Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Water SA
On-line version ISSN 1816-7950
Print version ISSN 0378-4738
Water SA vol.46 n.3 Pretoria Jul. 2020
http://dx.doi.org/10.17159/wsa/2020.v46.i3.8662
RESEARCH PAPERS
SPE-LC-PDA method development and application for the analysis of selected pharmaceuticals in river and wastewater samples from South Africa
NB Hlengwa; PN Mahlambi
Department of Chemistry, University of KwaZulu-Natal, King Edward Avenue, Scottsville, Private Bag X01, Pietermaritzburg, 3209, South Africa
ABSTRACT
The occurrence of pharmaceutical compounds in the environment has been a growing concern. However, there is little information on the concentration levels of pharmaceuticals in African countries. This indicates the need and the importance to develop sensitive methods that can detect these compounds at trace levels (ng/L-µg/L). This work reports on the development of solid phase extraction followed by liquid chromatography-photo diode array (SPE-LC-PDA) analysis. The method was then applied for the analysis of pharmaceutical compounds (ibuprofen, fenoprofen, naproxen, carbamazepine and diclofenac) in river and wastewater samples. The SPE recoveries obtained at 10 000 ng/L spike concentration ranged from 89-120% in distilled water, 79-110% in river water and 78-94% in wastewater. The LODs and LOQs ranged from 10.9-20.4 ng/L and 36.2-60.7 ng/L, respectively. Concentrations of the pharmaceuticals obtained in river water samples ranged from 60 to 32 900 ng/L; in wastewater they ranged from 70 to 66 900 ng/L. Higher concentrations of pharmaceuticals were detected in winter compared to the spring season.
Keywords: solid phase extraction, pharmaceuticals, liquid chromatography, photo diode array, wastewater
INTRODUCTION
Pharmaceutical compounds are drugs used for very important purposes, such as curing, diagnosis and treatment of minor and serious diseases (Archer et al., 2017). Pharmaceutical compounds have been classified as emerging contaminates as a result of the large amount of pharmaceuticals that enter the environment due to the increase in their consumption by animals and humans (Ascar et al., 2013). They have now been included in the list of ecologically active chemical entities under the global United Nations Environmental Programme (UNEP). Pharmaceuticals are not completely metabolized by our bodies; therefore, they are excreted from human bodies, as either metabolites or parent compounds via urine, faeces or sweat, into sewage, and hence make their way into wastewater treatment plants (WWTPs) (Mlunguza et al., 2019). Human waste streams and WWTPS have been regarded as the main source of pharmaceutical residues together with their metabolites entering the environment (Gracia-Lor et al., 2012). The wastewater treatment processes are not specifically intended to remove pharmaceuticals from wastewater; hence, these are disposed into the surface water with the treated WWTP effluents. Recent studies have shown that WWTPs partially remove pharmaceuticals with percentages ranging from negative removal up to 99% (Li et al., 2014; K'oreje et al., 2016; Madikizela et al., 2017; Gruchlik et al. 2018; Mosla et al., 2018; Mtolo et al., 2019). Also, due to the illegal dumping of unused and expired drugs, pharmaceutical compounds can make their way into the environment (Larivière et al., 2016; Madikizela et al., 2020).
Pharmaceuticals have been found to be present in wastewater effluent, drinking water, rivers and dams in various parts of the world (Gilart et al., 2014; López-Roldán et al., 2010; Kanama et al., 2018; Sibeko et al., 2019). Even though the analysis of pharmaceuticals has been conducted for almost a decade in African countries, there is still limited information with regards to their occurrence in the environment (K'oreje et al., 2020). This may be due to limited analytical facilities available; also there is a lack of suitable and sensitive methods that can be used to effectively analyse these organic pollutants (Agunbiade and Moodley, 2016). However, there has been some progress made in the past 3 years on the environmental monitoring and assessment of pharmaceuticals in African countries, and the number of published papers and review articles has steadily increased (including Madikizela et al., 2018; Mbhele et al., 2018; Mosla et al., 2018; Sibeko et al., 2019; Branchet et al., 2019; Mlunguza et al., 2019; Fekadu et al., 2019; Madikizela et al., 2020; K'oreje et al., 2020). A review on the occurrence of pharmaceuticals in freshwater aquatic environments in the African and European continents indicated that ibuprofen, carbamazepine, naproxen and diclofenac were amongst the top 10 frequently detected and quantified compounds. Naproxen was reported to be found at concentrations 171 times higher in African countries than on the European continent (Fekadu et al., 2019).
Amongst the various groups of pharmaceuticals, non-steroidal anti-inflammatory drugs (NSAIDs) are considered as one of the most used acidic pharmaceutical compounds worldwide, because they are easily accessible without any medical prescription needed. The most widely used NSAIDs are naproxen, fenoprofen, diclofenac and ibuprofen (Madikizela, 2017). They are known to have antipyretic, anti-inflammatory and analgesic properties (Wongrakpanich et al., 2018). However, with continuous exposure they may have negative effects on aquatic life and human health, even at trace levels. NSAIDs have also been found to have a negative effect on the species in the environment by affecting the functioning of the endocrine system. Xu et al. (2019) revealed a substantial decrease in triiodothyronine and thyroxine hormone levels in Zebrafish after 60 days of exposure to naproxen at environmentally relevant concentrations. Induced endocrine disruption, alterations of immunological parameters, and genotoxic effects in the mussel Mytilus galloprovincialis were observed after exposure to ibuprofen and diclofenac (Gonzalez-Rey and Beebianno, 2012; Mezzelani et al., 2018).
The sample clean-up and preparation process is one of the most important steps when dealing with organic pollutants in environmental matrices as they present at trace levels. Solid phase extraction (SPE) is usually used for sample preparation and pre-concentration due to its ability to reduce the matrix effect, enhance sensitivity and thus lower the detection limit (K'oreje et al., 2016; Zhang et al., 2017). Also, SPE uses less organic solvent and gives higher analyte recoveries (Manrique-Moreno et al., 2016). SPE Oasis HLB (hydrophobic-lipophilic balanced) cartridge is one of the most effective extraction and clean-up procedures for water samples. This is due to the presence of the neutral polar hook on the HLB cartridges which enhances the retention of polar analytes and thus improves their recoveries (Ntombela and Mahlambi, 2019). The commonly used chromatographic methods for the determination of pharmaceutical compounds are high performance liquid chromatography (HPLC) and gas chromatography (GC). HPLC has been widely used when analysing acidic pharmaceuticals because they are non-volatile, while the derivatization step is needed when GC is to be used in order to increase the volatility of the compounds, which may lead to the formation of unwanted compounds during the process (Sadkowska et al., 2017).
The aim of this study was therefore to develop solid phase extraction followed by liquid chromatography coupled-photodiode array detector (SPE-LC-PDA) method for simultaneous determination of the five most used pharmaceutical compounds (ibuprofen, naproxen, diclofenac, fenoprofen and carbamazepine) in river water and wastewater samples. These compounds were chosen due to their wide and consistent usage as therapeutic agents leading to their continuous release into the environment; they are frequently detected in wastewater and surface water worldwide. On the other hand, they have a negative health impact on humans as well as aquatic organisms even at trace levels; hence their monitoring in various water bodies is essential.
EXPERIMENTAL
Chemicals and reagents
Acetone (99.9%), acetonitrile (99.9%), dichloromethane (99.9%), methanol (99.9%) and ethyl acetate (99.9%) were purchased from Sigma-Aldrich (Steinheim, Germany). Carbamazepine (≥98%), fenoprofen (97%), ibuprofen (≥98%), naproxen (98%) and diclofenac (98%) were purchased from Sigma Aldrich (Durban, South Africa).
Instrumentation
The SPE manifold bought from Sigma Aldrich (Steinheim, Germany) was used for the cleaning up, purification and extraction of the compounds from water samples. SPE was connected to a pump manifold from Edwards (Munic, Germany). Oasis HLB cartridges (60 mg, 3 mL) purchased from Waters (Uppsala, Sweden) were used as SPE sorbents. LC-2020 equipped with Shim-Pack GIST C18-HP column (4.6 x 150 mm, 3 µm) coupled to a 2030/2040 PDA detector purchased from Shimadzu (Europe, Germany) was used for the analysis. The data were acquired at 229 nm. The mobile phase composition was kept as isocratic with 60:40 (acetonitrile: water in 0.1% formic acid) at a flow rate of 0.5 mL/min.
Preparation of standards
100 mg/L standard stock solution containing a mixture of target compounds (naproxen, carbamazepine, fenoprofen, diclofenac and ibuprofen) was prepared by transferring 10 mg of each analyte into a 100 mL volumetric flask and then dissolved this in acetonitrile. Standard working solutions with concentrations ranging from 10 000-1 000 000 ng/L were prepared from the stock solution and used for calibration of LC-PDA. All of the standard solutions prepared were stored at 4°C in the refrigerator and used within 24 h.
Sampling
The wastewater samples were collected in 5 wastewater treatment plants (WWTPs) in KwaZulu-Natal; 4 plants were in the Durban area (Amanzimtoti, Umhlathuzana, Umbilo and Northern WWTP) and 1 plant was in Pietermaritzburg area (Darvill).
The treatment plant receives wastewater from domestic and industrial sources that may contain pharmaceutical compounds. After the wastewater treatment, the effluent is released into the nearby rivers which could introduce the pharmaceuticals into the rivers as the treatment plants lack the proper facilities to completely remove organic pollutants.
Umbilo WWTP is designed to receive organic waste of 500 mg COD/L with a raw sewage inlet flow of 23 m3/day, but the load fluctuates up to 2 000 mg COD/L and receives an average of 650 mg COD/L. The treatment process in this plant consists of chlorination, mechanical and biological treatment. Umbilo treatment plant is surrounded by residential and industrial areas and it is near the Umbilo River where the effluent from the treatment plant is discharged (Poulsen and Lauridsen, 2005).
Darvill WWTP serves a population of over 300 000 people. It receives wastewater which is 30% from domestic and 70% from commercial and industrial sources as well as hospital facilities. The treatment plant is designed to receive about 65 ML/day of wastewater; however, it is currently receiving more than its designed capacity as it has a dry weather inflow load which is above 70 ML/day. The treatment plant process involves aeration by stirring the wastewater influent; thereafter it undergoes sedimentation followed by chlorination. It then discharges the treated effluent into the Msunduzi River (Agunbiade and Moodley, 2016; Matongo et al., 2015).
Umhlathuzana WWTP had been designed to treat inflow of 15 ML/day. It receives influent wastewater from two sources which are the Marianridge and Shallcross suburbs. Shallcross receives about 2 ML/day which is 100% domestic wastewater while Marianridge receives about 8 ML/day which is 30% industrial and 70% domestic. The operation unit consists of screens and grit chambers, the biological reactor which is employed as an extended aeration basin, followed by secondary clarification in secondary settling tanks. The effluent from the two sources is combined and treated with chlorine, and is then discharged into Umhlathuzana River. Umhlathhuzana WWTP receives industrial and domestic wastewater which could all contribute towards pharmaceutical concentrations (Mhlanga and Brouckaert, 2013).
Amanzimtoti WWTP is designed to receive 22 ML/day. Aman-zimtoti WWTP receives wastewater from industries and domestic sources. 2 000 kg/day of thin sludge is back-washed and the chlorine gas is used as a primary disinfectant. The plant discharges its effluent into the Mbokodweni River (Umgeni Water, 2013).
Northern WWTP has a design capacity of 58 ML/day and has undergone expansion to enlarge its treatment capacity to 66 ML/day and flow capacity to 99 ML/day. It discharges its treated effluent to the Mgeni River (Naidoo, 2013).
River water samples were collected from 4 sampling sites along the Msunduzi River in Pietermaritzburg (Camps Drift, College Road, YMCA, Bishopstowe and Woodhouse) (Fig. 1) and 4 sites on rivers in the Durban area (Umbilo, Umhlathuzana, Mbokodweni and Mgeni River), into which the investigated WWTPs discharge their effluent. There are a lot of activities that occur around the Msunduzi River, including agricultural, industrial, and domestic activities. There is also a wastewater treatment plant, which treats, among others, hospital and domestic waste, and discharges its treated effluent into the river. The river is used for illegal dumping of waste from informal settlements, and is very popular for its sporting activities which include fishing and canoeing. These activities occurring around the river could contribute to high pharmaceutical concentrations in the river. Sampling along the Msunduzi River was done during winter and spring seasons while for other sites samples were collected during the winter season only. Samples were collected using the grab sampling approach into pre-cleaned brown glass bottles. All the samples were collected in duplicate at each site; the duplicates were then combined to form a composite sample per site. They were then transported to the laboratory in a portable ice chest where they were kept in the refrigerator at 4°C and analysed within 72 h.
Sample preparation
The sample extraction was done using SPE (Oasis HLB 60 mg, 3 mL). SPE cartridges were conditioned with 2 mL of acetonitrile followed by equilibration with 2mL of distilled water. After conditioning, 50 mL of water sample was loaded through the cartridge to allow the analytes to be adsorbed onto the sorbent. 2 mL of distilled water was used to wash off the impurities and the cartridge was dried under vacuum for 10 min. The adsorbed analytes were then eluted with 2 mL of methanol. The extracts were concentrated to 1 mL with a gentle stream of nitrogen and analysed using liquid chromatography photo diode array (LC-PDA).
Optimization of the analytical method
Optimization of LC-PDA
The LC-PDA method from Versteeg (2014) was adopted and further optimized. The analytical method's conditions, such as flow rate, detector wavelength and mobile phase composition were optimized in order to obtain conditions that allow good separation of the analytes at reasonable retention times.
Optimization of SPE
The SPE conditions were adopted from Lin et al. (2005) and further optimized. SPE parameters that were optimized were sample pH, conditioning solvent, and sample volume, in order to obtain extraction conditions that will give higher recoveries for all the analytes. This was done by using distilled water, spiked with the target compounds to achieve a final concentration of 10 000 ng/L. River water and wastewater recovery tests were conducted under optimum conditions. All analyses were conducted in triplicate.
Validation of the analytical method
The method was validated in terms of the % recoveries, limit of detection (LOD), limit of quantification (LOQ), and linearity. The linearity was investigated between 100 and 1 000 000 ng/L. The LOD and LOQ were calculated as 3 and 10 times the signal to noise ratio, respectively. The recoveries were examined using distilled water samples spiked with 10 000 ng/L of pharmaceuticals. River water and wastewater were also spiked with 10 000 ng/L of pharmaceuticals to assess the recoveries of the compounds under optimum conditions.
RESULTS AND DISCUSSION
LC-PDA optimization
Initially, the mobile phase composition of 60:40% acetonitrile: water in 0.1% formic acid, 254 nm wavelength, and a flow rate of 0.5 mL/min were used. Under these conditions, ibuprofen was not detected; however, the other compounds were well separated with reasonable retention times (5.3-11.4 min). The detector wavelength was then changed to 229 nm and the other parameters were kept constant. At a wavelength of 229 nm, ibuprofen was detected at 12.3 min and good separation for the other analytes was observed with retention times that ranged from 5.4-12.3 min. The effect of the flow rate (0.2, 0.5 and 0.75 mL/min) was also investigated as it is one of the factors that influence peak broadening and peak resolution and thus affect analyte quantification (Meurs, 2016). The flow rate of 0.5 mL/min gave good separation for all of the compounds at reasonable retention times. A slow (0.2 mL/min) flow rate resulted in the longer retention time of the compounds with poor separation. On the other hand, the faster flow rate (0.7 mL/min) decreased the retention time of the compounds; however, a poor resolution was observed. Therefore, the optimum LC-PDA conditions were 60:40% acetonitrile:water in 0.1% formic acid, 229 nm as wavelength, 15 min run time and a flow rate of 0.5 mL/min.
SPE optimization
The effect of conditioning solvent
To investigate the effect of conditioning solvent on the recoveries of the analytes, acetonitrile, dichloromethane and methanol were used, because they are widely used solvents for the extraction of acidic pharmaceuticals (Gumbi et al., 2017). Acetonitrile (89-120%) and methanol (78-99%) gave higher recoveries for most of the compounds (Fig. 2). This indicated that they were both better able to remove the air present in the cartridges and fill the empty spaces in the adsorbent, and thus better penetrated through the pores of the sorbent (Camel, 2003). This resulted in the activation of the sorbent functional groups which allowed effective interaction between the analytes and the sorbent functional groups and thus increased the recovery of the analytes. Higher recoveries for methanol and acetonitrile compared to dichloromethane could be due to their higher polarity.
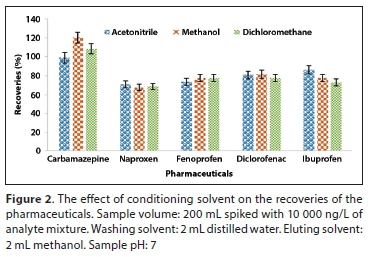
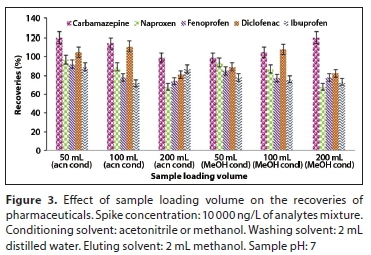
The effect of sample loading volume
The effect of sample loading volume was investigated using a 50, 100 and 200 mL of distilled water sample volume. Methanol and acetonitrile were both separately used as conditioning solvent. Higher recoveries were obtained when 50 mL sample was used with either acetonitrile or methanol as the conditioning solvent. However, the results obtained when acetonitrile was used (89-120%) were within the acceptable limit (80-120%) (Hooda and Wilkinson, 2019) for all target analytes and they were slightly higher compared to those obtained in methanol (78-119%) (Fig. 3). The decrease in recoveries when higher sample volume is used could be an indication that the capacity of the SPE sorbent has been exceeded, hence the cartridge became overloaded and no longer retained the analytes (Madikizela, 2017).
The effect of sample pH
The effect of sample pH was investigated in order to observe its effect on the recoveries of analytes. The pH investigated was acidic pH (2), basic pH (10) and neutral pH (7). The neutral pH gave higher recoveries (79-120%) while the basic and acidic pH gave lower recoveries (Fig. 4). The low recoveries at acidic pH could be due to the protonation of the H atom on the carboxylic group of pharmaceutical compounds (Gros et al., 2006). The reason for low recoveries at basic pH could be that the pH is higher than the pKa value of the compounds; therefore the compounds hydrolyse and thus exist in the anionic form which results in poor adsorption onto the cartridges (Versteeg, 2014; Madikizela, 2017). Higher recoveries obtained at neutral pH could be due to the fact that Oasis HLB cartridges contain lipophilic divinyl benzene and hydrophilic N-vinyl pyrrolidone, which allows sorbing at neutral pH thus improving the recoveries of the analytes (Madikizela et al., 2017). These results are in agreement with those reported in the literature (Lindholm-Lehto et al., 2018; Farré et al., 2008), where higher recoveries were obtained when neutral pH was used with Oasis HLB for the analysis of pharmaceuticals.

The washing and elution steps were not investigated since acceptable recoveries were obtained. This indicated that 2 mL of water as the washing solvent was effective in removing unwanted interferences and 2 mL of methanol as the eluting solvent was strong enough to completely elute the trapped analytes from the sorbent bed. Methanol is known to have high eluotropic and polarity strength which makes a good eluting solvent (Żwir-Ferenc and Biziuk, 2006). Acetonitrile was then taken as the optimum conditioning solvent. Similar findings were reported by Shraim et al. (2017), where acetonitrile was found to be the optimum conditioning solvent for the analysis of pharmaceuticals in wastewater, using Oasis HLB cartridges.
Analytical method validation
Method validation was done based on linearity, LOD, LOQ and recoveries. The linearity (R2) of the method was investigated using 100 000-1 000 000 ng/L of the analyte mixed standards. The calibration curves showed a good correlation for all target analytes with R2 between 0.998 and 1.000, which indicates good precision and accuracy of the optimized LC-PDA method. The LOD and the LOQ were calculated as 3 and 10 times the signal to noise ratio, respectively. The LODs ranged from 10.9-20.4 ng/L and the LOQs ranged from 36.2-68.1 ng/L (Table 1). The low values obtained for LODs and LOQs indicate good sensitivity of the method, which could allow the determination of pharmaceuticals at trace levels. The LOD and LOQ obtained in this study are slightly lower than those reported in literature for the analysis of similar compounds using HPLC coupled with photodiode array detector, which were 14.0-51.0 µg/L and 46.0-170.0 ng/L, respectively (Montagner and Jardim, 2011).
The recoveries obtained ranged from 78 to 120%, which is within the acceptable percentage recovery range of 70-120% (Hooda and Wilkinson, 2019). The recoveries were also shown to be independent of spike concentration in the samples, indicating the direct proportionality of the amount in the sample to that extracted, which is important for accurate quantification (Sibiya et al., 2012). The recoveries obtained in this work are higher than those obtained in the study conducted by Lin et al. (2005), (77-92%). This indicated the importance of optimizing the already published method before application to real samples. The accuracy and precision were expressed in terms of the relative standard deviation (RSD) and were found to be less than 10% (Table 1), which indicated that the optimized method has good precision and accuracy.
Application of the optimized method to real water samples
Concentrations obtained in wastewater samples
The influent samples had higher concentrations compared to the effluent samples (Table 2). This is expected since the influent is raw sewage that has not been cleaned to remove any contaminants (Nikolaou et al., 2007). The low concentration in the effluent could be due to either the adsorption of pharmaceuticals to the solid sludge or their degradation during the activated sludge treatment process, resulting in lower concentrations retained in the water (Grandclément et al., 2017).
Naproxen was found to be higher in the effluent than in the influent at Amanzimtoti. The NSAIDS are conjugated in the liver and are excreted as water-soluble glucuronides and sulfates. In WWTPs the glucuronides produced by bacteria hydrolyse the conjugated NSAIDs back to active parent compounds. Also, some pharmaceuticals are enclosed in faeces and are released into wastewater by microbial activities, which could be the reason for higher concentrations obtained in the effluent than in the influent, and this results in low or negative removal (Grandclément et al., 2017).
Ibuprofen was detected in most of the samples with the highest concentration in Umbilo influent (66 900 ng/L). This could be due to ibuprofen's high therapeutic dose (600 to 1 200 mg/d), of which 70-80% is excreted as the parent compound, or in the form of metabolites. The incompletely absorbed medication is excreted into the sewage system along with the unused drugs that may be disposed of via drains and toilets (Mlunguza et al., 2019) and hence find their way to the treatment plants. It could also be as a result of wastewater from industries, including pharmaceutical manufacturing, which has a high content of organic matter which the organic pollutants can adsorb onto. The plant is also designed for an organic load of about 500 mg COD/L but the treatment plant receives up to 2 000 mg COD/L which indicates that most of the time the plant is overloaded with organic matter. This results in high concentrations being detected in the wastewater samples (Poulsen and Lauridsen, 2005). The concentrations of ibuprofen in the treatment plants were higher in the influents than in the effluents and this could be because a significant portion of ibuprofen is degraded during wastewater treatment resulting in low concentrations in the effluent. Carbamazepine was found to be present in all wastewater samples except Umbilo and Northern effluents. Carbamazepine is an anticonvulsant pharmaceutical that is widely used in the treatment of epileptic patients. Huge amounts of carbamazepine are consumed by individuals as a result of a high maximum daily dose of 1 000 mg which may contribute to the presence of carbamazepine in wastewater even though epilepsy is not a very common disease (Du Plessis and Schloms, 2017; Kong et al., 2014). Also, carbamazepine presence could be due to its continuous release into the environment since it administered for a long time or even for life to the patients. Diclofenac was below detection limits in most of the samples, even though it has been reported to be one of the most commonly detected NSAIDs in the South African aquatic environment (Fekadu et al., 2019; Madikizela et al., 2020). Its low detection could be due to it being photochemically active; hence it might have degraded (Patrolecco et al., 2013).
Darvill WWTP was found to have the highest levels of all the analytes detected in the influent water. This could because the Darvill WWTP is the main treatment plant that treats wastewater for the population of over 300 000 people. It also treats industrial wastewater, as well as wastewater from hospitals and clinics; therefore high concentrations of the compounds are expected (Matongo et al., 2015). The concentrations obtained in this work for Darvill WWTP are higher for ibuprofen (260-66 009 ng/L) and lower for carbamazepine (70-24 000 ng/L) than those reported by Matongo et al. (2015) for the analysis of carbamazepine (910- 2 210 ng/L) and ibuprofen (58 700-117 500 ng/L) in Darvill WWTP. These differences in the concentrations observed in the same treatment plant could be due to different loads of pharmaceuticals that reach the plant at different times/years of sampling. The load is dependent on the consumption rate for each pharmaceutical, the excretion of un-metabolized pharmaceuticals as well as the resistance of pharmaceuticals to biodegradation (Madikizela, 2017).
Concentrations obtained in river water samples collected during the winter season
Ibuprofen was detected in all river water samples except for Umhlathuzana and Mbokodweni. Its concentrations ranged from 340 to 33 900 ng/L, with the highest concentration being for Mgeni River. The higher concentrations of ibuprofen could be because it is widely used as an antipyretic, analgesic and anti-inflammatory agent in South Africa and worldwide (Bessone, 2010; Fekadu et al., 2019; Madikizela et al., 2020). Carbamazepine was less frequently detected (30%), with the concentration ranging from 0.17 to 0.83 µg/L, which could be due to its low water solubility. However, other factors such as consumption and excretion rate may contribute to its low detection. The results obtained in this work are lower for ibuprofen (32 900 ng/L) when compared to those obtained by Matongo et al. (2015) for the analysis of ibuprofen (58 710 ng/L) in water samples from the same area (Msunduzi River, South Africa). On the other hand, for carbamazepine (830 ng/L), the concentrations are in the same order of magnitude as those observed by Matongo with a concentration of 910 ng/L (Matongo et al., 2015). However, the results obtained in this work are higher than those obtained in other South African water studies for ibuprofen, reaching up to 11 000 ng/L (Madikizela et al., 2017) and 1 400 ng/L (Sibeko et al., 2019). They are also higher than those reported for Greece by Nannou et al. (2015) for the analysis done for water samples from Kalamas River, Greece, where ibuprofen concentration was found to be 1 350 ng/L.
YMCA sampling point had the highest concentrations for most of the compounds detected. Higher concentrations could be due to illegal dumping of waste by residents, and possibly improper discharge of waste from clinics and hospitals around the area. In Camps Drift all the analytes were detected; however, they were below the quantification limits, with the exception of ibuprofen which was detected at a concentration of 22 000 ng/L. College Road and Woodhouse only had positive results for ibuprofen. In Woodhouse, the possible source of contamination could be Darvill WWTP effluent, since this site is just downstream from the treatment plant. All of the tested compounds had concentrations that were < LOD in the Umhlathuzana River. The results are shown in Table 3.
Effect of seasonal variations on the concentration levels of pharmaceuticals
Comparatively more pharmaceutical compounds were detected per sampling point along the Msunduzi River during spring (2-4) compared to winter (0-2), except ibuprofen which was detected in all samples during the winter season. However, most compounds showed higher concentrations during the winter season. The factors that influence the seasonal variation of pharmaceuticals in water include the water temperature, precipitation, irradiance, and dilution effect (Sun et al., 2015). The reason for high concentration levels of pharmaceuticals in winter could be due to low solar irradiation and low water temperature, which results in low photo-degradation and bio-degradation rates, respectively. Hence, there is a higher occurrence of pharmaceuticals in winter than in the spring (Sun et al., 2015). The dilution of compounds occurs mainly during spring, with significantly high rainfall, resulting in flooding and thus a decrease in the concentration of pharmaceuticals (Padhye et al., 2014; Osorio et al., 2016). On the other hand, rainfall can result in run-off washing the compounds from the dumpsites, leading to their discharge into the nearby rivers thus increasing their concentrations. Also, the differences in the rate of consumption of each pharmaceutical during each season contribute to differences in their concentration in the environment. These could be the reasons for the different seasonal concentrations detected.
Carbamazepine was detected for all sampling points during spring, except Camps Drift, with higher concentrations compared to the winter season. The presence of carbamazepine in river water could be due to its persistence in the environment. Also, it undergoes little or no degradation during conventional wastewater treatment processes, and hence is disposed of with the effluent to the receiving rivers (Ebele et al., 2017). These results are in agreement with those obtained by Guzel et al. (2018), where higher concentrations of carbamazepine were found during the spring season compared to other seasons.
Ibuprofen was detected at higher concentrations and at almost all sampling sites (95%) during both seasons, with the highest concentration in winter. The reason for this could be high water solubility and low volatility, which suggests a high mobility in the aquatic environment, resulting in it being commonly detected in the environment (Buser et al., 1999). Higher concentrations of ibuprofen in winter have also been reported in literature (Nadais et al., 2018; Lindholm-Lehto et al., 2016).
Fenoprofen frequency of detection was significantly lower (30%) in both seasons and it has been reported to be one of the least frequently detected NSAID pharmaceuticals in the environment (Küster and Adler, 2014; Fkhaida, 2014). This could be due to the conversion of acidic pharmaceuticals into other species during chlorination in the treatment plants, which leads to them being less detected in the environment (Mbhele et al., 2018). Diclofenac was also detected at many sampling points (60%) and with higher concentrations during the spring season. These results are in agreement with those reported in literature, where diclofenac concentrations were higher during the spring season (Loraine and Pettigrove, 2006; Guzel et al., 2018).
The rivers into which the investigated treatment plants discharge their effluent were found to have lower concentrations of pharmaceuticals than for the effluent, except in the case of the Mgeni River (Table 2). This could mean that the traces of pharmaceuticals disposed of as part of the WWTP effluents get diluted as they reach the rivers. It could also possibly indicate that the pharmaceuticals are photodegraded and hence transformed into other products, thus reducing their concentration levels detected in the river. Higher concentrations in the Mgeni River could be due to domestic discharges from informal settlements.
CONCLUSION
The SPE-LC-PDA method was successfully developed and applied for qualitative and quantitative analysis of pharmaceuticals in water matrices. The method recoveries ranged between 89 and 120%. Ibuprofen was detected at the highest concentrations in all samples. The concentrations obtained in real water ranged from 60-28 900 ng/L in river water and 70-66 900 ng/L in wastewater. Influent contained the higher concentrations (100-66 900 ng/L) of the analytes compared to effluent (70-9 450 ng/L). Concentrations recorded in effluent samples indicate that discharges from the treatment plants are one of the sources of environmental contamination by these compounds. The concentrations obtained during the winter season (60-28 900 ng/L) were higher compared to those obtained during the spring season (400-16 200 ng/L); however, spring had the greater number of pharmaceuticals detected. The concentrations found in this study were higher than those reported by other researchers, which is cause for alarm, as these contaminants are a growing concern due to their effect on aquatic life and human health. Also, these compounds do not have set allowable concentration limits; thus the results of this study can contribute towards the information on the concentration levels of pharmaceuticals in South Africa. This can help the policymakers to set allowable limits for South Africa.
ACKNOWLEDGEMENTS
The authors acknowledge the University of KwaZulu-Natal and National Research Foundation (NRF) of South Africa under Thuthuka [grant number 107091] for financial support.
REFERENCES
AGUNBIADE FO and MOODLEY B (2016) Occurrence and distribution pattern of acidic pharmaceuticals in surface water, wastewater, and sediment of the Msunduzi River, Kwazulu‐Natal, South Africa. Environ. Toxicol. Chem. 35 (1) 36-46. https://doi.org/10.1002/etc.3144 [ Links ]
ARCHER E, WOLFAARDT GM and VAN WYK JH (2017) Pharmaceutical and personal care products (PPCPs) as endocrine disrupting contaminants (EDCs) in South African surface waters. Water SA 43 (4) 684-706. https://doi.org/10.4314/wsa.v43i4.16 [ Links ]
ASCAR L, AHUMADA I, LÓPEZ A, QUINTANILLA F and LEIVA K (2013) Nonsteroidal anti-inflammatory drug determination in water samples by HPLC-DAD under isocratic conditions. J. Braz. Chem. Soc. 24 (7) 1160-1166. https://doi.org/10.5935/0103-5053.20130150 [ Links ]
BAROTI C, ROBERTA C, D'ASCENZO G, DI CORCIA A, GENTILI A and SAMPERI R (2000) Monitoring natural and synthetic estrogens at activated sludge sewage treatment plants and in a receiving river water. Environ. Sci. Technol. 34 (24) 5059-5066. https://doi.org/10.1021/es001359q [ Links ]
BESSONE F (2010) Non-steroidal anti-inflammatory drugs: What is the actual risk of liver damage? World J. Gastroenterol. 16 (45) 5651. https://doi.org/10.3748/wjg.v16.i45.5651 [ Links ]
BRANCHET P, ARIZA CASTRO N, FENET H, GOMEZ E, COURANT F, SEBAG D, GARDON J, JOURDAN C, NGOUNOU NGATCHA B, KENGNE I, CADOT E and GONZALEZ C (2019) Anthropic impacts on Sub-Saharan urban water resources through their pharmaceutical contamination (Yaoundé, Center Region, Cameroon). Sci. Total Environ. 660 886-898. https://doi.org/10.1016/j.scitotenv.2018.12.256 [ Links ]
BUSER HR, POIGER T and MÜLLER MD (1999) Occurrence and environmental behavior of the chiral pharmaceutical drug ibuprofen in surface waters and in wastewater. Environ. Sci. Technol. 33 (15) 2529-2535. https://doi.org/10.1021/es981014w [ Links ]
CAMEL V (2003) Solid phase extraction of trace elements. Spectrochim. Acta. Part B, Atomic Spectroscopy 58 (7) 1177-1233. https://doi.org/10.1016/S0584-8547(03)00072-7 [ Links ]
DU PLESSIS J and SCHLOMS B (2017) An investigation into the evidence of seasonal rainfall pattern shifts in the Western Cape, South Africa. J. S. Afr. Inst. Civ. Eng. 59 (4) 47-55. https://doi.org/10.17159/2309-8775/2017/v59n4a5 [ Links ]
EBELE AJ, ABDALLAH M A-E and HARRAD S (2017) Pharmaceuticals and personal care products (PPCPs) in the freshwater aquatic environment. Emerg. Contam. 3 (1) 1-16. https://doi.org/10.1017/S1464793105006950 [ Links ]
FARRÉ M, PETROVIC M, GROS M, KOSJEK T, MARTINEZ E, HEATH E, OSVALD P, LOOS R, LE MENACH K and BUDZINSKI H (2008) First interlaboratory exercise on non-steroidal anti-inflammatory drugs analysis in environmental samples. Talanta 76 (3) 580-590. [ Links ]
FEKADU S, ALEMAYEHU E, DEWIL R and VAN DER BRUGGEN B (2019) Pharmaceuticals in freshwater aquatic environments: a comparison of the African and European challenge. Sci. Total Environ. 654 324-337. https://doi.org/10.1016/j.scitotenv.2018.11.072 [ Links ]
FKHAIDA NM (2014) Fate of pharmaceutical compounds in Wadi Al Qilt catchment area. MSc thesis, Birzeit University. [ Links ]
GILART N, CORMACK P, MARCÉ RM, FONTANALS N and BORRULL F (2014) Selective determination of pharmaceuticals and illicit drugs in wastewaters using a novel strong cation-exchange solid-phase extraction combined with liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 1325 137-146. https://doi.org/10.1016/j.chroma.2013.12.012 [ Links ]
GONZALEZ-REY M and BEEBIANNO MJ (2017). Does non steroidal anti inflammetry (NSAID) ibuprofen induce antioxidant stress and endocrine disruption in mussel Mytilus galloprovincials (2012). Environ. Toxicol. Pharmacol. 33 361-371. https://doi.org/10.1016/j.etap.2011.12.017 [ Links ]
GRACIA-LOR E, SANCHO JV, SERRANO R and HERNÁNDEZ F (2012) Occurrence and removal of pharmaceuticals in wastewater treatment plants at the Spanish Mediterranean area of Valencia. Chemosphere 87 (5) 453-462. https://doi.org/10.1016/j.chemosphere.2011.12.025 [ Links ]
GRANDCLÉMENT C, SEYSSIECQ I, PIRAM A, WONG-WAH-CHUNG P, VANOT G, TILIACOS N, ROCHE N and DOUMENQ P (2017) From the conventional biological wastewater treatment to hybrid processes, the evaluation of organic micropollutant removal: a review. Water Res. 111 297-317. https://doi.org/10.1016/j.watres.2017.01.005 [ Links ]
GROS M, PETROVIĆ M and BARCELÓ D (2006) Development of a multi-residue analytical methodology based on liquid chromatography-tandem mass spectrometry (LC-MS/MS) for screening and trace level determination of pharmaceuticals in surface and wastewaters. Talanta 70 678-690. https://doi.org/10.1016/j.talanta.2006.05.024 [ Links ]
GRUCHLIK Y, LINGE K and JOLL C (2018) Removal of organic micropollutants in waste stabilisation ponds: A review. J. Environ. Manage. 206 202-214. https://doi.org/10.1016/j.talanta.2006.05.024 [ Links ]
GUMBI BP, MOODLEY B, BIRUNGI G and NDUNGU PG (2017) Assessment of nonsteroidal anti-inflammatory drugs by ultrasonic-assisted extraction and GC-MS in Mgeni and Msunduzi river sediments, KwaZulu-Natal, South Africa. Environ. Sci. Pollut. Res. 24 20015-20028. https://doi.org/10.1007/s11356-017-9653-6 [ Links ]
GUZEL EY, CEVIK F and DAGLIOGLU N (2018) Determination of pharmaceutical active compounds in Ceyhan River, Turkey: Seasonal, spatial variations and environmental risk assessment. Hum. Ecol. Risk Assess. 25 (8) 1980-1995. https://doi.org/10.1080/10807039.2018.1479631 [ Links ]
HOODA PS and WILKINSON J (2019) Environmental fate of emerging organic micro-contaminants. J. Appl. Sci. 9 2463. https://doi.org/10.3390/app9152997 [ Links ]
KANAMA KM, DASO AP, MPENYANA-MONYATSI L and COETZEE MAA (2018) Assessment of pharmaceuticals, personal care products, and hormones in wastewater treatment plants receiving inflows from health facilities in North West Province, South Africa. J. Toxicol. 2018 1-15. https://doi.org/10.1155/2018/3751930 [ Links ]
K'OREJE K, VERGEYNST L, OMBAKA D, DE WISPELAERE P, OKOTH M, VAN LANGENHOVE H and DEMEESTERE K (2016) Occurrence patterns of pharmaceutical residues in wastewater, surface water and groundwater of Nairobi and Kisumu city, Kenya. Chemosphere 149 238-244. https://doi.org/10.1016/j.chemosphere.2016.01.095 [ Links ]
K'OREJE K, OKOTH M, VAN LANGENHOVE H and DEMEESTERE K (2020) Occurrence and treatment of contaminants of emerging concern in the African aquatic environment: Literature review and a look ahead. J. Environ. Manage. 254 109752. https://doi.org/10.1016/j.jenvman.2019.109752 [ Links ]
KONG W, LIU N, ZHANG J, YANG Q, HUA S, SONG H and XIA C (2014) Optimization of ultrasound-assisted extraction parameters of chlorophyll from Chlorella vulgaris residue after lipid separation using response surface methodology. J. Food Sci. Technol. 51 (9) 2006-2013. https://doi.org/10.1007/s13197-012-0706-z [ Links ]
KÜSTER A and ADLER N (2014) Pharmaceuticals in the environment: scientific evidence of risks and its regulation. Phil. Trans. R. Soc. B 369 (1656) 20130587. https://doi.org/10.1098/rstb.2013.0587 [ Links ]
LARIVIÈRE A, LISSALDE S, SOUBRAND M and CASELLAS-FRANÇAIS M (2016) Overview of multiresidues analytical methods for the quantitation of pharmaceuticals in environmental solid matrixes: comparison of analytical development strategy for sewage sludge, manure, soil, and sediment samples. Anal. Chem. 89 (1) 453-465. https://doi.org/10.1021/acs.analchem.6b04382 [ Links ]
LIN WC, CHEN HC and DING WH (2005) Determination of pharmaceutical residues in waters by solid-phase extraction and large-volume on-line derivatization with gas chromatography-mass spectrometry. J. Chromatogr. A 1065 (2) 279-285. https://doi.org/10.1016/j.chroma.2004.12.081 [ Links ]
LINDHOLM-LEHTO PC, AHKOLA HS and KNUUTINEN JS (2018) Pharmaceuticals in processing of municipal sewage sludge studied by grab and passive sampling. Water Qual. Res. J. 53 (1) 14-23. https://doi.org/10.2166/wqrj.2018.022 [ Links ]
LÓPEZ-ROLDÁN R, DE ALDA ML, GROS M, PETROVIC M, MARTÍN-ALONSO J and BARCELÓ D (2010) Advanced monitoring of pharmaceuticals and estrogens in the Llobregat River basin (Spain) by liquid chromatography-triple quadrupole-tandem mass spectrometry in combination with ultra performance liquid chromatography-time of flight-mass spectrometry. Chemosphere 80 (11) 1337-1344. https://doi.org/10.1016/j.chemosphere.2010.06.042 [ Links ]
LORAINE GA and PETTIGROVE ME (2006) Seasonal variations in concentrations of pharmaceuticals and personal care products in drinking water and reclaimed wastewater in southern California. Environ. Sci. Technol. 40 (3) 687-695. https://doi.org/10.1021/es051380x [ Links ]
MADIKIZELA LM (2017) Determination of selected acidic pharmaceutical compounds in wastewater treatment plants. PhD thesis, University of the Witwatersrand, South Africa. [ Links ]
MADIKIZELA LM, NCUBE S and CHIMUKA L (2020) Analysis, occurrence and removal of pharmaceuticals in African water resources: A current status. J. Environ. Manage. 253 109741. https://doi.org/10.1016/j.jenvman.2019.109741 [ Links ]
MANRIQUE-MORENO M, HEINBOCKEL L, SUWALSKY M, GARIDEL P and BRANDENBURG K (2016) Biophysical study of the non-steroidal anti-inflammatory drugs (NSAID) ibuprofen, naproxen and diclofenac with phosphatidylserine bilayer membranes. Biochim. Biophys. Acta Biomembr. 1858 (9) 2123-2131. https://doi.org/10.1016/j.bbamem.2016.06.009 [ Links ]
MATONGO S, BIRUNGI G, MOODLEY B and NDUNGU P (2015) Pharmaceutical residues in water and sediment of Msunduzi River, KwaZulu-Natal, South Africa. Chemosphere 134 133-140. https://doi.org/10.1016/j.chemosphere.2015.03.093 [ Links ]
MBHELE ZE, NCUBE S anf MADIKIZELA LM (2018) Synthesis of a molecularly imprinted polymer and its application in selective extraction of fenoprofen from wastewater. Environ. Sci. Pollut. Res. 25 (36) 36724-36735. https://doi.org/10.1007/s11356-018-3602-x [ Links ]
MEURS J (2016) Flow rates in liquid chromatography, gas chromatography and supercritical fluid chromatography: a tool for optimization. J. Open Res. Softw. 4 (1) 32. https://doi.org/10.5334/jors.126 [ Links ]
MEZZELANI M, GORBI S, FATTORINI D, DERRICO G, CONSOLANDI G, MILANI M, BARGELLONI L and REGODI F (2018) Long term exposure to Mytilus galloprovincialis to diclofenac, ibuprofen and ketoprofen: Insightsinto bioavailability, biomakers and transcriptomic changes. Chemosphere 198 238-248. https://doi.org/10.1016/j.chemosphere.2018.01.148 [ Links ]
MHLANGA F and BROUCKAERT C (2013) Characterisation of wastewater for modelling of wastewater treatment plants receiving industrial effluent. Water SA 39 (3) 403-408. https://doi.org/10.4314/wsa.v39i3.9 [ Links ]
MLUNGUZA NY, NCUBE S, MAHLAMBI PN, CHIMUKA L and MADIKIZELA LM (2019). Adsorbents and removal strategies of non-steroidal anti-inflammatory drugs from contaminated water bodies. J. Environ. Chem. Eng. 7 (3) 103142. https://doi.org/10.1016/j.jece.2019.103142 [ Links ]
MONTAGNER CC and JARDIM WF (2011) Spatial and seasonal variations of pharmaceuticals and endocrine disruptors in the Atibaia River, São Paulo State (Brazil). J. Braz. Chem. Soc. 22 (8) 1452-1462. https://doi.org/10.1590/S0103-50532011000800008 [ Links ]
MOSLAH B, HAPESHI E, JRAD A and FATTA-KASSINOS D (2018). Pharmaceuticals and illicit drugs in wastewater samples in north-eastern Tunisia. Environ. Sci. Pollut. Res. 25 18226-18241. https://doi.org/10.1007/s11356-017-8902-z [ Links ]
MTOLO SP, MAHLAMBI PN and MADIKIZELA LM (2019) Synthesis and application of a molecularly imprinted polymer in selective solid-phase extraction of efavirenz from water. Water Sci. Technol. 79 356-365. https://doi.org/10.2166/wst.2019.054 [ Links ]
NADAIS H, LI X, ALVES N, COURAS C, ANDERSEN HR, ANGELIDAKI I and ZHANG Y (2018) Bio-electro-Fenton process for the degradation of non-steroidal anti-inflammatory drugs in wastewater. Chem. Eng. J. 338 401-410. https://doi.org/10.1016/j.cej.2018.01.014 [ Links ]
NAIDOO J (2013) Assessment of the impact of wastewater treatment plant discharges and other anthropogenic variables on river water quality in the EThekwini Metropolitan Area. Doctoral dissertation, University of KwaZulu-Natal, Durban. [ Links ]
NANNOU CI, KOSMA CI and ALBANIS TA (2015) Occurrence of pharmaceuticals in surface waters: analytical method development and environmental risk assessment. Int. J. Environ. Anal. Chem. 95 (13) 1242-1262. https://doi.org/10.1080/03067319.2015.1085520 [ Links ]
NIKOLAOU A, MERIC S and FATTA D (2007) Occurrence patterns of pharmaceuticals in water and wastewater environments. Anal. Bioanal. Chem. 387 (4) 1225-1234. https://doi.org/10.1007/s00216-006-1035-8 [ Links ]
NTOMBELA S and MAHLAMBI P (2019) Method development and application for triazine herbicides analysis in water, soil and sediment samples from KwaZulu-Natal. J. Environ. Sci. Health Part B 54 (7) 569-579. https://doi.org/10.1080/03601234.2019.1621113 [ Links ]
OSORIO V, LARRAÑAGA A, ACEÑA J, PÉREZ S and BARCELÓ D (2016) Concentration and risk of pharmaceuticals in freshwater systems are related to the population density and the livestock units in Iberian Rivers. Sci. Total Environ. 540 267-277. https://doi.org/10.1016/j.scitotenv.2015.06.143 [ Links ]
PADHYE LP, YAO H, KUNG'U FT and HUANG C-H (2014) Year-long evaluation on the occurrence and fate of pharmaceuticals, personal care products, and endocrine disrupting chemicals in an urban drinking water treatment plant. Water Res. 51 266-276. https://doi.org/10.1016/j.watres.2013.10.070 [ Links ]
PATROLECCO L, ADEMOLLO N, GRENNI P, TOLOMEI A, CARACCIOLO AB and CAPRI S (2013) Simultaneous determination of human pharmaceuticals in water samples by solid phase extraction and HPLC with UV-fluorescence detection. Microchem. J. 107 165-171. https://doi.org/10.1016/j.microc.2012.05.035 [ Links ]
POULSEN J and LAURIDSEN CL (2005) Modelling of the new works at Umbilo Sewage Purification Works with the WEST-program - plus an investigation of heavy metal content in the sludg. Masters Dissertation, Aalborg University, Denmark. [ Links ]
SADKOWSKA J, CABAN M, CHMIELEWSKI M, STEPNOWSKI P and KUMIRSKA J (2017) Environmental aspects of using gas chromatography for determination of pharmaceutical residues in samples characterized by different composition of the matrix. Arch. Environ. Protect. 43 (3) 3-9. https://doi.org/10.1515/aep-2017-0028 [ Links ]
SHRAIM A, DIAB A, ALSUHAIMI A, NIAZY E, METWALLY M, AMAD M, SIOUD S and DAWOUD A (2017) Analysis of some pharmaceuticals in municipal wastewater of Almadinah Almunawarah. Arab. J. Chem. 10 S719-S729. https://doi.org/10.1016/j.arabjc.2012.11.014 [ Links ]
SIBEKO PA, NAICKER D, MDLULI PS and MADIKIZELA LM (2019) Naproxen, ibuprofen, and diclofenac residues in river water, sediments and Eichhornia crassipes of Mbokodweni river in South Africa: an initial screening. Environ. Forensics 20 129-138. https://doi.org/10.1080/15275922.2019.1597780 [ Links ]
SIBIYA P, POTGIETER M, CUKROWSKA E, JÖNSSON JÅ and CHIMUKA L (2012) Development and application of solid phase extraction method for polycyclic aromatic hydrocarbons in water samples in Johannesburg area, South Africa. S. Afr. J. Chem. 65 206-213. [ Links ]
SUN J, LUO Q, WANG D and WANG Z (2015) Occurrences of pharmaceuticals in drinking water sources of major river watersheds, China. Ecotoxicol. Environ. Saf. 117 132-140. https://doi.org/10.1016/j.ecoenv.2015.03.032 [ Links ]
UMGENI WATER (2013) Infrastructure Master Plan 2013 volume 2 2013/2014-2043/44, September 2013. Umgeni Water, Pietermaritzburg. [ Links ]
VERSTEEG S (2014) The effects of select non-steroidal anti- inflammatory drugs on green and brown hydra. MSc thesis, University of Ontario Institute of Technology, Canada. [ Links ]
WONGRAKPANICH S, WONGRAKPANICH A, MELHADO K and RANGASWAMI J (2018) A comprehensive review of non-steroidal anti-inflammatory drug use in the elderly. Aging Dis. 9 (1) 143-150. https://doi.org/10.14336/AD.2017.0306 [ Links ]
XU C, NIU L, GUO H, SUN X, CHEN L, TU W, DAI Q, YE J, LIU W and LIU J (2019) Long term esposure tonthe non-steroidal anti inflammatory drug (NSAID) naproxen causes thyroid disruption in zebrafish at environmentally relevant concentrations. Sci. Total Environ. 1 (676) 387-395. https://doi.org/10.1016/j.scitotenv.2019.04.323 [ Links ]
ZHANG Y, LI Y, ZHU X-J, LI M, CHEN H-Y, LV X-L and ZHANG J (2017) Development and validation of a solid-phase extraction method coupled with HPLC-UV detection for the determination of biogenic amines in Chinese rice wine. Food Additives Contam. Part A 34 (7) 1172-1183. https://doi.org/10.1080/19440049.2017.1317366 [ Links ]
ŻWIR-FERENC A and BIZIUK M (2006) Solid phase extraction technique--trends, opportunities and applications. Pol. J. Environ. Stud. 15 (5) 677-690. [ Links ]
 Correspondence:
Correspondence:
PN Mahlambi
Email:Mahlambip@ukzn.ac.za
Received: 25 March 2019
Accepted: 8 June 2020














