Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Journal of Child Health
versão On-line ISSN 1999-7671
versão impressa ISSN 1994-3032
S. Afr. j. child health vol.17 no.3 Pretoria Set. 2023
http://dx.doi.org/10.7196/sajch.2023.v17i3.1969
RESEARCH
Nucleated red blood cells in neonates with hypoxic ischaemic encephalopathy treated with hypothermia: A worthwhile prognostic biomarker for clinicians in LMIC?
L MfingwanaI, II; J van ZylIII; J SmithIV, V; M RutherfordVI, VII; G T J KaliVIII, IX
IMPhil, Cert Paed Pulmonol; Department of Paediatrics and Child Health, Faculty of Medicine and Health Sciences, Stellenbosch University, Cape Town, South Africa
IIMPhil, Cert Paed Pulmonol; Tygerberg Hospital, Cape Town, South Africa
IIIMB ChB; Tygerberg Hospital, Cape Town, South Africa
IVMMed, PhD; Department of Paediatrics and Child Health, Faculty of Medicine and Health Sciences, Stellenbosch University, Cape Town, South Africa
VMMed, PhD; Tygerberg Hospital, Cape Town, South Africa
VIMB ChB, MRCP, FRCPH, FRCR, MD; Department of Paediatrics and Child Health, Faculty of Medicine and Health Sciences, Stellenbosch University, Cape Town, South Africa
VIIMB ChB, MRCP, FRCPH, FRCR, MD; Division of Bioengineering and Imaging Sciences, Department of Perinatal Imaging and Health, Centre for the Developing Brain, St Thomas' Hospital, King's College, London, UK
VIIIFCPaed, Cert Neonatol, PhD; Department of Paediatrics and Child Health, Faculty of Medicine and Health Sciences, Stellenbosch University, Cape Town, South Africa
IXFCPaed, Cert Neonatol, PhD; Tygerberg Hospital, Cape Town, South Africa
ABSTRACT
BACKGROUND: Neonatal hypoxic ischaemic encephalopathy (HIE) is a leading cause of term neonatal death worldwide, with a higher incidence in low- to middle-income settings
OBJECTIVE: To investigate whether nucleated red blood cell (nRBC) counts could predict severity of HIE and outcomes in term neonates treated with therapeutic hypothermia (TH
METHODS: We conducted a retrospective sub-study at Tygerberg Hospital in Cape Town, South Africa. The review included all cooled neonates' clinical records and blood samples from a National Health Laboratory Services database. One experienced neurodevelopmental expert assessed patients over a period of 12 months
RESULTS: Twenty-five files out of a total of 100 were excluded owing to missing data. In accordance with the Thompson HIE score, the cohort was classified as mild (56%), moderate (27%), and severe (17%). All included patients (n=75) had full blood counts within 6 hours of delivery. nRBC were detected in 52% of the samples. There was no correlation between nRBC category and HIE severity (p=0.265). Raised nRBCs (>30 cells/100 white blood cells (WBCs)) were more frequent in infants who died than in those who survived (p=0.008). Infants with nRBC counts >30 cells/100 WBCs had an increased likelihood of having cerebral palsy or impaired neurodevelopment (p=0.013
CONCLUSION: The study demonstrated a significant association between an early increase in nRBC counts in HIE infants treated with TH, and both short- and long-term outcomes. A larger multicentre study is required to better understand the relationship between nRBC counts and HIE in the era of cooling in our local setting
Neonatal hypoxic ischaemic encephalopathy (HIE) is a leading cause of term neonatal morbidity and mortality worldwide, with a higher incidence in low- and middle income countries (LMICs).[1] To date, therapeutic hypothermia (TH) is the only intervention shown to be effective in preventing adverse outcomes in neonatal HIE.[2] We previously demonstrated its safety and efficacy in a sub-Saharan setting.[3]
Current practice recommends clinical assessment of the neonate combined with evidence offetal compromise from cardiotocography (CTG), umbilical cord or infant blood gas pH, persistently low Apgar scores and a need for prolonged resuscitation to confirm a diagnosis of probable HIE.[4] After initiating TH within 6 hours of birth and continuing with it for 72 hours, supportive management of seizures, fluid balance, and haematological and cardiovascular anomalies is critical for achieving optimal outcomes.[4]
In LMIC settings, CTGs, blood gas machines and amplitude-integrated electroencephalography (aEEG) equipment may not be universally available, particularly at district-level hospitals in South Africa (SA), where a significant number of asphyxiated infants are born. Research is therefore necessary to identify simple markers to reliably distinguish infants exposed to intrapartum hypoxia who would develop significant HIE that may respond to TH from those in whom therapy would be pointless. In recent years, interest in the relationship between neonatal nucleated red blood cells (nRBCs) and perinatal asphyxia has increased.
In utero subacute or chronic hypoxia stimulates fetal erythropoiesis, resulting in an increased release of immature red blood cells into the fetal circulation. Reports suggest that it takes 4 to 5 hours from the onset of hypoxia to increase levels of erythropoietin, and then ~24 hours for nRBCs to appear on a peripheral blood film.[5] However, acute stress can lead to release of marrow stores of nRBCs in a shorter period.[6]
Although nRBCs can be found in neonatal blood following low-risk pregnancy and delivery, the nRBCs/100 white blood cells (WBCs) seldomly exceed 10 cells/100 WBCs.[7] Several factors that appear to increase nRBCs include asphyxia, prematurity, Rh-sensitisation, maternal diabetes mellitus and fetal growth restriction.[8,9] nRBCs, in combination with other biomarkers, may be useful in identifying those infants where there is no clear sentinel event.
Several published studies from both low- and high-income countries have reported an association between the number of neonatal nRBC and perinatal asphyxia,[7] demonstrating a significant association between the absence of fetal heart rate accelerations and elevated nRBCs in asphyxiated neonates,[10] as well as a correlation between increased nRBC counts, degree of acidosis and low Apgar scores.[11]
Tungalag et al.[8] found that increased cord blood nRBCs in asphyxiated infants predicted the occurrence and severity of HIE, and proposed its use as an inexpensive assessment tool in settings where no facilities for blood pH measurements exist.
Korst et al.[12] described nRBC >10 cells/100 WBCs as a marker for fetal asphyxia. Li et al.[13] concluded that both the nRBCs/100 WBCs and absolute nRBC count taken within 6 hours post birth in asphyxiated infants had a significant association with abnormal 2-week MRI (p=0.006) and 2-year neurodevelopmental sequelae (p=0.021), in both hypothermic and normothermic infants. The association was stronger for cerebral injury on MRI .
Most hospitals in SA, including district hospitals, have access to functional laboratory services via the National Health Laboratory Services (NHLS). Access to a full blood count (FBC) is therefore widely available. However, the ability of hospitals at different levels to identify nRBCs varies, with some only having the ability to identify the presence of nRBCs and requiring referral of the peripheral smear to a tertiary laboratory for analysis and quantification of nRBCs. While the ability to quantify nRBCs should not place additional demands on resources, the turn-around time varies from a minimum of 30 to 60 minutes to several hours depending on various logistical factors.
The purpose of the present study was to determine whether nRBC levels could be used to predict the severity of HIE in term neonates at a tertiary referral hospital in SA, and to determine whether nRBC levels could be used to predict both short-term (mortality) and longterm neurodevelopmental outcomes in HIE neonates treated with TH to identify those who might benefit from additional neuroprotection.
The aims of the study were to: (i) report the nRBC count in the first 6 hours in a cohort of neonates with HIE treated with therapeutic hypothermia (TH) at Tygerberg Hospital (TBH) between 2008 and 2011; (ii) determine the association between nRBC counts and severity of HIE as determined by the Thompson encephalopathy score; and (iii) determine the association between nRBC counts and mortality/neurodevelopmental outcome at 12 months.
Ethics
Ethics approval was obtained from the Human Research Ethics Committee (HREC) of Stellenbosch University. As this was a retrospective analysis of routinely collected data for which ethical permission had previously been obtained and no new patient interventions were to be performed, a waiver of individual informed consent was granted by Stellenbosch University's HREC (ref. no. S17/03/066). Permission to conduct the study was also obtained from the management of TBH.
Methods
This was a retrospective sub-study of a larger study that investigated TH in term neonates diagnosed with HIE.
The study was conducted at TBH, a tertiary hospital in Cape Town. Infants who were born at TBH (or referring hospitals) between November 2008 and November 2011 and who were admitted to the TBH neonatal intensive care unit (NICU) for cooling were included in the study based on the following criteria:
Infants >36 completed weeks' gestation with at least one of the following: Apgar <7 at 10 minutes of age; continued need for active resuscitation at 10 minutes of age; acidosis within 60 minutes of birth, defined as umbilical cord, arterial or capillary blood pH <7 or base deficit > 16.
Infants who met the aforementioned criteria were further evaluated on admission and every 24 hours for encephalopathy using the Thompson score.[14]
Exclusion criteria included neonates that did not meet cooling criteria, i.e.: presence of major congenital abnormalities; <36 weeks' gestation; birthweight <1.8 kg; or if they were >6 hours old at the time of assessment.
On admission to the NICU, demographic and clinical information such as birth weight, gestational age, sex, mode of delivery and Apgar scores were collected. Additionally, the Thompson score, blood gas analysis and lactate levels were documented and patients were classified into mild (HIE <10), moderate (HIE 11 - 14), and severe HIE (>15) based on their Thompson scores.
Patients were cooled for 72 hours with total body hypothermia using the Tecotherm TSmed 200N system (Tec Com Medizintechnik GmbH, Germany) and then gradually re-warmed over a minimum of 6 hours. Blood samples, including those for nRBC count determination, were collected within 6 hours of birth and repeated daily.
To determine individual nRBC data, the NHLS TrackCare database system was accessed. Blood samples were analysed retrospectively. nRBC counts were reported as nRBCs/100 WBCs. An automated system was used for detecting presence of and a threshold nucleated red blood cell level of 30 nRBCs/100 WBCs, using the Siemens Advia machine (Siemens Healthcare GmbH, Germany).
If the initial machine report was <30 nRBCs/100 WBCs, quantification of the exact number by microscopy was not performed. If >30 nRBCs/100 WBCs were reported, a smear was done by the haematologist for manual quantification and description of the nRBCs. This is the current operational protocol in the NHLS laboratory at TBH, which considers the laboratory's limited human resource capacity.
The first nRBC samples taken at the time of admission for cooling were used and compared with the first Thompson score after admission to the NICU (<6 hours of age). nRBC counts were stratified into three groups: nRBC = 0 (no nRBC detected by machine); nRBC <30 cells/100 WBCs (nRBC detected by machine but <30 cells), and nRBC >30 cells/100 WBCs.
The infants were transferred to the neonatal ward following successful TH and re-warming.
Long-term neurological assessment
Patients were assessed by a single neurodevelopmental specialist at the neonatal high-risk clinic at 3 and 12 months of age using a combination of neurological examinations with Amiel-Tison test positions[15] and the Bayley Scales of Infant Development Third Edition Screening Test (Bayley-111 screening test). Infants were considered severely impaired if they had motor dysfunction consistent with cerebral palsy, were deaf or blind or had significant developmental delay clinically and as defined by the risk category on the Bayley-111 screening test.
Statistical analysis
All relevant data were transferred to Microsoft Excel spread sheets (Microsoft Corp., USA). Patient identifiers and data variables were numerically coded and were analysed using Statistica software (StatSoft (Europe) GmbH, Germany). Descriptive data were reported as mean values with standard deviations (SDs), if normally distributed, and as medians with-ranges if the data were not normally distributed.
The association between nRBC proportion and the following outcomes were determined: severity of HIE as determined by maximum Thompson score on admission, neonatal death (short-term) and long-term neurological outcomes. Bivariate data were assessed using chi-squared test and Fisher's exact test for categorical variables. A p-value <0.05 was considered statistically significant.
Results
One hundred patient folders were reviewed for inclusion and 25 folders were excluded owing to significant missing data (Fig. 1).
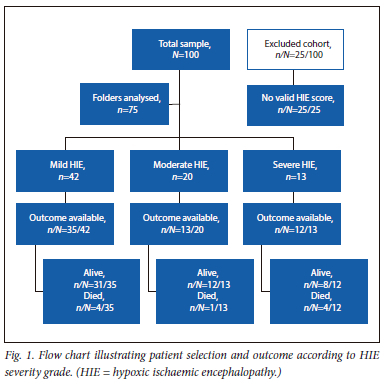
Both included and excluded patients' clinical characteristics were compared. The baseline characteristics and outcome of the 25 excluded patients did not differ significantly from those included in the analysis (Table 1). Ten of the excluded infants had nRBC done but no clinical outcome information. Fifteen patients had outcome information available, but no HIE grading. Of these 7 died, 4 had cerebral palsy or neurodevelopmental impairment and 4 were normal.
The majority of the infant population was male. The median birthweight of the included cohort was 3 117 g, and the median gestational age was 38.6 weeks. Fifty-six percent of included infants had mild HIE (Fig. 1). Only 36% of the cohort had documented outcomes and 12% of them were fatal (Table 1).
All included patients (n=75) had FBC done within the stipulated first 6 hours of delivery. nRBCs were detected in 52% (n=39) of the samples. nRBC distribution in the 75 samples is shown in Table 2.
Half of the neonates (21/42; 50%) with mild HIE had no nRBCs, compared with 55% (n/N=11/20) of neonates with moderate HIE and 31% (n/N=4/13) of those with severe HIE. The median nRBC value among infants with quantified nRBC (>30 cells/100 WBCs) demonstrated a rise from mild to severe HIE (Table 2), although there was no significant correlation between the category of nRBC (< or >30/100) and HIE severity (p=0.265).
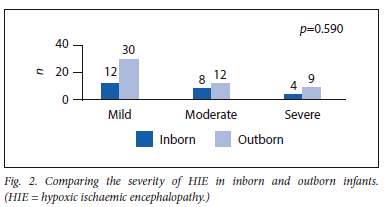
Twenty-four (32%) infants were inborn, while 51 (68%) were referred to TBH for treatment. Inborn infants were stratified as follows: mild (50%; n/N=12/24); moderate (33%; n/N =8/24); and severe HIE (17%; n/N=4/24). Outborn infants who were referred to TBH for cooling were similary stratified: mild (58.8%; n/N=30/51); moderate (23.5%; n/N=12/51) and severe HIE (17.7%; n=9/51). There was no statistically significant difference between the proportions of moderate to severe HIE in inborn and outborn infants (n/N=12/24 inborn v. n/N=21/51 outborn; p=0.473) (Fig. 2).
A comparison of the outcomes of the infants revealed that a higher nRBC count in the first 6 hours after delivery was associated with a worse outcome. Infants with nRBC counts >30 cells/100 WBCs at birth had an increased risk of cerebral palsy (CP) or impaired neurodevelopment at 1 year (p=0.013) and mortality (p=0.003) (Table 3).
Discussion
The present study demonstrated a significant relationship between an early significant increase in nRBCs (>30 cells/100 WBCs) in asphyxiated infants treated with TH, and both neonatal death and long-term neurodevelopmental impairment, but not with severity of HIE. We also found an association between low nRBC count (<30 cells/100 WBCs) and normal outcomes at 1 year.
We sought to determine whether we could define a threshold nRBC level that could be a useful predictive marker for infants who would develop moderate to severe HIE and qualify for cooling and those who would have a poor outcome, in a resource-constrained setting such as ours, where the majority of infants are referred for cooling. Our data were consistent with previously published literature that nRBCs can be used as a surrogate marker of birth asphyxia and predictor of long-term neurological outcome.
The presence of raised nRBC in all grades of HIE (Table 2) was comparable with the findings of Korst et al.,[12] who described nRBC counts of >10 cells/100 WBCs as a marker for fetal asphyxia.
The lack of significant association of nRBC counts >30 cells/100 WBCs with HIE severity may be related to the timing of clinical assessment, as it is known that HIE is an evolving process during its early stages.
We found an nRBC count >30 cells/100 WBCs to be a discriminator of outcome. Nine patients demised in the neonatal period in this study. A larger proportion of infants with nRBC counts of >30 cells/100 WBCs died compared with those with nRBC counts of <30 cells/100 WBC ((50% v. 10%; p=0.003). This outcome further supports available literature on the topic indicating that the higher the nRBC count, the more severe the short-term adverse outcomes, including instances where early-onset seizures are used as a marker of neurological injury.'161 Similarly, in the present study, all of the infants with nRBC counts >30 cells/100 WBCs who were assessed had poor neurodevelopmental outcomes.
Our findings of an association between raised nRBC counts and poor outcomes are similar to those of studies from other LMICs. Li et al.'131 concluded that both the nRBC count and absolute nRBC count taken within 6 hours of birth in asphyxiated infants had a significant predictive value for abnormal MRI findings (p=0.006) and 2-year neurodevelopmental impairment (p=0.021).
Boskabadi et al.[7] described a significant relationship between increased nRBC and early outcomes with HIE. Their mean (SD) nRBC per 100 WBC level in umbilical cord blood of newborns with HIE was 16.5 (6.4) per 100 WBCs, with a range of 3 - 25 nRBCs/100 WBCs, whereas it was 8.6 (7.01) nRBCs/100 WBCs in their control group, with a range of 1 - 26 nRBCs/100 WBCs. This difference was statistically significant (p<0.001).
Study strengths and limitations
The strengths of our study include the fact that data on cooled infants were collected prospectively, and all infants were routinely followed up for 1 year by a single experienced neurodevelopmental expert. Their outcomes were assessed using a standardised neurodevelopmental assessment tool (Bayley-111 screening test).
The findings of our study contribute to local knowledge and the growing interest in the potential association between nRBC, intrapartum hypoxia and its short- and long-term outcomes. To the best of our knowledge, it was the first such study to be conducted in the sub-Saharan region.
A limitation of our study was that we could not answer the primary questions of the study to the extent envisaged because prospectively collected data were retrospectively analysed. The post hoc nature of the study meant that we could not pre-specify how we required the nRBC to be analysed, and detailed analysis of nRBC counts of <30 cells/100 WBC was not available at the time the study was conducted. A further limitation was the small size of the final study sample.
Conclusion
The present study showed that nRBC levels >30 cells/100 WBCs had a significant correlation with early mortality and neurodevelopmental impairment at 1 year of age in this cohort. To gain a better understanding of the association between nRBC and HIE in the era of cooling in our setting, there is a need for a larger multicentre study which should include a description of normal term infant nRBC levels in uncomplicated pregnancies and seek to further delineate the association of nRBC with clinical outcomes before it can be applied to select patients for neuroprotective therapies.
Declaration. This manuscript was submitted in partial fulfilment of the requirements for completion of LM's MMed degree at Stellenbosch University.
Acknowledgements. LM is deeply grateful to the late Abongile Jojo (May 2023) who assisted with statistical analyses, all the staff at TBH who looked after the infants, and the Tygerberg NHLS laboratory staff who assisted with accessing the results.
Author contributions. LM: conceptualisation, data collection and analysis, as well as preparation of the manuscript. GTJK: conceptualisation, research supervision, data analysis and reviewing of manuscript. JvZ: neurodevelopmental assessments of infants and reviewing the manuscript. JS: conceptualisation and reviewing the manuscript. MR: reviewing of data and manuscript.
Funding. None.
Conflicts of interest. None.
References
1. Lawn JE, Cousens S, Zupan J. 4 million neonatal deaths: When ? Where? Why? Lancet 2005;365(9462):891-900. https://doi.org/10.1016/s0140-6736(05)71048-5 [ Links ]
2. Jacobs SE, Berg M, Hunt R, Tarnow-Mordi WO, Inder TE, Davis PG. Cooling for newborns with hypoxic ischaemic encephalopathy. Cochrane Database Syst Rev 2013;2013(1):CD003311. https://doi.org/10.1002/14651858.CD003311.pub3 [ Links ]
3. Kali GTJ, Martinez-Biarge M, van Zyl J, Smith J, Rutherford M. Management of therapeutic hypothermia for neonatal hypoxic ischaemic encephalopathy in a tertiary centre in South Africa. Arch Dis Child Fetal Neonatal Ed 2015;100(6):F519-F523. https://doi.org/10.1136/archdischild-2015-308398 [ Links ]
4. Ghosh B, Mittal S, Kumar S, Dadhwal V. Prediction of perinatal asphyxia with nucleated red blood cells in cord blood of newborns. Int J Gynecol Obstet 2003;81(3):267-271. [ Links ]
5. Christensen RD, Lambert DK, Richards DS. Estimating the nucleated red blood cell 'emergence time' in neonates. J Perinatol 2014;34(2):116-119. https://doi.org/10.1016/s0020-7292(03)00124-3 [ Links ]
6. Hermansen MC. Nucleated red blood cells in the fetus and newborn. Arch Dis Child Fetal Neonatal Ed 2001;84(3):F211-F215. https://doi.org/10.1136/fn.84.3.f211 [ Links ]
7. Boskabadi H, Maamouri G, Sadeghian MH, et al. Early diagnosis of perinatal asphyxia by nucleated red blood cell count: A case-control study. Arch Iran Med 2010;13(4):275-281. [ Links ]
8. Tungalag L, Gerelmaa Z. Nucleated red blood cell counts in asphyxiated newborns. Open Sci J Clin Med 2014;2(1):33-38. [ Links ]
9. McCarthy J, Capullari T, Thompson Z, Zhu Y, Spellacy W. Umbilical cord nucleated red blood cell counts: Normal values and the effect of labor. J Perinatol 2006;26:89-92. https://doi.org/10.1038/sj.jp.7211437 [ Links ]
10. Ferber A, Grassi A, Akyol D, O'Reilly-Green C, Divon MY. The association of fetal heart rate patterns with nucleated red blood cell counts at birth. Am J Obstet Gynecol 2003;188(5):1228-1230. https://doi.org/10.1067/mob.2003.352 [ Links ]
11. Hanlon-Lundberg KM, Kirby RS. Nucleated red blood cells as a marker of acidemia in term neonates. Am J Obstet Gynecol 1999;181(1):196-201. https://doi.org/10.1016/s0002-9378(99)70459-x [ Links ]
12. Korst LM, Phelan JP, Ahn MO, Martin GI. Nucleated red blood cell: An update on the marker for fetal asphyxia. AJOG 1996;175(4):843-846. https://doi.org/10.1016/s0002-9378(96)80010-x [ Links ]
13. Li J, Kobata K, Kamei Y, et al. Nucleated red blood cell counts: An early predictor of brain injury and 2-year outcome in neonates with hypoxic-ischemic encephalopathy in the era of cooling-based treatment. Brain Dev 2014;36(6):472-478. https://doi.org/10.1016/j.braindev.2013.06.012 [ Links ]
14. Thompson CM, Puterman AS, Linley LL, et al. The value of a scoring system for hypoxic ischaemic encephalopathy in predicting neurodevelopmental outcome. Acta Paediatr 1997;86(7):757-761. https://doi.org/10.1111/j.1651-2227.1997.tb08581.x [ Links ]
15. Gosselin J, Gahagan S, Amiel-Tison C. The Amiel-Tison neurological assessment at term: Conceptual and methodological continuity in the course of follow-up. Ment Retard Dev Disabil Res Rev 2005;11(1):34-51. https://doi.org/10.1002/mrdd.20049 [ Links ]
16. Blackwell SC, Refuerzo JS, Wolfe HM, et al. The relationship between nucleated red blood cell counts and early-onset neonatal seizures. Am J Obstet Gynecol 2000;182(6):1452-1457. https://doi.org/10.1067/mob.2000.106854 [ Links ]
 Correspondence:
Correspondence:
L Mfingwana
lunga@paedschest.co.za
Accepted 17 May 2023














