Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Journal of Psychiatry
versão On-line ISSN 2078-6786
versão impressa ISSN 1608-9685
S. Afr. j. psyc. vol.20 no.4 Pretoria Out./Dez. 2014
http://dx.doi.org/10.7196/sajp.574
ARTICLE
Magnetic resonance imaging study of corpus callosum abnormalities in patients with different subtypes of schizophrenia
E UnluI; E BagciogluII; M B AcayI; E KacarI; O TuramanlarIII; Y GonulIV; M CevikV; A AkpinarVI; K S CoskunII
IMD; Department of Radiology, Faculty of Medicine, Afyon Kocatepe University, Afyonkarahisar, Turkey
IIMD; Department of Psychiatry, Faculty of Medicine, Afyon Kocatepe University, Afyonkarahisar, Turkey
IIIMD; Department of Anatomy, Faculty of Medicine, Afyon Kocatepe University, Afyonkarahisar, Turkey
IVPT, PhD; Department of Anatomy, Faculty of Medicine, Afyon Kocatepe University, Afyonkarahisar, Turkey
VMD; Department of Psychiatry, Samsun Mental Health Hospital, Samsun, Turkey
VIMD; Department of Psychiatry, Faculty of Medicine, Suleyman Demirel University, Isparta, Turkey
ABSTRACT
BACKGROUND: Reductions in the size of the corpus callosum (CC) have been described for schizophrenia patients, but little is known about the possible regional differences in schizophrenia subtypes (paranoid, disorganised, undifferentiated, residual).
METHODS: We recruited 58 chronically schizophrenic patients with different subtypes, and 31 age-and-gender matched healthy controls. The callosum was extracted from a midsagittal slice from T1 weighted magnetic resonance images, and areas of the total CC, its five subregions, CC length and total brain volume were compared between schizophrenia subtypes and controls. Five subregions were approximately matched to fibre pathways from cortical regions.
RESULTS: Schizophrenia patients had reduced CC total area and length when compared with controls. Disorganised and undifferentiated schizophrenics had a smaller prefrontal area, while there was no significant difference for the paranoid and residual groups. The premotor/supplementary motor area was smaller in all schizophrenia subtypes. The motor area was smaller only in the disorganised group. A smaller sensory area was found in all subtypes except the residual group. Parietal, temporal and occipital areas were smaller in the paranoid and undifferentiated groups. Total brain volume was smaller in all schizophrenia subtypes compared with controls, but did not reach statistical significance.
CONCLUSION: These findings suggest that the heterogeneity of symptoms may lead to the different CC morphological characteristics in schizophrenia subtypes.
The corpus callosum (CC) is known to be the largest white-matter fibre tract in the brain. It connects the two cerebral hemispheres and facilitates interhemispheric communication.[1,2]The CC has been strongly implicated as an anatomical mediator of dysfunctional interhemispheric transfer in schizophrenia[3] and a number of psychiatric disorders, including bipolar affective disorder, depression,[4] antisocial personality disorder,[5] post-traumatic stress disorder,[6] autism[7] and attention deficit hyperactivity disorder.[8]
Rosenthal and Bigelow[9] first drew attention to an increased thickness of the CC in postmortem brains of schizophrenia patients. Later, in neuroimaging studies, CC size was found to be increased,[10] decreased[11] or unaltered[12] compared with healthy controls. In a meta-analysis of 28 magnetic resonance imaging (MRI) studies carried out by Arnone et al.,[13] they found that schizophrenia patients seem to show a reduction in CC area.[13]
The inconsistent findings may be a consequence of possible confounding factors, including age,[14] handedness,[15] gender,[16] chronicity of illness[17,18]and differences in CC partitioning schemes employed by the different studies.[19,20]In addition, it was suggested that it may also be owing to the heterogeneity in symptom profiles. [12,18]Schizophrenia is characterised by large heterogeneity,[12,18]and subtypes of schizophrenia may differ in neuroimaging-based measures of brain morphology.[21] Therefore, we planned to examine CC size on the basis of clinical subtype in schizophrenia, and we recruited the four subtypes of schizophrenia patients (paranoid, disorganised, undifferentiated and residual) who had different illness durations but were matched in age, sex and handedness. We hypothesised that the size of the CC may be different in the different subtypes of schizophrenia.
Objective
The objective of the current MRI study was to determine whether there were any differences in length and area of the CC among schizophrenia subtype patients and healthy subjects. Examining the association between schizophrenia subtypes and the size of the CC is likely to deepen our understanding of the pathophysiology of schizophrenia.
Methods
Patients
The study included 58 outpatients and inpatients with schizophrenia, comprising 44 males and 14 females (aged 18 - 50 years), who were seen in the Department of Psychiatry of Afyon Kocatepe University. All patients were diagnosed with schizophrenia according to the Diagnostic and Statistical Manual of Mental Disorders, 4th edition (text revised) (DSM-IV-TR)[22] on the basis of a clinical interview. Diagnosis of schizophrenia was established by two senior psychiatrists. Patients were divided into four groups according to DSM-IV-TR criteria: paranoid (n=17); disorganised (n=14); undifferentiated (n=20); and residual (n=7). The severity of psychotic symptoms was assessed using the positive and negative syndrome scale (PANSS), whose validity and reliability studies for the Turkish version were carried out by Kostakoglu et al.[23]
Controls
There were 31 normal healthy controls matched in age and sex (aged 23 - 62 years), comprising 22 males and 9 females, with no personal history of psychiatric illness or family history of psychosis, medical⁄neurological condition requiring regular medications or current exposure to psychotropic medications. They were screened with the non-patient version of the Structured Clinical Interview for DSM disorders (SCID).[24]
Exclusion criteria
Exclusion criteria for both groups were left-handedness, concurrent or previous DSM-IV-TR axis I disorder, a history of head trauma with loss of consciousness, a current medical illness (e.g. diabetes mellitus, hypertension or asthma) or known neurological disease, a lifetime history of substance dependence, or a history of any substance abuse or psychotic disorders in their first-degree relatives.
Only right-handed subjects as determined by the Oldfield handedness questionnaire[25] were included because handedness is an important factor that affects CC size.[26]
Ethics approval
The study protocol was approved by the local ethical committee. All subjects provided written informed consent after receiving an explanation of all relevant information related to study participation.
Imaging procedures
A 1.5 T Philips Intera Magnetic Resonance unit (Philips Medical Systems, The Netherlands) was used to obtain a set of T1 weighted sagittal scout images (TR = 500 ms; TE = 15 ms; FOV = 23 cm, slice thickness 5 mm; number of exitations = 2; matrix size = 256 × 256). The full sagittal series was reviewed, and on the consensus of two trained researchers who were blind to the study hypotheses, group assignments and subject identities, the correct slice was selected using the internal midsagittal landmark criteria of Woodruff et al.[17] The landmarks to identify the correct slice on the midline sagittal slice were: (i) a distinct outline of the CC; (ii) no white matter or only minimal white matter in the cortical mantle surrounding the CC; (iii) the interthalamic adhesion; and (iv) a transparent septum and visibility of the aqueduct of Sylvius.[17]
Segmentation and measurement of the CC area
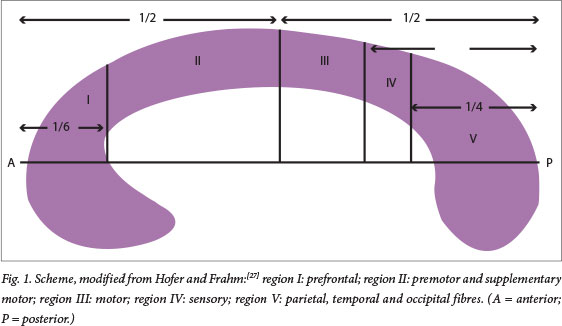
The CC was manually traced following its edge on the midsagittal slice of T1 images, where its structure appeared most remarkable. Anatomical measurements were conducted on the Aquarius Workstation using the AquariusNET program (TeraRecon Inc., USA). Morphometric measurements of the CC were conducted according to the same methods reported by Hofer and Frahm.[27]
Firstly, a line was drawn connecting the most anterior point of the genu with the most posterior point of the splenium, providing the overall callosal length. Subsequently, the traced CC was divided into five vertical partitions and each area was measured, as defined by Hofer and Frahm[27] (Fig. 1). The total and subregion areas and the maximum anterior-to-posterior length of the CC were measured. All callosal areas were expressed in square centimetres and length was expressed in centimetres. Total brain volume (gray and white matter plus cerebrospinal fluid) was also computed by the same raters to use as a covariate in order to minimise the effect of global brain size differences.
Inter- and intra-rater reliability were assessed using intra- and interclass correlation co-efficients on 10 randomly selected images. Intraclass correlation was 0.96 and 0.97 for the tracers. The intraclass correlation of intracranial brain volumes (ICVs) was 0.98. All these values are well within acceptable limits.
Statistical analyses
All statistical analyses were conducted via SPSS for Windows software, version 20.0 (SPSS Inc., USA). The two-tailed statistical significance level was set at p<0.05. Data were expressed as mean (standard deviation) values. The callosal measurement variables were observed to be in normal distribution, and homogeneity of variances were assumed to be equal according to the Shapiro-Wilk test. One-way analysis of variance (ANOVA) testing was used to compare CC length and total and subregional CC areas among the schizophrenia groups, and Tukey's b test results were used to determine post-hoc analysis. An independent sample t-test was used to define differences in CC measurements and ICV between total schizophrenia patients and controls. Pearson correlation co-efficients were used to examine the relationship between callosal measurements and age variables in all groups, and an independent sample t-test was used to define differences in CC measurements between the two gender groups. The correlations between age and CC measures were computed separately for each group.
Results
Participants
Demographic and clinical variables are presented in Table 1. There were no significant differences between patients and controls in age, sex or ICV. Patient and control groups had proportionally more males. All subjects were right-handed as assessed by the Oldfield handedness questionnaire.[25] There was no significant relationship between age and any callosal measure for the whole subject sample or for subgroups (male or female, healthy controls or patients) (Spearman correlation co-efficient, p>0.05). There was also no significant relationship between gender and callosal measures in patient and control groups.
In all groups of subjects, increasing age was associated with decreased CC area, but no significant relationship was determined between age and CC size (r=0.09; p=0.63). Also, there was no correlation determined between CC measures in the schizophrenia group (r=0.25; p=0.2). Total brain volume was smaller in total schizophrenia patients compared with healthy controls, but did not reach statistical significance (r=0.06; p=0.11). Therefore, we did not include age, gender or total brain volume as confounding factors, since there were no correlations between CC areas and sex or age.
Global CC
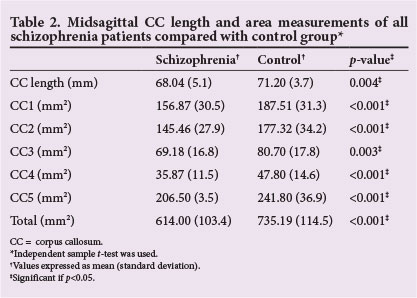
All schizophrenia patients were found to have smaller total subregional CC area and length compared with healthy groups (Table 2). There were no significant differences between schizophrenia subgroups in global CC area measurements among all schizophrenia patients. Furthermore, in all schizophrenia patients, the maximum anterior-to-posterior CC length was significantly smaller than in the healthy group (p<0.001). In addition, only the undifferentiated group had smaller length compared with the control group when comparing schizophrenia subgroups and the control group.
Subregional differences in the CC structure
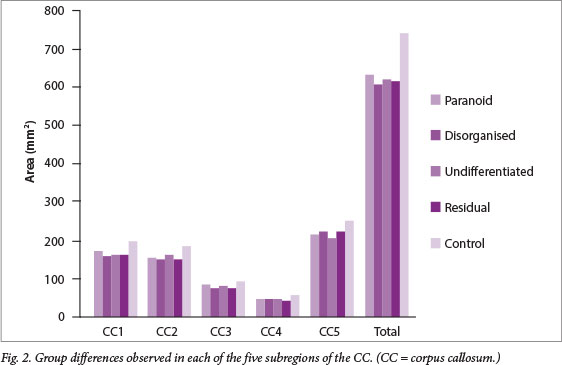
Table 3 displays the raw midsagittal CC area and length measurements for each of the five groups: paranoid, disorganised, undifferentiated, residual and control, and Fig. 2 shows the significant group differences observed in each of the five subregions of the CC.
CC1
Subregion CC1 was significantly smaller in the disorganised and undifferentiated schizophrenia group than in healthy controls (p=0.003, p=0.006, respectively). However, the CC1 area did not differ in paranoid and residual groups compared with the control group (p>0.05).
CC2
All schizophrenia groups (paranoid, disorganised, undifferentiated, residual) had a smaller sized callosal subregion CC2 than the control group (p=0.007, p=0.003, p=0.030, p=0.030, respectively). In addition, CC2 was the only area that was smaller in all groups compared with healthy controls.
CC3
Subregion CC3 was smaller for only the disorganised schizophrenia group compared with healthy controls (p=0.01). No significant differences were found between other schizophrenia groups and controls (p>0.05).
CC4
Subregion CC4 was smaller in the paranoid, disorganised and undifferentiated schizophrenia groups compared with healthy controls (p=0.01, p=0.04, p=0.04, respectively). There was no significant difference in the CC4 area in the residual group (p>0.05).
CC5
Subregion CC5 was smaller in paranoid and undifferentiated schizophrenia groups compared with healthy controls (p=0.05, p=0.007, respectively). Other comparisons of groups were not significant (p>0.05). In both patients and controls, no correlation was detected between PANSS scores and CC subregion volume.
Discussion
In the present study, we found that: (i) right-handed schizophrenia patients of different subtypes had significant differences in specific regional callosal measures; (ii) schizophrenia patients showed a smaller global CC area compared with healthy controls; and (iii) patients with undifferentiated schizophrenia had a smaller CC length in comparison with both healthy controls and other schizophrenia groups. Abnormally small areas of total CC and its subregions were found in a sample of four different subtypes of chronic schizophrenia patients, suggesting decreased density in white matter tracts in these specific callosal subregions. Therefore, schizophrenia patients may have connectivity disturbances between specific cerebral hemispheric regions and, as a result, show a heterogeneity in symptoms and different diagnostic criteria.
Monkey and human studies suggest a topographic mapping of cortical areas to specific CC regions, with prefrontal, premotor and motor fibres crossing through the rostral half, and parietal, temporal and occipital lobe fibres crossing through the caudal half of the CC.[28-30] These studies also indicate that the fibre composition differs according to the cortical regions connected. Highly myelinated, fast-conducting, large-calibre fibres connect primary and secondary sensory and motor areas, and poorly myelinated, small-calibre and slowly conducting fibres connect homologous prefrontal and temporoparietal association areas.[31,32]
A great deal of literature has reported a reduction in CC area in schizophrenia patients and suggested an association between CC abnormalities and the occurrence of schizophrenia.[11,13,14]However, previous studies have found no differences in CC area regarding age, gender, course of illness or handedness in schizophrenia patients.[14,33,34]A recent MRI study of the CC using different methodologies suggested that age, sex and clinical status may affect the shape of the CC in schizophrenia patients.[35] In our study, we found significant differences in the areas of CC subregions among schizophrenia subtypes. Similar to our findings, in a previous study it was suggested that neuroanatomic measures in the brain may be different among schizophrenia subtypes.[36] However, total and subareas of CC were not related to illness duration or illness severity in subtypes of schizophrenia in this study. Whitford et al.[37] found that measures of CC size were related to severity of psychotic symptoms in schizophrenia patients, which is inconsistent with our result. Walterfang et al. [38] found a reduction in the anterior genu region (prefrontal region) of the CC in first-episode schizophrenia patients, whereas reductions were also found in other regions of the CC connecting cingulate, temporal and parietal cortices in chronic schizophrenia patients. It seems that the changes of CC size in schizophrenia are more complex with confounding factors.
To our knowledge, this is the first study demonstrating the existence of structural abnormalities in all regions of the CC in different subtypes of schizophrenia. In our study, a specific objective was to examine the CC area in patients with schizophrenia subtypes, and we suggest that one of the ways to reduce the inconsistency of results is to focus specifically on groups and analyse region-specific differences in CC area. Our four patient groups and our control group were matched in age, sex and handedness. As mentioned above, this is important since these factors affect the CC area.
Another reason for discrepant results might be the parcellation of the CC. Older structural neuroimaging research on the CC was commonly based on classification criteria suggested by Witelson,[39]which divides the CC into seven subdivisions using criteria based on the results of experimental work with monkeys and humans. In our study, we compared the CC areas of schizophrenia patients with controls using a novel scheme proposed by Hofer and Frahm,[27] who used diffusion tensor imaging, and their partitioning scheme exactly mirrors the texture of the CC at the cellular level. Also, we used a manual method while tracing the CC, which has been the most frequently used method in clinical studies of CC morphometry for years.[40] More recent studies also used automated measurements of the human CC using MRI. However, in a study presenting a three-dimensional method for segmenting the CC in normal subjects and brain cancer patients with glioblastoma, researchers compared automatic and manual segmentation methods and obtained similar results with the two methods.[41] Also, Dewey et al.[40] found a strong correlation between manual and automated techniques in many structures measured in the brain. Furthermore, a study that tested the ability of conventional manual segmentation and two automatic segmentation methods for measuring hippocampal volume found that the manual method was more sensitive than both automated methods, and suggested that manual segmentation should still be used as the standard technique for volume measurements.[42] Furthermore, most of the automatic techniques are not completely automated and require manual intervention to outline the callosal boundaries.[43] The disadvantages of the manual method are: (i) results can be influenced by factors such as anatomical protocols, tracer experience, scan acquisition parameters and image quality; (ii) some authors consider that manual tracings are time consuming, but we think that this depends on the examined structure's complexity, and the CC is not a complex structure for tracing. Nevertheless, we consider that manual methods may be impractical in large cohort studies.
In accordance with the scheme proposed by Hofer and Frahm,[27] we distinguished five vertical partitions of the CC: region I, the anterior-most sixth of the CC, contains fibres projecting into the prefrontal region; region II, which is the remainder of the anterior half of the CC, contains fibres projecting to the premotor and supplementary motor areas; region III is defined as the posterior half minus the posterior-most third and comprises fibres projecting into the primary motor cortex; region IV, the posterior one-third minus the posterior one-fourth, contains fibres projecting to primary sensory cortices; and region V is defined as the posterior one-fourth of the CC, and contains fibres projecting to the parietal, temporal and occipital cortex.
The prefrontal cortex is a large area of the brain that takes up most of the frontal lobes in the right and left hemispheres. This part of the brain is highly developed in humans and gives us much of our intelligence and problem-solving ability. The prefrontal cortex plays a role in the regulation of complex cognitive, emotional and behavioural functioning, and has the ability to process both the current environment and past memories.[44] Brazo et al.[45] showed that there is a distinct cognitive impairment among subtypes of schizophrenia. In the current study, we found that the area of the CC1 (prefrontal area) was smaller in the disorganised and undifferentiated schizophrenia group than in the normal controls. We therefore hypothesise that patients with disorganised and undifferentiated schizophrenia have more affected cognitive functions than other schizophrenia subtypes. Also, our analysis showed that the prefrontal cortex was not affected in paranoid schizophrenia patients. This is in line with the findings of Goldstein et al.,[46] who described that paranoid schizophrenia patients tend to be more cognitively intact than patients with undifferentiated and residual subtypes of schizophrenia. Thus, we hypothesise that cognitive function of paranoid schizophrenia patients resembles that of healthy people.
The premotor cortex is a narrow area between the prefrontal and motor cortices. It projects directly to the spinal cord and may therefore play a role in the direct control of behaviour. It is involved in preparing and executing limb movements, and co-ordinates with other regions to select appropriate movements. In addition, the supplementary motor area is more involved in internally generated planning of movement, the planning of sequences of movement, and the co-ordination of the two sides of the body, such as in bimanual co-ordination movements. [47] All schizophrenia patients were associated with smaller areas of CC2 (premotor area), and the CC2 area was the only area that was smaller in all schizophrenia patients. Our findings are in line with previous studies that reported that the premotor cortex is the main disconnected area in patients with schizophrenia.[48] The primary motor cortex works in association with other motor areas to plan and execute simple movements. In the current study, we found that the area of the CC3 (motor area) was smaller in only the disorganised schizophrenia group compared with the normal controls. It is known that someone with disorganised schizophrenia will often have difficulties performing daily routine tasks, such as brushing their teeth, showering and dressing. Consequently, our findings may explain the difficulties of motor activities in disorganised schizophrenia patients who have a small CC3 area.
The primary somatosensory cortex is the main sensory receptive area for the sense of touch. In the current study, the CC4 (sensorial) area was found to be markedly smaller in all study patients except for those with the residual type of schizophrenia. In addition, the CC5 area, which contains fibres projecting to the visual and auditory cortex, was smaller in paranoid and undifferentiated schizophrenia patients. This is in line with the findings of Fukuzako et al.,[21] who described that undifferentiated subtypes have greater impairment in neuronal integrity or function in the temporal lobe than patients with other subtypes of schizophrenia.[21] In a functional MRI study, Simons et al.[49] reported that hallucinations are associated with abnormal brain activity in sensory areas. According to our data, in the residual group, the CC2 (premotor) area was the only affected area and the areas of the other four parts was not different from healthy subjects. This finding explains the lack of prominent positive symptoms (delusions and hallucinations) that are associated with sensory areas in residual schizophrenia. [50]
Our study was conducted using DSM-IV-TR criteria. As is known, schizophrenia subtypes have been removed from DSM-V (DSM, 5th edition). It is suggested that these subtypes are not useful to clinicians because patient symptoms often change from one subtype to another, and present overlapping subtype symptoms.[51] However, many studies have shown that schizophrenia is a heterogeneous pathophysiological disorder and its symptoms may be different at the biological level[52] and be associated with radiological abnormalities.[53] This study confirms that schizophrenia symptoms may play a potential role in differences in CC size.
Study limitations
Firstly, our sample size was small. Secondly, prior treatment of patients, which may change brain tissue volume, may be an issue.[54] Further studies with larger numbers of subjects who are free from antipsychotic medication are needed to overcome these limitations.
Conclusion
We found that CC area is significantly reduced in schizophrenia patients, and that this differs among schizophrenia subtypes. Further studies are needed to support our preliminary results about the relationship between CC area and the clinical characteristics of schizophrenia.
Acknowledgements. We thank teaching assistant lecturer Fugen Ozer Gonul (who is a graphic designer) for her figure preparation, and Yunus Yildiz, MD, for assistance with statistical analyses and manuscript preparation.
References
1. Gazzaniga MS. Cerebral specialization and interhemispheric communication: Does the CC enable the human condition? Brain 2000;123(7):1293-1326. [http://dx.doi.org/ 10.1093/brain/123.7.1293] [ Links ]
2. Quigley M, Cordes D, Turski P, et al. Role of the corpus callosum in functional connectivity. AJNR Am J Neuroradiol 2003;24(2):208-212. [ Links ]
3. David AS. Schizophrenia and the corpus callosum. Developmental, structural and functional relationships. Behav Brain Res 1994;64(1-2):203-221. [ Links ]
4. Brambilla P, Nicoletti M, Sassi RB, et al. Corpus callosum signal intensity in patients with bipolar and unipolar disorder. J Neurol Neurosurg Psychiatry 2004;75(2):221-225. [ Links ]
5. Raine A, Lencz T, Taylor K, et al. Corpus callosum abnormalities in psychopathic antisocial individuals. Arch Gen Psychiatry 2003;60(11):1134-1142. [http://dx.doi.org/10.1001/archpsyc.60.11.1134] [ Links ]
6. Kitayama N, Brummer M, Hertz L, Quinn S, Kim Y, Bremner JD. Morphologic alterations in the corpus callosum in abuse-related posttraumatic stress disorder: A preliminary study. J Nerv Ment Dis 2007;195(12):1027-1029. [http://dx.doi.org/10.1097/NMD.0b013e31815c044f] [ Links ]
7. Frazier TW, Hardan AY. A meta-analysis of the corpus callosum in autism. Biol Psychiatry 2009;66(10):935-941. [http://dx.doi.org/10.1016/j.biopsych.2009.07.022] [ Links ]
8. Hynd GW, Semrud-Clikeman M, Lorys AR, Novey ES, Eliopulos D, Lyytinen H. Corpus callosum morphology in attention deficit-hyperactivity disorder: Morphometric analysis of MRI. J Learn Disabil 1991;24(3):141-146. [ Links ]
9. Rosenthal R, Bigelow LB. Quantitative brain measurements in chronic schizophrenia. Br J Psychiatry 1972;121(562):259-264. [ Links ]
10. Nasrallah HA, Andreasen NC, Coffman JA, et al. A controlled magnetic resonance imaging study of corpus callosum thickness in schizophrenia. Biol Psychiatry 1986;21(3):274-282. [ Links ]
11. Bachmann S, Pantel J, Flender A, Bottmer C, Essig M, Schröder J. Corpus callosum in first-episode patients with schizophrenia: A magnetic resonance imaging study. Psychol Med 2003;33(6):1019-1027. [ Links ]
12. Guenther W, Brodie JD, Bartlett EJ, et al. Diminished cerebral metabolic response to motor stimulation in schizophrenics: A PET study. Eur Arch Psychiatry Clin Neurosci 1994;244(3):115-125. [ Links ]
13. Arnone D, Mcintosh AM, Tan GM, Ebmeier KP. Meta-analysis of magnetic resonance imaging studies of the corpus callosum in schizophrenia. Schizophr Res 2008;101(1-3):124-132. [http://dx.doi.org/10.1016/j.schres.2008.01.005] [ Links ]
14. Woodruff PW, McManus IC, David AS. Meta-analysis of corpus callosum size in schizophrenia. J Neurol Neurosurg Psychiatr 1995;58(4):457-461. [ Links ]
15. Preuss UW, Meisenzahl EM, Frodl T, et al. Handedness and corpus callosum morphology. Psychiatry Res 2002;116(1-2):33-42. [ Links ]
16. Highley JR, Esiri MM, McDonald B, Cortina-Borja M, Herron BM, Crow TJ. The size and fibre composition of the corpus callosum with respect to gender and schizophrenia: A post-mortem study. Brain 1999;122(1):99-110. [ Links ]
17. Woodruff PW, Pearlson GD, Geer MJ, Barta PE, Chilcoat HD. A computerized magnetic resonance imaging study of corpus callosum morphology in schizophrenia. Psychol Med 1993;23(1):45-56. [ Links ]
18. Tibbo P, Nopoulos P, Arndt S, Andreasen NC. Corpus callosum shape and size in male patients with schizophrenia. Biol Psychiatry 1998;44(6):405-412. [ Links ]
19. Höppner J, Kunesch E, Grossmann A, et al. Dysfunction of transcallosally mediated motor inhibition and callosal morphology in patients with schizophrenia. Acta Psychiatr Scand 2001;104(3):227-235. [http://dx.doi.org/10.1034/j.1600-0447.2001.00247.x] [ Links ]
20. John JP, Shakeel MK, Jain S. Corpus callosal area differences and gender dimorphism in neuroleptic-naive, recent-onset schizophrenia and healthy control subjects. Schizophr Res 2008;103(1-3):11-21. [http://dx.doi.org/10.1016/j.schres.2008.04.035] [ Links ]
21. Fukuzako H, Kodama S, Fukuzako T, et al. Subtype-associated metabolite differences in the temporal lobe in schizophrenia detected by proton magnetic resonance spectroscopy. Psychiatry Res 1999;92(1):45-56. [ Links ]
22. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th ed. Washington, DC: American Psychiatric Association, 1994. [ Links ]
23. Kostakoğlu AE, Batur S, Tiryaki A. Pozitif ve Negatif Sendrom Ölçeğinin (PANSS) Türkçe Uyarlamasının Geçerlik ve Güvenilirliği. Türk Psikoloji Dergisi 1999;14:23-32. [ Links ]
24. First M, Spitzer R, Gibbon M, Williams J. Structured Clinical Interview for DSM-IV Axis I Disorders (SCID-I), Clinician Version. Washington, DC: American Psychiatric Publishing, 1997. [ Links ]
25. Oldfield RC. The assesment and analysis of handedness: The Edinburgh inventory. Neuropsychologia 1971;9(1):97-113. [ Links ]
26. Witelson SF. The brain connection: The corpus callosum is larger in left-handers. Science 1985;229(4714):665-668. [ Links ]
27. Hofer S, Frahm J. Topography of the human corpus callosum revisited-comprehensive fibre tractography using diffusion tensor magnetic resonance imaging. Neuroimage 2006;32(3):989-994. [http://dx.doi.org/10.1016/j.neuroimage.2006.05.044] [ Links ]
28. de Lacoste MC, Kirkpatrick JB, Ross ED. Topography of the human corpus callosum. J Neuropathol Exp Neurol 1985;44(6):578-591. [ Links ]
29. Pandya DN, Rosene DL. Some observations on trajectories and topography of commissural fibres. In: Reeves AG, ed. Epilepsy and the Corpus Callosum. New York: Plenum, 1985:21-39. [ Links ]
30. Seltzer B, Pandya DN. Posterior parietal projections to the intraparietal sulcus of the rhesus monkey. Exp Brain Res 1986;62(3):459-469. [ Links ]
31. Aboitiz F. Brain connections: Interhemispheric fibre systems and anatomical brain asymmetries in humans. Biol Res 1992;25(2):51-61. [ Links ]
32. LaMantia AS, Rakic P. Axon overproduction and elimination in the corpus callosum of the developing rhesus monkey. J Neurosci 1990;10(7):2156-2175. [ Links ]
33. Highley JR, Esiri MM, McDonald B, Cortina-Borja M, Herron BM, Crow TJ. The size and fibre composition of the corpus callosum with respect to gender and schizophrenia: A post-mortem study. Brain 1999;122(1):99-110. [ Links ]
34. Bersani G, Quartini A, Iannitelli A, et al. Corpus callosum abnormalities and potential age effect in men with schizophrenia: An MRI comparative study. Psychiatry Res 2010;183(2):119-125. [http://dx.doi.org/10.1016/j.pscychresns.2010.04.011] [ Links ]
35. Joshi SH, Narr KL, Philips OR, et al. Statistical shape analysis of the corpus callosum in schizophrenia. Neuroimage 2013;64:547-559. [http://dx.doi.org/10.1016/j.neuroimage.2012.09.024] [ Links ]
36. Gur RE, Mozley PD, Shtasel DL, et al. Clinical subtypes of schizophrenia: Differences in brain and CSF volume. Am J Psychiatry 1994;151(3):343-350. [ Links ]
37. Whitford TJ, Kubicki M, Schneiderman JS, et al. Corpus callosum abnormalities and their association with psychotic symptoms in patients with schizophrenia. Biol Psychiatry 2010;68(1):70-77. [http://dx.doi.org/10.1016/j.biopsych.2010.03.025] [ Links ]
38. Walterfang M, Wood AG, Reutens DC, et al. Morphology of the corpus callosum at different stages of schizophrenia: Cross-sectional study in first-episode and chronic illness. Br J Psychiatry 2008;192(6):429-434. [http://dx.doi.org/10.1192/bjp.bp.107.041251] [ Links ]
39. Witelson SF. Hand and sex differences in the isthmus and genu of the human corpus callosum: A postmortem morphological study. Brain 1989;112(3):799-835. [ Links ]
40. Dewey J, Hana G, Russell T, et al. Reliability and validity of MRI-based automated volumetry software relative to auto-assisted manual measurement of subcortical structures in HIV-infected patients from a multisite study. Neuroimage 2010;51(4):1334-1344. [http://dx.doi.org/10.1016/j.neuroimage.2010.03.033] [ Links ]
41. Nazem-Zadeh MR, Saksena S, Babajani-Fermi A, et al. Segmentation of corpus callosum using diffusion tensor imaging: Validation in patients with glioblastoma. BMC Med Imaging 2012;12:10. [http://dx.doi.org/10.1186/1471-2342-12-10] [ Links ]
42. Coan AC, Kubota B, Bergo FP, Campos BM, Cendes F. 3T MRI quantification of hippocampal volume and signal in mesial temporal lobe epilepsy improves detection of hippocampal sclerosis. AJNR Am J Neuroradiol 2014;35(1):77-83. [http://dx.doi.org/10.3174/ajnr.A3640] [ Links ]
43. Herron TJ, Kang X, Woods DL. Automated measurement of the human corpus callosum using MRI. Front Neuroinform 2012;6:25. [http://dx.doi.org/10.3389/fninf.2012.00025] [ Links ]
44. Miller EK. The prefrontal cortex and cognitive control. Nat Rev Neurosci 2000;1(1):59-65. [ Links ]
45. Brazo P, Marié RM, Halbecq I, et al. Cognitive patterns in subtypes of schizophrenia. Eur Psychiatry 2002;17(3):155-162. [http://dx.doi.org/10.1038/35036228] [ Links ]
46. Goldstein G, Shemansky WJ, Allen DN. Cognitive function in schizoaffective disorder and clinical subtypes of schizophrenia. Arch Clin Neuropsychol 2005;20(2):153-159. [http://dx.doi.org/10.1016/j.acn.2004.03.008] [ Links ]
47. Roland PE, Larsen B, Lassen NA, Skinhøj E. Supplementary motor area and other cortical areas in organization of voluntary movements in man. J Neurophysiol 1980;43(1):118-136. [ Links ]
48. Bray T, Agius M. Soft neurological signs and schizophrenia - a looking glass into core pathology? Psychiatr Danub 2009;21(3):327-328. [ Links ]
49. Simons CJ, Tracy DK, Sanghera KK. Functional magnetic resonance imaging of inner speech in schizophrenia. Biol Psychiatry 2010;67(3):232-237. [http://dx.doi.org/10.1016/j.biopsych.2009.09.007] [ Links ]
50. Brown GG, Thompson WK. Functional brain imaging in schizophrenia: Selected results and methods. Curr Top Behav Neurosci 2010;4:181-214. [ Links ]
51. American Psychiatric Association. Schizophrenia: DSM-5 Fact Sheet 2013. Washington, DC: American Psychiatric Association, 2013. [ Links ]
52. Herken H, Uz E, Ozyurt H, Söğüt S, Virit O, Akyol O. Evidence that the activities of erythrocyte free radical scavenging enzymes and the products of lipid peroxidation are increased in different forms of schizophrenia. Mol Psychiatry 2001;6(1):66-73. [ Links ]
53. Pearlson GD, Marsh L. Structural brain imaging in schizophrenia: A selective review. Biol Psychiatry 1999;46(5):627-649. [ Links ]
54. Ho BC, Andreasen NC, Ziebell S, Pierson R, Magnotta V. Long-term antipsychotic treatment and brain volumes: A longitudinal study of first-episode schizophrenia. Arch Gen Psychiatry 2011;68:128-137. [http://dx.doi.org/10.1001/archgenpsychiatry.2010.199] [ Links ]
 Correspondence:
Correspondence:
E Unlu (edunlu@yahoo.com)














