo (RSD = 1.28; RDICP-MS = 7.39; %RDXRF = 4.47; averages; Figure 8). Hence, the following linear regressions allow calculation of Ga concentrations:
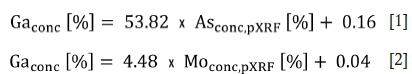
Antimony is another critical element enriched in the Tsumeb slag, although a certain portion has already been extracted. As direct analysis by pXRF of Sb is acceptable, but not of good precision (RSD = 3.53; %RDICP-MS = 23; %RDXRF = 10), indirect verification of pXRF-derived Sb data is recommended using Pb pXRF-derived data (Bero et al., 1993). The following linear formula allows the indirect calculation of Sb contents using Pb values:

Thus, a direct analysis of the critical element As enriched in Tsumeb granulated slag using low-cost on-site pXRF analysis is possible. In addition, indirect analysis of Ga and Sb is feasible, using As and Mo or Pb and As pXRF-derived data, respectively.
Reprocessing potential of Tsumeb slags
Slag compositions are influenced by the metallurgical process used and the composition of smelted ores and fluxes. At the Tsumeb smelter, ores of the Tsumeb deposit as well as other deposits close by in the Otavi Mountain Land were originally processed, which are well-known for their metal and metalloid enrichment (i.e. Pb, Cu, Zn, Ag, As, Sb, Cd, Co, Ge, Ga, Au, Fe, Hg, Mo, Ni, Sn, W and V) (Bowell, 2014; Frimmel, Deane and Chadwick, 1996; Melcher, 2003; Melcher, Oberthür and Rammlmair, 2006; Pirajno and Joubert, 1993).
Overall, approximately 30 Mt ore at 10% Pb, 4.3% Cu, and 3.5% Zn plus significant quantities of As, Sb, Ag, Cd, and Au were extracted from the Tsumeb deposit between 1907 and 1996 (Melcher 2003). Over time, these ores and other base metal ores from mines close by, and since 1996 from several deposits overseas, were processed in the Tsumeb smelter complex, producing Pb metal, blister Cu, and Zn concentrates with by-products Ag, Au, Ge, and Cd, and intermittently As and Sb (Kramer and Hultman, 1973; Kamona et al, 1999; Mapani et al., 2014). Therefore, the studied slags do not arise from the processing and smelting of a single ore. The chemical composition of the studied slags is the result of the metallurgical treatment of diverse base metal ores and reprocessing of older slag wastes. The Tsumeb smelter is one of few smelters worldwide that is capable of processing mixed Cu-Pb ore with high As contents (Mapani et al., 2014), and Tsumeb sulphidic ore is known for its arsenate content (Bowell, 2014). Thus, the very high As concentration detected within the metallurgical waste is a logical consequence; although some arsenic was temporarily extracted for production of arsenic trioxide (cf. Acid Plant Database, 2020). In the past, poor recovery technologies for base metals, trace metals, and metalloids as well as critical elements led to the observed element enrichments.
Tsumeb slags are enriched in the critical elements As, Bi, Ga, In, Sb, Sn, Te, and U. To date, the Southern African mineral processing and smelting industry has extracted Cu, Pb, Cd, and Sb from Tsumeb slags and flotation tailings (Guest, Svoboda, and Venter, 1988; Svoboda, Guest, and Venter, 1988). In addition, the recovery of Co from slags is possible, as demonstrated by Jones et al. (2002). Moreover, there are numerous studies that focus on recovery of trace elements from mining and processing residues using different technical approaches to recycle high-tech elements (e.g. Anand, Kanta Rao, and Jena, 1980; Gbor, Ahme, and Jia, 2000; Gbor, Hoque, and Jia, 2006; Tshiongo, Mbaya, and Maweja, 2011; Tümen and Bailey, 1990; Yang et al., 2010).
The Tsumeb copper slag is an under-exploited residue, from which metals, metalloids, and critical elements may be derived, e.g. Sn (Mapani et al., 2014). Successful recovery of raw materials from the existing waste would require reprocessing and adjustments to the existing metallurgical processes that would allow critical element extraction. The additional benefit of such waste valorization practices would be the prevention of metalliferous drainage and dust dispersion from the slag dump in the long term (Dundee Precious Metals Tsumeb, 2020; Ettler et al., 2009; Kríbek et al., 2014, 2018; Mapani et al., 2014). For example, the amorphous glassy slag matrix exhibits a certain susceptibility to leaching during weathering (Mostafa et al., 2001; Tshiongo, Mbaya, and Maweja, 2011). Thus, recovery of additional metals from nonferrous slags as by-products would result in two benefits: it could yield critical raw materials and prevent the development of significant environmental impacts from waste disposal.
Conclusion
This study aimed to chemically characterize the granulated copper slag at the Tsumeb smelter site and to demonstrate its potential as source of critical raw materials. In addition, it was shown that pXRF is a useful addition to the techniques for chemical characterization of pyrometallurgical wastes. The results of this study demonstrate that
(i) Tsumeb slags are enriched in base metals (Cu, Pb, Zn), trace metals and metalloids (Ba, Cd, Mo, S, Se), and critical elements (As, Bi, Ga, In, Sb, Sn, Te, U)
(ii) The critical element As can be determined in smelting slags by pXRF at excellent precision and accuracy
(iii) Other critical elements like Ga and Sb may be determined using element proxies (As, Mo, Pb) and simple linear regression functions
(iv) pXRF can be used as an additional low-cost tool for screening the chemical composition of smelting slags.
Acknowledgements
This work was supported by the German Federal Ministry of Education and Research (BMBF) and is part of the sub-Saharan based LoCoSu project; grant number 01DG16011. We thank staff of the Institute of Disposal Research of Clausthal University of Technology (Germany) for their help with the preparation of polished thin sections and access to the microprobe. Thanks to F. Türck for his patience and support during the many long days of pXRF analysis. Thanks to the Department of Processing at the RWTH Aachen University, in particular D. Gürsel and P. Ihl, for providing access to powder preparation equipment and their never-ending patience. Thanks to Dr. R. Ellmies (Gecko Namibia) for help and patience in the field.
Authors contribution
Conceptualization: B.G.L.; sampling: B.G.L., D.G., S.L.; methodology: D.G., S.L.; validation: S.L.; formal analysis: S.L., T.S.; data curation: S.L.; writing - original draft preparation: S.L.; writing - review and editing: B.G.L., D.G., T.S.; funding acquisition: B.G.L.
References
Acid Plant Database. 2020. http://www.sulphuric-acid.com/sulphuric-acid-on-the-web/acid%20plants/Namibia-Custom-Smelter.htm [accessed 19 May 2020]. [ Links ]
Anand, S., Kanta Rao, P., and Jena, P.K. 1980. Recovery of metal values from copper converter and smelter slags by ferric chloride leaching. Hydrometadlurgy, vol. 5. pp. 355-365. [ Links ]
Bero, B.N., von Braun, M.C., Knowles, C.R., and Hammel, J.E. 1993. The use of X-ray fluorescence to detect lead contamination of carpeted surfaces. Environment, Monitoring and Assessment, vol. 27. pp. 17-33. [ Links ]
Binnemans, K., Jones, P.T., Blanpain, B., van Gerven, T., and Pontikes, Y. 2015. Towards zero-waste valorisation of rare-earth-containing industrial process residues: a critical review. Journal of Cleaner Production, vol. 99. pp. 17-38. [ Links ]
Bowell, R.J. 2014. Hydrogeochemistry of the Tsumeb deposit: Implications for arsenate mineral stability. Reviews in Mineralogy and Geochemistry, vol. 79. pp. 589-627. [ Links ]
Bowell, R.J. and Mocke, H. 2018. Minerals new to Tsumeb. Communications of the Geological Survey of Namibia, vol. 19. pp. 20-46. [ Links ]
Dundee Precious Metals Tsumeb. 2020. https://www.dundeeprecious.com/English/Operating-Regions/Current-Operations/Tsumeb/Overview/default.aspx [accessed 19 May 2020]. [ Links ]
Ettler, V., Cervinka, R., and Johan, Z. 2009. Mineralogy of medieval slags from lead and silver smelting (Bohutin, Pribram district, Czech Republic): Towards estimation of historical smelting conditions. Archaeometry, vol. 51, no. 6. pp. 987-1007. [ Links ]
Ettler, V., Johan, Z., Kríbek, B., Sebek, 0., and Mihaljevic, M. 2009. Mineralogy and environmental stability of slags from the Tsumeb smelter, Namibia. Applied Geochemistry, vol. 24. pp. 1-15. [ Links ]
Ettler, V., Legendre, 0., Bodénan, F., and Touray, J.-C. 2001. Primary phases and natural weathering of old lead-zinc pyrometallurgical slag from Pribram, Czech Republic. The Canadian Mineralogist, vol., 39. pp. 873-888. [ Links ]
European Commission. 2010. Critical raw materials for the EU. Report of the ad-hoc working group on defining critical raw materials. European Commission, Brussels. [ Links ]
European Commission. 2017. Study on the review of the list of critical raw materials -criticality assessments. European Commission, Brussels. [ Links ]
Frimmel, H.E., Deane, J.G., and Chadwick, P.J. 1996. Pan-African tectonism and the genesis of base metal sulfide deposits in the northern foreland of the Damara orogen, Namibia. Society of Economic Geologists, Special Publications, vol. 4. pp. 204-217. [ Links ]
Gallhofer, D. and Lottermoser, B.G. 2018. The influence of spectral interferences on critical element determination with portable X-ray fluorescence (pXRF). Minerals, vol. 8, no. 320. doi: 10.3390/MIN8080320 [ Links ]
Gbor, P.K., Ahmed, I.B., and Jia, CO. 2000. Behaviour of Co and Ni during aqueous sulphur dioxide leaching of nickel smelter slag. Hydrometallurgy, vol. 57. pp. 13-22. [ Links ]
Gbor, P.K., Hooue, S., and Jia, CO. 2006. Dissolution behaviour of Fe, Co, and Ni from non-ferrous smelter slag in aqueous sulphur dioxide. Hydrometallurgy, vol. 81. pp. 130-141. [ Links ]
Guest, R.N., Svoboda, J., and Venter, W.J.C. 1988. The use of gravity and magnetic separation to recover copper and lead from Tsumeb flotation tailings. Journal of the South African Institute of Mining and Metallurgy, vol. 88, no. 1. pp 21-26. [ Links ]
JaroSítková, Α., Ettler, V., Mihaljevic, M., Kríbek, B., and Mapani, B. 2017. The pH-dependent leaching behaviour of slags from various stages of a copper smelting process: environmental implications. Journal of Environmental Management, vol. 187. pp. 178-186. [ Links ]
Jenner, G.A. 1996. Trace element geochemistry of igneous rocks: geochemical nomenclature and analytical geochemistry. Geological Association of Canada, Short Course Notes, vol. 12. pp. 51-77. [ Links ]
Jones, R.T., Denton, G.M., Reynolds, O.G., Parker, J.A.L., and van Tonder, G.J.J. 2002. Recovery of cobalt from slag in a DC arc furnace at Chambishi, Zambia. Journal of the South African Institute of Mining and Metallurgy, vol. 102, no. 1. pp. 5-9. [ Links ]
Kabbash, Α. and Smith, Α. 2016. Dundee's world-class acid plant demonstrates the latest in sustainable technology. Sulfuric Acid Today, vol. 2, no. 2. pp. 30-32. [ Links ]
Kamona, A.F., Lévêoue, J., Friedrich, G., and Haack, U. 1999. Lead isotopes of the carbonate-hosted Kabwe, Tsumeb, and Kipushi Pb-Zn-Cu sulphide deposits in relation to Pan African orogenesis in the Damaran-Lufilian fold belt of Central Africa. Mineralium Deposita, vol. 34. pp. 273-283. [ Links ]
Kramer, R. and Hultman, T. 1973. Tsumeb - a profile of United States contribution to underdevelopment in Namibia. Corporate Information Centre of the National Council of Churches, New York. [ Links ]
KrIbek, B., Majer, V., PaSava, J., Knésl, I., Kamona, F., Mapani, B., Mwiya, S., Kawali, L., Kandjii, L., and Keder, J. 2014. Impact of ore processing on the environment in the Tsumeb area, Namibia. Communications of the Geological Survey of Namibia, vol. 15. pp. 111-116. [ Links ]
KrIbek, B., Sípková, Α., Ettler, V., Mihaljevic, Μ., Majer, V., Knésl, I., Mapani, B., PenEek, V., Vanëk, Α., and Sracek, O. 2018. Variability of the copper isotopic composition in soil and grass affected by mining and smelting in Tsumeb, Namibia. Chemical Geology, vol. 493. pp. 121-135. [ Links ]
Lemère, B. 2018. A review of pXRF (field portable X-ray fluorescence) applications for applied geochemistry. Journal of Geochemical Exploration, vol. 188. pp. 350-363. [ Links ]
Lottermoser, B.G. 2002. Mobilization of heavy metals from historical smelting slag dumps, north Queensland, Australia. Mineralogical Magazine, vol. 66, no. 4. pp. 475-490. [ Links ]
Mapani, B., Ellmies, R., Hahn, L., Schneider, G., Ndalulilwa, Κ., Leonard, R., Zeeuw, Μ., Mwananawa, N., Uugulu, S., Namene, Ε., Amaambo, W., Sibanda, F., and Mufenda, Μ. 2014. Contamination of agricultural products in the surrounding of the Tsumeb smelter complex. Communications of the Geological Survey of Namibia,, vol. 15. pp. 92-110. [ Links ]
Melcher, F. 2003. The Otavi Mountain Land in Namibia: Tsumeb, germanium and snowball earth. Mitteilungen der Österreichischen Geologischen Gesellschaft, vol. 148. pp. 413-435. [ Links ]
Melcher, F., Oberthür, Τ., and Rammlmair, D. 2006. Geochemical and mineralogical distribution of germanium in the Khusib Springs Cu-Zn-Pb-Ag sulfide deposit, Otavi Mountain Land, Namibia. Ore Geology Reviews, vol. 28. pp. 32-56. [ Links ]
Mostafa, N.Y., El-Hemaly, S.A.S., Al-Wakeel, E.I., El-Korashy, S.A., and Brown, P.W. 2001. Characterization and evaluation of the hydraulic activity of water-cooled slag and air-cooled slag. Cement and Concrete Research, vol. 31. pp. 899-904. [ Links ]
Piatak, N.M., Parsons, M.B., and Seal II, R.R. 2015. Characteristics and environmental aspects of slag: a review. Applied Geochemistry, vol. 57. pp. 236-266. [ Links ]
Piercy, S.J. and Devine, M.C. 2014. Analysis of powdered reference materials and known samples with a benchtop, field portable X-ray fluorescence (pXRF) spectrometer: evaluation of performance and potential application for exploration lithogeochemistry. Geochemistry: Exploration, Environment, Analysis, vol. 14, no. 2. pp. 139-148. [ Links ]
Pirajno, F. and Joubert, B.D. 1993. An overview of carbonate-hosted mineral deposits in the Otavi Mountain Land, Namibia: implications for ore genesis. Journal of African Earth Sciences, vol. 16, no. 3. pp. 265-272. [ Links ]
Potysz, Α., van Hullebusch, E.D., Kierczak, J., Grybos, Μ., Lens, P.N.L., and Guibaud, G. 2015. Copper metallurgical slags - current knowledge and fate: a review. Critical Reviews in Environmental Science and Technology, vol., 45. pp. 2424-2488. [ Links ]
Rudnick, R.L. and Gao, S. 2003. Composition of the continental crust. Treatise on Geochemistry, vol. 3. pp. 1-64. [ Links ]
Sieber, J.R. and Pella, P.A. 1986. Improved determination of cobalt in steel by X-ray fluorescence analysis. X-ray Spectrometry, vol. 15. pp. 287-288. [ Links ]
Svoboda, J., Guest, R.N., and Venter, W.J.C. 1988. The recovery of copper and lead minerals from Tsumeb flotation tailings by magnetic separation. Journal of the South African Institute of Mining and Metallurgy, vol. 88, no. 1. pp. 9-19. [ Links ]
Tshiongo, N., Mbaya, R.K.K., and Maweja, Κ. 2011. Leaching kinetics of Cu, Co, Zn, Pb and Fe from copper smelting slags cooled in different ways after tapping. Proceedings of the VI Southern African Base Metals Conference, Phalaborwa, 18-21 July 2011. Southern African Institute of Mining and Metallurgy, Johannesburg. pp. 463-476. [ Links ]
Tümen, F. and Bailey, N.T. 1990. Recovery of metal values from copper smelter slags by roasting with pyrite. Hydrometallurgy, vol. 25. pp. 317-328. [ Links ]
USGS. 2018. Draft critical mineral list - summary of methodology and background information - US Geological Survey technical input document in response to Secretarial Order no. 3359. Open-File Report 2018-1021. https://doi.org/10.3133/ofr20181021 [ Links ]
Vítková, Μ., Ettler, V., Johan, Ζ., Kříbek, Β., Šebek, O., and Mihaljevic, Μ. 2010. Primary and secondary phases in copper-cobalt smelting slags from the Copperbelt Province, Zambia. Mineralogical Magazine, vol. 74, no. 4 pp. 581-600. [ Links ]
Wedepohl, K.H. 1969. Handbook of Geochemistry. 1st edn. Springer, BerlinHeidelberg. [ Links ]
Williams-Thorpe, O. 2008. The application of portable X-ray fluorescence analysis to archaeological lithic provenancing. Portable X-ray Fluorescence Spectrometry -Capabilitiesfor in situ Analysis. Potts, P.J. and West, M. (eds.). Royal Society of Chemistry. Cambridge, UK. pp. 174-205. [ Links ]
Yang, Ζ., Rui-Lin, Μ., Wang-Dong, N., and Hui, W. 2010. Selective leaching of base metals from copper smelter slag. Hydrometallurgy, vol. 103. pp. 25-29. [ Links ] ♦
 Correspondence:
Correspondence:
S. Lohmeier
Email: lohmeier@mre.rwth-aachen.de stephanielohmeier2017@gmail.com
Received: 15 Sep. 2020
Accepted: 5 Dec. 2020
Published: March 2021
ORCID
S. Lohmeier https://orchid.org/0000-0003-2556-2096
B.G. Lottermoser https://orchid.org/0000-0002-8385-3898
T. Schirmer https://orchid.org/0000-0003-3938-5860
D. Gallhofer https://orchid.org/0000-0003-2139-7847














