Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Water SA
On-line version ISSN 1816-7950
Print version ISSN 0378-4738
Water SA vol.43 n.2 Pretoria Apr. 2017
http://dx.doi.org/10.4314/wsa.v43i2.15
A comparison between Daphnia pulex and Hydra vulgaris as possible test organisms for agricultural run-off and acid mine drainage toxicity assessments
P Singh*; A Nel
Department of Zoology, University of Johannesburg, Auckland Park, Johannesburg 2006, South Africa
ABSTRACT
Bioassays, consisting of a diverse selection of organisms, aid in assessing the ecotoxicological status of aquatic ecosystems. Daphnia pulex and Hydra vulgaris are commonly used test organisms belonging to different trophic levels. The current study focused on comparing the sensitivity of H. vulgaris to D. pulex when exposed to geometric dilutions of two different water sources, the first (Site 1) from a source containing agricultural run-off and the second (Site 2), acid mine drainage. These sources were selected based on the contribution that the agricultural and mining sectors make to water pollution in South Africa. The bioassay method followed in this study was a modified version of the method described by the USEPA and additional peer-reviewed methods. The mortalities as well as morphological changes (H. vulgaris) were analysed using Microsoft Excel. The LC50-values were statistically determined using the EPA Probit Analysis Model and the Spearman-Karber analysis methods. Prior to being used, analysis of the physico-chemical properties, nutrients and metals of both water samples was performed. These results showed a relationship to the results obtained from the D. pulex and H. vulgaris bioassays, as Site 1 (lower concentration of contaminants) was less hazardous to both test organisms than Site 2 (higher concentration of contaminants). Both organisms can be used for ecotoxicity testing, with D. pulex being a more sensitive indicator of toxicity with regards to water sampled from the acid mine drainage site. Due to the sensitivities of sub-lethal endpoints observed over time, H. vulgaris may be used for chronic toxicity testing and D. pulex for acute toxicity testing.
Keywords: Hydra, Daphnia, toxicity, ecotoxicity, definitive toxicity test
INTRODUCTION
Man's production and use of chemicals and minerals, and his dependence thereon, has led not only to valuable products and services, but also to the release of numerous hazardous substances into the natural environment (Wharfe, 2005). Over the years, the agricultural and mining sectors have grown in South Africa. Although these sectors provide economic profits, the activities have resulted in increased pollution of South African water sources (Bezuidenhout, 2013). Irrigation and surface run-off water has been found to contain pesticides, fertilisers, harmful chemicals and/or pathogenic microorganisms (Britz and Sigge, 2012). Acid mine drainage (AMD) is characterised by a low pH, high metal concentration, high specific conductivity and a ferric oxyhydroxide precipitate commonly known as 'yellow boy' (Akcil and Koldas, 2006). The aims of ecotoxicology have evolved over the years, from establishing the concentrations at which chemicals exert adverse effects, estimating environmental risk based on measured toxicity endpoints, and predicting environmental concentrations for specific chemicals, to defining toxicant concentrations harmful for specific organism groups and/or for assemblages of species (Blaise and Fèrard, 2005).
The past few decades have produced and utilised a variety of bioassays to assess the toxicity and quality of the surrounding aquatic environment. These bioassays have involved the use of a diverse selection of organisms and can be conducted as acute, sub-chronic or chronic bioassays (Persoone and Janssen, 1993; Cairns, 1995; Slabbert and Venter, 1999; Persoone et al., 2003; Blaise and Fèrard, 2005; Goodfellow, 2005). These bioassays have aided in the establishment and promulgation of water quality criteria (regarding safe release of single chemicals into aquatic ecosystems), providing aquatic safety assessments for chemicals, biomonitoring initiatives, registration of pesticide products, assessing industrial and mine effluent, urban and agricultural run-off and the ranking of chemicals with respect to their hazardous potential (Bitton et al., 1995; Blaise and Fèrard, 2005).
Test organisms that have been used range from plants (Lepidium sativum, Lemna minor, Sorghum saccharatum, Sinapis alba) to a multitude of unicellular (Vibrio fischeri, Selenastrum capricornutum) and small multicellular organisms (Daphnia pulex, Hyalella azteca, Chironomus spp., Hydra spp., Poecilia reticulata) (Hall and Golding, 1998; Pardos et al., 1999; Gallagher et al., 2005; Goodfellow, 2005; Sanchez et al., 2005; Czerniawska-Kusza et al., 2006; Shuhaimi-Othman et al., 2010). Associated with each bioassay are lethal (mortality) and sub-lethal (e.g. growth inhibition, reproduction teratogenic effects) endpoints, which give an indication of expected toxicity of a contaminant(s) (Suter, 1995; Diaz-Baez and Dutka, 2005; Holdway, 2005; Jonczyk and Gilron, 2005).
Ecotoxicological testing has become a compulsory requirement today as many countries advocate its use to determine the toxicity of effluents, chemicals, metals, wastewaters and solid wastes, and to calculate limits for the discharge of these substances (Persoone et al., 2003). In South Africa, the National Water Act (Act 36 of 1998) (RSA, 1998) governs the protection, use, development, conservation, management and control of its water resources. Stemming from this Act was the National Toxicity Monitoring Programme (NTMP), whose responsibility is to measure, assess and regularly report on the state of South African water resources (Murray et al., 2003). The NTMP utilises and promotes the use of bioassays to assess the quality of water resources (Murray et al., 2004).
Globally, Hydra species have been used extensively for toxicity testing, i.e., assessing the toxicity of: pharmaceuticals (Pascoe et al., 2003; Quinn et al., 2008a; Quinn et al., 2008b; Quinn et al., 2009), bottled drinking waters (Arkhipchuk et al., 2006), chemicals such as glycol ethers (Bowden et al., 1995), Endosulfan (Pollino and Holdway, 1999), industrial effluents (Blaise and Kusui, 1997) and waste waters (Pardos et al. 1999), and metals (Beach and Pascoe, 1998; Pollino and Holdway, 1999; Karntanut and Pascoe, 2000, Karntanut and Pascoe 2002; Karntanut and Pascoe, 2005). Hydra species commonly used are H. viridissima, H. vulgaris, H. attenuata H. oligactis and H. pseudoligactis (Bell and Wolfe, 1985; Blaise and Kusui, 1997; Fukuhori et al., 2005; Holdway, 2005).
Holdway (2005) suggests that Hydra toxicity testing can be used to determine the teratogenic potential of chemicals in terms of the acute lethality, sub-lethality (morphological changes, behaviour and feeding response), chronic reproductive effects and Hydra regeneration effects that are displayed by the test organisms. Hence, these organisms are appropriate for acute and chronic bioassays. Acute toxicity tests are conducted over a maximum of 96 h with the only endpoint being survival/mortality. Hydra chronic toxicity tests can be conducted over a period 18-21 days and take into account survival, morphological changes and reproductive capacity (Arkhipchuk et al., 2006). Another test procedure, the Hydra reproduction test, occurs over 7 days and evaluates survival and population growth (Holdway, 2005). In a study by Pardos et al. (1999) H. attenuata displayed a higher sensitivity to wastewater when compared to the Microtox test (Vibrio fischeri).
Daphnia species have been widely used in aquatic ecotoxicology with D. pulex being one of the more preferred test species for a number of reasons: these organisms can be easily cultured and maintained; the age of the organisms is always known; biology of Daphnia has been thoroughly researched and documented; numerous studies have expressed the sensitivity of D. pulex to several chemicals (large toxicity database), their tolerance, ability to outcompete other species and provide the most toxicological information per unit effort (Sprules, 1972; Lynch, 1983; Pennak, 1989; Persoone and Janssen, 1993; Muller and Palmer, 2002; Jonczyk and Gilron, 2005). Daphnia species including D. pulex have been used internationally in acute and chronic toxicity tests, assessing the toxicity of potentially hazardous chemicals, and bio-monitoring of effluents discharged by industrial companies (Slabbert and Venter, 1999; Jonczyk and Gilron, 2005), municipal wastewater systems (Logue et al., 1989), produced- and receiving waters (Jonczyk and Gilron, 2005), insecticides (Wood and Stark, 2002; Stark and Vargas, 2003, 2005; Zalizniak and Nugegoda, 2006), and metals - zinc in a biotic ligand model (Clifford and McGeer, 2009), copper (Koivisto and Ketola, 1995), nickel (Kozlova et al., 2009; Leonard and Wood, 2013), and lead (Offem and Ayotunde, 2008). Daphnia have also been used in various chronic toxicity tests in which they were exposed for a period of 18-21 days. Here, the organisms' survival and total number of young produced were observed (Truter, 1994). D. pulex bioassay has also been found to be an alternative to the mouse bioassay due to its advantages and ability to detect cyanobacterial neurotoxins in raw water samples (Ferrao-Filho et al., 2010).
The current study focused on comparing the sensitivity of H. vulgaris to D. pulex in 96 h bioassays when exposed to geometric dilutions of water samples collected from a source containing agricultural run-off (Site 1) and a source containing acid mine drainage (Site 2).
METHODS
Test organisms and culture maintenance
D. pulex and H. vulgaris were obtained from laboratory monocultures in the Aquarium of the Department of Zoology at the University of Johannesburg. The test organisms were cultured in accordance with, and adapted from, the methodologies explained by Truter (1994) and USEPA (2002) for D. pulex, and Trottier et al. (1997) and Holdway (2005) for H. vulgaris.
The Daphnia and Hydra cultures were maintained in an environmental room with a constant temperature of 20 ± 1°C and a daily photoperiod of 16 h light and 8 h dark using ambient fluorescent lighting. A Daphnia stock solution (also known as Daphnia medium) was prepared and used for both cultures (Truter, 1994). Although previous studies utilised a Hydra medium for culturing Hydra (Blaise and Kusui, 1997; Beach and Pascoe, 1998; Holdway, 2005; Arkhipchuk et al., 2006), better culturing success was achieved for this study using the Daphnia medium (Truter 1994). The Daphnia cultures were kept in 3 L glass beakers and fed YTC (a suspension of commercial yeast, trout pellets and cerophyll) 3 times a week (Truter, 1994; U.S. EPA, 2002). The Hydra cultures were maintained in 1 L circular glass bowls and fed D. pulex and freshly-hatched Artemia salina nauplii 3 times a week (Sorgeloos and Persoone, 1975; Trotter et al., 1997; Holdway, 2005, Arkhipchuk et al., 2006). Feeding of test organisms were discontinued 48 h prior to and during the bioassays. This practice diminishes the risk of particles in the organism's digestive tract influencing the end result of the toxicity tests.
Water samples
Experiments were conducted using water samples from 2 different sites. Site 1 contained agricultural run-off and Site 2 contained acid mine drainage. Grab water samples were collected, transported on ice, and stored at 0-6°C prior to toxicity testing, as suggested by the relevant standard operating procedures (EC, 1996; USEPA, 2002). Water samples were collected from 2 different projects - one focusing on agricultural pollution and the other on AMD. Names of the sample locations are withheld.
Analytical techniques
Physico-chemical analysis of the water samples is required when conducting bioassays and was performed in 3 parts according to standard operational procedures. Firstly, physical parameters such as pH, dissolved oxygen (DO) (mg/L), percentage oxygen saturation, temperature and conductivity were quantified. These parameters were measured at the beginning of the test with the undiluted samples according to USEPA (2002). Secondly, photometric analysis was used to measure parameters such as ammonium, chloride, nitrate, nitrite, phosphate, sulphate, total hardness and turbidity (EC, 1996; USEPA, 2002). Thirdly, inductively coupled plasma spectrometry (ICP) was conducted to determine concentrations of metals such as aluminium, cobalt, iron, manganese, nickel, uranium and zinc in the water samples (EC, 1996; USEPA, 2002).
The metal concentrations found in the water samples were compared to the Target Water Quality Range (TWQR), Chronic Effect Value (CEV) and Acute Effect Value (AEV) guidelines described in DWAF (1996).
Toxicity test procedure and toxicity classification
The acute toxicity test procedure followed in this study was a modified version of the Daphnia method described in USEPA (2002) and Truter (1994), and incorporated aspects of Hydra toxicity testing (Trottier et al., 1997; Holdway, 2005; Arkhipchuk et al., 2006). The test duration was extended to 96 h, ensuring a better comparison between the two test species. Daphnia medium was used as the control- and dilution water in both bioassays. H. vulgaris and D. pulex were exposed to geometric dilutions (100%, 50%, 25%, 12.5% and 6.25%) of water samples from Sites 1 and 2. To enhance the accuracy of the results, exposures were done in triplicate. The 96th hour LC50-value was calculated for each test using Spearman-Karber analysis and the EPA Probit analysis model (Finney, 1971; Hamilton et al., 1977; Finney, 1978). The primary endpoint (lethal) observed for both bioassays was mortality at 24-h intervals spanning the duration of the bioassays. Morphological change, an additional secondary endpoint (sub-lethal), was observed for H. vulgaris at 24-h intervals spanning the duration of the bioassays. Modifications to the test method were aided by Truter (1994); Slabbert and Venter (1999); USEPA (2002); Holdway (2005); Jonczyk and Gilron (2005); and Arkhipchuk et al., 2006. The only modifications were testing both organisms to the same geometric dilution series, and exposing both test organisms for the same test duration, i.e., 96 h. This was done to create a common environment for comparing the organisms and to determine whether the test organism(s) were suitable for toxicity testing, as well as to compare which organism displayed a higher suitability to the ecotoxicity testing. All testing was done in an environmental room with the same controlled conditions described above. Daphnia were tested in 50 mL glass beakers with a final dilution volume of 40 mL. Hydra were tested in 500 mL glass beakers with a final dilution volume of 300 mL. Five (5) organisms were placed in each beaker, respectively.
Persoone et al. (2003) developed a water toxicity classification ranking system based on mortalities (percentage effect) of test organisms (Table 1). This system was used in this study to rank the water quality of the water samples based on the percentage mortality in the 100% concentration after the 96 h exposures.
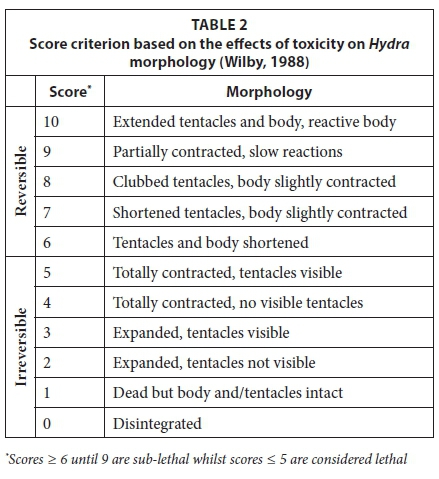
Hydras have been shown to display morphological changes in response to contaminants and an unfavourable environment (Wilby, 1988; Holdway, 2005; Quinn et al., 2009). Table 2, designed by Wilby (1988), illustrates the concept where the condition of the Hydra is given a score, based on the observed morphology. This score rated the effects of toxicity on the hydroid morphology. The score ranged from 10 (healthy, extended tentacles and body, body reactive) to 0 (body disintegrated). Any score ≤ 5 was concluded to be irreversible and the endpoint for lethality (Blaise and Kusui, 1997; Quinn et al., 2009). Although a subjective observation, previous studies by reputable authors in the ecotoxicity field have utilised this observation and score criterion. As a result, and similar to previous studies, it is only used as a secondary observation with the aim of informing the primary endpoint, i.e., mortality.
Statistical analysis
Statistical analysis of the data from the lethality exposure was performed. Spearman-Karber analysis and the EPA Probit analysis model were used to calculate the 96-h LC50-values with 95% confidence intervals (Finney, 1971; Hamilton et al., 1977; Finney, 1978). Graphical representation of the Hydra and Daphnia sensitivity to toxicity was done using Microsoft Excel. The calculation of the LC50 depends on certain factors. The Probit Method, a parametric statistical procedure, requires that the observed proportion mortalities should bracket 0.5, and 2 or more of the observed proportion mortalities must be between 0 and 1. The Spearman-Karber Method is recommended when the data does not fit the Probit model. It is a non-parametric statistical procedure for estimating the LC50 requiring that the smoothed adjusted proportion mortality for the lowest effluent concentration should be 0 and for the highest effluent concentration, 1 (USEPA, 2002).
RESULTS
Physical and chemical analysis of water from Site 1 (agricultural run-off) and Site 2 (acid mine drainage) was performed. Table 3 summarises the physico-chemical results that were obtained. For an aquatic ecosystem to optimally support life, a water source should ideally have a pH between 6.0 and 9.0, dissolved oxygen (DO) percentage greater than 40% (preferably 80%-120%) and a DO concentration greater than 4.0 mg/L (DWAF, 1996; U.S. EPA, 2002).
Parameters were measured in duplicate and are expressed as averages. Site 2 presented a lower pH and higher electrical conductivity than Site 1. Additionally, a high concentration of sulphates at Site 2 was noted which may be attributed to being impacted by acid mine drainage (Akcil and Koldas, 2006). Included in Table 3 is the rank of the water samples based on the toxicity (Persoone et al., 2003). Conforming to this hazard classification system, Site 1 presented no acute hazard whilst Site 2 presented a very high acute hazard.
Metal analysis of Sites 1 and 2 (Table 4) revealed elevated levels of aluminium, iron and manganese at Site 2 whilst Site 1 had low metal concentrations when compared to the water quality guidelines for aquatic ecosystems specified in DWAF (1996). Metal concentrations found in the water samples were compared to the TWQR, CEV and AEV guidelines specified in the water quality guidelines for aquatic ecosystems (DWAF, 1996).
The high levels of iron may not only be due to the geology of the sample's location but also as a result of acid mine drainage in the immediate vicinity/upstream (DWAF, 1996; Akcil and Koldas, 2006). From the available information in DWAF (1996), water quality from Site 1 did not exceed the TWQR, CEV and AEV guidelines whilst water from Site 2 had metals that exceeded the acceptable TWQR, CEV and AEV guidelines.
Figure 1 summarises the 96-h bioassay, comparing the responses of D. pulex and H. vulgaris at each concentration, using water sampled from Site 2. Throughout the exposure duration for Site 1, both test organisms showed zero percentage mortalities in all concentrations. This indicated that water from Site 1 poses no acute hazard to the two species of test organisms (USEPA, 2002; Persoone et al., 2003).
Percentage mortality of D. pulex at Site 2 increased with an increase in sample concentration and exposure time (Fig. 1). After 96 h, the percentage mortality of D. pulex from the lowest concentration to the highest concentration was 26.67%, 40%, 46.67%, 60% and 100%. After 96 h, the percentage mortality of H. vulgaris from the lowest concentration to the highest concentration was 0%, 0%, 0%, 6.67% and 100%.
After 24 h, all daphnids and hydras in the 100% concentration were dead (Fig. 1). A low percentage mortality was observed with H. vulgaris in the 50% sample concentration after 48 h, and remained the same throughout the duration of the test (Fig. 1). Exposure concentrations 6.25% and 12.5% resulted in zero mortality at the 48th hour, with a sudden increase in mortality between the 48th and 96th hour observations for D. pulex. D. pulex showed more sensitivity to the water from Site 2 as mortalities were observed from the 25% sample concentration at the 24-h reading and increased from this point onwards.
The number of hydras increased in the control, 6.25% and 100% concentrations at Site 1. This increase was due to budding (asexual reproduction) which occurs in favourable conditions. There was no change in the number of hydras for concentrations 12.5% to 50% of Site 1. In concentrations 25%, 12.5% and 6.25% (including the control) of Site 2 no mortalities were observed with H. vulgaris. There was, however, an increase in Hydra numbers through budding (not graphically presented) in both water samples during the test.
Statistical analysis of observations at the end of the bioassays is presented in Table 5. The Spearman-Karber and EPA Probit programs could not calculate the LC50 for both organisms exposed to water from Site 1 due to no significant mortalities (conditions as discussed in Methods section: Statistical analysis). Spearman-Karber and EPA Probit determined the LC50 for D. pulex exposed to water from Site 2 as 26.49% and 22.54%, respectively. Only the Spearman-Karber method could calculate the LC50 value for H. vulgaris exposed to water from Site 2, i.e., 70.71%, since the data did not fit the Probit model.
Morphological changes of H. vulgaris observed during the course of the bioassay were scored using the criteria developed by Wilby (1988) (Table 2) and are presented in Fig. 2. At Site 1 the score began to decrease after 72 h and ranged between 7 and 8 at the end of the test. At Site 2 the score began to decrease after 24 h from the 25% concentration onwards. At the end of the bioassay exposure, the hydra's scores at Site 2 ranged between 0 (in the highest concentration) and 9 (in the lowest concentration). From Fig. 2 it can be seen that H. vulgaris showed more sensitivity (morphological changes) at an earlier time at Site 2 than Site 1. This suggested that Site 2 has a higher toxicity than Site 1.
DISCUSSION
According to DWAF (1996) and USEPA (2002), for an aquatic ecosystem to support diverse life forms (and be suitable for biological toxicity testing), a water source should have a pH between 6.0 and 9.0, dissolved oxygen (DO) percentage greater than 40% (preferably 80%-120%) and a DO concentration greater than 4.0 mg/L. Physico-chemical analysis of the two water samples (Table 3) was performed in order to verify results obtained from the bioassays and aid in comparing the sensitivities of H. vulgaris and D. pulex (USEPA, 2002; Baderna et al., 2011). Site 1 (containing agricultural run-off) presented the constituents and water quality to support aquatic life and to be used for the bioassays, based on the results obtained in this study (Table 3). Site 2 (impacted by acid mine drainage) presented a pH of 2.6, as well as high levels of conductivity and sulphates (Table 3). At such levels the water sample may be rendered unsafe for aquatic life (DWAF, 1996; Lidman, 2005). Elevated levels of these parameters may have been as a result of acid mine drainage and a low pH (DWAF, 1996; Akcil and Koldas, 2006; Liang-qi et al., 2010). The dissolved oxygen measured for this sample was within limits for conducting toxicity tests as required by the standard operational procedure (USEPA, 2002). Both water samples were ranked according to a hazard classification system designed by Persoone et al. (2003) by comparing the response of the test organisms in the 100% concentration after 96 h. A consensus between the physico-chemical results and the results from the bioassay could be seen in Table 3, since water from Site 1 was ranked as Class I, having no acute hazard, whilst water from Site 2 presented a very high acute hazard (Class V).
Metal analysis of the water from Site 1 recorded lower metal concentrations than Site 2 (Table 4). Water from Site 2 presented elevated levels of aluminium, iron and manganese. High concentrations of certain metals such as iron could be attributed to the geology of the surrounding sampling area, acid mine drainage and physical properties of the water (DWAF, 1996; Akcil and Koldas, 2006; Dinelli et al., 2010; Liang-qi et al., 2010). pH values of less than 4.0 and higher than 6.5 increase the solubility of aluminium, which may explain the high concentration at Site 2 (DWAF, 1996). Metals present in the water from Site 1 fell within the limits for Target Water Quality Range (TWQR), Chronic Effect Value (CEV) and Acute Effect Value (AEV) guidelines. At Site 2, aluminium, manganese and zinc detected in the water exceeded the TWQR, CEV and AEV resulting in metals being present in concentrations that may be detrimental to aquatic life. This further correlated the physico-chemical results to that of the bioassays. The low pH and presence of other possible toxicants could have contributed to the high mortalities observed during this exposure (U.S. EPA, 2002).
The toxicity test method was a modified bioassay incorporating the methodologies explained in Truter (1994), Trottier et al. (1997), USEPA (2002), Holdway (2005) and Arkhipchuk et al. (2006). Doing so enhanced the comparison between the H. vulgaris and D. pulex as the only variables were the organisms themselves. Exposures were done in triplicate ensuring a more reliable statistical estimation of the toxicity of the samples and simultaneously minimising the effects of natural deaths to a certain degree. At Site 1, D. pulex and H. vulgaris showed no mortality in all the exposure concentrations (Fig. 1). From these results, it was concluded that the water from this sample was safe for their survival. According to Blaise and Ferard (2005), Hydra vulgaris and Daphnia pulex are representative aquatic invertebrates; therefore their sensitivities may be indicative of the toxicity of Site 1 and Site 2 to other aquatic invertebrates. The Hydra at Site 1 had population escalations which suggested a favourable environment, as asexual reproduction occurred (Mitchell and Holdway, 2000; Holdway, 2005). The rate at which asexual reproduction occurs when hydras are exposed to a water sample can be used as an indication of its toxicity (Mitchell and Holdway, 2000).
One hundred percent (100%) mortality was observed for both test organisms in the highest exposure concentration at Site 2 after 24 h (Fig. 1a). This observation could be attributed to the very low pH (2.6) and high level of conductivity, phosphates, metals and sulphates (Table 3 and 4) (Blaise and Kusui, 1997; Mitchell and Holdway, 2000; USEPA, 2002; Holdway, 2005). Blaise and Kusui (1997) found that a correlation exists between conductivity (contributed by the presence of metal ions) and the response of Hydra - an increase in conductivity spurs an increase in toxicity to Hydra. According to Mitchell and Holdway (2000), Hydra have been found to display sensitivity to metal and organic contaminants. It was found that further dilutions of water from Site 2 led to organisms being able to survive (Fig. 1). D. pulex mortality decreased with the decrease in concentration of the sample (Fig. 1). H. vulgaris showed a high sensitivity to the 100% concentration and a lower sensitivity to the 50% concentration of the sample. Further dilutions of water from Site 2 resulted in an increase in the Hydra populations (by budding). The dilution of acid mine water created a more favourable environment for the Hydra (Bell and Wolfe, 1985; Holdway, 2005). In a study by Loehr et al. (2006), dilution of wastewater discharges resulted in the effluent being less toxic to aquatic organisms when conducting WET (whole effluent toxicity) testing. The decrease in sensitivity could also have been as a result of the Hydra metabolising toxicants in the water sample (Quinn et al., 2009) and thereafter adapting themselves to the conditions. It was further observed in this experiment that D. pulex had a higher sensitivity to toxicants in water with poor water quality than the hydras.
The LC50-values were determined using the Spearman-Karber (SK) method and the EPA Probit analysis model (Finney, 1971; Hamilton et al., 1977; Finney, 1978). From Table 5 it is evident that the LC50 for both test organisms at Site 1 was incalculable due to no significant mortality rate (Refer to Methods section for conditions to the statistical programme). The LC50 for the daphnids at Site 2 was calculated as 26.49% (SK method) and 22.54% (Probit Model) (Table 5). The average of these two LC50 values suggested that a lethal concentration of 24.52% would kill off half the population of daphnids after 96 h. Using the SK method, the LC50 for the hydras at Site 2 was calculated as 70.71% but the 95% confidence limits were not reliable (Table 5). This was due to insignificant mortality response (variability in mortality) in the different exposure concentrations. The Probit model could not determine the lethal concentration for the hydras at Site 2, since there was no considerable mortality response to the different concentrations (Table 5).
The condition of test organisms is a reasonable sub-lethal endpoint as organisms should not only be able to survive but also to thrive in an aquatic environment. Such an endpoint was achieved with the Hydra bioassay. Scores presented in Fig. 2 that were less than and including 5 were considered a lethal endpoint (Wilby, 1988; Arkhipchuk et al., 2006). H. vulgaris showed an increased sensitivity to water from Site 1 after the 96th hour based on their morphological changes (Fig. 2). This may infer that if H. vulgaris had been exposed for a longer duration (chronic testing), higher sensitivity and even a population decrease may be observed as was experienced by Arkhipchuk et al. (2006). It also indicated that water from Site 1 presented a low hazard for aquatic life.
The Hydra's net population growth in the acid mine water (Site 2) (not graphically presented) did not necessarily indicate that they had no sensitivity to toxicity, since their morphologies presented scores that suggested their sensitivities over time (Fig. 2). After the 96th hour there was a greater variety of scores. Hence, even with water from Site 2, had the exposure time been extended, significant mortalities at the lower concentrations may eventually have been observed. This experiment provided a platform for future biological toxicology studies in South Africa as both H. vulgaris and D. pulex displayed sensitivity to water quality and proved to be suitable organisms for the acute toxicity testing method.
CONCLUSION
Advances in ecotoxicology have led to the development of various bioassays utilising a diverse selection of organisms belonging to different trophic levels. D. pulex and H. vulgaris have been and are currently used as test organisms. The focus of the study was to compare the sensitivities of both organisms and to suggest which organism(s) may be more applicable for acute toxicity testing of aquatic resources inundated/contaminated with either agricultural run-off or acid mine drainage. The study further focused on determining whether H. vulgaris may be used concurrently with D. pulex when observing possible effects of water samples on organisms belonging to two different trophic levels. This comparison was achieved by simultaneously exposing the organisms to 2 water samples following a modified bioassay method. The physico-chemical and metal analyses of the water samples showed a relationship to the results obtained from the bioassays: in the 100% sample concentration of Site 1 both organisms presented no mortality, whilst in that of Site 2, both organisms presented 100% mortality after 24 h. H. vulgaris showed a lesser degree of sensitivity for the endpoint mortality when compared to D. pulex but displayed morphological changes in response to toxicity, signifying sensitivity as a sub-lethal endpoint which can be useful for further studies. Hydra reproduces asexually in a favourable environment and this was observed in the water from Site 1 and dilutions of water from Site 2 where the number of Hydra increased. This may be a useful observation when assessing the acute hazard of a water sample and the effects of dilution on biota. In conclusion, both organisms can be used for ecotoxicity testing with D. pulex being a more sensitive indicator of toxicity. Due to the sensitivities observed over time, H. vulgaris may be used for chronic toxicity testing and D. pulex for acute toxicity testing.
ACKNOWLEDGEMENTS
The authors express their gratitude to the following people from the Department of Zoology: Prof B Jansen van Vuuren for the genetic sequencing of the Hydra and Mr M Mathonsi for technical support in the Aquarium of the Department of Zoology of the University of Johannesburg. This study was supported by the Department of Zoology of the University of Johannesburg.
REFERENCES
AKCIL A and KOLDAS S (2006) Acid mine drainage (AMD): causes, treatment and case studies. J. Cleaner Prod. 14 1139-1145. https://doi.org/10.1016/j.jclepro.2004.09.006 [ Links ]
ARKHIPCHUK VV, BLAISE C and MALINOVSKAYA MV (2006) Use of hydra for chronic toxicity assessment of waters intended for human consumption. Environ. Pollut. (Oxford, U.K.) 142 200-211. https://doi.org/10.1016/j.envpol.2005.10.012 [ Links ]
BADERNA D, MAGGIONI S, BORIANI E, GEMMA S, MOLTENI M, LOMBARDO A, COLOMBO A, BORDONALI S, ROTELLA G, LODI M and co-authors (2011) A combined approach to investigate the toxicity of an industrial landfill's leachate: Chemical analyses, risk assessment and in vitro assays. Environ. Res. 111 603-613. https://doi.org/10.1016/j.envres.2011.01.015 [ Links ]
BEACH MJ and PASCOE D (1998) The role of Hydra vulgaris (Pallas) in assessing the toxicity of freshwater pollutants. Water Res. 32 101-106. https://doi.org/10.1016/S0043-1354(97)00180-2 [ Links ]
BELL G and WOLFE LM (1985) Sexual and asexual reproduction in a natural population of Hydra pseudoligactis. Can. J. Zool. 63 851-856. https://doi.org/10.1139/z85-126 [ Links ]
BEZUIDENHOUT C (2013) A large scale study of microbial and physic-chemical quality of selected groundwaters and surface waters in the North-West Province, South Africa. WRC Report No. 1966/1/13. Water Research Commission, Pretoria. [ Links ]
BITTON G, RHODES K, KOOPMAN B and CORNEJO M (1995) Short-term Toxicity assay based on daphnid feeding behaviour. Water Environ. Res. 67 290-293. https://doi.org/10.2175/106143095X131493 [ Links ]
BLAISE C and KUSUI T (1997) Acute toxicity assessment of industrial effluents with a microplate-based Hydra attenuata assay. Environ. Toxicol. Water Qual. 12 53-60. https://doi.org/10.1002/(SICI)1098-2256(1997)12:1<53::AID-TOX8>3.0.CO;2-7 [ Links ]
BLAISE C and FÈRARD J-F (2005) Overview of contemporary toxicity testing. In: Blaise C and Fèrard J-F (eds.) Small-Scale Freshwater Toxicity Investigations: Volume 1. Springer, Dordrecht. [ Links ]
BOWDEN HC, WILBY OK, BOTHAM CA, ADAM PJ and ROSS FW (1995) Assessment of the toxic and potential teratogenic effects of four glycol ethers and two derivatives using the hydra regeneration assay and rat whole embryo culture. Toxicol. In Vitro 9 (5) 773-781. https://doi.org/10.1016/0887-2333(95)00054-C [ Links ]
BRITZ TG and SIGGE GO (2012) Quantitative investigation into the link between irrigation water quality and food safety: Volume I: Synthesis report. WRC Report No. 1773/1/12. Water Research Commission, Pretoria. [ Links ]
CAIRNS J (1995) The Genesis of ecotoxicology. In: Cairns J and Niederlehner BR (eds.) Ecological Toxicity Testing: Scale, Complexity, and Relevance. Lewis Publishers, Florida. [ Links ]
CLIFFORD M and MCGEER JC (2009) Development of a biotic ligand model for the acute toxicity of zinc to Daphnia pulex in soft waters. Aquat. Toxicol. 91 26-32. https://doi.org/10.1016/j.aquatox.2008.09.016 [ Links ]
CZERNIAWSKA-KUSZA I, CIESIELCZUK T, KUSZA G and CICHON A (2006) Comparison of the Phytotoxkit microbiotest and chemical variables for toxicity evaluation of sediments. Environ. Toxicol. 21 367-372. https://doi.org/10.1002/tox.20189 [ Links ]
DWAF (Department of Water Affairs and Forestry, South Africa) (DWAF) (1996) South African Water Quality Guidelines. Volume 7: Aquatic Ecosystems. Department of Water Affairs and Forestry, Pretoria. 145 pp. [ Links ]
DIAZ-BAEZ MC and DUTKA BJ (2005) Frameworks for the application of toxicity data. In: Thompson KC, Wadhia K and Loibner AP (eds.) Environmental Toxicity Testing. Blackwell Publishers Ltd, Oxford, United Kingdom. [ Links ]
DINELLI E, LIMA A, DE VIVO B, ALBANESE S, CICCHELLA D, and VALERA P (2010) Hydrogeochemical analysis on Italian bottled mineral waters: Effects of geology. J. Geochem. Explor. 107 317-335. https://doi.org/10.1016/j.gexplo.2010.06.004 [ Links ]
EC (Environment Canada) (1996) Biological Test Method: Acute Lethality Test Using Daphnia spp. EPS 1/RM/11. Environmental Technology Centre, Ontario. [ Links ]
FERRAO-FILHO AS, SOARES MCS, DE MAGALAHES VF and AZEVEDO SMFO (2010) A rapid bioassay for detecting saxitoxins using Daphnia acute toxicity test. Environ. Pollut. 185 2084-2093. https://doi.org/10.1016/j.envpol.2010.03.007 [ Links ]
FINNEY DJ (1971) Probit Analysis (3rd edition). Cambridge University Press, London. 333 pp. [ Links ]
FINNEY DJ (1978) Statistical Method in Biological Assay (3rd edn). Charles Griffin & Co. Ltd, London. 508 pp. [ Links ]
FUKUHORI N, KITANO M and KIMURA H (2005) Toxic effects of Bisphenol A on sexual and asexual reproduction in Hydra oligactis. Arch. Environ. Contam. Toxicol. 48 495-500. https://doi.org/10.1007/s00244-004-0032-1 [ Links ]
GALLAGHER JS, DUKE BM and RODGERS JH-JR (2005) Responses of Hyalella azteca and Ceriodaphnia dubia to reservoir sediments following chelated copper herbicide applications. J. Aquat. Plant Manage. 43 95-99. [ Links ]
GOODFELLOW WL (2005) The aquatic environment. In: Thompson KC, Wadhia K and Loibner AP (eds.) Environmental Toxicity Testing. Blackwell Publishers Ltd, Oxford, United Kingdom. https://doi.org/10.1002/9781444305531.ch5 [ Links ]
HALL JA and GOLDING LA (1998) Standard methods for whole effluent toxicity testing: development and application. NIWA Report for the Ministry for the Environment, New Zealand. Report no. MFE80205. National Institute of Water and Atmospheric Research Ltd, Hamilton. [ Links ]
HAMILTON MA, RUSSO RC and THURSTON RV (1977) Trimmed Spearman-Karber method for estimating median lethal concentrations. Environ. Sci. Technol. 11 (7) 714-719. https://doi.org/10.1021/es60130a004 [ Links ]
HOLDWAY DA (2005) Hydra population reproduction toxicity test method. In: Blaise C and Fèrard J-F (eds.) Small-scale Freshwater Toxicity Investigations: Volume 1. Springer, Dordrecht, Netherlands. 395-411. https://doi.org/10.1007/1-4020-3120-3_12 [ Links ]
JONCZYK E and GILRON G (2005) Acute and chronic toxicity testing with Daphnia sp. In: Blaise C and Fèrard J-F (eds.) Small-scale Freshwater Toxicity Investigations: Volume 1. Springer, Dordrecht, Netherlands. 337-393. https://doi.org/10.1007/1-4020-3120-3_11 [ Links ]
KARNTANUT W and PASCOE D (2000) A comparison of methods for measuring acute toxicity to Hydra vulgaris. Chemosphere 41 1543-1548. https://doi.org/10.1016/S0045-6535(00)00068-0 [ Links ]
KARNTANUT W and PASCOE D (2002) The toxicity of copper, cadmium and zinc to four different Hydra (Cnidaria: Hydrazoa). Chemosphere 47 1059-1064. https://doi.org/10.1016/S0045-6535(02)00050-4 [ Links ]
KARNTANUT W and PASCOE D (2005) Effects of removing symbiotic green algae on the response of Hydra viridissima (Pallas 1776) to metals. Ecotoxicol. Environ. Saf. 60 301-305. https://doi.org/10.1016/j.ecoenv.2004.04.001 [ Links ]
KOIVISTO S and KETOLA M (1995) Effects of copper on life-history traits of Daphnia pulex and Bosmina longirostris. Aquat. Toxicol. 32 255-269. https://doi.org/10.1016/0166-445X(94)00094-7 [ Links ]
KOZLOVA T, WOOD CM and MCGEER JC (2009) The effect of water chemistry on the acute toxicity of nickel to the cladoceran Daphnia pulex and the development of a biotic ligand model. Aquat. Toxicol. 91 221-228. https://doi.org/10.1016/j.aquatox.2008.11.005 [ Links ]
LEONARD EM and WOOD CM (2013) Acute toxicity, critical body residues, Michaelis-Menten analysis of bioaccumulation, and ionoregulatory disturbance in response to waterborne nickel in four invertebrates: Chironomus riparius, Lymnaea stagnalis, Lumbriculus variegatus and Daphnia pulex. Comp. Biochem. Physiol., Part C 158 10-21. https://doi.org/10.1016/j.cbpc.2013.03.008 [ Links ]
LIANG-QI L, CI-AN S, XIAN-LI X, YAN-HONG L and FEI W (2010). Acid mine drainage and heavy metal contamination in groundwater of metal sulphide mine at arid territory (BS Mine, Western Australia). Trans. Nonferrous Met. Soc. China 20 1488-1493. https://doi.org/10.1016/S1003-6326(09)60326-5 [ Links ]
LIDMAN U (2005) The nature and chemistry of toxicants. In: Thompson KC, Wadhia K and Loibner AP (eds.) Environmental Toxicity Testing. Blackwell Publishers Ltd, Oxford, United Kingdom. https://doi.org/10.1002/9781444305531.ch3 [ Links ]
LOEHR LC, BEEGLE-KRAUSE C-J, GEORGE K, MCGEE CD, MEARNS AJ and ATKINSON MJ (2006) The significance of dilution in evaluating possible impacts of wastewater discharges from large cruise ships. Mar. Pollut. Bull. 52 681-688. https://doi.org/10.1016/j.marpolbul.2005.10.021 [ Links ]
LYNCH M (1983) Ecological genetics of Daphnia pulex. Evolution 37 (2) 358-374. https://doi.org/10.2307/2408344 [ Links ]
LOGUE CL, KOOPMAN B, BROWN GK and BITTON G (1989) Toxicity screening in a large, municipal wastewater system. Res. J. Water Pollut. Control Fed. 61 (5) 632-640. [ Links ]
MITCHELL FM and HOLDWAY DA (2000) The acute and chronic toxicity of the dispersants Corexit 9527 and 9500, water accommodated fraction (WAF) of crude oil, and dispersant enhanced WAF (DEWAF) to Hydra viridissima (green hydra). Water Res. 34 (1) 343-348. https://doi.org/10.1016/S0043-1354(99)00144-X [ Links ]
MULLER WJ and PALMER CG (2002) The use of Daphnia spp. and indigenous river invertebrates in whole effluent toxicity testing in the Vaal Catchment. WRC Report No. 815/1/02. Water Research Commission, Pretoria. [ Links ]
MURRAY K, SLABBERT L and MOLOI B (2003) Needs assessment and development framework for a tested implementation plan for the utilisation and execution of a National Toxicants Monitoring Programme (NTMP). Department of Water Affairs and Forestry, Pretoria. 50 pp. [ Links ]
MURRAY K, HEATH R and ALBERTUS A (2004) Design of a South African National Toxicity Monitoring Programme for inland surface waters. Proceedings of the 2004 Water Institute of Southern Africa (WISA) Biennial Conference, 2-4 May 2004, Cape Town. [ Links ]
OFFEM BO and AYOTUNDE EO (2008) Toxicity of lead to freshwater invertebrates (Water fleas; Daphnia magna and Cyclop sp.) in fish ponds in a tropical floodplain. Water, Air, Soil Pollut. 192 39-46. https://doi.org/10.1007/s11270-008-9632-0 [ Links ]
PARDOS M, BENNINGHOFF C, GUÉGUEN C, THOMAS R, DOBROWOLSKI J and DOMINIK J (1999) Acute toxicity assessment of Polish (waste) water with a microplate-based Hydra attenuata assay: a comparison with the Microtox®test. Sci. Total Environ. 243-244 141-148. https://doi.org/10.1016/S0048-9697(99)00369-1 [ Links ]
PASCOE D, KARNTANUT W and MULLER T (2003) Do pharmaceuticals affect freshwater invertebrates? A study with the cnidarians Hydra vulgaris. Chemosphere 51 521-528. https://doi.org/10.1016/S0045-6535(02)00860-3 [ Links ]
PENNAK RW (1989) Freshwater Invertebrates of the United States Protozoa to Mollusca. (3rd edn). John Wiley and Sons, New York. [ Links ]
PERSOONE G and JANSSEN CR (1993) Freshwater invertebrate toxicity tests. In: Calow P (ed.) Handbook of Ecotoxicology: Volume 1. Blackwell Science Ltd., London. [ Links ]
PERSOONE G, MARSALEK B, BLINOVA I, TOROKNE A, ZARINA D, MANUSADZIANAS L, NALECZ-JAWECKI G, TOFAN L, STEPANOVA N, TOTHOVA L and CO AUTHORS (2003) A practical and user-friendly toxicity classification system with microbiotests for natural and wastewaters. Environ. Toxicol. 18 (6) 395-402. https://doi.org/10.1002/tox.10141 [ Links ]
POLLINO CA and HOLDWAY DA (1999) Potential of two hydra species as standard toxicity test animals. Ecotoxicol. Environ. Saf. 43 309-316. https://doi.org/10.1006/eesa.1999.1796 [ Links ]
QUINN B, GAGNÉ F and BLAISE C (2008a) An investigation into the acute and chronic toxicity of eleven pharmaceuticals (and their solvents) found in wastewater effluent on the cnidarians, Hydra attenuata. Sci. Total Environ. 389 306-314. https://doi.org/10.1016/j.scitotenv.2007.08.038 [ Links ]
QUINN B, GAGNÉ F and BLAISE C (2008b) The effects of pharmaceuticals on the regeneration of the cnidarians, Hydra attenuata. Sci. Total Environ. 402 62-69. https://doi.org/10.1016/j.scitotenv.2008.04.039 [ Links ]
QUINN B, GAGNÉ F and BLAISE C (2009) Evaluation of the acute, chronic and teratogenic effects of a mixture of eleven pharmaceuticals on the cnidarians, Hydra attenuata. Sci. Total Environ. 407 1072-1079. https://doi.org/10.1016/j.scitotenv.2008.10.022 [ Links ]
RSA (Republic of South Africa) (1998) National Water Act (Act No. 36 of 1998). Department of Water Affairs and Forestry, South Africa. Government Gazette no. 19182. Government Printers, Cape Town. [ Links ]
SANCHEZ P, ALONSO C, FERNANDEZ C, VEGA MM, GARCIA MP and TARAZONA JV (2005) Evaluation of a multi-species test system for assessing acute and chronic toxicity of sediments and water to aquatic invertebrates: Effects of pentachlorophenol on Daphnia magna and Chironomus prasinus. J. Soils Sediments 5 (1) 53-58. https://doi.org/10.1065/jss2004.10.114 [ Links ]
SHUHAIMI-OTHMAN M, NADZIFAH Y and AHMAD AK (2010) Toxicity of copper and cadmium to freshwater fishes. World Acad. Sci. Eng. Technol. 65 869-871. [ Links ]
SLABBERT JL and VENTER EA (1999) Biological assays for aquatic toxicity testing. Water Sci. Technol. 39 (10-11) 367-373. https://doi.org/10.1016/S0273-1223(99)00300-5 [ Links ]
SORGELOOS P and PERSOONE G (1975) Technological improvements for the cultivation of invertebrates as food for fishes and crustaceans. II. Hatching and culturing of the brine shrimp, Artemia salina L. Aquaculture 6 303-317. https://doi.org/10.1016/0044-8486(75)90110-6 [ Links ]
SPRULES WG (1972) Effects of size-selective predation and food competition on high altitude zooplankton communities. Ecology 53 (3) 375-386. https://doi.org/10.2307/1934223 [ Links ]
STARK JD and VARGAS RI (2003) Demographic changes in Daphnia pulex (Leydig) after exposure to the insecticides spinosad and diazinon. Ecotoxicol. Environ. Saf. 56 334-338. https://doi.org/10.1016/S0147-6513(02)00074-X [ Links ]
STARK JD and VARGAS RI (2005) Toxicity and hazard assessment of fipronil to Daphnia pulex. Ecotoxicol. Environ. Saf. 62 11-16. https://doi.org/10.1016/j.ecoenv.2005.02.011 [ Links ]
SUTER GW (1995) Endpoints of interest at different levels of biological organization. In: Cairns J and Niederlehner BR (eds.) Ecological Toxicity Testing: Scale, Complexity, and Relevance. Lewis Publishers, Florida. [ Links ]
TROTTIER S, BLAISE C, KUSUI T and JOHNSON EM (1997) Acute toxicity assessment of aqueous samples using a microplate-based Hydra attenuata assay. Environ. Toxicol. Water Qual. 12 265-271. https://doi.org/10.1002/(SICI)1098-2256(1997)12:3<265::AID-TOX10>3.0.CO;2-9 [ Links ]
TRUTER E (1994) Method for estimating the chronic toxicity of a chemical or water sample to the Cladoceran Daphnia pulex. Institute for Water Quality Studies, Department of Water Affairs and Forestry, Pretoria. 11 pp. [ Links ]
USEPA (United States Environmental Protection Agency) (2002) Methods for measuring the acute toxicity of effluents and receiving waters to freshwater and marine organisms (5th edn). EPA 821/R-02/012, October 2002. USEPA Office of Water, Washington DC. 266 pp. [ Links ]
WHARFE J (2005) Historical perspective and overview. In: Thompson KC, Wadhia K and Loibner AP (eds) Environmental Toxicity Testing. Blackwell Publishers Ltd, United Kingdom. https://doi.org/10.1002/9781444305531.ch1 [ Links ]
WILBY OK (1988) The Hydra regeneration assay. Proceedings of workshop organised by Association Francaise de Teratologie, France. 108-124. [ Links ]
WOOD B and STARK JD (2002) Acute toxicity of drainage ditch water from a Washington State cranberry-growing region to Daphnia pulex in laboratory bioassays. Ecotoxicol. Environ. Saf. 53 273-280. https://doi.org/10.1006/eesa.2002.2210 [ Links ]
ZALIZNIAK L and NUGEGODA D (2006) Effect of sublethal concentrations of chlorpyrifos on three successive generations of Daphnia carinata. Ecotoxicol. Environ. Saf. 64 207-214. https://doi.org/10.1016/j.ecoenv.2005.03.015 [ Links ]
Received 22 July 2014
Accepted in revised form 27 March 2017
* To whom all correspondence should be addressed. Tel: + 27 76 819 4518; e-mail: prasheensingh@yahoo.com














