Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Child Health
On-line version ISSN 1999-7671
Print version ISSN 1994-3032
S. Afr. j. child health vol.16 n.1 Pretoria Apr. 2022
http://dx.doi.org/10.7196/sajch.2022.v16.i1.1825
RESEARCH
The epidemiology and treatment outcomes of clubfoot in a South African tertiary academic hospital
M ThiartI; C FennII; J du ToitIII; M BurgerIV
IMB ChB, FCOrth (SA); Division of Surgical Sciences, Department of Orthopaedics, Faculty of Medicine and Health Sciences, Stellenbosch University and Tygerberg Hospital, Cape Town, South Africa
IIMB ChB; Division of Surgical Sciences, Department of Orthopaedics, Faculty of Medicine and Health Sciences, Stellenbosch University and Tygerberg Hospital, Cape Town, South Africa
IIIMB ChB; Division of Surgical Sciences, Department of Orthopaedics, Faculty of Medicine and Health Sciences, Stellenbosch University and Tygerberg Hospital, Cape Town, South Africa
IVMSc, PhD; Division of Surgical Sciences, Department of Orthopaedics, Faculty of Medicine and Health Sciences, Stellenbosch University and Tygerberg Hospital, Cape Town, South Africa
ABSTRACT
BACKGROUND: Clubfoot is a common congenital condition with a global incidence estimated at 1 in 1 000 live births. There is a paucity of information regarding the epidemiology and incidence of clubfoot in South Africa
OBJECTIVES: To describe the epidemiology, clinical characteristics, and the treatment outcomes of children with clubfoot who were treated at a tertiary hospital in South Africa. A secondary objective was to determine the incidence rate of clubfoot in our direct catchment area (within 40 km from Tygerberg hospital) for the study period
METHODS: We conducted a retrospective cohort study of all records of children with clubfoot from 2014 - 2018. Demographic and clinical information, treatment progression and short-term outcomes including early relapse were collected. A subset of the total cohort, including only clubfoot patients from the direct catchment area of the hospital, together with the number of live births in the direct catchment area, was utilised to calculate the incidence rate of clubfoot in our setting
RESULTS: A total of 197 patients were included over a 60-month period. Two-thirds of the patients were male (63.5%; n=125). A positive family history was reported in 12.2% (n=24) of cases, and 88.8% (n=175) and 11.2% (n=22) of patients were diagnosed as having idiopathic clubfoot or syndromic clubfoot, respectively. Relapsed clubfoot was diagnosed in 22.9% (n=40) and 22.7% (n=5) of patients with idiopathic and syndromic clubfoot, respectively. The overall incidence rate of idiopathic clubfoot in our direct catchment area was 1.02 per 1 000 live births
CONCLUSION: The epidemiology, treatment outcomes and incidence rates observed at our institution are like those reported globally. We report a low positive family history and relapse rate, which could be under reported and should be the focus of future investigations
Clubfoot (also called congenital talipes equinovarus) is one of the most common musculoskeletal congenital conditions that causes impairment in mobility, globally.[1] A clubfoot is characterised by a rigid varus and equinus deformity of hindfoot, a cavus midfoot and a forefoot in supination and adduction.[1-3] This condition can either be idiopathic (80% of cases) with a 25% familial occurrence or associated with other conditions (termed syndromic clubfoot), including myelomeningocele, amniotic band syndrome and arthrogryposis.[4]
Clubfoot can be treated successfully using the current gold standard, Ponseti treatment,[5-8] which entails weekly casting of the affected foot to manipulate and stretch the foot into the correct position. Ponseti treatment should commence as soon as possible after birth as the bones are cartilaginous and the soft tissues are more amenable to stretching.[9] Subsequently, late presentation leads to increasing difficulty in the treatment process due to the resistance of ossified bones and contracted fibrotic soft tissue. Ponseti treatment has been shown to be resource appropriate in the developing world[10] with unsuccessful treatment often being reported to be as a result of non-compliance with the schedule.[11]
The global birth prevalence of clubfoot is estimated to be 1 in 1 000 live births,[4,7,8,12] with males being more affected than females at a 2:1 ratio. It is estimated that 80% of children born with clubfoot live in developing countries.[1,7,8] Kromberg et al.[13]reported the incidence rate to be between 1.55 and 6.93 per 1 000 live births in South African (SA) infants, differing between black and white children. However, the authors admit to a high risk of bias in these figures.[13] Du Toit et al.[14]tried to report the prevalence in rural Zululand in 1971, but reported that the follow-up was poor and that data collection was frustrating. This historical survey highlighted that children present at all ages and that follow-up is unreliable.[14] Due to the paucity of recent SA literature on clubfoot, the local burden of clubfoot on the healthcare system, its epidemiology and incidence of this condition are unknown.
The aim of this study was therefore to describe the epidemiology, clinical characteristics, and the early treatment outcomes of children with clubfoot, who are treated at a tertiary hospital in SA. A secondary objective was to determine the incidence rate of clubfoot in our direct catchment area (within 40 km) for the study period.
Methods
We conducted a retrospective review of our academic institution's clinical records from 2014 - 2018 of all clubfoot children, including idiopathic and syndromic/neuropathic clubfeet. Children diagnosed with postural clubfoot as well those diagnosed with cerebral palsy were excluded. Information related to patient demographics such as age at first presentation, sex, type of clubfoot and address was collected. The distance to the clinic was extrapolated from the reported address, and this information was later utilised to identify a subset of the total cohort to increase the accuracy of the incidence rate calculation, as described later. Family history of clubfoot was noted together with any associated physical abnormalities.
Information related to treatment, treatment progression and outcome was recorded. This included the age at each visit, the treatment phase, the documented Pirani score together with range of motion (abduction and dorsiflexion), tenotomy requirements and other clinical notes. Treatment progression and outcomes are reported for idiopathic and syndromic clubfoot. In the case of patients with relapsed clubfoot, the presence of non-compliance was recorded if reported.
Data was analysed using Statistica v13.5 (TIBCO software, USA) and is presented as means ± standard deviation (if normally distributed), medians with interquartile ranges (IQR) (if not normally distributed) or as frequencies and counts, as appropriate. To calculate the incidence rate of clubfoot at our setting, the number of live births in our institution's direct catchment area for the study period was obtained from local government. All patients within our direct catchment area (within a 40 km radius) were included in the calculation. The crude incidence rate was calculated for the study period with the following formula:
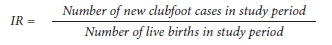
Results
A total of 197 patient were included over the 60-month study period. Two-thirds (63.5%; n=125) of the patients were male. The median (IQR) age at presentation was 5 (2.0 - 12.0) weeks, with 88.8% (n=175) of patients diagnosed as having idiopathic clubfoot and 12.2% (n=24) reporting a positive family history. Although patients had to travel a median of 20.9 km, the distance patients had to travel to attend the clinic ranged from 2.1 - 325 km, with some patients from outlying areas travelling to our clinic for treatment (Table 1).
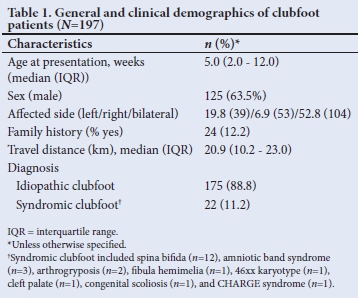
The mean Pirani score at presentation was similar between idiopathic and syndromic clubfeet (Table 2), with idiopathic clubfoot requiring a median (IQR) of 6 (4 -9) casts compared with syndromic clubfoot, which required a median (IQR) of 7 (4 - 11) casts before commencing the next phase of treatment (Table 2). The maximum number of casts required for a single patient with idiopathic clubfoot was 35, and 16 for syndromic clubfoot.

A total of 22.9% (n=40) of idiopathic clubfoot patients relapsed during the maintenance phase, of whom 20.6% underwent repeat Ponseti serial manipulation and casting and 8.6% had a repeat Achilles tenotomy (Table 3). All syndromic patients who relapsed during the maintenance phase (22.7%) underwent repeat Ponseti serial manipulation and casting as well as Achilles tenotomies (Table 3).
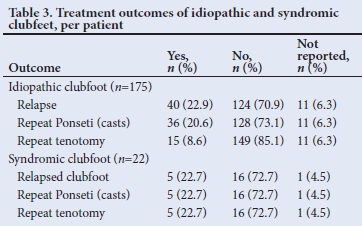
When combining all patients who had relapsed clubfoot (idiopathic clubfoot (n=40) and syndromic clubfoot (n=5)), compliance with the treatment regimen was problematic in 22.2% (n=10) of patients (data not shown). Compliance issues, as reported in clinical notes, included not attending follow-up visits or not complying with bracing instructions.
During the 60-month study period, the total number of live births in our drainage area was 159 348 ('Access to Info' Birth statistics). A total of 162 patients with idiopathic clubfoot resided within a radius of 40 km from the institution. Therefore, the overall incidence rate of idiopathic clubfoot was calculated to be 1.02 per 1 000 live births over the 5-year study period.
Discussion
The aim of the present study was to describe the epidemiology and early treatment outcomes of clubfoot patients treated at a tertiary hospital in SA. Although clubfoot is commonly seen in orthopaedic clinics in SA, there is limited literature providing epidemiological and treatment outcome information regarding clubfoot.
The first main finding of the present study was that the demographic and clinical characteristics of our patients are largely in agreement with the reported global literature, with similar frequencies of male patients (63.5%), bilaterally affected feet (52.8%) and idiopathic clubfeet (88.8%) observed in our setting. A study by Firth et al.[15] reported similar results in the private sector, with 62% male prevalence, 17% syndromic and a right foot predominance at 55%.
The prevalence of syndromic clubfeet in the present study was 11.2%, which is lower than the globally reported prevalence of 20%.[4] Syndromic clubfeet children in the present study included spina bifida as the most common diagnosis (n=12). Street et al.[l6]also reported similar results, which is unsurprising since SA patients populations are broadly similar, albeit from different geographic regions.
A positive family history of clubfoot is reported in 10.5 - 35% of clubfoot patients, with the lower rates specifically reported in developing countries such as Haiti,[17] Sri Lanka[18] and Zimbabwe.[19] In Caucasian populations, the reported positive family history typically ranges between 24 - 30%,[3] while family histories as high as 54% are reported in Polynesian populations.[3] In the present study, we reported a positive family history in only 12.2% of our patients. A study by Malagelada et al.[5]considered the impact of clubfoot treatment on caregivers and parents, and also reported a low positive family history (10%).[5] Firth et al.[15]reported a 16% positive family history based on their early Ponseti results in a SA clinic. Possible reasons for the apparent low rate of positive family histories in SA includes English not being the first language of most of our patients, leading to possible misinterpretation, and the potential of non-disclosure due to fear of stigmatisation, which is still prevalent in our setting. However, it is worthwhile noting that no instances of siblings presenting with clubfoot were reported in this study.
Various studies have reported the range of casts required to fully correct the clubfoot to be between 3 - 8 casts, [1,5,17,20-22] which is in agreement with the median of 6 or 7 for idiopathic and syndromic clubfoot, respectively, as reported in the present study. A study performed in Bangladesh[10] reported that the use of >10 casts was typically attributed to atypical clubfeet, older neglected clubfeet, incidents where cast breakage was common and in cases where complications such as pressure sores were observed. Firth et al.[15] reported an average of 6.5 casts, similar to our results.
A systematic review in 2018 reported that syndromic clubfeet needed on average more casts, had an increased tenotomy incidence, increased relapse rate and a less successful outcome than idiopathic clubfoot feet.[23] Interestingly, the mean Pirani scores at the time of first presentation were similar between the idiopathic and the syndromic clubfoot patients. In addition, a similar treatment protocol of a median number of 6 - 7 casts was utilised for both idiopathic and syndromic clubfeet. A possible explanation for this observation could however be that no reference to 'the atypical clubfoot' was made in the treatment of any of the patients. The 'atypical clubfoot' typically includes a deep medial crease extending to the lateral side, high cavus and a short hyperextended first ray.[24,25] Most of our syndromic clubfeet patients were children with spina bifida. This condition requires fewer casts to obtain correction when compared with arthrogryposis,[23] and could be another reason why the number of casts to correction were similar between the two groups in our study.
The reported rate of tenotomy requirements during Ponseti treatment ranges from 30 - 98%, globally.[17,26] Only 42.3% and 59.1% of idiopathic and syndromic clubfoot patients, respectively, underwent an Achilles tenotomy. The tenotomy rate of 44.2% (idiopathic and syndromic combined) in our clinic is similar to those reported in Nigeria[20] and Haiti,[17,26,27] both developing countries. In contrast, the national Bangladesh clubfoot study reported a tenotomy rate of 76%[10] while Firth et al.[15] reported a tenotomy rate of 74%. These differences could be attributed to the frequent rotation of orthopaedic registrars, leading to treatment variability. Although this forms part of the daily reality in SA clubfoot clinics, a clear understanding of the underlying pathology, adequate training as well as meticulous casting technique remains vital in the successful treatment of clubfoot.
A total of 22.8% of patients required treatment for early relapse. This is a relatively low frequency when compared with the global data, which report relapse rates ranging from 6.6 - 45%.[26] A possible explanation of this low frequency could be that we report specifically on early relapse (i.e. patients who relapse during the treatment and/or maintenance phase). We did not report on the long-term relapse rate in the present study; therefore, our numbers should be interpreted with caution. Interestingly, Firth et al.[15]also reported recurrence requiring recasting as 23%, which is similar to our results, despite having a higher tenotomy rate than the current series. A study by Avilucea et al.reported tenotomy rates of 93.5% in New Mexico, USA. Furthermore, this study reported a relapse rate of 26% in rural patients.[11] Interestingly, the authors reported that the use of bracing was very different between the rural and urban groups and they speculated that the relapse rate was associated with the distance from the clinic as well as cultural factors.[11]
Similarly, barriers to successful treatment in Uganda have been reported to include the availability of braces, costs linked to travel, overwhelming poverty and lack of spousal support.[28] The situation in Vietnam is similar, with transportation and distance to clinics proving to be consistent, universal problems obscuring the treatment process.[7] SA is no exception, with poverty also being a constant concern in our setting, and although the majority of patients live within 20 km of the clinic, we report cases of patients having to travel >300 km in one direction to obtain treatment. The socioeconomic burden of having a child with clubfoot includes loss of income to attend treatment appointments as well as sympathy from the parents regarding bracing, potentially decreasing compliance. These are valid concerns and realities that need to be addressed in developing world settings, including ours.[7]
The incidence rate of idiopathic clubfoot is variable depending on the geographical area, with a global incidence rate estimated at 0.6 - 1.5 per 1 000 live births.[29] Uganda, a developing country, has a slightly higher reported incidence rate of 1.2 per 1 000 live births.[30] Similar rates have been reported in developed countries, with the UK reporting incidence rate ranging between 0.89 -1.24 per 1 000 live births.[31] It should however be noted that certain populations such as the Maori or other Pacific islanders have a substantially higher incidence rate (6 - 7 per 1 000 live births),[30,31] while the Chinese[1,8] population has a surprising low reported incidence rate of 0.39 per 1 000 live births. The incidence rate observed at our institution falls within the global range, with 1.02 clubfoot cases per 1 000 live births. We were surprised by this given the genetic heterogenicity in SA. However, the incidence rate is similar to areas inhabited by populations of predominantly African and European descent. The higher incidence rates are found in Maori and Pacific islanders' descent, and neither of these groups are prominently represented in SA. These differences highlight the role of genetic factors, which are thought to be involved in the aetiology of clubfoot and requires further investigation.
Study strengths and limitations
The strength of this study is that the results represent the reality of a clubfoot clinic, resulting in a realistic impression. In addition, this is the first study to report the incidence rate of clubfoot in our setting. Considering that our clinic is the only clinic providing treatment for clubfoot in our direct catchment area, the incidence rate is considered an accurate representation of our population.
There are various limitations to this study. The retrospective nature of this study results in areas of missing data and poor records, which made subsequent interpretation difficult. In addition, the Pirani score is a subjective scoring system, which has variable results due to multiple clinicians seeing the patients over the course of their treatment regimen. Similarly, the Ponseti casting technique allows for slight variation between clinicians, which affects the treatment outcomes. Although these are not limitations of this study itself, but rather of the treatment process, these limitations influence the results reported in the present study. Additionally, patients who are lost to follow-up could not be accounted for in the present study and therefore the reported relapse rate might not be accurate. The reasons for relapse were dependant on the details provided in clinical notes. Therefore, the true rate of non-compliance could be much higher than what is reported in the present study, and future prospective research should investigate the reasons for relapse in the SA population.
Conclusion
In conclusion, we report that the epidemiology, early treatment outcomes and incidence rates observed at our institution are similar to those reported globally. However, we report a low frequency of positive family history as well as a low relapse rate, which could potentially be underreported and should be the focus of future studies.
Declaration. None.
Acknowledgements. We would like to thank Rishay Padayachey for capturing the data.
Author contributions. MT conceptualised the study and wrote the manuscript. MT, MB, CF collected data and MB and MT analysed the data. MB and JdT revised the manuscript. All authors approved the final manuscript for publication.
Funding. None.
Conflicts of interest. None.
References
1. Smythe T, Kuper H, Macleod D, et al. Birth prevalence of congenital talipes equinovarus in low- and middle-income countries: A systematic review and meta-analysis. Trop Med Int Heal 2017;22(3):269-285. https://doi.org/10.1111/tmi.12833 [ Links ]
2. Zionts LE, Sangiorgio SN, Cooper SD et al. Does clubfoot treatment need to begin as soon as possible? J Paediatr Orthop 2016;36(6):558-564. https://doi.org/10.1097/bpo.0000000000000514 [ Links ]
3. Miedzybrodzka Z. Congenital talipes equinovarus (clubfoot): A disorder of the foot but not the hand. J Anat 2003;202(1):37-42. https://doi.org/10.1046/j.1469-7580.2003.00147.x [ Links ]
4. Dobbs MB, Gurnett CA. Genetics of clubfoot. J Paediatr Orthop 2012;21(1):7-9. https://doi.org/10.1097/bpb.0b013e328349927c [ Links ]
5. Malagelada F, Mayet S, Firth G, et al. The impact of the Ponseti treatment method on parents and caregivers of children with clubfoot: A comparison of two urban populations in Europe and Africa. J Child Orthop 2016;10(2):101-107. https://doi.org/10.1007/s11832-016-0719-7 [ Links ]
6. Gao R, Tomlinson M, Walker C. Correlation of Pirani and Dimeglio scores with number of Ponseti casts required for clubfoot correction. J Paediatr Orthop 2014;34(6):639-642. https://doi.org/10.1097/bpo.0000000000000182 [ Links ]
7. Wu V, Nguyen M, Nhi HM, et al. Evaluation of the progress and challenges facing the Ponseti method program in Vietnam. Iowa Orthop J 2012;32:125-134. [ Links ]
8. Wallander HM. Congenital clubfoot: Aspects on epidemiology, residual deformity and patient reported outcome. Acta Orthop 2010;81(339):1-25. https://doi.org/10.3109/17453671003619045 [ Links ]
9. Ponseti IV, Smoley EN, Brand RA. The classic congenital clubfoot: The results of treatment. J Bone Jt Surg Am 1963;45:261-344. https://doi.org/10.2106/00004623-196345020-00004 [ Links ]
10. Ford-Powell VA, Barker S, Khan MSI, et al. The Bangladesh clubfoot project: The first 5 000 feet. J Paediatr Orthop 2013;33(4):e40-44. https://doi.org/10.1097/bpo.0b013e318279c61d [ Links ]
11. Avilucea FR, Szalay EA, Bosch PP, et al. Effect of cultural factors on outcome of Ponseti treatment of clubfeet in rural America. J Bone Jt Surgery-American 2009;91(3):530-540. https://doi.org/10.2106/jbjs.h.00580 [ Links ]
12. Alvarado DM, McCall K, Hecht JT, et al. Deletions of 5' HOXC genes are associated with lower extremity malformations, including clubfoot and vertical talus. J Med Genet 2016;53(4):250-255. https://doi.org/10.1136/jmedgenet-2015-103505 [ Links ]
13. Kromberg JGR, Jenkins T. Common birth defects in South African blacks. South African Med J 1982;62(17):599-602. [ Links ]
14. du Toit SN. A one-year survey of a rural orthopaedic clinic in Zululand. S Afr Med J 1973;47(45):2189-2190. [ Links ]
15. Firth GB, Eltringham M, Shnier G. Early results of the Ponseti technique for a clubfoot clinic in South Africa. SA Orthop J 2009;8(4):67-71. [ Links ]
16. Street MR, Ramguthy Y, Firth G. Do Ponseti plasters delay gross motor milestones of South African children treated for idiopathic clubfeet? SA Orthop J Winter 2016;15(2):49-52 https://doi.org/10.17159/2309-8309/2016/v15n2a8 [ Links ]
17. Qudsi R, Selzer F, Hill SC, et al. Clinical outcomes, and risk-factor analysis of the Ponseti method in a low-resource setting: Clubfoot care in Haiti. PLoS ONE 2019;14(3).e0213382. https://doi.org/10.1371/journal.pone.0213382 [ Links ]
18. Wijayasinghe SR, Abeysekera WYM, Dharmaratne TSS. Descriptive epidemiology of congenital clubfoot deformity in Sri Lanka. J Coll Physicians Surg Pakistan 2018;28(2):166-168. https://doi.org/10.29271/jcpsp.2018.02.166 [ Links ]
19. Smythe T, Chandramohan D, Bruce J, et al. Results of clubfoot treatment after manipulation and casting using the Ponseti method: Experience in Harare, Zimbabwe. Trop Med Int Heal 2016;21(10):1311-1318. https://doi.org/10.1111/tmi.12750 [ Links ]
20. Adegbehingbe OO, Adetiloye AJ, Adewole L, et al. Ponseti method treatment of neglected idiopathic clubfoot: Preliminary results of a multi-centre study in Nigeria retrospective study. World J Orthop 2017;8(8):624-630. https://doi.org/10.5312/wjo.v8.i8.624 [ Links ]
21. Miller NH, Carry PM, Mark BJ, et al. Does strict adherence to the Ponseti method improve isolated clubfoot treatment outcomes? A two-institution review. Clin Orthop Relat Res 2016;474(1):237-243. https://doi.org/10.1007/s11999-015-4559-4 [ Links ]
22. Besselaar AT, Kamp MC, Reijman M, et al. Incidence of congenital idiopathic clubfoot in the Netherlands. J Paediatr Orthop 2018;27(6):563-567. https://doi.org/10.1097/bpb.0000000000000525 [ Links ]
23. De Mulder T, Prinsen S, Van Campenhout A. Treatment of non-idiopathic clubfeet with the Ponseti method: A systematic review. J Child Orthop 2018;12:575-581. https://doi.org/10.1302/1863-2548.12.180066 [ Links ]
24. Ponseti IV, Zhivkov M, Davis N, et al. Treatment of the complex idiopathic clubfoot. Clin Orthop Relat Res 2006;451(451):171-176. https://doi.org/10.1097/01.blo.0000224062.39990.48 [ Links ]
25. Matar HE, Beirne P, Bruce CE, et al. Treatment of complex idiopathic clubfoot using the modified Ponseti method: Up to 11 years follow-up. J Paediatr Orthop 2017;26(2):137-142. https://doi.org/10.1097/bpb.0000000000000321 [ Links ]
26. Gelfer Y, Wientroub S, Hughes K, et al. Congenital talipes equinovarus. Bone Joint J 2019;101(6):639-645. https://doi.org/10.1302/0301-620x.101b6.bjj-2018-1421.r1 [ Links ]
27. Vo NQD, Huynh NM. Mid-term results of Ponseti management for an idiopathic congenital clubfoot at a single center in Vietnam. J Paediatr Orthop 2016;25(3):253-257. https://doi.org/10.1097/bpb.0000000000000267 [ Links ]
28. McElroy T, Konde-Lule J, Neema S, et al. Understanding the barriers to clubfoot treatment adherence in Uganda: A rapid ethnographic study. Disabil Rehabil 2007;29(11-12):845-855. https://doi.org/10.1080/09638280701240102 [ Links ]
29. Ansar A, Rahman AE, Romero L, et al. Systematic review and meta-analysis of global birth prevalence of clubfoot: A study protocol. BMJ Open 2018;8(3):19246. https://doi.org/10.1136/bmjopen-2017-019246 [ Links ]
30. Pirani S, Naddumba E, Mathias R, et al. Towards effective Ponseti clubfoot care: The Uganda sustainable clubfoot care project. Clin Orthop Relat Res 2009;467(5):1154-1163. https://doi.org/10.1007/s11999-009-0759-0 [ Links ]
31. Macnicol MF. The management of club foot: Issues for debate. J Bone J Surg 2003;85(2):167-170. https://doi.org/10.1302/0301-620x.85b2.14067 [ Links ]
 Correspondence:
Correspondence:
M Thiart
marithiart@sun.ac.za; mari.thiart18@gmail.com
Accepted 23 February 2021














