Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
South African Journal of Child Health
versión On-line ISSN 1999-7671
versión impresa ISSN 1994-3032
S. Afr. j. child health vol.8 no.1 Pretoria feb. 2014
CASE REPORT
Salivary gland anlage tumour - a rare cause of congenital nasal obstruction
T DanillerI; J GoedhalsII; R Y SeedatI
IMB ChB, MMed ORL, FCORL (SA); Department of Otorhinolaryngology, University of the Free State and Universitas Academic Hospital, Bloemfontein, South Africa
IIMB ChB, MMed Anat Path; Department of Anatomical Pathology, University of the Free State and National Health Laboratory Service, Bloemfontein, South Africa
ABSTRACT
Salivary gland anlage tumour, also known as congenital pleomorphic adenoma, is a rare salivary hamartoma that presents in the nasopharynx of neonates. It is important to distinguish this lesion from other causes of neonatal respiratory distress. Treatment usually involves simple excision, and recurrence is rare. No cases of malignant progression have been reported in the literature. We report an interesting case referred to our unit a day after birth with respiratory distress.
Salivary gland anlage tumour (SGAT), or congenital pleomorphic adenoma, is a rare salivary hamartoma that presents in the nasopharynx of neonates. We report an interesting case referred to us a day ater birth with a suspected diagnosis of choanal atresia.
Case report
After an uncomplicated pregnancy, a term (37 weeks) baby was delivered to a healthy primigravid woman by caesarean section for fetal distress at a small district hospital in the Free State Province, South Africa. The Apgar scores were reported to be 8 and 9 at 1 and 5 minutes, respectively. The birth weight was 2 840 g. The baby became distressed ater delivery with intercostal recession and intermittent cyanosis, but appeared to be normal otherwise with no syndromic stigmata. A diagnosis of suspected choanal atresia was made owing to inability to pass a nasogastric tube through either of the choanae, and the baby was referred to the neonatal intensive care unit (NICU) at Universitas Hospital, the tertiary referral centre for the Free State.
On arrival, the baby maintained good oxygen saturation after placement of an oropharyngeal airway, despite copious secretions. The baby was stabilised and special investigations were performed as deemed necessary. We were unable to perform flexible endoscopy successfully at the bedside.
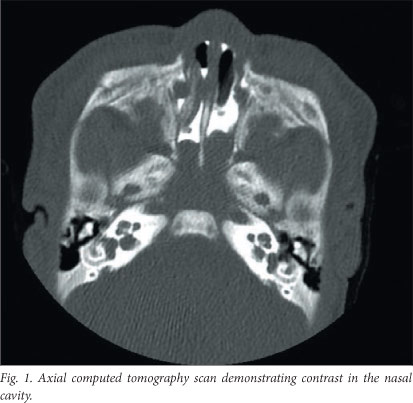
A computed tomography (CT) scan, with contrast placed into the nostrils, showed a nasopharyngeal soft-tissue mass of heterogeneous density, with no flow-through into the nasopharynx (Fig. 1). No intracranial extension or bony erosion of the skull base was evident, but unfortunately we did not perform ancillary magnetic resonance imaging (MRI). The baby was booked for urgent examination under anaesthesia. Examination in theatre revealed a very narrow right nasal passage, and it was impossible to pass the nasal endoscope into the posterior choana. Endoscopy of the left nostril revealed a pinkish-grey soft-tissue mass protruding into the posterior choana, which was biopsied. On elevation of the soft palate, a soft-tissue mass was visible and seemed to prolapse out of the nasopharynx and into the oropharynx after biopsy.
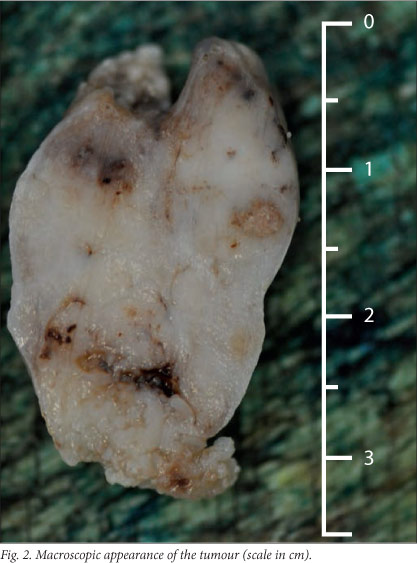
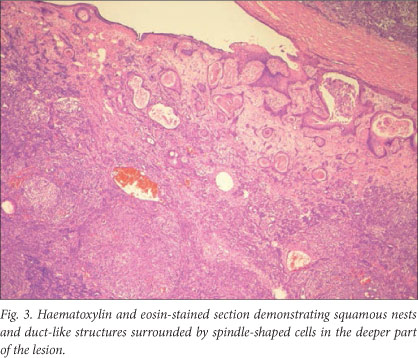
Histopathological examination revealed a lesion composed of duct-like structures surrounded by solid nodules and sheets of spindle-shaped cells, compatible with a diagnosis of SGAT. Immunohistochemical stains for AE1/AE3, MNF116 and EMA were positive and stains for p63, actin and calponin demonstrated scattered intervening positive cells. Stains for S100, desmin, CD34 and myogenin were negative. The mass was removed via a combined transnasal transoral approach. Bleeding was minimal and excision appeared complete on macroscopic examination (Fig. 2). The mass measured 32x20x10 mm, was well circumscribed, and on cut section was solid and white in colour. The appearance on histological examination was consistent with that of the biopsy specimen, with squamous nests and duct-like structures surrounded by solid groups of plump spindle-shaped cells, confirming the diagnosis of SGAT (Fig. 3).
The baby was admitted to the NICU postoperatively and intubated for approximately 12 hours. The condition was stable on discharge, and follow-up thus far has been unremarkable.
Definitive histological examination later confirmed excision of the entire tumour, the features of which were unchanged from the initial report.
Discussion
The list of possible causes for nasal obstruction in the neonatal period is a long one, the most common being nasal mucosal oedema, infectious causes such as syphilis (rare nowadays), bony abnormalities such as pyriform aperture stenosis, choanal atresia, cystic lesions such as a dacrocystocele or Rathke's pouch cyst, Thornwaldt cyst, and other developmental midline nasal lesions such as a meningo-encephalocele or glioma. Neoplastic lesions are usually benign, and include dermoids, hamartomas, teratomas and haemangiomas. Malignant lesions such as neuroblastoma, rhabdomyosarcoma or lymphoma may be seen, but are extremely rare, and metastatic disease even more so. Nasopharyngeal obstruction due to adenoidal hypertrophy is uncommon in the neonatal period.[1] Iatrogenic and traumatic causes should also be sought. Clinical examination and the history should assist in deciding on further management and investigations.
Examination of the upper aero-digestive tract by means of fibre-optic endoscopy is advantageous, as anterior rhinoscopy is not always helpful. Failing this, an examination under anaesthesia is recommended. Because the position of the nasopharynx makes it difficult to access, multiple diagnostic approaches may be required for visualisation.[2]Anatomical evaluation by means of CT and MRI, preferably both, are essential prior to surgical intervention.[3,4] Intracranial extension can be evaluated and more common pathology identified by the nature of enhancement of the mass, its size, and other necessary information gleaned from such studies.
Our patient had a presumed diagnosis of choanal atresia. Radiological examination, however, hinted at the possibility of an alternative diagnosis. Although there was demonstrable failure of contrast to pass through the posterior choanae, a soft-tissue mass was visible in the nasopharynx. An important finding was the lack of intracranial extension. MRI would have been beneficial in delineating this more clearly, owing to the lack of ossification of the skull base in this age group.[4] No bony remodelling or overt abnormalities were present on the CT scan.
SCAT is ararehamartoma that presents as a midline nasopharyngeal mass in neonates, with only a few cases previously reported.[1-10] The most recent articles in a PubMed search cite approximately 24 known cases.[1] The incidence of the tumour is difficult to estimate, as it is relatively uncommon.
SCAT was first described in the literature by Dehner et al.[3] in 1994. Prior to this description, similar lesions had been described in the literature as early as 1980. These lesions were probably also SGATs.[1-10]
Patients typically present with respiratory distress if nasal obstruction is bilateral, as neonates are obligate nasal breathers.[1,2,5] Patients with unilateral obstruction may present later, but most cases are seen within the first two weeks of life.[1] The tumour has a male predilection and arises from the minor salivary gland tissue on the nasopharyngeal surface of the soft palate, usually pedicled to the posterior septum or posterior nasopharyngeal wall.[1-10] This pedicle may easily be torn with probing, resulting in dislodgement and possible complete and unexpected airway obstruction. Treatment includes simple excision, with no recurrences reported in the literature.[1-10] No risk of malignancy has been reported, but a high mitotic index may mimic a malignant tumour.[10] Periodic follow-up is advisable.
Conclusion
Nasal obstruction in neonates can lead to life-threatening respiratory distress. In this case, we were misled by the initial clinical findings, which suggested a diagnosis of choanal atresia. A CT scan demonstrated a soft-tissue mass filling the nasopharynx, which was proven to be a SGAT and was successfully excised.
Ethics approval
Research ethics committee approval was received.
References
1. Tinsa F, Boussetta K, Bousnina S, et al. Congenital salivary gland anlage tumor of the nasopharynx. Fetal Pediatr Pathol 2010;29(5):323-329. [http://dx.doi.org/10.3109/15513811003796961] [ Links ]
2. Antunes MB, Javia LR, Ransom ER, Kazahaya K. Salivary anlage tumor of the nasopharynx: A case report and review of the literature. Int J Pediatr Otorhinolaryngol Extra 2011;6(2):69-71. [http://dx.doi. org/10.1016/j.pedex.2010.03.005] [ Links ]
3. Dehner LP, Valbuena L, Perez-Atayde A, Reddick RL, Askin FB, Rosai J. Salivary gland anlage tumor ('congenital pleomorphic adenoma'): A clinicopathologic, immunohistochemical and ultrastructural study of nine cases. Am J Surg Pathol 1994;18(1):25-36. [http://dx.doi.org/10.1097/00000478-199401000-00003] [ Links ]
4. Morgensen MA, Lin AC, Chang KW, Berry GJ, Barnes PD, Fischbein NJ.
Salivary gland anlage tumor in a neonate presenting with respiratory distress: Radiographic and pathologic correlation. Am J Neuroradiol 2009;30(5):1022-1023. [http://dx.doi.org/10.3174/ajnr.A1364]
5. Herrmann BW, Dehner LP, Lieu JE. Congenital salivary gland anlage tumor: A case series and review of the literature. Int J Pediatr Otorhinolaryngol 2005;69(2):149-156. [http://dx.doi.org/10.1016/j.ijporl.2004.08.014] [ Links ]
6. Cohen EG, Yoder M, Thomas RM, Salerno D, Isaacson G. Congenital salivary gland anlage tumor of the nasopharynx. Pediatrics 2003;112(1):e66-e69. [http://dx.doi.org/10.1542/peds.112.1.e66] [ Links ]
7. Boccon-Gibod LA, Grangeponte MC, Boucheron S, Josset PP, Roger G, Berthier-Falissard ML. Salivary gland anlage tumor of the nasopharynx: A clinicopathologic and immunohistochemical study of three cases. Fetal Pediatr Pathol 1996;16(6):973-983. [http://dx.doi.org/10.1080/15513819609168721] [ Links ]
8. Michal M, Sokol L, Mukensnabl P. Salivary gland anlage tumor: A case with widespread necrosis and large cyst formation. Pathology 1996;28(2):128-130. [http://dx.doi.org/10.1080/00313029600169723] [ Links ]
9. Har-El G, Zirkin HY, Tovi F, et al. Congenital pleomorphic adenoma of the nasopharynx (report of a case). J Laryngol Otol 1985;99(12):1281-1287. [http://dx.doi.org/10.1017/S0022215100098546] [ Links ]
10. Marien A, Maris M, Verbeke S, et al. An unusual tumour causing neonatal respiratory distress. B-ENT 2012;8(2):149-151. [ Links ]
 Correspondence:
Correspondence:
T Daniller
(danillert@ufs.ac.za)














