Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Child Health
On-line version ISSN 1999-7671
Print version ISSN 1994-3032
S. Afr. j. child health vol.9 n.1 Pretoria Jan. 2015
RESEARCH
The feasibility of determining the position of an endotracheal tube in neonates by using bedside ultrasonography compared with chest radiographs
S H de KockI; S F OttoII; G JoubertIII
IMB ChB, HED; Department of Clinical Imaging Sciences, Faculty of Health Sciences, University of the Free State, Bloemfontein, South Africa
IIMB ChB, MMed Rad (Diag), LLD, MPH; Department of Clinical Imaging Sciences, Faculty of Health Sciences, University of the Free State, Bloemfontein, South Africa
IIIBA, MSc; Department of Biostatistics, Faculty of Health Sciences, University of the Free State, Bloemfontein, South Africa
ABSTRACT
BACKGROUND: Neonates in our neonatal intensive care unit (NICU) receive a large amount of radiation with X-rays (XRs) being done daily, even more often with reintubation, repositioning of endotracheal tubes (ETTs) and confirmation thereof, which has been our NICU policy for many years
OBJECTIVE: To investigate the feasibility of determining the position of ETTs in neonates by using bedside ultrasonography (BUS), and to compare the results with those obtained from chest XR (CXR) findings
METHODS: A prospective, cross-sectional study was done on intubated neonates in the NICU at Universitas Academic Hospital, Bloemfontein, to determine the position of ETTs by using BUS
RESULTS: Thirty intubated patients included in this study had a median age of 13.5 days and a median weight of 1.6 kg. Ninety-three per cent of ETT placements were considered optimal when visualised by BUS, while 73.3% were considered to be placed optimally when CXR was viewed. When CXR and BUS findings were compared regarding optimal placing, the agreement was poor (κ=0.10; 95% confidence interval -0.2 - 0.4). In four patients, the distance from the aortic arch to the tip of the ETT was outside the expected range of 1.5 - 2.2 cm: in two patients it was <1.5 cm (6.7%) and in the other two >2.25 cm (6.7%). BUS measurements were done mainly in extended head (53.3%) or neutral (36.7%) position
CONCLUSION: Although poor agreement between CXR and BUS findings was obtained, possibly because of handling of patients with secondary shifting of ETTs, BUS was found not to be comparable with CXR, but an alternative feasible method to determine the optimal position of ETTs in the trachea in neonates when using other reference points, with the added advantage of no radiation exposure
The current situation in our neonatal intensive care unit (NICU) at Universitas Academic Hospital, Bloemfontein, is that all neonates undergo a daily routine X-ray (XR) (either chest or abdominal, or both). Those neonates who are intubated are X-rayed more often, for example, with reintubation, repositioning of endotracheal tubes (ETTs) or when deterioration of their condition is noted. This has been our NICU policy for many years. Consequently, neonates are exposed to high levels of radiation, which should be reduced where possible.
A literature search suggested that a study to determine the position of an ETT using bedside ultrasonography (BUS) would be feasible and possible. Several studies have confirmed that BUS can accurately determine the placing of ETTs in a paediatric ICU setting.[1-3] Ultrasound (US) and chest radiography agreed on ETT placement in 83% of cases, with US having a sensitivity of 91% and a specificity of 50%.[4] Hosseini et al.[5] used right subcostal US with good results to evaluate diaphragmatic motion for secondary confirmation of ETT position. The overall accuracy of BUS was 98.1% (95% confidence interval (CI) 93.0 - 100%).[5] A kappa of 0.85 indicated a very good agreement between the bedside upper airway ultrasonography and waveform capnography.[6] In a Cochrane review, Schmvlzer et al.[7] compared techniques to ascertain correct ETT placement in neonates, including US, but found insufficient evidence to determine the most effective technique.
Having a US machine readily available in our NICU, we considered it appropriate to optimise available resources in order to expedite service delivery efficiently with a quicker examination that clinicians could do themselves, with real-time alterations and confirmation of ETT positioning. Therefore, the objective of the study was to investigate the feasibility of determining the position of ETTs in neonates by using BUS, and to compare the results with those obtained from chest XR (CXR) findings.
Methods
A prospective, cross-sectional study was done on intubated neonates in the NICU at Universitas Academic Hospital, Bloemfontein, over a 3-month period, to determine the position of ETTs by using BUS.
The procedure and purpose of the investigation were discussed with patients' mothers or caregivers, from whom written consent was obtained. An information document regarding the study and contact details of the researcher was supplied.
Exclusion criteria included patients whose mothers did not give consent and patients with whom the medical team was actively busy with resuscitation and intervention.
At approximately 6 o'clock every morning, each patient in the NICU undergoes a routine CXR, done by radiographers on night duty. For this particular study, intubated patients received an additional US conducted by the first author, a radiology registrar, after radiographers had completed routine XRs in the NICU. This approach contributed to the time delay between the two methods of examination. A Toshiba (Japan) Nemio XG US machine with a small curvilinear probe (6 MHz) was used to determine the position of the ETT by BUS. Sagittal and axial images were obtained.
During a pilot study on five neonates, before official data collection commenced, it was determined that the ETT could be visualised well with BUS, although BUS could not determine the exact level of the tip of the ETT according to the thoracic vertebrae, as was the case with CXR. Keeping this in mind, it was decided to apply relevant surface anatomy. Anatomically, the thyroid lies anterior and lateral to the most proximal part of the trachea at the C6/7 vertebral level.[8] The aortic arch begins and ends at the manubrio sternal junction at the T4/5 level.[8] The bifurcation of the trachea in neonates is at the T3 - T5 level. In ~85% of neonates, the tracheal bifurcation occurs at T4 level.[9] The ideal position of an ETT in a neonate with the head in neutral position is at the T1/2 level,[9,10] and when applying surface anatomy, inferior to the level of the thyroid and superior to the aortic arch.
The patients included in this study were mainly premature babies with low birth weight (LBW). The mean thoracic vertebral body height was 5 mm, and the mean intervertebral disk height was 2.5 mm, giving a distance of ~7.5 mm from the inferior end of one thoracic vertebra to the inferior end of the next. Optimal measured distances from the aortic arch to the tip of the ETT (as obtained by BUS) correlated with distances of 1.5 - 2.25 cm (T1/2 level) in our patient population.
Two data forms, A and B, were used to capture information on each patient. The reference points noted above were used by the first author on Form A to record the position of the ETT. The correct patient information was noted at the top of both forms, also by the first author, who handed Form B to an independent 'blind' radiologist, who assessed the early morning 06h00 CXR and noted the position of the ETTs on Form B.
Statistical analysis of data was done by the Department of Biostatistics, University of the Free State. Results were summarised by frequencies and percentages (categorical variables) and medians (numerical variables, owing to skew distributions).
Before commencement of the study, ethical approval was obtained from the Ethics Committee, Faculty of Health Sciences, University of the Free State. Permission to conduct the study was also obtained from the Clinical Head of the Universitas Academic Hospital in Bloemfontein. Written parental consent was obtained for every participant.
Results
The 30 patients included in this study had a median age of 13.5 (range 2 - 38) days, and a median weight of 1.6 (1.2 - 3.1) kg. A median delay of 48 minutes between CXR and BUS occured. Table 1 outlines the positions of ETTs according to the early morning CXR, showing the exact position/level of the ETTs in relation to the vertebral bodies. As shown, 73.3% of ETTs were considered to be placed optimally when CXR was viewed, with optimal being defined by the radiologist as being between C7/T1 disk space and T2/3 disk space.
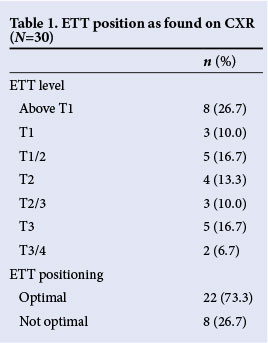
Table 2 outlines the position of the ETTs as observed with BUS and the patient's head position during BUS. According to BUS findings, 93.3% of ETTs were placed optimally, which was defined (objectively) as being at the lower level of the thyroid or just below the thyroid.
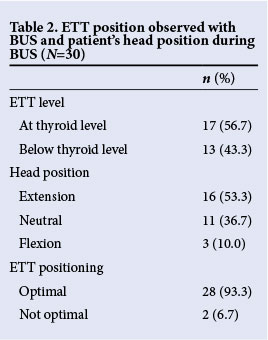
Table 3 compares CXR and BUS findings regarding optimal placing. The agreement between these findings was poor (κ=0.10; 95% CI -0.2 - 0.4).
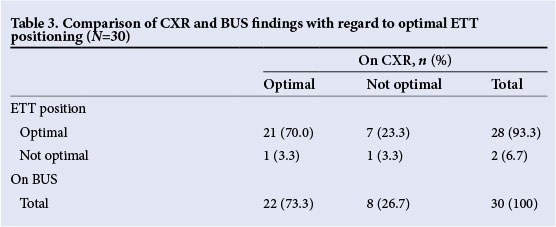
Table 4 shows the measured distances from the aortic arch to the tip of the ETT as obtained by BUS. The thoracic vertebral body height of our specific patient population, including the intervertebral disk space, was ~7.5 mm. The estimated distance from T4 (level of aortic arch) to T2 was 1.5 cm, and 2.25 cm from T4 to T1. Ideally, the tip of the ETT should be ~1.5 -2.25 cm from the level of the aortic arch (at T1/T2 level). In two (6.7%) patients, the measurements were <1.5 cm (T3 level) and in another two (6.7%), it was >2.25 cm (C7 level).
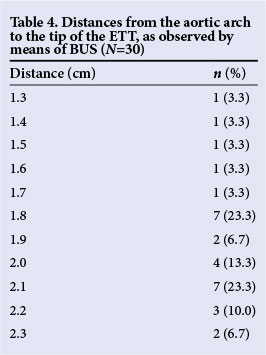
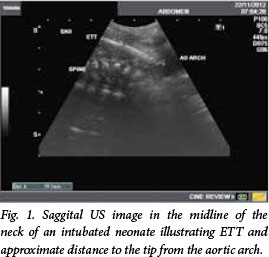
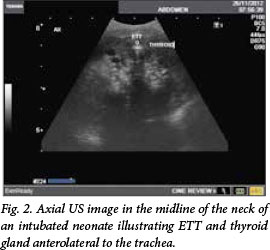
Fig. 1 shows a saggital US image in the midline of the neck of an intubated neonate, illustrating ETT and approximate distance to the tip from the aortic arch. Fig. 2 shows an axial US image in the midline of the neck of an intubated neonate, illustrating ETT and thyroid gland anterolateral to the trachea.
Discussion
ETTs in all the patients included in this study could be visualised very well using BUS, which was found feasible and easy to use when applying relevant surface anatomy to determine the optimal position. It can also be used in real time during intubation, without the risk of radiation exposure. If the position of ETT according to BUS and clinical evaluation is unsatisfactory, alterations can be made immediately in real time and confirmed by using BUS, making the procedure time- and cost-effective.
Repositioning of ETTs by handling the babies or altering their head positions from flexion (most CXR head positions) to extension was most likely the main reason why BUS and CXR findings were not comparable.[11] Because small babies have short necks and a relatively small acoustic window in which to do BUS, head position should at least be neutral, although extension is the ideal position to visualise ETTs with BUS. Another possible reason for poor correlation between CXR and BUS was because the head positions of participants were not standardised or recorded when CXRs were performed for this study, a limitation which was not anticipated before the study commenced.
As noted in the Methods, ETTs should be above the arch of the aorta, and below or at the lower level of the thyroid to be correctly in position. Both of these reference points can be observed easily and very well with BUS. These reference points stay constant for every baby independent of weight or size.
Study limitations
Possible measurement errors included repositioning of the ETT during the interval between the CXR and BUS examinations. An ETT can move up to 3.1 mm with neck flexion and up to 7.4 mm with extension in LBW infants,[11] the latter being the ideal position for BUS to visualise the ETT optimally.
Conclusion
Although anatomical reference points for BUS and CXR determination of ETT positioning are not comparable, BUS was found to be an easy, feasible alternative method to determine the optimal position of ETTs in the trachea of neonates when using relevant surface anatomical reference points. It can also be particularly useful in limiting radiation exposure of neonates. More refinement of this method is needed; doctors attending to this patient population would need to be trained to use the technique themselves and to gain experience and confidence in performing the procedure, which is a method worthwhile exploring.
Acknowledgements. We would like to thank Dr W Harmse, diagnostic radiologist, for interpretation of CXRs, and Dr D Struwig, medical writer, Faculty of Health Sciences, University of the Free State, for technical and editorial preparation of the manuscript.
References
1. Hsieh KS, Lee CL, Lin CC, Hwang TC, Weng KP, Lu WH. Secondary confirmation of endotracheal tube position by ultrasound image. Crit Care Med 2004;32(9 Suppl):S374-S377. [http://dx.doi.org/10.1097/01.CCM.0000134354.20449.B2] [ Links ]
2. Galicinao J, Bush AJ, Godambe SA. Use of bedside ultrasonography for endotracheal tube placement in pediatric patients. Pediatrics 2007;120(6):1297-1303. [http://dx.doi.org/10.1542/peds.2006-2959] [ Links ]
3. Dennington D, Vali P, Finer NN, Kim JH. Ultrasound confirmation of endotracheal tube position in neonates. Neonatology 2012;102(3):185-189. [http://dx.doi.org/10.1159/000338585] [ Links ]
4. Kerrey BT, Geis GL, Quinn AM, Hornung RW, Ruddy RM. A prospective comparison of diaphragmatic ultrasound and chest radiography to determine endotracheal tube position in pediatric emergency department. Pediatrics 2009;123(6):e1039-1044. [http://dx.doi.org/10.1542/peds.2008-2828] [ Links ]
5. Hosseini JS, Talebian MT, Ghafari MH, Eslami V. Secondary confirmation of endotracheal tube position by diaphragm motion in right subcostal ultrasound view. Int J Crit Illn Inj Sci 2013;3(2):113-117. [http://dx.doi.org/10.4103/2229-5151.114269] [ Links ]
6. Adi O, Chuan TW, Rishya M. A feasibility study on bedside upper airway ultrasonography compared to waveform capnography for verifying endotracheal tube location after intubation. Crit Ultrasound J 2013;5(1):7. [http://dx.doi.org/10.1186/2036-7902-5-7] [ Links ]
7. Schmvlzer GM, Roehr CC. Techniques to ascertain correct endotracheal tube placement in neonates. Cochrane Database Syst Rev 2014;9:CD010221. [http://dx.doi.org/10.1002/14651858.CD010221.pub2] [ Links ]
8. Butler P, Mitchell AWM, He ally JC. Applied Radiological Anatomy. 2nd ed. Cambridge: Cambridge University Press, 2006:2-4. [ Links ]
9. Blayney MP, Logan DR. First thoracic vertebral body as reference for endotracheal tube placement. Arch Dis Child Fetal Neonatal Ed 1994;71(1):F32-F35. [http://dx.doi.org/10.1136/fn.71.1.F32] [ Links ]
10. Thayyil S, Nagakumar P, Gowers H, Sinha A. Optimal endotracheal tube tip position in extremely premature infants. Am J Perinato 2008;25(1):13-16. [http://dx.doi.org/10.1055/s-2007-995221] [ Links ]
11. Rost JR, Frush DP, Auten RL. The effects of neck position on endotracheal tube location in low birth weight infants. Pediatr Pulmonol 1999;27(3):199-202. [http://dx.doi.org/10.1002/(SICI)1099-0496(199903)27:3%3C199::AID-PPUL8%3E3.0.C0;2-0] [ Links ]
 Correspondence:
Correspondence:
S H de Kock
sharonhdekock@yahoo.com














