Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
South African Journal of Child Health
versión On-line ISSN 1999-7671
versión impresa ISSN 1994-3032
S. Afr. j. child health vol.9 no.4 Pretoria nov. 2015
http://dx.doi.org/10.7196/sajch.2015.v9i4.814
ARTICLE
The effect of lactose-free formula feeds on growth responses among severely malnourished HIV-infected children in Durban, South Africa
E BinkaI; D Montoya-FontalvoI; M HealyI; M SobieszczykII; P LaRussaI; R BobatIII; M ArcharyIV
IMD; Department of Medicine, Columbia University College of Physicians and Surgeons, Columbia University, New York, USA
IIMD, MPH;Department of Medicine, Columbia University College of Physicians and Surgeons, Columbia University, New York, USA
IIIMB ChB, FCPaed, MD;Paediatric Infectious Diseases Unit, Department of Paediatrics and Child Health, Nelson R Mandela School of Medicine, University of KwaZulu-Natal, Durban, South Africa
IVMB ChB, DCH (SA), FCPaeds, Paeds ID (SA); Paediatric Infectious Diseases Unit, Department of Paediatrics and Child Health, Nelson R Mandela School of Medicine, University of KwaZulu-Natal, Durban, South Africa
ABSTRACT
BACKGROUND: The co-occurrence of HIV infection and severe malnutrition contributes to high rates of morbidity and mortality among children in resource-limited settings. Lactose-free, ready-to-use therapeutic feeds (RUTFs) may be most appropriate in this population because of underlying mucosal damage secondary to inflammation and infection.
OBJECTIVES: To describe the effect of lactose-free RUTFs on the growth parameters of severely malnourished HIV-infected children in Durban, South Africa (SA).
METHODS: This was a prospective, observational study of nutritional recovery in HIV-infected, severely malnourished children, aged 6 months to 5 years, who received lactose-free RUTFs following admission to King Edward VIII Hospital in Durban, SA. The primary outcome was nutritional recovery, defined as 15% weight gain from enrolment to end of study. Secondary outcomes included z-scores for weight-for-height, weight-for-age, height-for-age, triceps skinfold thickness (SFT) and subscapular SFT calculated at baseline and 7, 14, 30 and 45 days after admission. Univariate analysis was done to compare outcomes among antiretroviral therapy (ART)-naive and ART-experienced children; the effect of ART on nutritional recovery was evaluated in a logistic regression model
RESULTS: A significant improvement in most nutritional parameters was found at 45 days; 59% of children attained nutritional recovery. There was no significant difference in the proportion of children reaching recovery based on ART status at admission (p=0.08).
CONCLUSION: Lactose-free formula feeds may be an effective strategy for nutritional rehabilitation of severely malnourished and HIV-infected children in resource-limited settings. It remains to be determined how ART initiation affects nutritional recovery in these children.
Severe acute malnutrition (SAM) affects 13 million children under the age of 5 years and leads to 1 - 2 million preventable paediatric deaths annually.[1] The World Health Organization (WHO) defines SAM as the presence of at least one of: (i) weight-for-height more than 70% below the age-specific median or three or more standard deviations (SDs) below the mean in a normally distributed population; (ii) mid-upper arm circumference <115 mm; or (in) the presence of bilateral lower extremity pitting oedema of nutritional origin.[2,3]
In developing countries, there is often an overlap between HIV infection and malnutrition. The prevalence of SAM is approximately three-fold higher in HIV-infected than HIV-uninfected children,[4] and SAM is often the precipitating event that prompts healthcare-seeking behaviour among HIV-infected children. Co-occurrence of SAM and HIV increases morbidity, including stunted physical and cognitive development, and mortality[5,6]
The WHO recommends the use of two specific formula diets as treatment for SAM: the F-75 (75 kcal-th or 315 kJ/100 mL) during the initial phase of treatment and the F-100 (100 kcal-th or 420 kJ/100 mL) during the rehabilitation phase after the child's appetite has returned.[2] Countries such as South Africa (SA) have developed local, ready-to-use therapeutic feeds (RUTFs) similar in nutritional composition to the WHO-recommended F-100 formula. Unlike the F-100, RUTFs are not water based, thus minimising potential contamination or barriers associated with water acquisition.[6,7] Furthermore, the WHO-recommended feeds used to treat SAM are high in lactose and may not be optimal for HIV-infected children who tend to present with diarrhoea and malabsorption. Few studies have investigated the effect of the local and lactose-free RUTFs on the growth responses and nutritional recovery of severely malnourished HIV-infected children.[8] Our study describes the effect of a lactose-free RUTF on growth responses of severely malnourished HIV-infected children in Durban, SA.
Methods
Patients
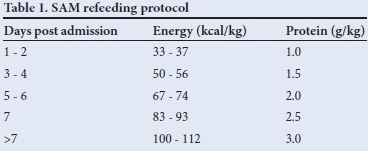
Children admitted to the King Edward VIII Hospital paediatric wards were screened during two time periods: between February and March 2011 and between December 2011 and April 2012. Those who were 6 months - 5 years of age, confirmed by HIV DNA polymerase chain reaction or HIV ELISA to be HIV-infected, and met the WHO definition of SAM, were eligible for enrolment in this prospective, observational study. Antiretroviral (ARV) therapy (ART)-experienced children were defined as children who reported taking ARVs at the time of admission; ART-naive children were defined as children who were not taking ARVs at admission. All patients were fed with an age-appropriate, lactose-free RUTF based on the postadmission day (Table 1).
Study procedures
Parents and guardians were approached to obtain written informed consent. The consent form was read to them in English or IsiZulu with the use of a translator. Measurements of weight, height, and biceps, triceps, subscapular and mid-thigh skinfold thickness (SFT), and demographic and pertinent clinical information were obtained at baseline and at days 7, 14, 30 and 45 post enrolment. SFT measurements were done with a Harpenden Skinfold Caliper (Baty International, UK). The study was approved by the Institutional Review Board of Columbia University and the Biomedical Research Ethics Committee at the University of KwaZulu-Natal, as well as by the hospital management.
Statistical analysis
Indices of growth response and recovery were calculated using the WHO Anthro software (version 3.2.2). Z-scores for weight-for-height (WHZ), weight-for-age (WAZ), height-for-age (HAZ), triceps SFT (TSZ) and subscapular SFT (SSZ) were calculated at baseline and at follow-up days 7, 14, 30 and 45. Nutritional recovery was defined as >15% weight gain from enrolment through to end of study period or by day 45. Data were captured in Microsoft Access (USA). A binary logistic regression model was applied to data, including age, gender, CD4 cell count and percentage, baseline weight, ART status at baseline, presence of pedal oedema and WHZ >70% as independent variables. Analysis was performed using SPSS version 19 (IBM, USA).
Results
Baseline demographics of participants are shown in Table 2. Sixteenof 30 children had CD4% available at baseline. Of these, 12 (75%) were severely immunosuppressed (CD4% <15%). Of 30 children, eight had HIV-1 viral loads available at baseline; median log10 HIV-1 RNA was 6.3 (interquartile range (IQR) 5.7 - 6.9). Fifteen out of 30 children (50.0%) were on ARVs at enrolment.
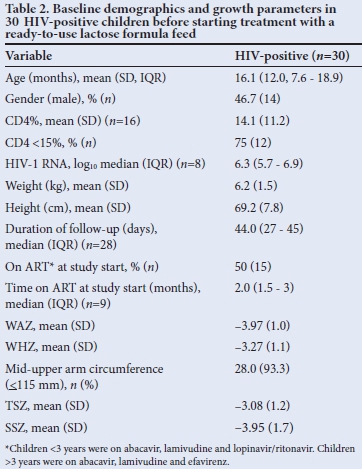
Baseline growth parameters for participants are shown in Table 2. Mean baseline growth parameters indicated that the children were severely malnourished: mean (SD) WAZ -3.97 (1.0), mean WHZ -3.27 (1.1), mean TSZ -3.08 (1.2) and mean SSZ -3.95 (1.7). Twenty-four of 30 had a WHZ <70% of the median, suggesting that this was more of an acute presentation of severe malnutrition rather than a chronic state where stunting would be more prevalent. Baseline comorbidities are shown in Table 3.

Twenty-two of the 30 children had evaluations through to study end at day 45. Three died of opportunistic infections and five were lost to follow-up. Of the study subjects, 59.0% («=13) attained nutritional recovery while 41.0% («=9) remained malnourished. There was a significant improvement from baseline to day 45 («=22 patients) in WAZ (1.34 (1.90), £=0.003), TSZ (1.43 (1.60), p=0.0005) and SSZ (2.02 (2.50), p=0.001), a trend to significance for WHZ (0.95 (2.20), p=0.060) and a statistically insignificant improvement for HAZ (0.76 (0.50), p=0.100). The mean rate of weight gain was 3.5 (6.9) g/kg/day but increased to 4.7 (5.0) g/kg/day after excluding the three deceased.
A comparison of baseline characteristics and clinical outcomes between ART-experienced and ART-naive children at the start of the study showed no significant differences (Table 4). However, in univariate analysis, ART-naive children had a slightly higher likelihood of reaching nutritional recovery at day 45 (60% v. 40%; p=0.08) than children who were ART-experienced. In logistic regression analysis, being ART-naive at presentation was associated with nutritional recovery at day 45 (OR 2.25, p=0.465), although this was not statistically significant (Table 5). Demographic factors, baseline weight, and CD4 cell percentage were not associated with nutritional recovery at day 45.


Discussion
Current WHO guidelines for the treatment of SAM do not make specific formula recommendations for refeeding children who are both severely malnourished and HIV-infected. With HIV infection, there is mucosal damage secondary to infection or inflammation leading to chronic diarrhoea, protein-losing enteropathy, carbohydrate deficiencies and fat malabsorption.[7] Therefore, lactose-free formulas may be appropriate alternatives for refeeding regimens in this population. Our results showed that HIV-infected children on lactose-free RUTFs gained an average of 3.5 g/kg/day. This figure is similar to the average weight gain of severely malnourished children known to be HIV-infected in previous studies of lactose-containing formulas recommended by the WHO. Ndekha et al.[9] and Sandige et al.[10] studied children in Malawi who were severely malnourished and HIV-infected; both studies reported an average weight gain of 3.1 (2.7) g/kg/ day and 3.6 (4.7) g/kg/day, respectively. Our study showed that children refed with lactose-free RUTF gained weight in a similar fashion. However, our study was limited owing to policies of the institution that did not make it possible to directly compare lactose-free with lactose-containing formulas. We were also limited by our inability to confirm the presence oflactose maldigestion in our patients at presentation.
The WHO recommends that the minimum rate of weight gain at the end of a nutritional rehabilitation period should be 5.0 g/kg/day. After excluding values for the deceased children, our mean weight gain increased to 4.7 g/kg/day, approaching WHO-expected rates. This occurred despite the expectation that severely malnourished children who are HIV-infected should gain less weight or gain less rapidly than a normally distributed population, given the malabsorptive state and mucosal damage created by HIV infection.
Even when using high-energy RUTFs to manage SAM in HIV-infected children, mortality rates of ~38% at 4 - 6 weeks are too high.[111 Our mortality rates were slightly lower. At the end of our 45-day study period, three patients had died (10% of our study population). Although the use of RUTF diets has been shown to lead to significant and sustained weight gain in HIV-infected children, this improvement has not always been associated with recovery in CD4 cell count or improvement in survival.[11,12] One major limitation of our study was that we were not able to track the changes in CD4 cell count and percentage in our patients to determine if there was any association between nutritional recovery and improvement in CD4 cell count or percentage.
We compared the baseline characteristics and clinical outcomes ofchildren on ART and those not on ART prior to the study. We observed no statistically significant differences between the two groups, although this may be due to our small sample size. We also investigated the effect of concurrent ART use on attaining nutritional recovery using a multivariate logistic regression model. Based on this model, being ART-naive during acute illness may have been associated with nutritional recovery, although this result did not reach statistical significance. We hypothesise that the acute state of severe malnutrition and the concurrent use of ART with associated gastrointestinal side-effects could compound the malabsorptive state in the gastrointestinal tracts of these children, leading to suboptimal absorption of nutrients and ART. Other studies have shown that early initiation of ART leads to improved nutritional status in HIV-infected children.[13-151 Further research needs to be conducted to determine the right timing for initiation of ART in severely malnourished and HIV-infected children.
Conclusion
Lactose-free formula feeds may be an effective strategy for nutritional rehabilitation of severely malnourished and HIV-infected children in resource-limited settings. Future studies should directly compare the effectiveness of both types of formulas in promoting nutritional recovery and explore how the timing of ART initiation with respect to acute refeeding affects nutritional recovery in these children.
References
1. Collins S, Dent N, Binns P, Bahwere P, Sadler K, Hallam A. Management of severe acute malnutrition in children. Lancet 2006;368(9551):1992-2000. [http://dx.doi.org/10.1016/S0140-6736(06)69443-9] [ Links ]
2. World Health Organization. Management of severe malnutrition: A manual for physicians and other senior health workers. Geneva: World Health Organization, 1999. [ Links ]
3. World Health Organization/World Food Programme/United Nations System Standing Committee on Nutrition/United Nations Children's Fund. Community-based management of severe acute malnutrition. Geneva/Rome/Geneva/New York: World Health Organization/World Food Programme/ United Nations System Standing Committee on Nutrition/United Nations Children's Fund, 2007. [ Links ]
4. Prendergast A, Bwakura-Dangarembizi MF, Cook AD, et al. Hospitalization for severe malnutrition among HIV-infected children starting antiretroviral therapy. AIDS 2011;25(7):951-956. [http://dx.doi.org/10.1097/QAD.0b013e328345e56b] [ Links ]
5. Chinkhumba J, Tomkins A, Banda T, Mkangama C, Fergusson P. The impact of HIV on mortality during in-patient rehabilitation of severely malnourished children in Malawi. Trans R Soc Trop Med Hyg 2008;102(7):639-644. [http://dx.doi.org/10.1016/j.trstmh.2009.04.02] [ Links ]
6. Miller T, Orav EK, Martin SR, Cooper ER, McIntosh K, Winter HS. Malnutrition and carbohydrate malabsorption in children with vertically transmitted human immunodeficiency virus 1 infection. Gastroenterol 1991;100(5):1296-1302. [ Links ]
7. Hendricks KM. Ready-to-use therapeutic food for prevention of childhood undernutrition. Nutr Rev 2010;68(7):429-435. [http://dx.doi.org/10.1111/j.1753-4887.2010.00302.x] [ Links ]
8. Biggs C. Clinical dietetic practice in the treatment of severe acute malnutrition in a high HIV setting. J Hum Nutr Diet 2013;26(2):175-181. [http://dx.doi.org/10.1111/jhn.12003] [ Links ]
9. Ndekha MJ, Manary MJ, Ashorn P, Briend A. Home-based therapy with ready-to-use therapeutic food is of benefit to malnourished, HIV-infected Malawian children. Acta Paediatr 2005;94(2):222-225. [ Links ]
10. Sandige H, Ndekha MJ, Briend A, Ashorn P, Manaray MJ. Home-based treatment of malnourished Malawian children with locally produced or imported ready-to-use food. J Pediatr Gastroenterol Nutr 2004;39(2):141-146. [ Links ]
11. Heikens GT, Bunn J, Amadi B, et al. Case management of HIV-infected severely malnourished children: Challenges in the area of highest prevalence. Lancet 2008;371(9620):1305-1307. [http://dx.doi.org/10.1016/S0140-6736(08)60565-6] [ Links ]
12. Rollins NC, van den Broeck J, Kindra G, Pent M, Kasambira T, Bennish ML. The effect of nutritional support on weight gain of HIV-infected children with prolonged diarrhoea. Acta Paediatr 2007;96(1):62-68. [ Links ]
13. Kim MH, Cox C, Dave A, et al. Prompt initiation of ART with therapeutic food is associated with improved outcomes in HIV-infected Malawian children with malnutrition. Acquir Immune Defic Syndr 2012;59(2):173-176. [http://dx.doi.org/10.1097/QAI.0b013e3182405f8f] [ Links ]
14. Sutcliffe CG, van Dijk JH, Munsanje B, et al. Weight and height z-scores improve after initiating ART among HIV-infected children in rural Zambia: A cohort study. BMC Infect Dis 2011;11(1):54. [http://dx.doi.org/10.1186/1471-2334-11-54] [ Links ]
15. Weigel R, Phiri S, Chiputula F, et al. Growth response to antiretroviral treatment in HIV-infected children: A cohort study from Lilongwe, Malawi. Trop Med Int Health 2010;15(8):934-944. [http://dx.doi.org/10.1111/j.1365-3156.2010.02561.x] [ Links ]
 Correspondence:
Correspondence:
M Archary
archary@ukzn.ac.za














