Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Industrial Engineering
On-line version ISSN 2224-7890
Print version ISSN 1012-277X
S. Afr. J. Ind. Eng. vol.25 n.2 Pretoria Aug. 2014
CASE STUDIES
Multi-objective optimisation in carbon monoxide gas management at Tronox KZN Sands
J. Stadler; Bekker*
Department of Industrial Engineering Stellenbosch University, South Africa. Jb2@sun.ac.za
ABSTRACT
Carbon monoxide (CO) is a by-product of the ilmenite smelting process from which titania slag and pig iron are produced. Prior to this project, the CO at Tronox KZN Sands in South Africa was burnt to get rid of it, producing carbon dioxide (CO2). At this plant, unprocessed materials are pre-heated using methane gas from an external supplier. The price of methane gas has increased significantly; and so this research considers the possibility of recycling CO gas and using it as an energy source to reduce methane gas demand. It is not possible to eliminate the methane gas consumption completely due to the energy demand fluctuation, and sub-plants have been assigned either CO gas or methane gas over time. Switching the gas supply between CO and methane gas involves production downtime to purge supply lines. Minimising the loss of production time while maximising the use of CO arose as a multi-objective optimisation problem (MOP) with seven decision variables, and computer simulation was used to evaluate scenarios. We applied computer simulation and the multi-objective optimisation cross-entropy method (MOO CEM) to find good solutions while evaluating the minimum number of scenarios. The proposals in this paper, which are in the process of being implemented, could save the company operational expenditure while reducing the carbon footprint of the smelter.
OPSOMMING
Koolstofmonoksied (CO) is 'n newe-produk van die ilmenietsmeltproses waardeur titaniumslak en ru-yster geproduseer word. Voor die uitvoering van hierdie projek is die CO gas wat deur Tronox KZN Sands, Suid-Afrika produseer is, verbrand om daarvan ontslae te raak. Die verbranding het weer koolstofdioksied produseer. By hierdie aanleg word ongeprosesseerde materiale voorverhit met metaangas wat van 'n eksterne verskaffer aangekoop word. Die prys van metaangas het beduidend toegeneem, en die moontlikheid om CO te herbruik in die plek van metaangas is in hierdie studie ondersoek. Weens die variasie in vraag na metaangas, kan die gebruik daarvan nie volledig uitgeskakel word nie; dus moet of CO of metaangas aan sub-aanlegte oor tyd toegeken word. Sodra daar 'n oorskakeling tussen die twee gassoorte plaasvind, moet die toevoerlyne eers gespoel word; en dit veroorsaak verlies van produksietyd. 'n Multidoelwit-optimeringprobleem het dus ontstaan waarin verlies aan produksietyd minimeer word terwyl CO gasgebruik maksimeer word. Hierdie probleem bevat sewe besluitveranderlikes, en scenario's is met rekenaarsimulasie ge-evalueer. Rekenaarsimulasie en die kruis-entropie metode vir multidoelwit-optimering is toegepas om goeie oplossings te verkry terwyl die minimum aantal scenario's evalueer is. Die voorstelle, wat tans in die proses is om implementeer te word, sal die onderneming operasionele koste bespaar en die koolstofspoor verklein.
1 INTRODUCTION
In a minerals processing environment, stable production processes, cost minimisation, and energy efficiency are key to operational excellence, safety, and profitability. At an ilmenite smelter, typically found in the mineral sands industry, it is no different. Management of the ilmenite smelting process is a complex, multi-variable challenge involving high costs and safety risks.
A by-product of ilmenite smelting is superheated carbon monoxide (CO) gas, or furnace offgas. This gas is flammable and extremely poisonous to humans. At the same time, CO is a potential energy source for various on-site heating applications. Re-using furnace off-gas can increase the energy efficiency of the energy-intensive smelting process, and can save the cost of procuring other gas for heating purposes.
In this paper we show how computer simulation and multi-objective optimisation using the cross-entropy method (MOO CEM) were successfully applied in a project to near-optimise the use of CO gas at the Tronox KZN Sands operation in South Africa. The research contribution focuses on the applicability of the MOO CEM to an industrial problem.
2 MINERAL SANDS INDUSTRY
The mineral sands industry forms an important link in the macro-economic value chain, as it is a key source industry for a variety of consumer industries as well as for the construction, motor, and aerospace industries. Mineral sands mining and beneficiation involve unique, sophisticated processes and technology that contribute to the complex operational and legislative environment associated with the industry.
Most mineral sands deposits are found in unconsolidated fossil shorelines, mostly several hundreds of metres to tens of kilometres inland from the present coastline. Mineral sands ore bodies essentially fall into two categories based on the mode of deposition: alluvial (deposited by water) or aeolian (deposited by wind) [1]. These deposits are found on almost every continent, with the most significant ones occurring in South Africa, Australia, and North America. The industry consists of two principal product streams: titanium dioxide (TiO2) minerals in the form of rutile, ilmenite, and leucoxene; and zircon [2].
3 TRONOX KZN SANDS
Tronox KZN Sands is one of three active mineral sands operations in South Africa, sand is located on the north coast of the KwaZulu-Natal (KZN) province near the town of Richards Bay. The company is a newly-acquired business unit of Tronox Limited, a chemicals firm listed on the New York Stock Exchange. The company started in 1994 as the Iscor Heavy Minerals project, with the first product being produced in 2001. During 2012 the managing share was sold by the South African top-40 mining company, Exxaro Resources, to US-based Tronox. The KZN business unit forms part of the bigger Mineral Sands division of Tronox, which includes another operation in South Africa (Tronox Namakwa Sands) and one in Western Australia (Tronox Northern Operations). Tronox is now the world's largest fully-integrated producer of titanium dioxide pigment, the second-largest producer of zircon, and the third-largest producer of titanium feedstock [3].
Tronox KZN Sands is a major exporter to Europe, North America, and the Far East, thereby earning foreign exchange for South Africa. The business unit employs around 1,050 staff members, with annual revenue exceeding US$175 million per annum, with the potential to more than double the current annual revenue with the commissioning of the new Fairbreeze mine near Mtunzini in KZN, scheduled for the second half of 2014 [4].
4 PROCESS DESCRIPTION
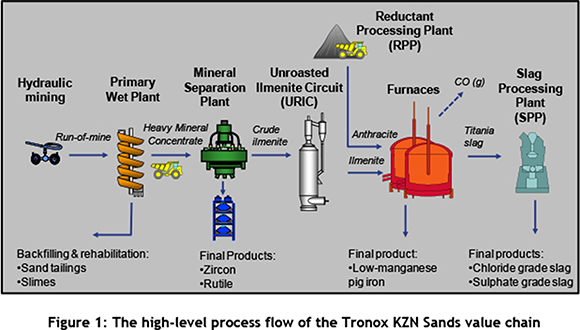
Hydraulically-mined material is called run-of-mine (ROM), and is concentrated to produce heavy mineral concentrate (HMC) in the Primary Wet Plant at the Hillendale mine near Richards Bay. The HMC, containing ilmenite, rutile, and zircon, is transported from the mine to the Mineral Separation Plant where rutile and zircon are produced as final products and crude ilmenite as an intermediate product. Rutile is exported for the production of pigment, refractory ceramics, titanium metal, and welding rods, while zircon is primarily used for the manufacture of ceramics, tile glazing, and refractories. The crude ilmenite is dried and further upgraded in the Unroasted Ilmenite Circuit (URIC) where smelter-grade ilmenite is produced. Anthracite is offloaded from external suppliers at the Reductant Processing Plant (RPP) where it is dried and prepared for smelting. The smelting of ilmenite, together with anthracite, in two 36 MW direct-current arc furnaces (smelters) produces different grades of low-manganese pig iron as final products for foundry markets, and titania slag as an intermediate product. The latter still requires cooling, crushing, drying, and separation into different size fractions at the Slag Processing Plant (SPP) before being shipped as titanium feedstock to Tronox pigment plants or being sold to external pigment producers. The Mineral Separation Plant, URIC, smelter, and SPP are all situated at the Central Processing Complex (CPC) outside the town of Empangeni. Figure 1 depicts the high-level process flow of the value chain at Tronox KZN Sands; the URIC, RPP and SPP are the focus of this study.
5 CO GAS PRODUCTION AND THE USE OF ON-SITE GAS
This research focuses on the ilmenite smelting process, and specifically the by-product stream of CO gas. The basic chemical reaction inside the furnaces can be shown as
FeTiO3 + C  Fe + TiO2 + CO (g).
Fe + TiO2 + CO (g).
Before this project began, all the CO gas produced at the furnaces was flared (i.e., burnt) at the emission stacks at the top of the two furnaces. The combustion of CO gas in atmospheric air produces carbon dioxide (CO2). The volumetric flow of CO gas produced is approximately 9,000 m3/h, resulting in roughly 10,000 tons/month of CO2 being emitted. This is comparable to the carbon footprint of 15 round trips between London and Hong Kong per month by a Boeing 747-400 carrying 420 passengers. The flaring of CO gas contributes a significant 86 per cent of direct CO2 emissions, and adds roughly 24 per cent to the total carbon footprint of the KZN business unit.
The drying of feed material that enters the URIC, RPP, and SPP requires gas as a fuel source for the burners at these plants. The CPC has access to a pipeline of methane gas from an external supplier, but was designed with the intention of using the furnace off-gas on-site. For a number of strategic and operational reasons, however, the CO gas has not been used at the dryers for sustainable periods over the first decade of operation. Before this project began, the last previous attempt was in 2006. This implied that methane gas was continually being purchased and the CO gas was being flared.
6 OPPORTUNITY FOR IMPROVEMENT AND POTENTIAL BENEFITS
Using CO gas to reduce the consumption and thus the cost of buying in methane gas had been a possibility since the inception of the plant. In 2007, a project was initiated to re-evaluate the feasibility of this concept. Ilmenite smelting is known to be a complex pyro-metallurgical process that constantly challenges production metallurgists [5].
The initial investigation pointed out several challenges to implementing this solution. First, CO gas had never been used at the dryers for more than three consecutive months because of the historic unstable availability of CO gas, a result of process instabilities at the furnaces [6]. Due to the inconsistent CO gas availability, too many undesirable operational problems were foreseen in replacing the methane gas with CO gas. One of the main anticipated challenges was that, at times of an undersupply of CO gas, switchovers between CO and methane gas would be unacceptably disruptive to production. The existing technology at the CPC did not allow for fully-automated switchovers. Before and after every switchover, a section of the gas line would need to be purged with nitrogen to prevent flashbacks. Every switchover would result in lost production time, and required many hours to manage the switchover and purging.
Second, there is no pressure vessel at the CPC that would maintain a buffer capacity of CO gas to smooth fluctuations in supply. Since 2007, the high-level business priorities that drive the capital budget and deployment of project resources have not allowed for the construction of such a pressure vessel. This was still the case in the short- to medium-term in 2013.
Third, CO gas is highly poisonous, while also being colourless and odourless [7]. Implementing the project would imply that many new areas, and staff, would be exposed to CO gas at the CPC. The health and safety risks associated with the use of CO gas would result in additional costs and management responsibility. This awareness made management even more hesitant to prioritise the project in the face of a project funnel with numerous other, equally-pressing, lower-risk projects that needed to be done.
In the last instance, because the CO gas had not been distributed sustainably to the dryers for more than ten years, the distribution infrastructure had deteriorated to a state of inoperability, and did not comply with the latest regulations. In addition, at the time of re-evaluating the project, the staff's knowledge of operating and maintaining the CO gas system was limited.
On the positive side, between 2007, when the re-evaluation started, and 2013, operational efficiency and stability at the furnaces had improved significantly and, with that, the availability and stability of CO gas supply. This improvement can be ascribed to increased process knowledge and the implementation of various continuous improvement projects. All this supported the case for change. Figure 2 indicates how the produced volumes of CO gas per furnace had increased from 2010 to 2012. Notice how volume per minute increased from 2010 to 2012 for each furnace.
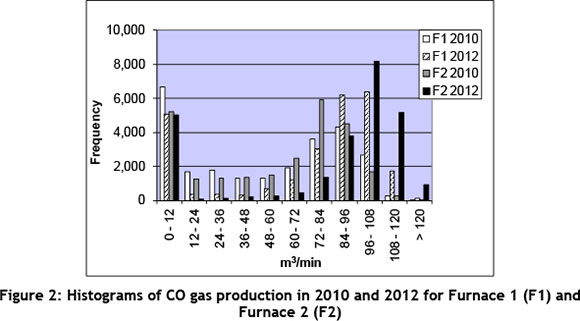
Further, CO gas from furnace operations has been used successfully with significant economic and environmental benefits in a number of different industries around the world, including some applications in South Africa. In the steel industry particularly, CO gas from furnace operations (furnace CO gas) is used for the pre-heating of scrap, for the continuous charging of furnace feed material ([8]), and for model predictive control in the automation of manually-controlled variables such as the forced-draught fan power and the air-entrainment slip-gap width for the furnace off-gas process [9].
Another industry application that uses furnace CO gas is the co-generation of electricity. Particularly in South Africa, where the national electricity utility experiences capacity shortages, investigating the use of furnace off-gases as a primary fuel source in power generation facilities has become increasingly important. Other producers of furnace CO gas in South Africa that produce the volumes required for electricity generation include Samancor Ferrochrome in Middelburg (Mpumalanga Province), Tronox Namakwa Sands in Saldanha (Western Cape Province), and Richards Bay Minerals in Richards Bay (KZN Province) [10]. Since 2011, a 13 MW co-generation project has been under way at the Tronox Namakwa Sands operation.
A visit to the Eramet ilmenite smelter in Tyssedal, Norway, showed the successful use of furnace CO gas in a pre-reduction process. These industry examples of the successful use of furnace CO gas gave further impetus to the project becoming a reality at Tronox KZN Sands.
The initial investigation identified two main potential benefits that could be gained from the successful implementation of the project:
1. A reduction of up to 90 per cent in the consumption of methane gas, with a potential cost-saving exceeding US$170 000 per month, at 2012 methane gas prices and an exchange rate of ZAR8.50 to US$1.
2. A reduction of over 5 per cent in the direct CO2 emissions from the CPC, equating to a reduction of roughly 1.4 per cent in the carbon footprint. This has many qualitative advantages, and could even result in a financial benefit in the form of carbon credits or cuts in carbon taxes.
7 SOLUTIONS CONSIDERED AND APPROACH SELECTED
The challenges that needed to be addressed to ensure the successful implementation of the project were split into two groups: investigative challenges and practical challenges. Before addressing the practical challenges, such as upgrades to the distribution infrastructure (pipelines, valves, burners, etc.), optimising the switchover control mechanisms, and training the staff, it was necessary to confirm whether the supply of CO gas was sufficient and stable enough to meet the demand for gas at the burners. The Business Improvement section of the business, consisting of a small team of industrial engineers, was tasked with modelling and studying the CO gas supply-demand situation. Recommendations were to be provided to management and the project implementation team.
With access to different Operations Research/Management Science tools, the Business Improvement team considered the most appropriate way to model and study the problem. Existing static Excel models of the CO gas supply and demand, as well as metallurgical mass balance models, which model furnace behaviour and product streams empirically, were considered as part of the pre-evaluation. We concluded that the existing models of the CO gas system were inadequate to investigate the problem. The main shortcomings of the existing Excel models of CO gas supply versus demand were that they catered for neither the dynamic behaviour nor the stochastic nature of the supply of and demand for CO gas. These models could indicate that in, say, an average month, there would be sufficient CO gas for the demand under certain conditions, but they provided no insight into the near-instantaneous supply-demand balance of CO gas. Due to the risk of operational disruptions caused by a negative CO gas balance occurring for short periods (a few minutes or hours), an evaluation based on averages was not satisfactory. The empirical metallurgical furnace models modelled physical and chemical properties and events in the process well, but provided no insight into the downstream operational issues associated with the distribution and utilisation of CO gas. Discrete-event simulation was therefore selected as the appropriate method to study the CO gas problem. The continuous gas flow was approximated through discrete time intervals of one minute's duration.
8 CONCEPT MODEL AND DESIGN OF EXPERIMENT
A concept model of the supply-demand CO gas problem was developed. Two furnaces produced the CO gas from the supply side, while on the demand side there were three consumers of CO gas. The furnaces operate independently of one another. The level and stability of CO gas supply is a direct function of furnace availability and operational stability. The consumers are the URIC, RPP, and SPP, where drying of feed material occurs. The instantaneous demand for gas is a function of the combination of the plants set to receive CO gas, the feed rate and type of feed material into the plants, and whether or not the plants are operational at that time. Another factor affecting the supply-demand balance was the availability of the CO gas distribution plant. Cleaned CO gas is compressed at this plant before being distributed to the gas-consuming plants. The distribution plant is the pivot between the furnaces and the gas-consuming plants. The distribution of CO gas is governed by a prioritisation logic programmed into a digital control system. Figure 3 shows the basic conceptual model developed for the problem.
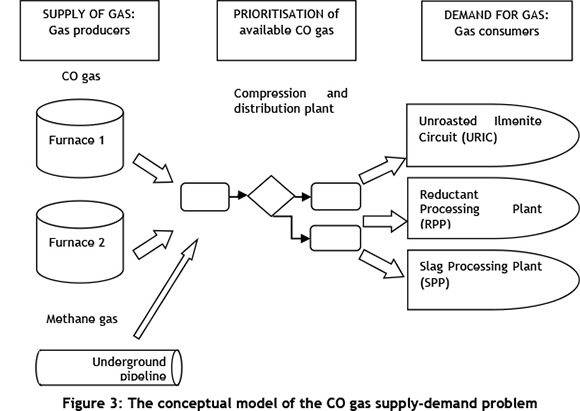
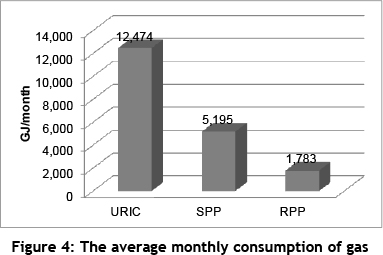
The graph in Figure 4 shows the average monthly requirements for gas energy in gigajoules (GJ) at the different plants. The gas consumption of a plant is dependent on the energy required per ton of feed material it receives. The URIC has the highest priority in the prioritisation logic, followed by the SPP and then the RPP.
A computer simulation model of the CO gas supply-demand problem was developed from the concept model in Rockwell Software's Arena® package while following the simulation process prescribed by Banks [11]. The process was modelled as a terminating system with a duration of 168 hours (one week).
Input data for the simulation model was collected from the operations management system database of Tronox KZN Sands. This allowed for the analysis of different sets of input data to determine statistical distributions for certain input variables. The detailed input data analysis will not be discussed here; we found, for example, that the CO gas production rate at Furnace 2 follows a beta distribution (65+beta(2.39,10) m3/min), and the URIC mean time to repair (MTTR) is lognormal distributed (lognormal(116, 492) minutes). From the 16 input variables identified, seven were identified as control variables, or decision variables (DVs), as shown in Table 1.

Note that the URIC, SPP, and RPP are all part of the process, and none is more profitable than the other. The values of DV4-7 cannot be decided, but they were varied during the study to provide target availabilities for the maintenance team.
Prioritisation logic governing the simulation model was available from the on-site digital control system manuals [12]. During the modelling process it became clear that the interaction between system components required the problem to be modelled as a multi-objective problem (MOP). The two main conflicting output variables, or objectives, selected from a list of 15 output variables are shown in Table 2.objective problem (MOP). The two main conflicting output variables, or objectives, selected from a list of 15 output variables are shown in Table 2.

Although it may seem that the two objectives identified for the optimisation of the CO gas problem can both easily be expressed in monetary value, which would render the study a single-objective problem, this would be an oversimplified assumption. First, translating the production time lost during a switchover at the different plants into a monetary value is not an arbitrary process, mainly because most of the plants involved produce intermediate products into stockpiles/buffers. This skews the monetary value of that product stream at any given moment. Second, and probably more relevant, the operational personnel explicitly requested an estimate of any expected operational disruptions that would occur should the project be implemented. This was to determine which configuration(s) resulted in minimum production loss. The production loss estimations could be used in profit/loss calculations if needed.
9 SIMULATION MODEL VALIDATION
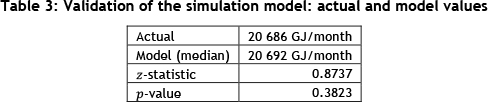
Before performing simulation production runs, the minimum number of replications required was calculated [13], followed by validation (ensuring the correct model was built to represent reality satisfactorily) and verification (ensuring that the model was built correctly, with no syntax or run-time errors) of the model. The model was validated by comparing the estimated gas energy consumed in the model (in GJ/month) to the actual gas energy consumed for the same scenario and time period. We could not prove that the gas consumption is normally distributed, and so used the Wilcoxon rank sum test [13] to compare the model output to the real system value. The Matlab test results are shown in Table 3 for a significance level of a = 5%. It is concluded that the model is an adequate approximation of the real process, since p > a.
10 SCENARIO DEFINITION
Another step that was needed before simulation production runs could be performed was to define the scenarios to be evaluated with the simulation model. These scenarios were defined as different combinations of the seven decision variables in Table 1.
First, we needed to evaluate each combination of gas-consuming plants to find a good or optimum combination. This resulted in seven possible on-off combinations of DV1, DV2, and DV3 (e.g., DV1=1, DV2=0, DV=0). The more plants set to receive CO gas, the higher the demand, particularly the peak demand, and therefore the higher the likelihood of switchovers (and thus lost production time) at the plants involved, but also the higher the possibility of a larger cost saving. In the case of an undersupply of CO gas, the prioritisation logic determines that the lowest-priority plant will be switched from CO gas to methane gas.
The next consideration was the availability of the CO gas distribution plant, DV4: increasing its availability over the long term would increase the CO gas available for distribution, but this would incur maintenance costs that need to be offset against the cost saving on methane gas. The last consideration incorporated into the scenarios was the availability of the gas-consuming plants, i.e., DV5, DV6, and DV7. If a certain combination of plants is set to receive CO gas and one of the plants experiences downtime, the prioritisation logic dictates that the available CO gas will be distributed to the highest-priority users that are operational at that moment. Downtime at a high-priority user will also allow a lower-priority user that is operating on methane at that time to switch back to CO gas. Incorporating all the feasible combinations of the seven DVs into the simulation study resulted in a search space of 7,560 scenarios to be evaluated.
11 NEAR-OPTIMISATION WITH A META-HEURISTIC
Simulation does not necessarily guarantee the optimisation of the system under study, but primarily serves as a powerful analysis tool to observe system behaviour over time, and to test the impact of different scenarios in a less costly way than making actual system changes [14]. Thus, in order to find the values of system parameters (particularly DVs) that would yield the best scenario, some optimisation technique was required. Further, due to the multi-objective nature of the problem, in which two objectives of equal weight are to be optimised, a meta-heuristic capable of multi-objective optimisation (MOO) needed to be employed.
We selected the cross-entropy method (CEM), described in Rubinstein and Kroese [15] and De Boer et al. [16], as the optimisation technique to be used in conjunction with the simulation model. The CEM is a relatively recently-developed and computationally-powerful meta-heuristic. Due to the multi-objective nature of the problem, we selected the MOO CEM algorithm proposed by Bekker and Aldrich [17]. The algorithm is new, and few applications to industry problems are found in the literature. Application of this algorithm to an industry problem would therefore add to the body of knowledge, and potentially provide further evidence of the algorithm's suitability as an MOO technique, particularly for solving dynamic stochastic, real-world problems.
12 SIMULATION PRODUCTION RUNS AND RESULTS
With the simulation study defined, the production runs were performed. We did an exhaustive enumeration of all 7,560 feasible scenarios for research purposes, which was followed by an optimisation exercise using the MOO CEM algorithm. For each scenario, a point estimator and interval estimator were estimated according to the procedure set by Law and Kelton [14]. The exhaustive enumeration yielded the true Pareto solution set, while an approximated Pareto front was obtained with the MOO CEM algorithm in significantly less computation time: we found a good solution when it was set 30 times faster. In terms of the performance of the MOO CEM algorithm, the difference in computation time illustrates the efficiency of the MOO CEM-approximated Pareto solution set.
Figure 5 shows the approximated Pareto front obtained with the MOO CEM algorithm, versus the values obtained in the exhaustive enumeration, for 2010 data.
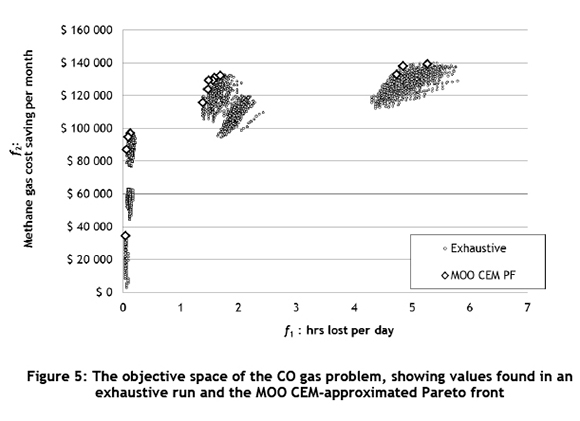
Figure 6 shows how the MOO CEM-approximated Pareto front (containing 14 values) compares with the true Pareto front (containing 22 values), for 2010 data.
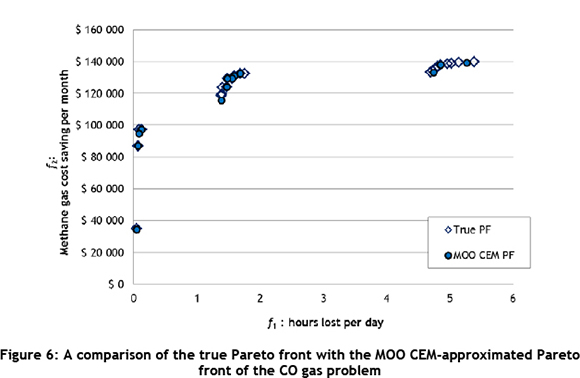
Figure 7 shows how the MOO CEM-approximated front changed when using 2012 data in the simulation. Clearly shown in this Figure is the increase in the expected cost saving and the decrease in the expected hours lost due to improved furnace operation and thus increased availability of CO gas.
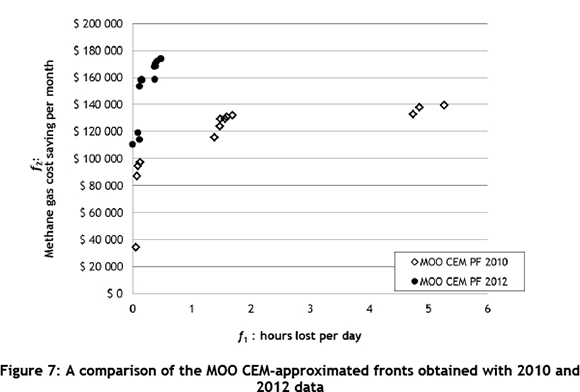
The members of the approximate Pareto front for 2012 in Figure 7 have point estimators for f1 and f2 with their 95 per cent confidence interval half-width values hw(f1) and hw(f2). These are shown in Table 4, based on 10 pseudo-independent simulation replications.
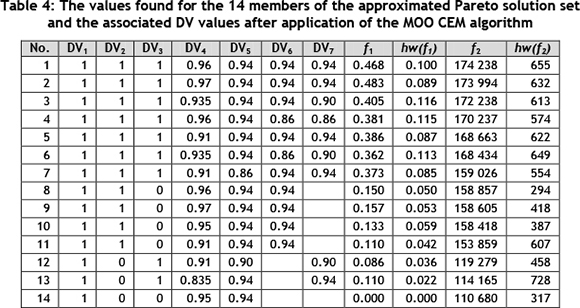
The approximate Pareto solution set can be interpreted as follows. Say, for example, that management decides that at least US$150,000 per month must be saved on methane gas. As shown in row 11 of Table 4, this will require the CO gas to be distributed to the URIC and the SPP while maintaining the gas distribution plant at 91 per cent, the URIC at 94 per cent, and the SPP at 94 per cent. Plant managers can expect a lost production time of 0.11 hours (seven minutes) per plant per day due to switchovers. This implies less than one expected switchover per day. It would be prudent to advise plant operators at the URIC and SPP to prepare for these switchovers to minimise disruptions.
Another approach is to select the solution with the highest 'utility' from the approximate Pareto set. The simplest technique is to use Simple Additive Weighting [18]. In this method, there are m solutions each with η objectives. The value of a competing scenario or solution is given by
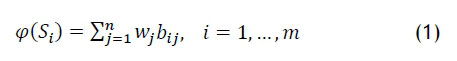
with wj the weight assigned by the decision-maker to objective j, and the larger the value of φ, the better the solution is relative to its competitors. The bij in (1) are normalised values of the objective function values [18]. Beneficial objectives are normalised with
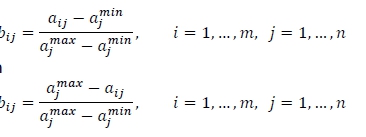
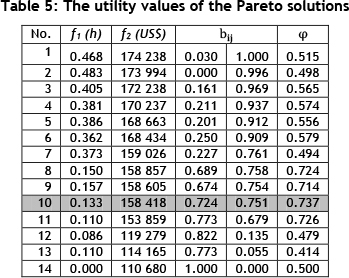
and cost objectives with where αjmax= max{a1j, a2j,..., amj} and ajmin = min{a1j,a2j,...,amj}. The utility values are shown in Table 5 for equal weights with f1 a cost objective (production downtime) and f2 a beneficial objective (monetary saving). Given these, solution number 10 has the highest utility. This means that in combination, the lost production hours and the amount saved on methane gas per month for solution 10 (φ(S10)=0.737) is the 'best' value for the decision-maker.
13 CONCLUSIONS AND RECOMMENDATIONS FOR PROJECT IMPLEMENTATION
The results obtained from the simulation study and optimisation allowed the following conclusions to be drawn:
1. The application of discrete-event simulation proved worthwhile, as it allowed for a much more thorough investigation of the CO gas problem compared with the results obtained with the business's existing static Excel models. While incorporating stochastic variables, the expected values of the objectives were estimated within 95 per cent confidence intervals.
2. Despite the lack of buffer capacity, benefits can be achieved through the dynamic prioritisation of CO gas. With the prevailing operational conditions and current equipment configuration at the business, it is not necessarily optimal to operate all the gas-consuming plants on CO gas, and definitely not optimal to distribute CO gas to none of those plants. The optimum combinations of plants to receive CO gas were found such that production disruptions would be minimised and savings maximised.
3. The approximate Pareto solution set obtained with the MOO CEM-algorithm clearly indicates the trade-off between the two objectives. In particular, it was quantitatively shown how setting more plants to receive CO gas increased the expected lost production time at all the plants. Although this had been anticipated, the results quantified the extent of the potential loss. The Pareto front also makes clear the availabilities at which plants should be maintained to achieve the optimum benefit. The approximate Pareto solution set therefore serves as an effective management decision-making aid.
Based on the conclusions discussed above, the following recommendations were made to management and the project implementation team:
1. With reference to the approximate Pareto solution set, it is recommended that the distribution of CO gas gradually be increased to include more plants over time. It is recommended that the CO gas initially be distributed to one plant only, even though the intention is eventually to operate all the plants on CO gas. This recommendation is based on the awareness that, in practice, gaining knowledge and experience in initially operating a new system successfully at just one plant is likely to contribute to plant personnel gaining confidence more quickly, enabling them eventually to operate the full system. The Pareto front indicates that a decision initially to distribute the CO gas to one plant only means that the URIC should be the first to receive CO gas: see row 14 of the Pareto set in Table 4. This is expected to result in a cost saving of more than US$110,000 per month, with an expected production time loss of less than one minute per day (roughly one switchover per week), and thus minimal disruptions during the learning process. Downtimes at the furnaces, particularly planned downtimes, should be communicated to URIC staff in advance to allow the URIC to switch to methane gas as seamlessly as possible.
2. After success is proven at the URIC, the CO gas can be distributed to any of the combinations of plants that form part of the Pareto solution set. The importance of maintaining the availabilities of the distribution plant and gas-consuming plants at the values prescribed in the Pareto solution set must be understood if the indicated cost savings and minimised switchovers are to be achieved.
3. The hazardous nature of CO gas cannot be emphasised enough. All measures need to be taken and capital made available to implement the required control measures such as signage, fixed CO gas monitors, and alarms on the digital control system to minimise this risk.
13.1 Implementation
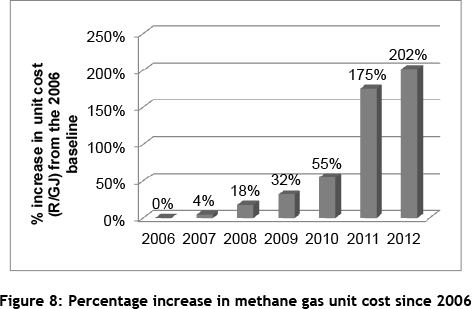
The implementation of this project was delayed for a number of years, but top management started focusing on it in early 2011 when the unit cost of methane gas increased rapidly, as shown in Figure 8. A further contributing factor was the increased pressure on the business to implement 'green projects' to lower its environmental impact, particularly its carbon footprint. The completion of the simulation study and the presentation of recommendations to management came at the right time, and resulted in the drawing up of a management-approved implementation plan in 2011.
The practical challenges listed earlier in this article were addressed by the plant managers and the project implementation team as follows:
1. Appropriate pressure and thickness tests of the CO gas distribution infrastructure were conducted at the URIC and RPP, and are in process at the SPP.
2. Capital was and is currently being spent on repairing the distribution infrastructure and ensuring compliance with the relevant South African Pipeline Gas Association (SAPGA) standards.
3. Responsibility for the maintenance of the distribution infrastructure has been clarified between plant managers.
4. Updating of Standard Task Procedures (STPs) and training manuals for operating and maintaining the CO gas system began in 2011, and was completed for the URIC and the RPP. Staff training was done in parallel.
5. Management took note of the expected disruptions due to switchovers. The safe and swift handling of switchovers was made part of staff training.
Factors beyond the control of the Business Improvement team resulted in the infrastructure reparations, compliance testing, and certification being completed first at the RPP. As a result, the CO gas system was commissioned at the RPP first. Despite this not being in line with the recommendation to commission the URIC first, it proved to be a major step forward in actually implementing the project after more than 10 years of flaring all the CO gas. Initially, commissioning the RPP only allowed management to phase in the implementation while having the luxury of troubleshooting the newly-commissioned system one plant at a time. The commissioning of the URIC on CO gas started in November 2012 with short CO gas runs and troubleshooting. Full commissioning of the URIC was completed in the fourth quarter of 2013. Capital was approved for upgrades to the SPP CO gas infrastructure by March 2013, while the conversion of the SPP burner was completed by December 2013. Upgrades to and certification of the CO gas pipelines at the SPP were still underway during December 2013.
13.2 Benefits realised to date
Some benefits have already been realised. First, the mere fact that the implementation of this project was approved and became the focus of management for the first time in the life of the business is a significant milestone. Second, the fact that the full upgrade and certification of the CO gas distribution infrastructure is in process is another significant milestone. Having all the relevant staff trained in operating and maintaining the CO gas system and updating the training material are further significant benefits. The RPP was commissioned in April 2012, and run-time on CO gas has steadily been increased. At the time of writing this article, the RPP had been processing on average 74 per cent of its feed per month on CO gas from June 2012 onwards, while implementation of the project at the URIC had just been completed, and at the SPP it was still in process. Implementation of the project has so far resulted in a cost saving of US$22,400 per month over the first 18 months of operation.
This project has become an intra-company example of Tronox's commitment to a greener economy.
13.3 Further Investigation
The following possibilities were identified for further investigation. These are expected to result in even greater economic and environmental benefits:
1. Investigate the use of a pressure vessel to maintain a CO gas buffer to ensure constant CO gas supply. As an interim means to improve the steady availability of gas at the burners, the full automation of switchovers should be investigated.
2. Investigate the co-generation of electricity. If the current trend of rising electricity prices in South Africa continues, this option might become lucrative. Not only is the cost benefit expected to be higher than using the CO gas for drying feed material, but this option has the potential to decrease the carbon footprint of Tronox KZN Sands by up to 24 per cent. Lessons can be learnt from the co-generation project at the Tronox Namakwa Sands operation.
3. Investigate the re-commissioning of the furnace pre-heaters that also operate on CO gas. This will increase the energy efficiency of the furnaces by reducing the MWh/ton of ilmenite smelted. This also has the potential to decrease the carbon footprint, in that less electric energy is required to produce the same tons of titania slag from preheated feed material.
13.4 Further research
The ranking and selection of the scenarios entering the Pareto set must be refined. In this work we accepted solutions that rank high numerically, but the statistical significance of differences should be evaluated as in [Error! Bookmark not defined.] and in [19].
ACKNOWLEDGEMENTS
We acknowledge the Tronox KZN Sands management team, particularly the Central Processing Plant Operations managers and the Planning and Optimisation manager for their support of the project. We also acknowledge the Central Processing Plant Production and Maintenance superintendents for providing valuable insight into operational issues, and for playing a vital role in the implementation of the project. Finally, the Tronox KZN Sands Projects department deserves acknowledgement for taking responsibility for the detailed planning and implementation of the project.
REFERENCES
[1] Jones, G. 2009. Mineral sands: An overview of the industry. Unpublished. Capel: Iluka Resources Limited. [ Links ]
[2] Oomardath, V. 2009. Overview of the mineral sands industry. Unpublished. Empangeni: Exxaro KZN Sands Limited. [ Links ]
[3] www.tronox.com. Retrieved from http://www.tronox.com/our-company/. Accessed on 26 April 2013. [ Links ]
[4] www.miningweekly.com. Retrieved from http://www.miningweekly.com/article/revised-fairbreeze-bar-submitted-in-search-of-project-approval-2012-03-16. Accessed on 26 April 2013. [ Links ]
[5] Zietsman, J.H. & Pistorius, P.C. 2006. Modelling of an ilmenite-smelting DC arc furnace process. Minerals Engineering, 19, pp. 262-279. [ Links ]
[6] Rajkumar, K. 2012. Post implementation review - CO gas utilisation at URIC and reductant. Empangeni: Tronox KZN Sands Limited. [ Links ]
[7] Penney, D. 2000. Carbon monoxide toxicity. Boca Raton: CRC Press LLC. [ Links ]
[8] Turkdogan, E. 1996. Fundamentals of steelmaking. London: Institute of Materials, Minerals and Mining. [ Links ]
[9] Bekker, J.G., Craig, I.K. & Pistorius, P.C. 2000. Model predictive control of an electric arc furnace off-gas process. Control Engineering Practice, 8, pp. 445-455. [ Links ]
[10] Schubert, E.S. & Gottschling, R. 2011. Co-generation: A challenge for furnace off-gas cleaning systems. In: Jones, R.T. & den Hoed, P. (eds) Southern African pyrometallurgy. Johannesburg: Southern African Institute of Mining and Metallurgy. [ Links ]
[11] Banks, J. 1998. Handbook of simulation. Wiley-Interscience. [ Links ]
[12] Brouwer, G., Janse van Vuuren R. & Volk B. 2003. Functional description Kumba Heavy Minerals Empangeni CPC-Smelter Furnace Off-Gas Distribution Circuit 783. Johannesburg: Howden Process Compressors Limited. [ Links ]
[13] Salkind, N.J. (ed.) 2007. Wilcoxon signed ranks test. In: Encyclopedia of Measurement and Statistics. Thousand Oaks, CA: Sage Publications, Inc. pp. 1052-54. [ Links ]
[14] Law, A.M. & Kelton, W.D. 2000. Simulation modeling and analysis. 3rd edition, McGraw-Hill. [ Links ]
[15] Rubinstein, R.Y. & Kroese, D.P. 2004. The cross-entropy method: A unified approach to combinatorial optimization, Monte-Carlo simulation, and machine learning. New York: Springer erlag. [ Links ]
[16] De Boer, P.T., Kroese, D.P., Mannor, S. & Rubinstein, R.Y. 2005. A tutorial on the cross- entropy method. Annals of Operations Research, 134, pp. 19-67. [ Links ]
[17] Bekker, J. & Aldrich, C. 2011. The cross-entropy method in multi-objective optimisation: An assessment. European Journal of Operational Research, 211, pp. 112-121. [ Links ]
[18] Ma, J., Fan, Z. & Huang, L. 1999. A subjective and objective integrated approach to determine attribute weights. European Journal of Operational Research, 112, pp. 397-404. [ Links ]
[19] Lee, L.H., Chey, E.P., Chen, S.Y. & Goldsman, D. 2010. Finding the non-dominated Pareto set for multi-objective simulation models. IIE Transactions, 42(9), pp. 656-674. [ Links ]
* Corresponding author














