Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Child Health
On-line version ISSN 1999-7671
Print version ISSN 1994-3032
S. Afr. j. child health vol.9 n.2 Pretoria Jan./Apr. 2015
http://dx.doi.org/10.7196/SAJCH.859
RESEARCH
The use of nasal CPAP at the Charlotte Maxeke Johannesburg Academic Hospital
C JardineI; D E BallotII
IMB BCh; Department of Paediatrics and Child Health, University of the Witwatersrand and Charlotte Maxeke Johannesburg Academic Hospital, Johannesburg, South Africa
IIMB BCh, FCPaed (SA), PhD; Department of Paediatrics and Child Health, University of the Witwatersrand and Charlotte Maxeke Johannesburg Academic Hospital, Johannesburg, South Africa
ABSTRACT
BACKGROUND: Nasal continuous positive airway pressure (NCPAP) is well established as a treatment for hyaline membrane disease (HMD) and other respiratory diagnoses in neonates. NCPAP is an affordable intervention that reduces the number of neonatal admissions to the intensive care unit (ICU) for ventilation. At the Charlotte Maxeke Johannesburg Academic Hospital (CMJAH) we have been using NCPAP since April 2006.
OBJECTIVES: To review the use of early NCPAP in our hospital setting.
METHODS: This was a retrospective, descriptive study of all neonates >750 g admitted to CMJAH between 1 January 2013 and 31 July 2014, who received NCPAP within 72 hours of birth. The characteristics and the survival of all infants who received NCPAP were described using univariate analysis.
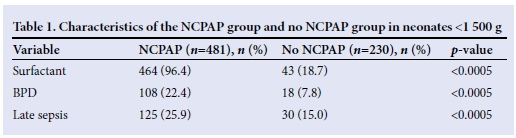
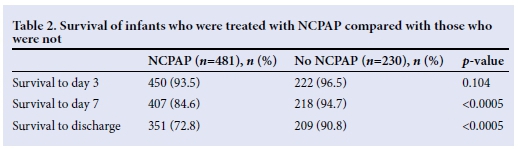
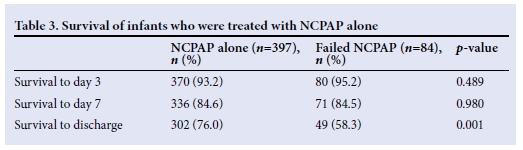
RESULTS: The NCPAP group (n=481) of neonates <1 500 g was significantly associated with surfactant use (p<0.0005), bronchopulmonary dysplasia (p<0.0005) and late sepsis (p<0.0005). The survival to day 7 and to discharge of infants treated with NCPAP was significantly decreased (p<0.0005). NCPAP alone (without ventilation) improved the survival to discharge (p=0.001). The survival was 95.4% in the >1 500 g infants, compared with 87.6% in the very low birth weight infants and 55.2% in the extremely low birth weight infants.
CONCLUSION: NCPAP is an effective intervention for HMD; it is both cost-effective and easy to use in a resource-limited setting, and reduces the morbidity and mortality associated with ICU admission.
Nasal continuous positive airway pressure (NCPAP) has become widely used and accepted as a treatment for hyaline membrane disease (HMD) since its first introduction in 1971.[1,2] HMD affects premature babies and causes considerable morbidity and mortality. The use of NCPAP on its own and in combination with the use of surfactant has been shown to improve the outcomes of HMD in premature infants.[3]
The incidence of HMD is related to the degree of prematurity of the lungs, and therefore the incidence increases as the gestational age (GA) decreases. The use of antenatal steroids given to mothers where a premature delivery is expected has reduced the incidence and severity of HMD.[4] The administration of surfactant for severe HMD has been shown to improve lung compliance, facilitate weaning off supplemental oxygen and result in fewer neonates needing ventilatory support. Surfactant replacement therapy also significantly reduces the mortality from HMD.[5]
NCPAP is not only used in HMD but is also used to treat apnoea of prematurity, respiratory distress due to other aetiologies, some types of upper airway obstruction, and can sometimes be used as an alternative to endotracheal intubation or as a weaning mode of ventilation.[1] The continuous positive pressure provided by NCPAP helps to support and distend the alveoli, preventing their collapse. This results in the recruitment of alveoli, which improves the ventilation perfusion mismatch of the lungs and increases the functional residual capacity and tidal volume, thereby improving oxygenation in the neonate.[1] NCPAP also exerts a distending pressure on the larger airways, thus stabilising them and preventing upper airway collapse.[1] The early use of NCPAP in premature neonates reduces the need for surfactant and results in fewer neonates needing to be intubated and ventilated.[3,6,7] This decreases ventilator-induced lung injury and results in fewer cases of bronchopulmonary dysplasia (BPD).[3,7] Adverse effects of NCPAP use, including 'CPAP belly syndrome',[8] nasal septal necrosis and pneumothorax, may sometimes occur.
NCPAP is an affordable intervention that is easy to use in a resource-poor setting. The successful use of NCPAP could result in a considerable reduction in the costs of neonatal care, as there would be a reduction in ventilation and neonatal intensive care unit (NICU) admissions, as well as a decrease in poor outcomes associated with prolonged ventilation. Recently, there have been a number of studies reporting on the survival of extremely low birth weight (ELBW) infants (birth weight < 1 000 g). At Charlotte Maxeke Johannesburg Academic Hospital (CMJAH), it was found that the use of NCPAP in ELBW infants was not associated with an improvement in survival.[9] This study was done using data from neonates < 900 g admitted between January 2006 and December 2010, a period during which fewer ELBW infants were receiving NCPAP than currently. A previous study at CMJAH, which assessed the determinants of survival in very low birth weight (VLBW) neonates (birth weight <1 500 g) found that NCPAP was associated with improved survival.[10] Kirsten et al.[11] at the Tygerberg Children's Hospital in the Western Cape found that NCPAP practised together with InSurE (intubating to give surfactant and then extubating) in a neonatal high-care ward with limited resources improved the survival of ELBW infants.
NCPAP was introduced into the neonatal unit at the CMJAH in April 2006. It is now used as the first line of ventilatory support in neonates with HMD. The objective of this study was to review the use of early NCPAP in neonates at CMJAH.
Methods
This was a retrospective, descriptive study of all neonates with a birth weight >750 g admitted to the neonatal unit at CMJAH between 1 January 2013 and 31 July 2014, who received NCPAP within 72 hours of birth. Neonates were excluded if there was insufficient information, if NCPAP was used post extubation from conventional mechanical ventilation (CMV) or if they had major congenital abnormalities. All in-born babies, babies born at maternity outpatient units, or babies born before arrival (BBA) were initially observed in the transitional care unit for assessment before being admitted to the neonatal unit, and were included in our statistics. T-piece resuscitators were not available in the labour ward nursery, and bag mask valve resuscitation was used for babies who required resuscitation at birth. The babies were observed for 1 - 2 hours in the transitional care unit and then a decision was made by the attending doctor on whether or not they required NCPAP and surfactant. NCPAP with early rescue surfactant was offered to neonates with respiratory failure due to HMD as per the neonatal unit's protocol. Respiratory failure was defined as an oxygen saturation <88% in 60% supplemental oxygen, respiratory acidosis on arterial blood gas or clinical signs of severe respiratory distress, including indrawing of the sternum and tachypnoea. Due to insufficient NCPAP machines, not all neonates who qualified for NCPAP could receive it. Neonates who required NCPAP were admitted to the high-care neonatal ward where there were nine Bubble NCPAP (Fisher and Paykel, New Zealand) machines available; NCPAP was not initiated in the NICU. If NCPAP was not available, babies were given surfactant and placed on nasal prong oxygen. The neonatal unit's policy at the time of the study was that neonates > 900 g would qualify for ventilation in the NICU if required.
Database
The neonatal records at CMJAH are kept on the REDCap (Research Electronic Data Capture) electronic neonatal database.[12] REDCap is a secure, web-based program that has been designed to aid data capture for the purposes of clinical audit and quality improvement. Data are collected upon discharge of patients and entered into the REDCap database. The information is verified at several different stages of collection. The following data were collected from the database: (i) maternal data -antenatal steroids, place and mode of delivery, multiple gestation; and (ii) infant data -gestational age, birth weight, gender, place of birth, 5-minute Apgar score, necrotising enterocolitis (NEC), intraventricular haemorrhage (IVH), NCPAP with or without surfactant or CMV, respiratory diagnosis, duration of NCPAP and ventilation, late sepsis (occurring after day 3), BPD (defined by oxygen requirement at 28 days of age), nasal septal necrosis, pneumothorax, and outcome (death or survival) at discharge.
Statistical analysis
The data were entered into an MS Excel (Microsoft, USA) spreadsheet and imported into statistical software package SPSS version 19 (IBM, USA). Categorical variables were described using frequencies and percentages, while continuous variables were described using means and standard deviations (SDs). The data were also stratified into birth weight categories (ELBW < 1 000 g, VLBW < 1 500 g, low birth weight (LBW) <2 500 g and weight > 2 500 g), and the proportion of infants in each category receiving NCPAP was determined. Neonates <1 500 g who had not received NCPAP were compared with those who had received NCPAP, with regard to characteristics and survival to discharge. IVH was graded according to the sonographic grading system described by Papile et aí.[13] Grades 1 and 2 of IVH were considered together as 'mild' and grades 3 and 4 as 'severe. The NEC category included grades 2 and 3 of NEC, according to modified Bell's staging criteria.[14] Infants were regarded as having birth asphyxia if they had Apgar scores of < 5 at 5 minutes. Babies on NCPAP who developed respiratory failure and required ventilation in the NICU were regarded as having failed NCPAP. Babies who were transferred out and those who were discharged home directly were grouped together as survivors for the purpose of analysis. Univariate analysis was used to compare the characteristics of the two groups, and survival was described. Categorical variables were compared using χ2 tests, and continuous variables using unpaired t-tests (as the distribution was normal). A p-value of < 0.05 was considered to be significant. In neonates > 1 500 g, the characteristics, respiratory diagnosis and survival of those who received NCPAP were described.
Ethics
Ethics approval for the study was granted by the Human Research Ethics Committee at the University of the Witwatersrand, Johannesburg (clearance certificate number M140403).
Results
Infants <1 500g
There were 748 VLBW infants admitted over the 19-month period. Information was not available on 9 infants, 18 infants had major birth defects, NCPAP was used as a weaning mode of ventilation in 3 infants and NCPAP was started after 72 hours in 7 infants. All of the above were excluded, leaving a total of 711 infants in the study.
The majority were female (n=378, 53.2%). The mean birth weight was 1 158 (SD 220) g and GA was 29.5 (2.6) weeks. The majority (n=380, 53.4%) were born by emergency caesarean section, and most (n=591, 83.1%) were inborn. Antenatal steroids were given in 275 (38.7%) cases. NCPAP was provided to 481 (67.7%) of the infants in total. Most babies («=397, 82.5%) coped on NCPAP alone; in 84 (17.5%), NCPAP failed. Overall, there were 560 (78.8%) survivors. The mean duration of NCPAP was 2.58 (3.52) days with a maximum duration of 35 days. Nasal septal necrosis occurred in 27/481 (5.6%) and pneumothorax occurred in 5/481 (1%) of the infants who received NCPAP. Surfactant was given to 507 (71.3%) infants in total; 43 (8.4%) of these received surfactant without NCPAP. Birth asphyxia occurred in 79 (11.1%) infants.
VLBW 1 000 - 1 499 g
In this category, there were 517 infants, of whom 453 (87.6%) survived. A total of 308 (59.6%) infants received NCPAP, and surfactant was given to 329 (63.6%) infants. There were 41 (8%) infants with birth asphyxia. The use of NCPAP was significantly associated with death (p=0.001, odds ratio (OR) 0.41, 95% confidence interval (CI) 0.23 - 0.73). Overall, 252 (81.8%) infants coped on NCPAP alone, while 56 (18.2%) infants failed NCPAP.
ELBW 750 - 999 g
In this category, there were 194 infants, of whom 107 (55.2%) survived. A total of 173 (89.2%) infants received NCPAP, and surfactant was given to 178 (91.8%) infants. There were 38 (19.6%) infants with birth asphyxia. NCPAP was not significantly associated with survival (p=0.261, OR 0.72, 95% CI 0.39 - 1.35). Overall,
145 (83.8%) infants coped on NCPAP alone,
while 28 (16.1%) infants failed NCPAP (these were all >900 g and thus were offered ventilation).
NCPAP v. no NCPAP
The entire group of neonates between 750 and 1 499 g was divided into two groups: those who had received NCPAP and those who had not. The characteristics of the two groups were compared and significant differences are shown in Table 1. There was no significant difference in the babies with regards to CMV, pneumothorax, antenatal steroids, IVH or NEC. Mild IVH occurred in 69 (14.4%) infants in the NCPAP group compared with 18 (7.8%) infants not receiving NCPAP, and severe IVH occurred in 20 (4.2%) infants receiving NCPAP compared with 7 (3%) infants not receiving NCPAP. These differences were not significant. The survival of the infants in the two groups was compared in Table 2. The use of NCPAP was associated with an increased risk of death by day 7 and at discharge. When the successful use of NCPAP alone was compared with those who failed NCPAP, there was a significantly higher rate of survival to discharge (Table 3).
Infants > 1 500 g
A total of 1 997 neonates weighed >1 500 g; 1 570 infants did not receive NCPAP, 154 had a major birth defect, 7 had insufficient information and 5 had NCPAP as a weaning mode from CMV. There were thus 261 infants in the NCPAP group.
The majority were males (n=164, 62.8%). Most of the infants were inborn (n=217, 83.1%). In 129 (49.4%) infants, the mode of delivery was by emergency caesarean section. In the LBW (1 500 - 2 499 g) category, there were 220 (84.3%) infants, and in the >2 500 g category, there were 41 (15.7%) infants. The mean birth weight was 2 044 (528) g, with a maximum of 5 250 g, and the mean GA was 33.5 (2.9) weeks with a minimum of 28 weeks. Overall, 249 (95.4%) infants survived. Surfactant was given to 210 (80.5%) infants. The mean duration of NCPAP was 1.62 (1.7) days, with a minimum of <1 day and a maximum of 31 days. There were 33 (12.6%) infants who failed NCPAP. This group of larger infants (> 1 500 g) represented 35.1% (261/742) of all the babies who received NCPAP over the study period. The respiratory diagnosis in the majority of cases was HMD (n=213, 81.6%) but also included transient tachypnoea of the newborn (TTN), congenital pneumonia, meconium aspiration syndrome (MAS) and apnoea (Table 4).

Discussion
This was the first study from our unit on how we are using NCPAP in all our neonates from 750 g to 5 250 g. Previously, we had investigated the use of NCPAP as it relates to the survival of both ELBW and VLBW infants in our unit, but we had not determined the characteristics of the infants receiving NCPAP across all weight categories.
The majority of the babies in the ELBW category received NCPAP (89.2%). It was expected that all the babies in this weight category would have had HMD, requiring NCPAP and surfactant replacement, and it is encouraging that we managed to provide NCPAP to most patients. Only 16.1% of the ELBW infants and 18.2% of the VLBW infants failed NCPAP and were given ventilation. However, in the ELBW category, only infants >900 g would have been offered CMV. These figures are appropriate for a resource-poor setting in a high-care nursery such as ours. We have only nine Bubble NCPAP machines available, but most of our babies were able to receive NCPAP. This may be attributed to the fact that babies were able to wean off of the NCPAP relatively quickly, allowing a high turnover of these machines. With the successful use of NCPAP, far fewer premature babies are requiring NICU admission for ventilation. This is both cost-saving and greatly reduces morbidity and mortality associated with the NICU. Our ICU is shared between neonates, paediatrics and paediatric surgery patients. There was a concern that lowering our ICU admission weight category to 900 g would inundate the ICU with premature babies, but - largely due to NCPAP - this has not been the case. However, it is important to note that although babies who require NCPAP do not need NICU admission, they do require adequate high-care facilities with well-trained nursing staff, as they need close monitoring and active weaning off the NCPAP.
In the larger (>1 500 g) babies, only 12.6% failed NCPAP. In these babies, we were using NCPAP for other indications such as TTN, congenital pneumonia, MAS and apnoea, but in the majority of cases it was used for HMD in larger premature babies.
It is disappointing to note that in this study there was no improvement in the use of antenatal steroids since the study done in 2010[10] (38.7% in this study v. 36% in the 2010 study). This is largely due to missed opportunities and late presentation once the mother was already in preterm labour.[15]
The use of NCPAP was significantly associated with a higher mortality to day 7 (p<0.0005) and discharge (p<0.0005). The increased mortality with NCPAP use may be due to other factors affecting survival, such as late sepsis and NEC, which VLBW infants are prone to. There is also a selection bias, as NCPAP is given to babies with more severe respiratory illness and not routinely offered to all premature babies. Within the NCPAP group, NCPAP alone (without CMV)
improved survival to discharge (p=0.001). Prolonged ventilation is associated with increased morbidity, and infants who fail NCPAP have a more severe degree of HMD with higher mortality.
Surfactant, BPD and late sepsis were shown to be significantly associated with the use of NCPAP. The overall incidence of 22.4% BPD was higher than the 8.8% reported in our hospital in 2010.[10] More babies are now offered NCPAP and are surviving long enough to develop BPD than in 2010, where over a 1-year period only 96/474 (20.3%) VLBW infants received NCPAP. VLBW infants are prone to late-onset sepsis[16] and the use of NCPAP has also been associated with this.[17] Limited nursing staff and overcrowding in the neonatal nursery further compound this problem.
There was a very low rate of complications of NCPAP in our study, with only five infants developing a pneumothorax, and 27/481 infants developing nasal septal necrosis, while IVH was not found to be significantly associated with NCPAP. When compared with the Vermont Oxford Network, our rate of pneumothoraces was lower (1% v. 4%) and our NCPAP use (67.7% v. 73.5%) was also lower.
Conclusion
NCPAP is cost-effective and easy to use in a resource-poor environment with a small number of Bubble NCPAP machines being able to treat a large number of neonates with HMD. This effectively releases ICU beds, thereby increasing the number of beds that are available for ventilatory support for neonatal and general paediatric patients.
References
1. Sankaran K, Adegbite M. Noninvasive respiratory support in neonates: A brief review. Chin J Contemp Pediatr 2012;14(9):643-652. [ Links ]
2. Roberts CL, Badgery-Parker T, Algert CS, Bowen JR, Nassar N. Trends in the use of neonatal CPAP: A population-based study. BMC Pediatrics 2011;11:89. [http://dx.doi.org/10.1186/1471-2431-1189] [ Links ]
3. Pelligra G, Abdellatif MA, Lee SK. Nasal continuous positive airway pressure and outcomes in preterm infants: A retrospective analysis. Paediatr Child Health 2008;13(2):99-103. [ Links ]
4. Wong D, Abdel-Latif ME, Kent AL, et al. Antenatal steroid exposure and outcomes of very premature infants: A regional cohort study. Arch Dis Child Fetal Neonatal Ed 2014;99(1):F12-F20. [http://dx.doi.org/10.1136/archdischild-2013-304705] [ Links ]
5. Polin RA, Carlo WA. Surfactant replacement therapy for preterm and term neonates with respiratory distress. Pediatrics 2014;133(1):156-163. [http://dx.doi.org/10.1542/peds.2013-3443] [ Links ]
6. Saxena A, Thapar RK, Sondhi V, Chandra P. Continuous positive pressure for spontaneously breathing premature infants with respiratory distress syndrome. Indian J Pediatr 2012;70(9):1185-1191. [http://dx.doi.org/10.1007/s12098-012-0722-z] [ Links ]
7. Miksch RM, Armbrust S, Pahnke J, Fusch C. Outcome of very low birth weight infants after introducing a new standard regime with the early use of nasal CPAP. Eur J Pediatr 2008;167(8):909-916. [http://dx.doi.org/10.1007/s00431-007-0646-1] [ Links ]
8. Jaile JC, Levin T, Wung JT, Abramson SJ, Ruzal-Shapiro C, Berdon WE. Benign gaseous distension of the bowel in premature infants treated with nasal continuous airway pressure: A study of contributing factors. AJR Am J Roentgenol 1992;158(1):125-127. [ Links ]
9. Kalimba EM, Ballot DE. Survival of extremely low-birth-weight infants. S Afr J CH 2013;7(1):13-16. [http://dx.doi.org/10.7196/SAJCH.488] [ Links ]
10. Ballot DE, Chirwa TF, Cooper PA. Determinants of survival in very low birth weight neonates in a public sector hospital in Johannesburg. BMC Pediatrics 2010;10:30. [http://dx.doi.org/10.1186/1471-2431-10-30] [ Links ]
11. Kirsten GF, Kirsten CL, Henning PA. The outcome of ELBW infants treated with NCPAP and InSurE in a resource-limited institution. Pediatrics 2012;129(4):e952-e959. [http://dx.doi.org/10.1542/peds.2011-1365] [ Links ]
12. Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap): A metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform 2009;42(2):377-381. [http://dx.doi.org/10.1016/j.jbi.2008.08.010] [ Links ]
13. Papile LA, Burstein J, Burstein R, Koffler H. Incidence and evolution of subependymal and intraventricular hemorrhage: A study of infants with birth weights less than 1 500 g. J Pediatr 1978;92(4):529-534. [ Links ]
14. Neu J. Necrotizing enterocolitis: The search for a unifying pathogenic theory leading to prevention. Pediatr Clin North Am 1996;43(2):409-432. [ Links ]
15. Ballot DE, Ballot NS, Rothberg AD. Reasons for failure to administer antenatal corticosteroids in preterm labour. S Afr Med J 1995;85(10):1005-1007. [ Links ]
16. Stoll BJ, Hansen N, Fanaroff AA, et al. Late-onset sepsis in very low birth weight neonates: The experience of the NICHD Neonatal Research Network. Pediatrics 2002;110(2 Pt 1):285-291. [ Links ]
17. Graham PL, Begg MD, Larson E, Della-Latta P, Allen A, Saiman L. Risk factors for late onset Gram-negative sepsis in low birth weight infants hospitalized in the neonatal intensive care unit. Pediatr Infect Dis J 2006;25(2):113-117. [http://dx.doi.org/10.1097/01.inf.0000199310.52875.10] [ Links ]
 Correspondence:
Correspondence:
C Jardine
carlsjardine@gmail.com














