Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Health SA Gesondheid (Online)
On-line version ISSN 2071-9736
Print version ISSN 1025-9848
Health SA Gesondheid (Online) vol.22 Cape Town 2017
http://dx.doi.org/10.1016/j.hsag.2016.11.001
FULL LENGTH ARTICLE
Supply chain solutions to improve the distribution of antiretroviral drugs (ARVs) to clinics in rural areas: A case study of the QwaQwa district
Mamolise MokhesengI, 1; Gideon S. HornI, *, 1; Aileen G. KlopperII, 1
IPO Box 77000, Nelson Mandela Metropolitan University, Port Elizabeth, 6031, South Africa
IIFaculty of Health Sciences, PO Box 77000, Nelson Mandela Metropolitan University, Port Elizabeth, 6031, South Africa
ABSTRACT
This article serves as a case study based on research that was performed in the QwaQwa district in the Free State Province where the distribution of ARVs to the regional Manapo hospital, as well as between the hospital and its peripheral clinics, was interrupted and inconsistent due to problems in the supply chain. An unreliable and interrupted ARV supply chain creates the risk of virus reactivation and eventual patient mortality.
The objectives of the study were to explore the problems experienced with the ARV distribution practices at the Manapo hospital, and to recommend ways in which the distribution of ARVs can be improved so that patients can receive an uninterrupted supply. The nature of the topic researched dictated the use of mainly the quantitative research method.
The main problems identified include: Wrong and no uniform practice of ordering stock by the hospital and the clinics; lack of reliable, structured transportation from the depot to the hospital; as well as poor inventory management and poor overall communication. Recommendations to address the problems include: Implementing a supply chain planning and design process; improving inventory management and warehousing practices; implementing more effective and reliable distribution and transportation processes; as well as improving supply chain coordination and overall communication.
Keywords: ARV distribution, Fixed order quantity policy, Inventory management, Supply chain management, Warehousing practice
1. Introduction
Research has shown that people living with the Human Immunodeficiency Virus (HIV) and the Acquired Immunodeficiency Syndrome (AIDS) can lead normal and healthy lives on condition that they receive their antiretroviral medication (ARVs) on a continuous, uninterrupted basis (Rang, Dale, Ritter, & Moore, 2003, pp. 657, 659-650; Sande & Volberding, 1999, p. 97). With uninterrupted Antiretroviral Treatment (ART) the level of HIV in an infected person's body is kept at minimum levels; any further weakening of the immune system is stopped; the virus can become dormant; and the lives of HIV and AIDS infected persons prolonged (Schouten, 2011,p. 1).
However, the treatment of HIV and AIDS is life-long and, once initiated, it is imperative for patients to obtain an uninterrupted monthly supply of their ARVs from health facilities such as hospitals and clinics so that their medication can be taken on a daily basis for the rest of the person's life. When the use of the ARVs is interrupted there is a potential risk of virus reactivation. Unfortunately, the supply of ARVs to hospitals and clinics is often interrupted due to unreliable, ineffective (not at satisfactory levels) and inefficient (too costly) procurement and distribution systems, which results in high levels of morbidity and mortality (Management Sciences for Health, 2006, p. 228). In a study undertaken by Mori and Owenya (2014, p. 1) on ARV distribution in Tanzania, they found that stock-outs of ARVs due to inefficient supply systems, quantification problems and short expiry duration, caused patients to change their ART regimens, thereby increasing the risk of the emergence of drug-resistant HIV strains. According to Nsimba and Irundi (2010, p. 14), medication non-adherence has been associated with increased secondary bacterial and parasite resistance in Sub-Saharan African countries.
This article is part of a study that was performed in the QwaQwa district in the Free State Province. Here the distribution of ARVs to the regional Mofumahadi Manapo Mopeli Regional hospital (Manapo hospital for short), as well as between the hospital and its peripheral clinics, was interrupted and inconsistent due to problems in the supply chain, making Highly Active Antiretroviral Treatment (HAART) very difficult. With the hospital not receiving its ordered quantities on time, it failed to provide clinics with an uninterrupted supply of ARVs. This resulted in patients experiencing adherence problems, often causing treatment failure (death). This interrupted and inconsistent supply of ARVs to patients due to supply chain problems, is the research problem that was addressed in this study. Therefore, the research question is the following: What are the reasons for the interrupted and inconsistent supply of ARVs from the hospital and clinics to the patients?
2. Purpose of the article
The purpose of the article is to explore the effectiveness (whether patients are well served) and efficiency (at the lowest possible cost) of the supply of ARVs between the pharmaceutical warehouses/depots, the hospital and the clinics.
3. Objectives of the article
Based on the research problem and the research question indicated above, the objectives of this article, similar to the objectives of the study on which it is based, are the following:
3.1. To explore and describe the supply chain problems experienced with the ARV distribution practices at the Manapo hospital.
3.2. To recommend ways in which the distribution of ARVs can be done more effectively and efficiently at the hospital, in the district, in rural areas, and the country at large, so that patients can receive an uninterrupted supply of ARVs.
4. ARV treatment in practice
In November 2003, the South African government, through the document called the Operational Plan for Comprehensive HIV and AIDS Care, Management and Treatment (OPCCMT), initiated the supply of ARVs through public health facilities to HIV patients as part of the Comprehensive Care, Management and Treatment (CCMT) programme (National Department of Health (NDoH) (2003), pp. 13-14, 17).
Based on the document and in terms of current policy, the provincial departments of health, on the conclusion of open tenders, appoint the successful bidders as contractors who are responsible for procuring ARVs from the manufacturers and supplying these to provincial regional pharmaceutical warehouses/depots (pharmaceutical warehouses for short). The manufacturer maintains a warehouse from which orders are dispatched to the pharmaceutical warehouses according to a prescribed requisition procedure (see Fig. 1). Each hospital is allocated a budget which is used to order its required quantities of ARVs from these pharmaceutical warehouses.
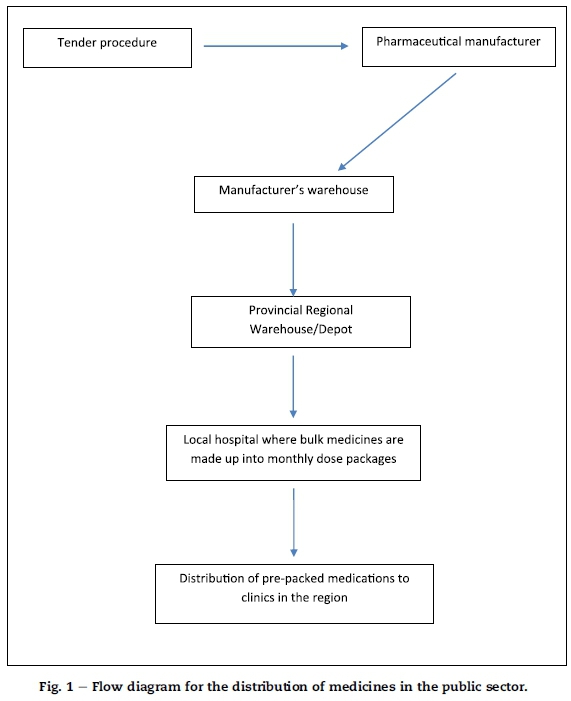
The pharmaceutical warehouse, on request, distributes the medication to the local hospitals where the bulk medications are made up into pre-packed monthly packages for further distribution to the clinics in the region. A hospital such as the Manapo hospital would thus order ARVs from the nearby pharmaceutical warehouse, and have these transported to and stored at the hospital. From the hospital the drugs are distributed to various peripheral clinics for easy access by patients. The release of these medications to the clinics follows a requisition procedure in which each clinic will request a specified number of packages according to the number of patients requiring the medication.
This is a relatively simple process for chronic medications where the demand is likely to be fairly constant, for example, ARVs, anti-hypertensive drugs and anti-epileptic drugs. A small excess will be maintained in case any new patients are enrolled at the clinic. Certain medications will be ordered according to the specific procedure with due cognisance of available supplies, for example, antibiotics, analgesics and cold medications.
When HIV patients are initiated onto ARV treatment at hospitals and clinics, they are issued with their first month's supply of ARVs, and given a three-week return date to report back to their initiation centres. The reasons for the three-week return date include: performing a pill-count of the balance of the medication to check whether there are any missed doses; to assess and further educate the patient on the management of the drug's adverse effects; to provide further counselling on the importance of adherence and compliance; to issue the following month's supply of ARVs; and to inform the patients about the down-referral system, that is, where to collect their continuous supply of ARVs. Most importantly, a three-week return date is given to ensure that patients collect the following month's ARVs supply while they still have a few days of medication remaining from the previous collection to avoid treatment interruption. However, the uninterrupted supply of ARVs to patients can only be achieved through a well-managed supply chain - a topic discussed next.
5. Short explanation of the concept supply chain management
Supply chain management is a wide and encompassing field of study, therefore only the main aspects and key concepts pertaining to this article, are briefly outlined in the following paragraphs.
A supply chain is a network of business organisations, called supply chain partners, that work together to convert and move goods from the raw material stage to the end user (Verma & Boyer, 2010, p. 19). Hugos (2011, p. 4) defines supply chain management as the coordination of production, location, inventory and transportation activities among supply chain partners, to achieve the best mix of responsiveness and efficiency for the market being served. Therefore, in the case of distributing ARVs, each supply chain partner involved, namely the contractor, manufacturer, warehouse, transporter, hospital and clinic, has to play a coordinated and integrated role to ensure the uninterrupted distribution of ARVs to the eventual patients.
Unfortunately, there were problems in obtaining and distributing the ARVs between the pharmaceutical warehouse, the Manapo hospital and the peripheral clinics, causing patients to be without medication. The hospital claimed the warehouse maintained inadequate stock-keeping practices and was therefore unreliable in the timely delivery of the drugs. On the other hand, the hospital was accused of lacking sufficient and suitable warehousing facilities; that the staff did not have proper inventory management procedures in place; and that the transport system between the hospital and the peripheral clinics was unreliable. All these supply chain issues resulted in an unsatisfied eventual customer -who in this case is the patient in need of treatment.
The four main components of supply chain management, with all four being applicable to this article, include: supply chain planning and design; inventory management and warehousing; distribution and transportation; as well as supply chain coordination. Strategic and careful planning and design is often seen as the most important aspect because, as Pienaar and Vogt (2012, pp. 34-45) argue, the success of any business organisation depends on whether the design of its own business strategy is well integrated with that of the specific supply chain within which it operates. Proper supply chain planning and design goes a long way in eliminating problems such as inferior customer service issues, communication problems, relatively too high or too low inventory levels for the level of customer service to be achieved, poor coordination of effort, as well as long production and delivery lead-times. The concept lead-time refers to the elapsed time between the placement of an order and the receipt of the order (Jacobs, Berry, Whybark, & Vollmann, 2011, pp. 197, 280).
The four main operational areas to address during strategic supply chain planning and design are customer seruice leuels, facility location, inventory decisions and transport strategy (Ballou, 2004, pp. 39-41). A supply chain model should only be designed once the strategic planning on these four areas has been completed. Therefore, selecting the proper design greatly affects the efficiency and effectiveness of the supply chain.
Strongly linked to supply chain planning and design are the so-called supply chain drivers, which are the buildingblocks of a well-integrated supply chain. The major supply chain drivers relate to flexibility, responsiveness, reliability, availability and lowest delivery costs (efficiency) (Fawcett, Ellram, & Ogden, 2007, pp. 32-35). All five of these drivers play important interrelated roles to ensure an efficient and effective supply chain and, therefore, an uninterrupted supply of, amongst others, ARVs.
Flexibility is required when problems are experienced around the structured supply chain operations. For example, if the vehicle allocated for the transportation of goods (ARVs in this case) breaks down, alternative transport has to be available (Pienaar & Vogt, 2012, p. 41). Responsiveness, on the other hand, makes the firm more flexible and responsive to customers' changing requirements by designing new and/or improved products or services (Wisner, Keah-Choon, & Leong, 2012, pp. 24-25).
Reliability represents the notion that a product and the supply chain of which it forms part, can be relied on. Availability is the continuous accessibility of the product offered by the business to its customers. Product availability has to be ensured when demand occurs at any given time (Hugos, 2011, p. 57). The last supply chain driver, which is lowest delivery costs, while highly valued by customers, presents managers with unrelenting pressure to reduce costs by sourcing from the most efficient suppliers (Pienaar & Vogt, 2012, pp. 40-43).
The key focus areas and key performance indicators (KPIs) used to assess whether the drivers are in place and also to assess progress in terms of supply chain success, sustain-ability and effectiveness, include (Pienaar & Vogt, 2012, pp. 45-47):
5.1. Operational efficiency and effectiveness
This ensure the smooth running of supply chain operations. The stock ordering processes, for example, are considered efficient and effective if the stock is delivered within the specified time, at the correct place, at a competitive price, is of the right quality and in the correct quantity. This is known as the five rights of Logistics. In relation to this article, it means that ARVs need to be delivered and distributed at the right time, the right place, the right price, the right quality and the right quantity.
5.2. Inventory management and optimisation
The required stock levels at all stocking points, for example, the warehouses at the pharmaceutical warehouse and the hospital, should be maintained at all times, taking into account factors such as lead-times, replenishment cycles and safety stock. There should also be a system in place that easily tracks the location and movement of stock at all points. This requires good communication and information (computer) systems.
Therefore, a consistent data structure has to be established throughout the supply chain to facilitate effective and efficient decision-making and execution. Data on factors such as stock quantities on order, lead-times, stock balances in the warehouse, and all other relevant documentation should always be accurate and available for retrieval. In terms of this article, the proper management of inventory and warehousing is key to a successful ARV drug distribution system. Warehousing is an essential requirement for not only the storage of inventory, but also to facilitate the proximity of the stock to the customers and the preparation of ordered stock. Furthermore, a warehouse must have a proper schedule of the dates and times to make deliveries; which batch of consignment to deliver; and shipment documentation to accompany the consignment being transported when scheduling deliveries.
5.3. Risk management
The objective of risk management is to anticipate, avoid and neutralise risks. For example, if an ARV supplier cannot supply the order of a customer such as a pharmaceutical warehouse, there should be measures or contingency actions in place to deal effectively with such uncertainties and complexities to ensure that the customer's order is completely filled as soon as possible.
5.4. Relationship management
Mutually beneficial relationships with suppliers, management and customers have to be established and maintained. A long-term relationship should be maintained with only those suppliers that have high performance scores. Efficient distribution, for example, relies on an efficient transportation system, which requires good communication between the ARV supplier, the distributor and the customer - the latter being the pharmaceutical warehouse or hospital. Decisions also have to be made and agreement reached on the mode of transport to be used for efficient transportation. According to Chopra and Meindl (2010, p. 403), the most important operational decision related to transportation in the supply chain is the routing and scheduling of deliveries. The decision when selecting the appropriate route has to be based on the geographical area, the distance between the sourcing and stocking point, and the safety of the route.
A lack of coordination in a supply chain often occurs when each operational stage or supply chain partner has their own set of objectives that are in conflict with the other stages or partners, or when the information moving between the stages or partners is delayed and/or distorted. When the different stages do not function as a whole, each stage or partner will optimise only its own objectives, without considering the impact on the complete chain. The result is a loss of supply chain coordination with negative consequences for every partner and the final customer (patient).
The so-called bullwhip effect is one of the main reasons for a lack of coordination in a supply chain. The bullwhip effect refers to a situation where members upstream in a supply chain are faced with a high degree of uncertainty and variability about the time pattern and volume of replenishment orders to be received from members downstream (Ireland & Crum, 2005,p. 6; Chandrasekaran, 2012, p. 370). Demand information is distorted and fluctuations in orders at all stages in the supply chain occur because each partner has a different assessment of what demand looks like (Johnson, Leenders, & Flynn, 2011, p. 223). In other words, the clinic does not know the correct quantity of ARVs to order from the hospital, resulting in the hospital not ordering or ordering the wrong quantity from the pharmaceutical warehouse, which again results in the pharmaceutical warehouse not ordering or ordering the incorrect quantity from the ARV manufacturer. This results in problems such as stock-outs, product obsolescence due to excessive stock levels, and/or incorrect production batches by the ARV manufacturer. All these problems lead to unacceptable customer service levels and eventual treatment failure (death).
Having been previously employed in the QwaQwa district as a pharmacy manager and experiencing ARV distribution problems during her time of employment there, the main researcher of this article undertook the study to identify the reasons for the interruption problems, and to recommend solutions and standard operating procedures (SOPs) for the Manapo hospital and rural areas in general. The results of the study should enable the Manapo and other rural hospitals to improve their ARV drug distribution practices and the management of their ARV supply chains so that patients can have uninterrupted access to their monthly supply of ARVs, thereby increasing drug adherence and prolonging lives.
6. Research methodology
The nature of the topic researched dictated the use of mainly the quantitative research method, with the study being descriptive and contextual in nature. According to De Vos, Strydom, Fouche, and Delport (2011, pp. 96, 167, 251), descrip-tiue research uses a data analysis method that entails the organisation, display and interpretation of data which results in presenting a picture of the specific details of a situation. It focuses on the how and why questions - in this case the specific details of the ARV drug ordering, receipt and distribution practices followed at the Manapo hospital. Contextual research was used to contextualise the objectives of the study as the investigation took place in a specific setting (the QwaQwa rural district) under certain circumstances (the interrupted supply of ARVs at the Manapo hospital) (Babbie & Mouton, 2001, p. 272).
The research methods utilised in this study included: judgement sampling as a non-probability sampling method; a self-administered questionnaire as the measuring instrument to measure the study variables; and statistical methods to analyse the data collected. The target population of the study comprised of rural hospitals, as well as pharmacists and pharmacist assistants who work at these rural hospitals and are involved in the distribution of ARVs. The Manapo hospital, together with its pharmacists and pharmacist assistants who worked at the hospital before and at the time of the study, was the sample of the article. According to the Manapo hospital pharmacy records of staff departmental allocations and duty roster, a total of 21 pharmacists and pharmacist assistants were allocated to work in the ARV department to perform pharmaceutical-related functions. Therefore, the sample size was restricted to the 21 pharmacists and pharmacist assistants working in the QwaQwa district.
The questionnaire was constructed after secondary sources in the form of books, journals, electronic databases, the Internet, as well as various governmental reports and policies, including the Free State Department of Health (FSDoH) Inventory Management Policy (2012), had been extensively consulted. Furthermore, the main researcher consulted with a statistician, who is a staff member in the Statistical Consultation Unit at the Nelson Mandela Metropolitan University (NMMU) in Port Elizabeth, to clarify and discuss the content and analysis of the questionnaire. The final questionnaire consisted of a total of 34 open-ended questions. The open-ended questions were structured in such a manner that the main researcher could assess the opinions of the respondents for each of the variables she saw necessary to address the purpose of the study.
After designing and developing the questionnaire, it was pre-tested. The main researcher had a pilot run with five pharmacists who gave feedback on the questions and the researchers then did some minor rewording and rephrasing with regard to some of the questions. After these minor adjustments had been done, the researchers were satisfied and convinced that the questionnaire questions were clear and unambiguous, and that the respondents would be able to complete the questionnaire without any difficulty and at their own convenience, as advised by Welman, Kruger, and Mitchell (2005, p. 153). The questionnaire had a covering letter to inform the respondents of the reasons for and objectives of the study, as well as stating that their identity would not be disclosed and the responses treated as anonymous.
A copy of the questionnaire, together with the cover letter, a consent form, and a spare envelope were hand-delivered by the main researcher to each respondent who still worked at the Manapo hospital at the time of the study. For those respondents who had retired and/or moved to other places, the consent forms and questionnaires were sent via e-mail, mailed or faxed. The format of the questionnaire made it easy for the responses to be coded into numeric data that could be analysed. The role of the main researcher was to collect and collate the data, which was then edited, coded, captured in Microsoft Excel and imported into the Statistica Version 9.0 package for analyses. A reliability test was not conducted as the sample was not sufficient to conduct Cronbach Alpha testing.
Two types of validity are applicable to this study, namely content validity and face validity. Content validity refers to the extent (adequacy) to which the measuring instrument (a questionnaire in this case) measures the actual concepts related to the topic. Face validity refers to the fact that the questions are unambiguous to the respondents and therefore the gathered information will be valid (De Vos et al. 2011,p. 167). As explained earlier, the researchers ensured face validity through means of a pilot study where certain questions were slightly adapted, as well as by formulating questions that addressed the defined study objectives. Face validity and content validity were also ensured as follows: the NMMU statistician assisted the main researcher in the design of the questionnaire and with the conversion of the data collected from the questionnaires into numerical data for ease of analysis; a comprehensive literature review was undertaken on which the questionnaire questions were based; as well as the 17 years of practical experience gained by the main researcher in the specific field of study. With the main researcher having left the district as employee by the time the study was done, there was no conflict of interest, neither response bias from the respondents. Response bias was further avoided by the fact that all the members of the sample were invited to participate in the survey without any incentives being offered. Participation was totally voluntaryand no person was forced in any way to participate in the survey.
As far as ethical issues are concerned, approval to conduct the study in the QwaQwa district was obtained from the Head of Department of the Free State Department of Health. As advised by Neuman (2011, p. 152), confidentiality and anonymity were ensured by the main researcher by holding the respondents' information in confidence and by releasing the data in a way that did not permit anyone to link specific individuals to the gathered information. Each questionnaire had a unique tracking number assigned to each respondent. The main researcher kept a list of the respondents' names and tracking numbers which was only used to identify the non- respondents for follow-up purposes.
7. Empirical results and discussion
This section presents the empirical findings of the study with the aid of Tables 1-6. The tables are based on the summaries of the questionnaire responses and presented in terms of the study objectives and the lay-out of the questionnaire used in the study. Of the sample size of 21 possible respondents identified, 20 responded, giving a response rate of 95.2%.
Based on the empirical results, three main reasons for the interrupted supply of ARVs to patients were identified and are outlined below.
7.1. The hospital and the clinics did not order stock according to official policy
In general industry practice the ordering of stock is done in terms of either the fixed order quantity (FOQ) approach or the fixed order interval (FOI) approach. The fixed order quantity model involves re-ordering a fixed amount of a product each time re-ordering takes place. Organisations using this approach generally need to develop a minimum stock level to determine when to re-order the fixed quantity. This is called the (predetermined) re-order point. When the stock on hand reaches this predetermined level, the fixed order quantity (also called the economic order quantity or EOQ) is "automatically" ordered. Basically, the predetermined ordering point/level triggers the next order. When only a fixed amount of a given item is ordered at one time, it helps to limit re-order mistakes, conserve storage space, and prevent unnecessary expenditures that would tie up funds that could be better utilised elsewhere (Coyle, Langley, Novack, & Gibson, 2013, pp. 353, 370).
On the other hand, the fixed order interval approach, also called the fixed period or fixed review period approach, involves ordering varying quantities of inventory at fixed or regular intervals. Generally, the varying amounts ordered depend on how much of the product is in stock and available at the time of the review, meaning that order sizes are normally based on the amount on hand at the time of review (Coyle et al., 2013, pp. 353, 370).
The ordering of stock according to predetermined reorder points - the first-mentioned FOQ approach - is the official Inventory Management Policy of the Free State Department of Health (FSDoH) (2012, p. 15). However, when studying Tables 1 and 2 which list the responses to the questions applicable to the ordering of stock by the hospital from the pharmaceutical warehouse, as well as by the clinics from the hospital, respectively, it is clear that neither official policy nor a uniform, specific order pattern was followed. For example, Table 1 shows that the majority of the respondents (65%) indicated that hospital staff ordered stock from the warehouse on order dates (the FOI approach); 25% of the respondents ordered stock only when safety levels were reached; and 10% of the respondents ordered stock only when needed. According to Table 2, the majority of the respondents (70%) indicated that clinic staff also placed orders mainly according to order dates, while the remaining 30% indicated that clinics placed orders with the hospital only when the stock was depleted (20%) or when convenient (10%).
The ordering of ARVs according to order dates, as practiced by the majority of the respondents, is seen as a main reason why the correct order quantities are not delivered timeously and stock-outs are experienced. The problem is that there might seem to be enough stock at the time of review and ordering, but the stock could become depleted quicker than expected when demand increases unexpectedly.
Table 1 also shows that stock was ordered through the use of manual forms by 45% of the respondents. Some 25% used only the computer system to order the stock, while 30% used both the computer and the manual forms to order the stock from the pharmaceutical warehouse. The use of manual forms could also explain why 35% of the respondents indicated that no information was available on lead-times (see Table 1), while 65% of the respondents could not access information on expected delivery dates. The literature review states that orders should rather be placed electronically (E-procurement) to reduce the lead-times associated with order placement and to improve information transfer and retrieval (Chandrasekaran, 2012, p. 424). Electronic placement also reduces the overall transmitted time.
As shown in Table 2, the majority of the respondents (78%) used in-house transport to deliver the drugs from the hospital to the clinics. The remaining 22% of the respondents alternated between the use of in-house and outsourced transport to deliver ARVs to the clinics.
It was observed that the majority of the respondents (72%) arranged for transport telephonically to request delivery of the drugs from the hospital to the clinics. This type of booking was considered acceptable as the main transport utilised by the respondents was in-house transport. Electronic booking systems would, of course, have been preferable if the dominant transport system was outsourced.
7.2. Lack of reliable, structured transportation from the pharmaceutical warehouse to the hospital
Table 3 lists the results of the questions applicable to the transportation of stock from the pharmaceutical warehouse to the Manapo hospital. The transport system was found to be unreliable as 70% of the respondents indicated that transport only sometimes (not always) arrive according to the scheduled dates and times. This causes delays in the delivery of ARVs, with only 30% of respondents indicating that they receive their orders within the expected time frames, and only 35% always receiving the correct order quantities. Of further concern is the fact that 36% of the respondents ascribed the delays to the unavailability of stock at the pharmaceutical warehouse, with the lack of staff at the pharmaceutical warehouse seen by 32% of the respondents as problematic. Some 18% of the respondents identified the cause of the delays in the delivery of ARVs as the unavailability of transport vehicles to deliver the drugs, while 14% blamed it on the late placement of orders at the pharmaceutical warehouse.
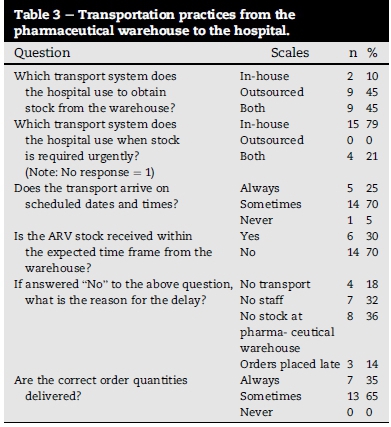
Outsourced transport was utilised for the delivery of the stock from the pharmaceutical warehouse by 45% of the respondents, while 45% of the respondents used both in-house and outsourced transport to obtain the stock from the warehouse. The remaining 10% used only the in-house transport to obtain their orders from the pharmaceutical warehouse. Urgent deliveries were carried out using the in-house transport by 79% of the respondents, while the remaining 21% used both in-house and outsourced transport for urgent deliveries.
7.3. Poor inventory management and poor overall communication
The results of the questions pertaining to inventory management and warehouse practices at the hospital are shown in Table 4. The hospital experienced a high occurrence of stock shortages, with stock-outs often extending over a period of one to three months. Incorrect quantities (53%) and delivered stock having short expiry dates (74%) were the main concerns of the respondents on the status of the stock received from the pharmaceutical warehouse, while 17% and 11% respectively received damaged stock or experienced a break in the cold chain storage. All four of the mentioned inventory-related problems compromise the quality of the ARVs and the success of the inventory management process.
The FSDoH Inventory Management Policy (2012) prescribes two forms of stock take to be performed, namely bi-annual (on 31 March and 30 September) and in-house spot-checks to be carried out on a quarterly basis. However, the empirical results shown in Table 4 give no indication of bi-annual stocktaking being performed at the Manapo hospital. The majority of the respondents (65%) performed stock-taking on a monthly basis, while 25% did it quarterly and 10% on a weekly basis. Also, 85% of the respondents captured the ARV stock levels on stock-cards, while only 15% recorded the stock levels on their computer system. Data is more easily accessible for retrieval when computerised. Furthermore, the process of double-checking orders before issuing ARVs to clinics, which is a basic warehouse operational requirement, was standard practice for only 25% of the respondents.
Although there were a high percentage of respondents who kept records of stock balances at the hospital (78%) and stock issues to clinics (82%), only 50% of the respondents kept records of stock balances at clinics, while the other 50% did not perform any record keeping at all. This was not satisfactory as the hospital staff are the sole providers of medications to the clinics, and therefore need reliable information with regard to the stock levels at the clinics in order to accurately calculate orders from the clinics. The results also show that the total percentage of the respondents who did not maintain any records of stock returns, or the reasons for the returns, or corrective steps to be taken, were calculated at 33%, 24% and 29%, respectively.
A main reason for the poor levels of inventory management is the poor levels of communication and information sharing among the supply chain partners. The majority of the respondents (61%) indicated that there were no meetings or forums held to share information between the warehouse, the hospital and the clinics on matters related to the distribution of ARVs. The remaining 39% of the respondents indicated that they had attended meetings either on a monthly basis (22%) or every 3 months (17%). This made access to accurate information on the lead-times of orders placed and the dates of scheduled deliveries difficult, and this in turn distorted the order cycle processes.
8. Recommendations
As discussed earlier, the literature study undertaken for this article identified the following supply chain processes as being critical for supply chain success: supply chain planning and design; inventory management and warehousing; distribution and transportation; and supply chain coordination. The recommendations proposed to address the main problems identified, are outlined according to these processes.
8.1. Abide by official policy and implement a supply chain planning and design process
As the main topic of this article was the interruptions in the delivery of ARVs and finding solutions for the problems, supply chain planning and design needs to start with abiding by the official FSDoH Inventory Management Policy, that is, following the Fixed Order Quantity approach, with stock to be ordered at a predetermined level. This will ensure more consistency in the timely delivery of ARVs in thecorrectquantities,aswellaslessinterruptionsinthe availability of stock being distributed to the clinics, thereby better meeting patients' requirements of a daily intake.
In line with official policy, a further recommendation on inventory decisions is the use of the pull strategy as opposed to the push strategy currently in existence. The pull strategy facilitates the ordering of stock when pre-determined levels are reached, thereby minimising stock obsolescence (Bowersox, Closs, Cooper, & Bowersox, 2013, p. 174). The latter problem (stock obsolescence) is associated with the push strategy where stock is ordered in advance in anticipation of being used.
8.2. Improve inventory management and warehousing practices
The staff at the pharmaceutical warehouse, the hospital and the clinics should be trained on the importance and optimum methods of stock management, warehousing practices and thorough record keeping of stock. Record keeping of stock discrepancies also needs attention, while the double-checking of orders to be issued to the clinics will limit errors that could arise along the order cycle. Furthermore, electronic stocksheets or stock-registers instead of manual stock-cards should be used as it facilitates easier and faster retrieval of information when required. The use of manual forms should be limited and only used as back-up when the computer systems are off line. The practice of bi-annual stock-taking, which is a requirement of the FSDoH's Inventory Management Policy, should also be introduced and adhered to at the hospital.
In line with the literature review, the capacity of the hospital's warehouse needs to be determined so that the space can be better utilised. Properly utilised space, through carefully planned inventory management and distribution operations, is important to ensure a more consistent availabilityof stock. A Warehouse Management System (WMS/computer system) needs to be put in place to get maximum use of the warehouse and to manage inventory better. For the WMS to assist in establishing more effective warehousing operations, the hospital staff should ensure that the following practices are adhered to in the warehouse:
8.2.1 Keep a time schedule for the preparation of clinic orders to ensure the smooth running of operations.
8.2.2 Maximise on the use of space allocated to each store's activity.
8.2.3 Minimise the time taken to carry out the store's operational activities.
8.2.4 Keep track of the location of patients' treatments that are still being prepared and packaged, as well as the location of completed clinic orders.
8.2.5 Promote the order-picking accuracy, that is, the preparation and packaging of patients' treatment and its staging, prior to delivery to the clinics.
Furthermore, the pharmaceutical warehouse needs to ensure the consistent availability of stock. The delivery of the correct quantities is stressed throughout the literature review, and particular emphasis is placed on the fact that the effective management of inventory starts with the receipt of the correct quantities.
8.3. Implement an effective order cycle (replenishment) system
The recommendations to ensure the effective and efficient management of inventory and warehousing are centred on a successful order cycle system. Inventory management can be improved by placing, processing and preparing orders prior to shipment to the various clinics through an effective order cycle system. To facilitate an effective order cycle system, the following parameters should be ensured:
8.3.1 The completion of an order cycle within the stipulated cycle time.
8.3.2 The performance of frequent stock spot-checks at both the hospital and the clinics to ensure sufficient stock levels and order quantities.
8.3.3 Less variability in the length of the order cycles and lead-times to minimise the levels of safety stock being kept at the hospital.
8.3.4 Determining more dependable and consistent lead-times to ensure lower inventory costs, higher product quality, sufficient inventory levels and less stock-outs. Dependable lead-times also reduce errors such as the duplication of order processing and/or the shipment of incorrect quantities.
8.4. More effective and reliable distribution and transportation processes
The Manapo hospital is the distribution centre (DC) that serves as a storage location for ARVs and as a transfer point of treatment to the peripheral clinics in the QwaQwa district. However, the distribution and transportation of ARVs from the hospital as DC should be improved to ensure a less interrupted and more consistent supply of the drugs by increasing the reliability of the transportation system.
Decisions should be made on which transport system, either the in-house or the outsourced transportation mode, will be the dominant format and which will be the back-up or alternative format for both the warehouse (main) deliveries and urgent deliveries. The routing and scheduling, for example, as well as the dates and times the trucks are expected to make deliveries, must be precisely planned and executed to ensure the uninterrupted and consistent delivery of the stock. As part of improved routing and scheduling, the correct shipment documentation must accompany the orders of ARVs transported to the peripheral clinics, so that any form of delay in the timely delivery of the drugs can be reduced or eliminated to avoid stock shortages.
As shown in Table 5, outsourced transport, which is a facility at the warehouse, is recommended for use as the dominant transport for the main deliveries, and the in-house transport used as the dominant transport for urgent warehouse deliveries, main clinic orders and urgent clinic deliveries.
In an effort to improve the delivery of orders, a recommendation is provided in Table 6 to improve the routing and scheduling of ARV delivery from the hospital to the peripheral clinics. A total of ten peripheral clinics are associated with the issue of ARVs to patients in the QwaQwa district. Therefore, as shown in Table 6, the clinics were grouped according to their geographical location so that a single shipment could be carried out to clinics closest to each other.
In terms of Table 6, orders for clinics 1, 2 and 3 are processed in the first week, orders for clinics 4, 5 and 6 are processed in the second week, orders for clinics 7 and 8 are processed in the third week, and orders for clinics 9 and 10 in the fourth week. It should be noted that three of the ten clinics each are allocated to weeks 1 and 2, and that only two clinics each are allocated to weeks 3 and 4. Taking into consideration that weeks 3 and 4 fall within the pay period of the 15th and end of the month, the distribution tasks are kept at a minimum for any uncertainties that may occur such as staff absenteeism.
According to Table 6, clinics submit their monthly orders on Mondays as per the schedule. Once the orders are received, transport is then booked for delivery every Thursday. The hospitals prepare and package patients' treatments on Tuesdays and Wednesdays. On Thursdays, all the necessary shipment documentation is prepared to accompany the treatment for transport to the clinics. Fridays are open for the staff to perform all other ARV-related functions such as stock spot-checks. It is important to emphasise that flexibility should be exercised around the planned schedule without offsetting the complete structure.
8.5. Improved supply chain coordination and overall communication
In order to improve supply chain coordination, the accuracy of information shared between the warehouse, the hospital and the clinics is of the utmost importance as it promotes trust between the supply chain partners and facilitates a good working relationship. The recommendations for supply chain coordination are as follows:
8.5.1 There should be shared sets of objectives and accurate information transferred between the pharmaceutical warehouse, the hospital and the clinics. Therefore, open communication channels with regard to factors such as lead-times, the routing and scheduling of deliveries, as well as fluctuations in orders, should be established to minimise the occurrence of low inventory levels and/or stock-outs experienced by the hospital at times.
8.5.2 Coordination between the flow processes of an order cycle, from the placing of an order by the hospital, to the shipment of the patients' treatment by the hospital to the clinics, is key to successful supply chain operations. This again calls for open and regular communication between all the supply chain partners who are responsible for the various ARV distribution activities.
9. Conclusion
The implementation of an uninterrupted and consistent ARV supply chain is not negotiable as patients' lives are at stake. All the role-players at the provincial regional pharmaceutical warehouse, the Manapo hospital and the peripheral clinics need to take responsibility for implementing a proper supply chain planning and design process; improving inventory management and warehousing practices; implementing more effective and reliable distribution and transportation processes; as well as improving supply chain coordination and overall communication.
Research significance
The results of the article should enable the Manapo and other rural hospitals in South Africa to improve their ARV drug distribution practices and the management of their ARV supply chains so that patients can have uninterrupted access to their monthly supply of ARVs, thereby increasing drug adherence and prolonging lives.
Author contribution
• Mrs Mokheseng was the Master's student who undertook the study on which the article is based.
• Prof Gideon S. Horn was the study leader.
• Mrs Aileen G. Klopper was the co-study leader.
REFERENCES
Babbie, E., & Mouton, J. (2001). The practice of social research (2nd ed.). Cape Town: Oxford University Press. [ Links ]
Ballou, R. H. (2004). Business logistics/supply chain management: Planning, organizing, and controlling the supply chain (5th ed.). New Jersey: Pearson Prentice Hall. [ Links ]
Bowersox, D. J., Closs, D. J., Cooper, M. B., & Bowersox, J. C. (2013). Supply chain logistics management (4th ed.). New York: McGraw- Hill. [ Links ]
Chandrasekaran, N. (2012). Supply chain management. New Delhi: Oxford University Press. [ Links ]
Chopra, S., & Meindl, P. (2010). Supply chain management: Strategy, planning and operation (4th ed.). Boston: Pearson Hall. [ Links ]
Coyle, J. J., Langley, C. J., Jr., Novack, R. A., & Gibson, B. J. (2013). Managing supply chains: A logistics approach (9th ed.). Ohio: South-Western. [ Links ]
De Vos, A. S., Strydom, H., Fouche, C. B., & Delport, C. S. L. (2011). Research at grass roots: For the social sciences and human service professions (4th ed.). Pretoria: Van Schaik. [ Links ]
Fawcett, S. E., Ellram, L. M., & Ogden, J. A. (2007). Supply chain management: From vision to implementation. New Jersey: Pearson Prentice Hall. [ Links ]
Free State Department of Health (FSDoH) Inventory Management Policy. (2012). Inventory management policy. Free State: Government Printer, 10(1), 13-15. [ Links ]
Hugos, M. H. (2011). Essentials of supply chain management (3rd ed.). New Jersey: John Wiley & Sons. [ Links ]
Ireland, R. K., & Crum, C. (2005). Supply chain Collaboration: How to implement CPFR® and other best collaborative practices. Florida: J. Ross. [ Links ]
Jacobs, F. R., Berry, W. L., Whybark, D. C., & Vollmann, T. E. (2011). Manufacturing planning and control for supply chain management (6th ed.). New York: McGraw-Hill/Irwin. [ Links ]
Johnson, P. F., Leenders, M. R., & Flynn, A. E. (2011). Purchasing and supply management (14th ed.). New York: McGraw-Hill. [ Links ]
Management Sciences for Health. (2006). HIV/AIDS pharmaceutical management Training: Respondents guide. United States Agency International Development (USAID). Unpublished manuscript. [ Links ]
Mori, A. T., & Owenya, J. (2014). Stock-outs of antiretroviral drugs and coping strategies used to prevent changes in treatment regimens in the Kinondoni district, Tanzania: A cross- sectional study. Journal of Pharmaceutical Policy and Practice., 7(3), 1-12. [ Links ]
National Department of Health (NDoH). (2003). Operational plan for comprehensive HIV and AIDS care, management and treatment for South Africa [Pdf]. Pretoria: National Department of Health. Available at: http://www.hsph.harvard.edu/population/aids/southafrica.aids.03.pdf (Accessed 27 November 2013). [ Links ]
Neuman, W. L. (2011). Social research methods: Qualitative and quantitative approaches (7th ed.). Boston: Allyn & Bacon. [ Links ]
Nsimba, S. E. D., & Irundi, H. C. (2010). Barriers to ARV adherence among HIV/AIDS positive persons taking anti-retroviral therapy in two Tanzanian regions 8-12 months after program initiation. Journal of AIDS & Clinical Research,14-28. December 15. [ Links ]
Pienaar, W. J., & Vogt, J. J. (2012). Business logistics management: A value chain perspective (4th ed.). Cape Town: Oxford University Press. [ Links ]
Rang, H. P., Dale, M. M., Ritter, J. M., & Moore, P. K. (2003). Pharmacology (5th ed.). London: Churchill Livingstone. [ Links ]
Sande, M. A., & Volberding, P. A. (1999). The medical management of AIDS (6th ed.). Philadelphia: Saunders. [ Links ]
Schouten, E. J. (2011). Antiretroviral drug supply challenges in the era of scaling up ART in Malawi. Journal ofthe International AIDS Society,1-15. [ Links ]
Verma, R., & Boyer, K. K. (2010). Operations & supply chain management: World class theory and practice. Australia: South- Western/Cengage learning. [ Links ]
Welman, J. C., Kruger, F., & Mitchell, B. (2005). Research methodology (3rd ed.). Cape Town: Oxford University Press. [ Links ]
Wisner, J. D., Keah-Choon, T., & Leong, G. K. (2012). Supply chain management: A balanced approach (3rd ed.). Ohio: South- Western/Cengage learning. [ Links ]
Received 8 September 2015
Accepted 4 November 2016
* Corresponding author.E-mail addresses: mamolisem@gmail.com (M. Mokheseng), Gideon.horn@nmmu.ac.za (G.S. Horn), Gail.klopper@nmmu.ac.za (A.G. Klopper).
1 All three authors are affiliated to the Nelson Mandela Metropolitan University in Port Elizabeth.














