Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
South African Journal of Child Health
versión On-line ISSN 1999-7671
versión impresa ISSN 1994-3032
S. Afr. j. child health vol.8 no.3 Pretoria sep. 2014
RESEARCH
Fluoroscopic guided benign oesophageal stricture dilatation in children: 12 years' experience
A ReindersI; M J van WykII
IMB ChB, DCH (SA); Department of Clinical Imaging Sciences, University of the Free State, Bloemfontein, South Africa
IIMB ChB, MMed Diag Rad, FC Rad D (SA); Department of Clinical Imaging Sciences, University of the Free State, Bloemfontein, South Africa
ABSTRACT
BACKGROUND: Benign oesophageal strictures (BOSs) are a debilitating health concern in the paediatric populations of developing countries, which lead to impaired weight gain. Various non-surgical methods may be used to address these strictures.
OBJECTIVES: To compare the success and complications of fluoroscopically guided Savary-Gilliard bougienage (SGB) with balloon dilatation and the combination of both methods for the treatment of BOSs in children at Universitas Hospital, Bloemfontein, South Africa.
METHODS: A retrospective review of the patient notes on all children <12 years who underwent oesophageal dilatation for benign strictures from January 2001 to June 2012 was performed. Interventional radiologists performed the dilatations on children under general anaesthesia either by angioplasty balloon, SGB or a combination of both. Postdilatation contrast swallows were done to confirm the absence of perforation.
RESULTS: A total of 432 oesophageal dilatations was performed on 63 children aged <12 years. Of these, 36 were males (57%), and 71% were exclusive balloon dilatation, 19% exclusive SGB and 10% utilised both techniques. Five dilatations failed (1.2%) and no complications were documented. Average follow-up dilatations needed per procedure were 8.3 after SGB, 7.2 after balloon dilatation, and 4.2 after a combined method (p<0.05). Strictures due to caustic ingestion required, on average, more dilatations (n=11) compared with those in oesophageal atresia (n=6).
CONCLUSION: Fluoroscopically guided dilatation of benign oesophageal strictures by either balloon catheter dilatation or SGB or a combination of the two methods is a safe and effective treatment in our paediatric population.
Benign oesophageal strictures (BOSs) are a debilitating health concern in the paediatric populations of developing countries, and lead to impaired weight gain, second only to malnutrition.[1]
Since the early seventeenth century, treatment of these strictures was undertaken with tapered candles by the people of Bijayah - a small centre in North Africa - birthing the term 'bougienage.[2] Surgical techniques soon followed, but were marred by a postoperative complication rate of as much as 20%. The complications included, among others, anastomotic leakage, mediastinitis and stricture formation.[2] Alternative techniques were only considered in the early 1980s with the introduction of a Gruentzig-type balloon dilatation technique, boasting a lower complication rate, decreased number of dilatations and ease of use.[1]
Several aetiologies for BOSs have been documented, ranging from stenosis secondary to trachea-oesophageal fistula repair (18 -50%), caustic ingestion (38%), achalasia (5%), epidermolysis bullosa (<1%) and gastro-oesophageal reflux, to name a few.[3,4] All have showed a good 1-year follow-up success rate with balloon dilatation techniques.[1-5]
Differences in successful BOS balloon dilatations were noticed with certain aetiologies, varying from superior results after trachea-oesophageal fistula repair or gastro-oesophageal reflux disease'11 to poorer success rates in BOSs secondary to caustic ingestion (33%).[5] One study argued that balloon dilatation decreased hospital stay and postprocedural complications in comparison with bougienage dilatations.[5] In the available literature, bougie-nage dilatations in the paediatric population are few and far between, in part for fear of the 'axial shearing forces' exerted on the already stenotic oesophagus in contrast to the 'radial force' exerted during balloon dilatation,'11 and in part because of good results obtained with the alternative balloon technique.'1-4,61 Even though previous reports in a similar subset of adult patients treated with Savary-Gilliard bougienage (SGB) did show a low complication rate, corresponding data for paediatric patients are still lacking.[2]
Objective
The objective of this retrospective review is to describe the technical success and procedural complications of fluoroscopically guided SGB and balloon dilatation for the treatment of BOSs in children aged <12 years at our tertiary centre.
Methods
Ethical review board clearance as well as institutional approval were obtained to perform the review.
A retrospective review of all the procedure and clinical notes as well as contrast swallow reports on children <12 years who underwent oesophageal dilatation for benign strictures during the period from January 2001 to June 2012 at Universitas Hospital, Bloemfontein, was performed.
Interventional radiologists performed the dilatations. As per institutional protocol, the patients were fasted to empty the stomach in preparation for the procedure. All procedures were performed under general anaesthesia with endotracheal intubation. A per-oral antegrade approach was used in all patients. The oesophageal stricture was traversed using a 0.035 inch angled glide wire (Terumo Medical Corporation, Japan), under fluoroscopic guidance (Fig. 1).
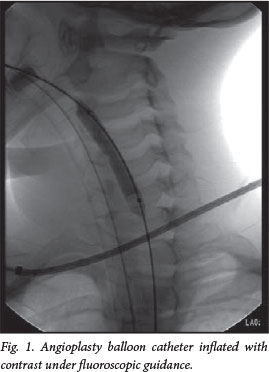
The guide wire was securely curled up in the stomach and exchanged for an Amplatz Super Stiff guide wire (Boston Scientific, USA) via a multipurpose catheter. Gruentzig-type angioplasty balloon catheters (Fig. 2) of increasing size were placed over the stiff guide wire and correctly positioned under fluoroscopic guidance. Balloons ranging from 4 mm to 14 mm were used in the procedures. The size of the balloon was chosen in accordance with the interpedicular distance or rule of thumb as described in previous papers.[7] These balloons were inflated by hand, with diluted radio-opaque contrast, at the stenotic segment. Inflation was sustained for 60 seconds. Initially, a waist was noted at the stenosis, which obliterated on progressive dilatations. Dilatations were augmented or replaced by SGB (Fig. 3) in older children where the maximal available balloon size (14 mm) had been reached but the stricture had not adequately dilated. All strictures, no matter the aetiology, were initiated with balloon dilatations and later augmented with SGB according to the interventional radiologist's discretion. The end-point of dilatation was reached as soon as the waist disappeared and resistance was encountered on passage of the balloon catheter or bougie. The maximum size of the balloon corresponded to the diameter of the adjacent normal oesophagus. Post dilatation, a suction catheter was inserted over the guide wire and then aspirated on retraction to evacuate secretions. A water-soluble contrast swallow was performed after the procedure, in the awake patient, to exclude oesophageal perforation. The patients were generally discharged on the same day as soon as the effects of anaesthesia had worn off and they could tolerate oral feeds. Repeat dilatations were performed at scheduled intervals as dictated by the radiologist and as mandated by the patient's symptoms. Each child was assigned an individual set of dilatation balloons, which were resterilised for future use after each dilatation procedure.

The patients' charts were reviewed and the following data were obtained: gender, age at first dilatation, dilatation date, cause of stricture, maximum balloon or bougie size, time interval between serial dilatations, technical success and complications.
Safety was defined as the absence of complications within 48 hours post oesophageal dilatation. Any unexpected adverse event relating to the dilatation resulting in morbidity or mortality was recorded. Technical success was achieved when a balloon could negotiate a stricture and a dilatation could be performed. A stenosis impossible to cross with even the smallest balloon was regarded as a failed procedure. Efficacy was defined as primary oesophageal patency after dilatation.
Results were summarised by frequencies and percentages (categorical variables) and means and standard deviations or percentiles (numerical variables, depending on the distribution of the data). Groups were compared using χ2 or Fisher's exact tests for categorical variables and ANOVA, t-tests, Kruskal-Wallis tests, or Mann-Whitney tests for numerical variables.
Results
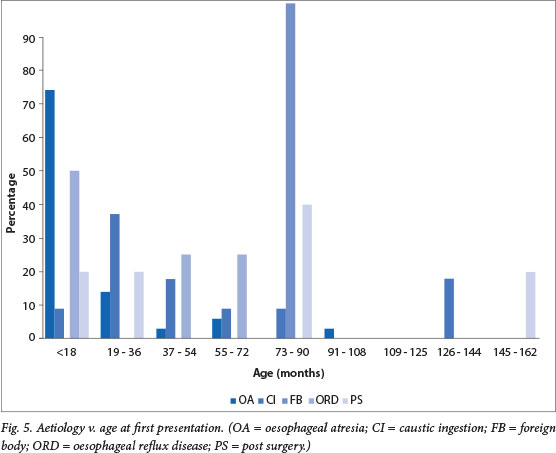
During the period from January 2001 to June 2012, 432 dilatations were performed on 63 paediatric patients aged <12 years, of which 36 were males (57%) and 27 were females (43%). Most children presented at <18 months of age (51%) (Fig. 4). The youngest patient was 21 days old and the oldest was 12 years and 3 months. The median age was 17.7 months. Females tended to present earlier, with 92% presenting within 54 months of age, v. only 65% of male patients.

Causes for oesophageal strictures included post-surgical stenosis secondary to oesophageal atresia post repair (59%), caustic ingestion (20%), oesophageal reflux disease (9%) surgical causes (8%) and foreign body ingestion (4%).
When aetiology was related to age, a marked predominance in oesophageal atresia in the age group <18 months (74%) was seen. Caustic ingestion had an increased prevalence in children between 18 and 36 months of age (37%) and foreign body ingestion in children 73 - 90 months of age (100%) (Fig. 5).
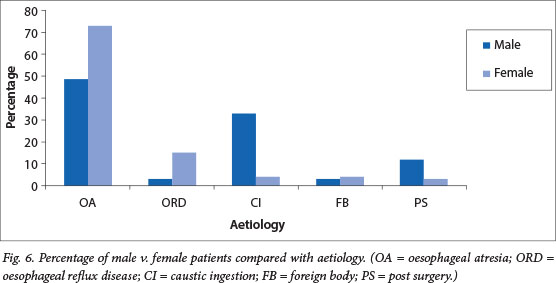
When aetiology was compared with gender, oesophageal atresia was the predominant cause in female patients (73%). Caustic ingestion was noted to be higher in males (52%), with oesophageal reflux disease predominating in female patients (24% v. 3% in male patients) (Fig. 6).
Of the total (N=432) number of dilatations performed, 427 dilatations (98.8%) were regarded as successful. A total number of 305 (71%) were exclusive balloon dilatations, 81 (19%) received SGB alone and 41 (10%) required a combination of both methods.
Failed dilatations were noted in five patients (1.2%). Reasons for unsuccessful dilatations were not explained in the patient notes. No complications were noted during the procedure for any patients. The average number of dilatations per child was 6 for oesophageal atresia and 11 for caustic ingestion.
On average, the follow-up number of dilatations needed after index balloon dilatation were 8.3 times after SGB, 7.16 times after balloon dilatation, and 4.15 times after the combined method (p<0.05). Median number of dilatations per patient per cause is depicted in Fig. 7. The median follow-up time after dilatation was noted to be shorter in patients treated with only balloon dilatation (1.8 months) v. SGB (3 months). The median follow-up time of the combined method was noted to be 2.6 months.

Discussion
The treatment of unassisted and fluoroscopically assisted BOS, by means of balloon or rigid dilatation techniques, has been well described.[1-5] In the past, fluoroscopically assisted balloon dilatation techniques have formed the mainstay and primary treatment option in children with oesophageal strictures in many parts of the world.[1-4,8] Although different aetiological factors cause such strictures, two main categories exist,[4] namely strictures post surgical repair of oesophageal atresia (OA) - with or without trachea-oesophageal fistula - and others, of which caustic ingestion and gastro-oesophageal reflux disease predominate.
The incidence of oesophageal atresia between the sexes differed slightly, with a male to female ratio of 1.3:1.[9] In our study population, OA predominated as the main cause of BOS in children <18 months old, and showed a marked female predominance of 2.7:1, which was statistically significant (p<0.01). Serhal et al.[8] alluded to the fact that the presence of post-OA repair oesophageal strictures could be linked to the primary gap length and anastomotic tension repair. As noted by Brown and Tam in Lan et al.,[10] gastro-oesophageal reflux disease is also a common concurrent factor in patients with oesophageal atresia, increasing the likelihood of a stricture postoperatively. We concur with this, as 24% of the female and 5% of the male patients in the OA group also had gastro-oesophageal reflux disease. Caustic ingestion followed as the second most common aetiology, but only presented at an older age and was mainly seen in male patients, whereas foreign body ingestion and postsurgical complications only accounted for a small number of cases.
Although international literature denotes balloon dilatation to be superior to SGB in the management of benign oesophageal strictures in children,[1-7] especially as a result of caustic injury,[11] we noted no complications with either balloon or SGB dilatation techniques despite the aetiology. This is currently better than the reported international standard, which is benchmarked at a complication rate of less than 2% (Table 1).[7]
We concur with Serhal et al.,[8] who previously described bougienage dilatation in a similar subset of OA patients to be >85% effective when done by a skilled operator.
Another contentious issue is that of conscious sedation v. general anaesthesia (GA) when performing the dilatation procedure. Even though Yeming et al.[12] described the significant advantage of balloon dilatation over SGB to be the omission of GA, we did not note any GA-related complication in our population. Intubation may have the added benefit of protecting the patient's airway from aspiration during the procedure.
We demonstrated a 98.8% success rate with only five failed dilatations out of the 432 performed. The reasons for the failed dilatations were not elaborated on in the patient notes.
No statistically significant differences in median follow-up times were noted with the balloon, bougie or combined method when compared with one another (p=0.1). This could either have been due to the lack of standard follow-up dilatation protocol or due to overly concerned parents, increasing the number of hospital visits. However, fewer follow-up dilatations were needed with the combined method (both balloon and SGB dilatations) (4.15 dilatations) v. patients treated with either balloon (8.3 dilatations) or SGB alone (7.16 dilatations), which was statistically significant. We postulate this to be due to the combined cylindrical and shearing forces exerted on the oesophagus, likely causing microtears and subsequent mucosal remodulation in both axial and longitudinal directions, with improved functionality and dilatation durability as the outcome.
Owing to the fact that we assigned a specific set of angioplasty balloons to each individual child, costs were markedly reduced. As far as we are aware, this study constitutes one of the largest dilatation series to date. Our technique has not been described in the paediatric population as of yet and, according to our dataset of patients, the combined method of dilatation proved to be a safe addition to balloon or SGB dilatations.
Study limitations
Our study was limited by a number of factors. First, there was a lack of a standardised follow-up programme post dilatation procedure. The time interval between repeat dilatations was decided according to the specific interventional radiologist's discretion and this made the quantification of durability of the dilatation procedure impossible. It was also unclear what guideline for follow-up was given to the parent by the treating physician, other than to return on dysphagia. A second limitation was the retrospective nature of this review. Insufficient explanation of the technical failures was obtained from the notes. The retroviral status of our patients was also not available in the clinical notes. Therefore, we could not draw a direct causal link between HIV status and aetiologies, which might have skewed our results. Lastly, none of our patients had preprocedural gastroscopies to conirm or deny the presence or absence of candida oesophagitis, which could have potentially explained more regular hospital visits by patients. These limitations should be addressed in future studies.
Conclusion
We conclude that, in our population of children, a meticulous, fluoroscopically guided oesophageal dilatation by means of balloon catheter, SGB or a combination of both is a safe and effective treatment for benign oesophageal strictures. The combined method, however, results in fewer repeat dilatations. These results may be valuable in guiding successful benign oesophageal dilatation techniques in future.
Acknowledgements. We would like to acknowledge and thank Prof. G Joubert (Department of Biostatistics), Dr W S Harmse and Dr E Loggenberg (diagnostic radiologists, Department of Clinical Imaging Sciences) from the University of the Free State for their valued contributions in the preparation and inal layout of this article.
References
1. Ko HK, Shin JH, Song HY, et al. Balloon dilatation of anastomotic strictures secondary to surgical repair of oesophageal atresia in a pediatric population: Long-term results. J Vasc Interv Radiol 2006;17(8):1327-1333. [http://dx.doi.org/10.1097/01.RVI.0000232686.29864.0A] [ Links ]
2. Khanna S, Khanna S. Management of benign oesophageal strictures in children. Indian J Otolaryngol Head Neck Surg 2008;60(3):218-222. [http://dx.doi.org/10.1007/s12070-008-0081-2] [ Links ]
3. Weintraub JL, Eubig J. Balloon catheter dilatation of benign oesophageal strictures in children. J Vasc Interv Radiol 2006;17(5):831-835. [http://dx.doi.org/10.1097/01.RVI.0000217964.55623.19] [ Links ]
4. Fasulakis S, Andronikou S. Balloon dilatation in children for oesophageal strictures other than those due to primary repair of oesophageal atresia, interposition or restrictive fundoplication. Pediatr Radiol 2003;33(10):682-687. [http://dx.doi.org/10.1007/s00247-003-1011-9] [ Links ]
5. Lisy J, Hetkova M, Snajdauf J, Vyhnanek M, Stanislav T. Long term outcomes of balloon dilatation of oesophageal strictures in children. Acad Radiol 1998;5(12):832-835. [http://dx.doi.org/10.1016/s1076-6332(98)80243-2] [ Links ]
6. Said M, Mekki M, Golli M, et al. Balloon dilatation of anastomotic strictures secondary to surgical repair of oesophageal atresia. Br J Radiol 2003;76(901):26-31. [http://dx.doi.org/10.1259/bjr/64412147] [ Links ]
7. Jayakrishnan VK, Wilkinson AG. Treatment of oesophageal strictures in children: A comparison of fluoroscopically guided balloon dilatation with surgical bouginage. Pediatr Radiol 2001;31(2):98-101. [http://dx.doi.org/10.1007/s002470000368] [ Links ]
8. Serhal L, Gottrand F, Sfeir R, et al. Anastomotic stricture after surgical repair of oesophageal atresia: Frequency, risk factors, and efficacy of oesophageal bougie dilatations. J Pediatr Surg 2010;45(7):1459-1462. [http://dx.doi.org/10.1016/j.jpedsurg.2009.11.002] [ Links ]
9. Dumon JF, Meric B, Sivak MV Jr, Fleischer D. A new method of esophageal dilatation using Savary Gilliard Bougies. Gastrointest Endosc 1985;31(6):379-382. [http://dx.doi.org/10.1016/S0016-5107(85)72252-3] [ Links ]
10. Lan LCL, Wong KKY, Sprigg A, et al. Endoscopic balloon dilatation of oesophageal strictures in infants and children: 17 years' experience and a literature review. J Pediatr Surg 2003;38(12):1712-1715. [http://dx.doi.org/10.1016/j.jpedsurg.2003.08.040] [ Links ]
11. Allmendinger N, Hallisey MJ, Markowitz SK, Hight D, Weiss R, McGowan G. Balloon dilation of oesophageal strictures in children. J Pediatr Surg 1996;31(3):334-336. [ Links ]
12. Yeming W, Somme S, Chenren S, Huiming J, Ming Z, Liu DC. Balloon catheter dilatation in children with congenital and acquired oesophageal anomalies. J Pediatr Surg 2002;37(3):398-402. [http://dx.doi.org/10.1053/jpsu.2002.30844] [ Links ]
 Correspondence:
Correspondence:
A Reinders
(antoinette.reinders@gmail.com)














