Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
Water SA
versión On-line ISSN 1816-7950
versión impresa ISSN 0378-4738
Water SA vol.43 no.2 Pretoria abr. 2017
http://dx.doi.org/10.4314/wsa.v43i2.13
Aqueous solubility of Cr(VI) compounds in ferrochrome bag filter dust and the implications thereof
SP du PreezI; JP BeukesI, *; WPJ van DalenI; PG van ZylI; D PaktuncII; MM Loock-HattinghI
IChemical Resource Beneficiation, North-West University, Potchefstroom Campus, Private Bag X6001, Potchefstroom 2520, South Africa
IICanmetMINING, Natural Resources Canada, 555 Booth St., Ottawa, ON, K1A0G1, Canada
ABSTRACT
The production of ferrochrome (FeCr) is a reducing process. However, it is impossible to completely exclude oxygen from all of the high-temperature production process steps, which may lead to unintentional formation of small amounts of Cr(VI). The majority of Cr(VI) is associated with particles found in the off-gas of the high-temperature processes, which are cleaned by means of venturi scrubbers or bag filter dust (BFD) systems. BFD contains the highest concentration of Cr(VI) of all FeCr wastes. In this study, the solubility of Cr(VI) present in BFD was determined by evaluating four different BFD samples. The results indicate that the currently applied Cr(VI) treatment strategies of the FeCr producer (with process water pH ≤ 9) only effectively extract and treat the water-soluble Cr(VI) compounds, which merely represented approximately 31% of the total Cr(VI) present in the BFD samples evaluated. Extended extraction time, within the afore-mentioned pH range, proved futile in extracting sparingly-soluble and water-insoluble Cr(VI) species, which represented approximately 34% and 35% of the total Cr(VI), respectively. Due to the deficiencies of the current treatment strategies, it is highly likely that sparingly water-soluble Cr(VI) compounds will leach from waste storage facilities (e.g. slimes dams) over time. Therefore, it is critical that improved Cr(VI) treatment strategies be formulated, which should be an important future perspective for FeCr producers and researchers alike.
Keywords: hexavalent chromium, Cr(VI), ferrochromium, ferrochrome, bag filter dust, smelter waste
INTRODUCTION
Since its discovery in 1798, chromite has remained the only commercially viable source of new chromium (Cr) units (Murthy et al. 2011; Riekkola-Vanhanen, 1999). Approximately 90% to 95% of mined chromite is used in the metallurgical industry for the production of various grades of FeCr, which is produced through the carbo-thermic reduction of chromite ore (Riekkola-Vanhanen, 1999). About 80% to 90% of ferrochrome (FeCr) is consumed by the stainless-steel industry, primarily as high-carbon or charge-grade FeCr (Murthy et al. 2011). Stainless steel is a vital alloy in various applications (Gasik, 2013), with the Cr content of stainless steel increasing its corrosion resistance. South Africa holds the largest chromite deposits and is the second-largest FeCr producing country (ICDA, 2012), with 14 FeCr smelters (Beukes et al., 2012).
Cr is present as Cr(III) in chromite ore, while Cr(0) occurs in the produced FeCr. Although completely unintended, small amounts of Cr(VI) are formed during FeCr production, which may be present in waste materials (Beukes et al., 2010). Cr(VI) is generally regarded as carcinogenic (IARC, 2012), with specifically airborne exposure to Cr(VI) being associated with cancer of the respiratory system (Proctor et al., 2002). In contrast to Cr(VI), Cr(III) and Cr(0) are not classified as carcinogenic. Cr(III) is in fact used a dietary supplement for certain human health abnormalities (Hininger et al., 2007).
The main types of waste generated during FeCr production are slag, sludge from wet venturi scrubbers and bag filter dust (BFD) (Van Staden et al., 2014). Slag-to-FeCr generation ratios of 1.1:1 up to 1.9:1 are common in this industry, with ratios varying according to different production technologies employed (Beukes et al., 2010; Niemelä and Kauppi, 2007). Although the volumes of slag generated are large, the Cr(VI) content of the slag is usually very low (Beukes et al., 2010; Niemelä and Kauppi, 2007). Additionally, FeCr slag can be used in various commercial applications (Niemelä and Kauppi, 2007; Zelić, 2005; Lind et al., 2001; Riekkola-Vanhanen, 1999). Sludge, which is generated during the wet venturi scrubbing of closed furnace off-gas (Beukes et al., 2010; Niemelä et al., 2004) does not usually contain significant Cr(VI) concentrations (Gericke, 1998). However, in contrast to slag and sludge, BFD generated during the cleaning of off-gas from open/semi-closed FeCr furnaces contains significant levels of Cr(VI) (Beukes et al., 2012; Maine et al., 2005; Gericke, 1998), which cannot be disposed without proper Cr(VI) treatment (Beukes et al., 2012). After the BFD is treated to reduce Cr(VI), it is usually disposed in fit-for-purpose waste facilities such as slimes dams.
Treatment of BFD (and indeed any Cr(VI) containing FeCr waste) usually involve the aqueous reduction of Cr(VI) with an appropriate inorganic reducing agent such as Fe(II) (Beukes et al., 2012; Seaman et al., 1999; Buerge and Hug, 1997) and S(IV) (Beukes et al., 2001 and 1999), or bacterial reduction (Dhal et al., 2013; Molokwane et al., 2008). Although Cr(VI) can also be reduced with numerous organic compounds (Kassem, 2010; March, 1992), this is usually avoided due to the potential solubilisation of Cr(III) (Beukes et al., 2012; Apte et al., 2006). In this paper, the leaching potential of Cr(VI) from BFD is investigated and it is proven that this can be a major limitation in the effectiveness of the conventional Cr(VI) treatment used by industry.
MATERIALS AND METHODS
Materials
Numerous factors can influence the composition of BFD, including the production technology employed (e.g. open or semi-closed furnace technology, type of filters used in the bag filter plant), physical separation in the bag filter plant itself (e.g. some compartments containing finer material than others), metallurgical operating conditions (e.g. basic or acid slag operating conditions) and composition of feed materials (e.g. chemical and physical differences of ores). Therefore, four different BFD samples were obtained from FeCr producers in South Africa. These producers preferred to remain anonymous and therefore the samples are merely referred to as BFD samples A, B, C and D.
All chemicals used were analytical grade reagents. Standard Cr(VI) solutions were prepared from a 1 009 ± 5 mg·mL-1 aqueous chromate (CrO42-) analytical solution (Spectrascan, distributed by Teknolab AB, Sweden), which were used for calibration and verification of the analytical technique employed. The post-column reagent that was used during Cr(VI) analysis was prepared using 1,5-diphenylcarbazide (DPC) (FLUKA), 98% sulfuric acid (Rochelle Chemicals) and HPLC grade methanol (Ace). Solutions of sodium hydroxide (Merck) and perchloric acid (Merck) were used to adjust the pH of aqueous solutions/mixtures. According to solubility data, Cr(VI) chemicals can be classified as water soluble, sparingly water soluble and water insoluble (IARC, 2012; Ashley et al., 2003). Ammonium sulphate with 99% purity (Merck SA) and 25% ammonia solution (Associated chemical enterprises) were used to prepare a 0.05 M (NH4)2SO4 - 0.05 M NH4OH extraction buffer that is capable of leaching all water-soluble and sparingly water-soluble Cr(VI) compounds (Ashley et al., 2003). Sodium hydroxide (Promark chemicals) and anhydrous sodium carbonate (Merck SA) were used to prepare a 3 wt% Na2CO3 - 2 wt% NaOH extraction buffer, to quantitatively extract all Cr(VI) in the BFD samples, including the water-insoluble fraction (Ashley et al., 2003). Ultra-pure deionized water (resistivity, 18.2 MΩcm-1), produced by a Milli-Q water purification system, was used for all dilutions and aqueous extractions. 99.999% pure nitrogen gas (N2) (AFROX) was used to provide an inert environment during leaching. Hydrophilic PVDF 0.45 µm filters (Millipore Millex, USA) were used for the filtration of solutions.
BFD sample characterisation
A Malvern Mastersizer 2000 was used to determine the particle size distribution of the BFD samples. A diluted suspension of material was ultra-sonicated for 1 min prior to the particle size measurement, in order to disperse the individual particles and to prevent the use of a chemical dispersant. Laser obscuration was kept at 10% to 15%, while mechanical stirring was set to 2 000 r·min-1. Ten measurements were made for each sample and the average taken.
Scanning electron microscopy with energy dispersive x-ray detectors (SEM-EDS) was used to perform morphological and chemical characterisation of the BFD particles. FEI Quanta 250 FEG ESEM incorporating Oxford Inca X-Max 20 EDS system with a 15 kV electron beam at a 10 mm working distance was used. BFD samples were prepared with two different procedures for SEM analysis. Firstly, samples were mounted onto a specimen stub with carbon-coated tape and subsequently gold coated in order to determine the general BFD particle characteristics, e.g., size and shape. In order to determine the chemical compositions of the BFD samples by SEM-EDS, the samples were set in resin and polished before being analysed. The SEM-EDS analysis was performed by scanning a wide rectangular area of the sample surface in a raster-like pattern for 60 s. Spot analyses were also performed to determine the composition of individual particles present in the BFD.
X-ray diffraction (XRD) analyses were performed on BFD samples ground to < 45 µm using a Rigaku D/MAX 2500 rotating-anode powder diffractometer with Cu Kα radiation at 50 kV, 260 mA, a step-scan of 0.02°, and a scan rate at 1 min-1 in 2 h from 5° to 70°. Phase identifications were made using JADE v.9.3 coupled with the ICSD and ICDD diffraction databases.
Trace metal analysis
Inductively coupled plasma mass spectroscopy (ICP-MS) (Agilent 7500CE ICP-MS with Octopole Reaction System) was used to determine the total metal concentrations, irrespective of oxidation state, present in aqueous leach solutions. These solutions were obtained as described in the next section.
Cr(VI) extraction and analysis
For each BFD sample, 0.50 g was transferred into a 100 mL glass beaker, which contained 50 mL of deionized water. The initial pH of the deionized water had been adjusted to a predetermined value. Leaching solutions were purged with N2 for 10 min prior to Cr(VI) extraction to remove dissolved oxygen from the solution, which may cause unwanted Cr(III) oxidation (Ashley et al., 2003). Each beaker and its contents were placed on a magnetic stirrer and agitated for a period of 2 h. A steady stream of N2 was continually used to purge the suspension, ensuring an inert environment during leaching. After Cr(VI) extraction, the leaching solutions were filtered through hydrophilic Millipore 0.45 µm PVDF filters to remove particulate matter, followed by another pH measurement to ensure that no pH drift occurred. Thereafter the filtrates were transferred to 100 mL A-grade volumetric flasks and diluted to a final volume of 100 mL with pH-adjusted deionized water. 50 mL of each solution was used for aqueous trace metal analyses, while the remaining 50 mL was used for Cr(VI) analysis. All pH measurements were conducted with a HANNA - HI 991001 pH meter with a temperature probe.
In addition to Cr(VI) extraction at specific pH values, Cr(VI) extractions were also performed using hot plate digestion. A buffer of 0.05 M (NH4)2SO4 and 0.05 M NH4OH was used to leach water-soluble and sparingly water-soluble Cr(VI) compounds, while a buffer of 3 wt% Na2CO3 and 2 wt% NaOH was used to leach total Cr(VI) (Ashley et al., 2003; NIOSH 7605, 2003). The afore-mentioned buffer (Na2CO3 - NaOH) enhances the dissolution of insoluble Cr(VI) compounds at higher pH levels by the addition of carbonate (CO32-), as indicated in Eq. 1. In the presence of a large excess of CO32- the equilibrium shifts quantitatively to the right of Eq. 1 and insoluble Cr(VI) becomes soluble (Ashley et al., 2003).
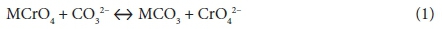
with M = cation associated with chromate
The Cr(VI) concentrations of aliquots were determined with a Thermo Scientific Dionex ICS 3000 ion chromatograph (IC) according to the procedure described by Loock et al. (2014) and Du Preez et al. (2015), which was derived from Dionex Application Update 179 (2011), Dionex Application Update 144 (2003), and Thomas et al. (2002). The IC system was equipped with a Dionex IonPac AG7 4 x 50 mm guard column and a Dionex IonPac AS7 4 x 250 mm analytical separation columns. A 1 000 µL injection coil and a 2 x 375 µL knitted reaction coil coupled to a UV-visible absorbance detector set at a wavelength of 530 nm were used. 250 mM ammonium sulphate was used as an eluent and an isocratic pump carried the eluent and the injected sample through the system. 2 mM DPC solution was used for post column coloration, which was delivered into the system by an AXP pump. The flow rates of the eluent and colorant were 1.0 mL·min-1 and 0.5 mL·min-1, respectively. A six-point calibration was performed for Cr(VI), ranging from 5 to 75 µg·L-1. The correlation coefficient of this linear calibration line was > 99.96%. The detection limit of this method at a confidence level of 98.3%, which was calculated according to the method specified by Skoog et al. (2013), was experimentally determined as 0.9 µg·L-1 by Loock-Hattingh (2016). The accuracy of the specific analytical method (and the specific instrument used) was recently indicated (Venter et al., 2016) by analysis of a Community Bureau of Reference (former reference materials program of the European Commission) BCR No 545 welding dust filter certified reference material (individual identification No B7-36, purchased on 25 January 2013), for which a value of 39.7 mg·g-1 Cr(VI) was obtained, which was within the uncertainty of the reference material that was defined as the half width of the 95% confidence interval of the mean of 40.2 mg·g-1 Cr(VI).
X-ray absorption spectroscopy (XAS)
XAS experiments were carried out at PNC-CAT's bending magnet beam-line 20-BM of the Advanced Photon Source (APS), Argonne, IL, USA. Finely ground and homogenized BFD samples were placed as monolayers on tapes and loaded into Plexiglass sample holders. Experiments were carried out at room temperature both in transmission and fluorescence modes using ion chambers filled with nitrogen and helium gases, and a Canberra 13-element detector. A Cr foil was used for energy calibration. Between 4 and 6 scans were collected from each sample and the reference materials. Data reduction and analysis were performed with ATHENA (Ravel and Newville, 2005). The least squares fitting of the XANES spectra were made with LSFitXAFS (Paktunc, 2004). XAS, including both X-ray absorption near-edge spectroscopy (XANES) and extended X-ray absorption fine structure (EXAFS) regimes, was employed to determine the sources and quantitative speciation of Cr in the BFD samples and possible cation association of CrO42- that could be leached and precipitated from the BFD samples. Precipitates were prepared by contacting 50 g of BFD with 1 L of deionized water for 4 consecutive 24 h contacting periods. After 24 h contact, the water was separated from the BFD by means of filtration, after which the BFD were re-contacted with fresh water and the procedure repeated. All four leachates were combined and then allowed to evaporate at room temperature in a fume cupboard circulated with filtered air to prevent contamination, producing a solid residue that was analysed with XAS spectroscopy techniques.
RESULTS AND DISCUSSION
Particle size analysis
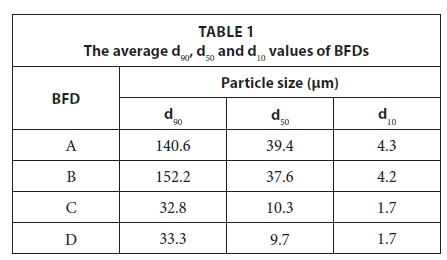
The d90, d50 and d10 of the particle size distribution of the different BFD samples are presented in Table 1. The d90 is defined as the equivalent particle size for which 90% of the particles are smaller, with definitions of d50 and d10 derived similarly. The particle size analyses indicated that the d90 of all four BFD samples ranged between 32.8 and 152.2 μm, with the A and B samples being coarser than the C and D samples. The particle sizes reported here are similar to BFD particle sizes reported previously (Van Staden et al., 2014; Beukes et al., 2010).
SEM observations
Figures 1(a), 1(b), 1(c) and 1(d) indicate SEM micrographs of BFD samples of A, B, C and D. Each of these figures present combined images, with the larger images indicating cross-sectional polished micrographs. The smaller images (in the bottom right-hand corners) represent micrographs of the BFDs pressed onto stubs with adhesive carbon tape. It is evident from these micrographs that the BFD samples consisted mainly of very small particulate matter (mostly < 40 µm), with some larger particles of up to approx. 200 µm. It was previously shown that the larger unevenly-shaped particles observed in BFD are unreacted or partially-reacted feed materials such as chromite, fluxes or reductants, while the very small spherical particles represent the actual off-gas particulates (Van Staden et al., 2014; Beukes et al., 2010; Beukes et al., 1999). However, to confrim that unreacted/partially-reacted chromite particles were present in the BFD samples, one of the unevenly-shaped grey particles, marked with a white box in Fig. 1(a) (BFD sample A), was analysed with SEM-EDS, which revealed Cr and Fe contents of 30.7 and 20.8 wt%, respectively. These correspond to a Cr/Fe ratio of 1.48 which is comparable to typical South African chromite ore (Cramer et al., 2004). The SEM micrographs also revealed that the BFD samples A and B (Figs. 1(a) and 1(b)) contained more unreacted/partially-reacted larger raw material particles when compared to C and D (Figs. 1(c) and 1(d)). The SEM observations confirmed the particle size analysis results (Table 1), which indicated that the A and B samples were coarser than C and D. As previously indicated, the presence of a greater number of larger raw material particles in the BFD could be ascribed to the differences in the technology applied or the feed materials used at different production facilities. For instance, some FeCr smelters utilize a cyclone to remove larger and denser particles before the off-gas is cleaned in the bag filter plant, while others merely use a drop-out box that is less effective.
SEM-EDS chemical analyses of the BFD samples are presented in Table 2. These results should be considered as semi-quantitative, with O content determined by difference. Carbon (C) was not analysed, since a carbon-based resin was used to set the BFD samples prior to polishing. Cr content of the samples varied between 3.28% (Sample C) and 7.62% (Sample B). As expected, the elements with lower elemental mass that are typically associated with actual off-gas particles, such as magnesium (Mg), aluminium (Al), silicon (Si) and zinc (Zn), were present in significant concentrations in all of the BFD samples. Of particular interest was that no noteworthy amounts of Na were detected in any of the BFD samples. It is well known that alkali roasting of chromite is an industrial process for the production of sodium chromate, the formation efficiency of which depends on the amount of O2 and alkali present, as well as the temperature during the production process (Antony et al., 2001). Maine et al. (2005) proposed that Cr(VI) could mainly be associated with alkali metals in FeCr BFD, but did not present any evidence to support this statement. However, the lack of Na in the BFD samples cannot be considered as evidence that anionic Cr(VI) (CrO42-) is not associated with significant amounts of Na since the SEM-EDS technique has limitations with respect to Na analysis.
XRD analysis
XRD of the BFDs indicated that all of the samples comprised mainly of the same crystalline compounds. In the order of decreasing abundance, they are chromiun spinel ((Fe2+,Mg)(Cr,Fe3+)2O4), quartz (SiO2), halite (NaCl), anorthite (CaAl2Si2O8), osakaite (Zn4SO4(OH)6·5H2O) and forsterite (Mg2SiO4). The similarities in crystalline phases of the investigated BFDs indicate that the samples originated from a similar type of process (i.e. FeCr production with BFD off-gas cleaning).
XAS characterisation
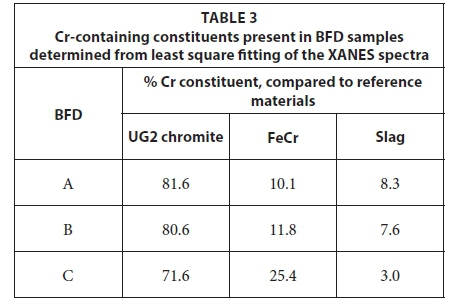
As is evident from Fig. 2(a) XAS spectra of the BFD samples collected at Cr K-edge indicate that they are broadly similar. The spectral features at the edge and above are similar to those of chromite from the upgraded upper group 2 (UG2) chromite layer of the Bushveld Complex in South Africa, suggesting that the unreacted chromite is the dominant Cr carrier in the furnaces where the BFD samples A, B and C were collected. BFD sample D was not characterized by XAS. Other important sources of Cr in the BFD samples include ferrochrome and slag. Figure 2(b) presents the least squares fitting of the Cr K-edge XANES spectra, indicating that the BFD samples consists of chromite, FeCr and FeCr slag, as represented by reference materials, i.e., UG2 chromite, FeCr and FeCr slag obtained from a FeCr smelter in South Africa. The BFD samples are dominated by chromite in terms of Cr concentrations, with chromite abundances ranging from 72% to 82% by weight (Table 3). The remaining Cr concentrations are tied to FeCr and slag.
Trace metal leachability
It is generally accepted that higher concentrations of most heavy and transitional metals will leach out of a solid matrix (e.g. waste and soil) at low pH values (Ding et al., 2014; Lee et al., 2012). This was also observed experimentally, as indicated in Figs. 3 and 4, which present the log concentrations of alkali and alkali earth (Fig. 3), as well as transition and heavy (Fig. 4) metals as a function of leach solution pH. The higher solubility at lower pH was especially evident at pH ≤ 3. Elements occurring in low concentrations in the BDF samples (< 10 mg·kg-1 BFD) were excluded from all figures to prevent congestion of the graphs, and Na contents were excluded from Figs 3(a) and (b) at pH > 7, since NaOH solution was used to increase the pH of the leach solutions.
When comparing metal concentrations presented in Fig. 3 to concentrations presented in Fig. 4, it is evident that most alkali and alkali earth metals, excluding Ba, were present in much higher concentrations than transition and heavy metals, except for Zn that had concentrations comparable to alkali and alkali earth metals, especially at very low pH. The results presented in Figs 3 and 4 will be contextualized further in the following sections.
Cr(VI) leachability
Figure 5(a) presents the leachable Cr(VI) concentrations as a function of leach solution pH, as well as the two solubility fractions, i.e., combined water-soluble and sparingly water-soluble Cr(VI), as well as total Cr(VI) determined. Figure 5(b) indicates the same data normalized to percentage, based on the total extractable Cr(VI) of each sample. From these results, it is evident that the amount and percentage of leachable Cr(VI) increased with an increase in leach solution pH at pH ≥ 9. This is in contrast to the results obtained for all the other metals (Figs 3 and 4) for which the leachability increased as the leach solution pH decreased.
From the data presented in Figs 3, 4 and 5, it is impossible to shed any light on the possible association of anionic CrO42- with cations, to indicate the identity of the Cr(VI) compounds that are present in the BFD samples. However, the observed increase of leachable Cr(VI) with an increase in pH (Fig. 5(b)) is an indication that not only water-soluble (e.g. Na2CrO4, K2CrO4), but also sparingly water-soluble (e.g. CaCrO4, SrCrO4) and waterinsoluble (e.g. BaCrO4, PbCrO4) Cr(VI) compounds occur in the samples evaluated. This data can also be used to estimate the average water-soluble, sparingly water-insoluble and water-insoluble fractions. As is evident from Fig. 5(b), there was no statistically significant difference in the extractable Cr(VI) at pH ≤ 9, with approximately 31% of the total Cr(VI) being extracted in this range. This represents the water-soluble Cr(VI) fraction. On average, approximately 65% of the total Cr(VI) was present as a combination of water-soluble and sparingly water-soluble compounds. The difference between these two factions, i.e. 34%, therefore represents the percentage of sparingly water-soluble Cr(VI) compounds. The difference between the total Cr(VI) (100%) and the combined water-soluble and sparingly water-soluble fractions (65%) revealed the presence of approximately 35% water-insoluble Cr(VI) compounds in the BFD samples evaluated.
The above-mentioned results and, in particular, the presence of substantial fractions of sparingly water-soluble and water-insoluble Cr(VI) compounds, contradict the general perception that total Cr(VI) extraction from ferrochrome BFD can be achieved by neutral or acidic aqueous extractions. For instance, Maine et al. (2005) indicated that > 100 h of extraction is required with neutral water to completely extract Cr(VI) from BFD. Gericke (1995) maintained that 24 h leaching with aqueous solutions of pH 2 to 6 is adequate for Cr(VI) extraction from BFD. Bulut et al. (2009) stated that only 30 min was required to dissolve all Cr(VI) present in BFD and that the solution pH did not play a significant role. However, none of these authors used buffers and extraction methods to ensure extraction of sparingly water-soluble and water-insoluble Cr(VI) compounds (Ashley et al., 2003).
Apart from the above-mentioned possible underestimation of the amount of Cr(VI) present in BFD by previous studies, the retention times applied in some studies (Maine et al., 2005; Gericke, 1995) are unpractical. Some FeCr producers pump their BFD sludge (BFD after being contacted with water) directly to a thickener to separate the solid particles from the process water, while others might have a small surge tank to regulate the pumping of the afore-mentioned BFD sludge to the thickener. Figure 6 indicates a simplified schematic illustration to demonstrate a typical process flow of BFD.
Typically, chemical Cr(VI) treatment takes place just prior to the BFD sludge entering the thickener. Currently, ferrous sulphate reduction of Cr(VI) is the most commonly applied treatment method (Beukes et al., 2012). However, Fe(II) that is not consumed to reduce Cr(VI) is oxidized within a couple of minutes to Fe(III), at the pH levels applicable to FeCr process water (Beukes et al., 2012; He et al., 2004; Buerge and Hug., 1997; Fendorf and Li, 1996). Therefore, the effective residence time of the BFD particles in the process water, during which Cr(VI) extraction can take place, is far less than the 2 h extraction time applied in this study. Considering the afore-mentioned description, it is evident that it is highly likely that the short extraction times associated with current Cr(VI) treatment strategies of FeCr producers only allow the extraction of the water-soluble Cr(VI) fraction.
Previously Maine et al. (2005) attempted to explain the slow release of Cr(VI), beyond the initial fast solubilisation of a certain fraction. These authors postulated that most of the Cr(VI) condenses onto the surface of the rounded fly ash type BFD particles, which can dissolve very rapidly in water. They further suggested that a small fraction of Cr(VI) might be trapped by the glassy surface of the BFD particles, which resulted in the slow release of the remaining Cr(VI). However, the results presented in this paper prove that the slow release, following the rapid initial dissolution of a fraction of the Cr(VI), can rather be attributed to the presence of substantial fractions of sparingly water-soluble and water-insoluble Cr(VI) compounds.
According to a survey conducted by Beukes et al. (2012) at various FeCr producers, the pH of FeCr process waters usually varies between 6.2 and 9.0. As indicated in Fig. 5(b), as well the discussions thereafter, approximately 31% of the total Cr(VI) is leached at pH ≤ 9, which correlates with the water-soluble Cr(VI) fraction. In order to confirm that the currently applied Cr(VI) treatment strategies extract only the water-soluble Cr(VI) fraction, even if extended extraction times are applied, the solid residue obtained from the evaporation of 4 successive 24 h extractions of the BDF samples at pH 7 were characterized with XAS and presented in Fig. 7. The pH level of 7 was chosen to avoid potential complications from pH adjustments (i.e. addition of CO32- and/or OH- altering cation association of CrO42- at higher pH levels).
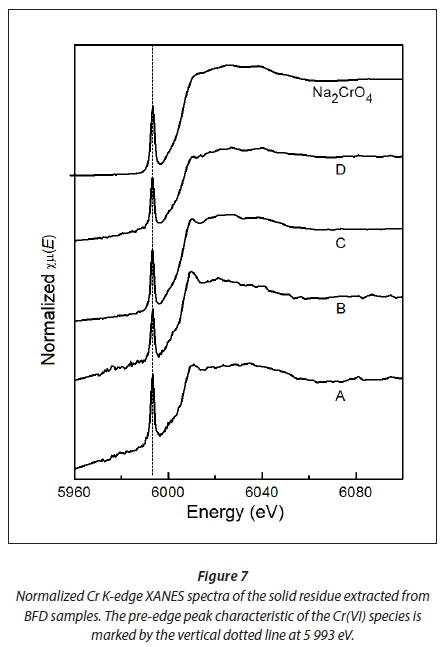
As is evident from Fig. 7, the XANES spectra of the solid residue of all four BFD samples investigated (A, B, C and D) indicated that the Cr present in the samples is dominated by Cr(VI) species. The spectra display a strong similarity to the spectrum of Na2CrO4, which is a water-soluble Cr(VI) species. This does not necessarily imply that Na2CrO4 was the sole source of Cr(VI) in the leachate precipitates of the BFD samples. Due to the low Cr concentrations, EXAFS spectra were not optimal and the results should therefore be regarded as qualitative, rather than quantitative. These results also did not permit modelling of the local coordination environment of the Cr(VI) species. Notwithstanding the afore-mentioned limitations, the presence of Cr(VI) in all BFD leachate precipitates is unequivocal; additionally, the Cr(VI) species were similar to Na2CrO4.
CONCLUSIONS
The results presented in this paper indicate that the currently applied Cr(VI) treatment strategies of FeCr producer (with process water pH ≤9) only effectively extract and treat the water-soluble Cr(VI) compounds from the BFD, which represent approximately 31% of the total Cr(VI) present in the BFD samples evaluated. Extended extraction times of 4 consecutive 24 h extractions, proved futile in extracting sparingly water-soluble and water-insoluble Cr(VI) species within the afore-mentioned pH range, as confirmed with XAFS.
Considering the above-mentioned, it is critical that improved Cr(VI) treatment strategies be formulated that would extract all of the Cr(VI) from BFDs. The results presented in this paper suggest that extraction at pH 13 would be required to solubilise the sparingly water-soluble Cr(VI) species from BFD. This will ensure that sparingly water-soluble Cr(VI) compounds do not leach from the waste storage facilities (e.g. slimes dams). Otherwise, there is the potential of Cr(VI) releases over time, which has been proven to results in environmental Cr(VI) pollution (Loock-Hatting, 2016; Loock-Hatting et al., 2015; Loock et al., 2014). However, it is highly unlikely that such a high process-water pH would be economically feasible. Additionally, it is well known from literature that Fe(II), currently the most commonly employed chemical reductant of Cr(VI), oxidizes very quickly with dissolved oxygen at such high pH levels. Therefore, the effectiveness of this and other potential reductants at elevated pH levels needs to be investigated. Furthermore, the need to extract the water-insoluble Cr(VI) species prior to chemical reduction should be investigated in greater detail, in order to assess the likelihood of such species being mobilised over extended time periods relevant to waste disposal facilities.
ACKNOWLEDGEMENTS
The authors of this paper wish to thank the four FeCr production facilities that provided the BFD case study samples that were used in this study. The financial assistance of the National Research Foundation (NRF) towards the PhD study of SP du Preez (grant number: 101345) is hereby acknowledged. Opinions expressed and conclusions arrived at, are those of the authors and are not necessarily to be attributed to the NRF. XAS experiments were performed at the PNC-CAT beamline of the Advanced Photon Source, Argonne National Laboratory which is supported by the US Department of Energy under Contracts W-31-109-Eng-38 (APS) and DE-FG03-97ER45628 (PNC-CAT). The XAS experiments were also supported by the Natural Sciences and Engineering Research Council of Canada through a MRS grant to the Canadian Light Source.
REFERENCES
ANTONY M, TATHAVADKAR V, CALVERT C and JHA A (2001) The soda-ash roasting of chromite ore processing residue for the reclamation of chromium. Metall. Mater. Trans. B. 32 987-995. https://doi.org/10.1007/s11663-001-0087-6 [ Links ]
APTE AD, TARE V and BOSE P (2006) Extent of oxidation of Cr(III) to Cr(VI) under various conditions pertaining to natural environment. J. Hazardous Mater. 128 164-174. https://doi.org/10.1016/j.jhazmat.2005.07.057 [ Links ]
ASHLEY K, HOWE AM, DEMANGE M and NYGREN O (2003) Sampling and analysis considerations for the determination of hexavalent chromium in workplace air. J. Environ. Monit. 5 707-716. https://doi.org/10.1039/b306105c [ Links ]
BEUKES JP, DAWSON NF and VAN ZYL PG (2010) Theoretical and practical aspects of Cr(VI) in the South African ferrochrome industry. J. S Afr. Inst. Min. Metall. 110 743-750. [ Links ]
BEUKES JP, PIENAAR JJ and LACHMANN G (2000) The reduction of hexavalent chromium by sulphite in wastewater - An explanation of the observed reactivity pattern. Water SA 26 393-395. [ Links ]
BEUKES JP, PIENAAR JJ, LACHMANN G and GIESEKKE EW (1999) The reduction of hexavalent chromium by sulphite in wastewater. Water SA 25 363-370. [ Links ]
BEUKES JP, VAN ZYL PG and RAS M (2012) Treatment of Cr(VI)-containing wastes in the South African ferrochrome industry - a review of currently applied methods. J. S. Afr. Inst. Min. Metall. 112 347-352. [ Links ]
BUERGE IJ and HUG SJ (1997) Kinetics and pH dependence of chromium (VI) reduction by iron (II). Environ. Sci. Technol. 31 1426-1432. https://doi.org/10.1021/es960672i [ Links ]
BULUT VN, OZDES D, BEKIRCAN O, GUNDOGDU A, DURAN C and SOYLAK M (2009) Carrier element-free co-precipitation (CEFC) method for the separation, pre-concentration and speciation of chromium using an isatin derivative. Anal. Chim. Acta. 632 35-41. https://doi.org/10.1016/j.aca.2008.10.073 [ Links ]
CRAMER LA, BASSON J and NELSON LR (2004) The impact of platinum production from UG2 ore on ferrochrome production in South Africa. J. S. Afr. Inst. Min. Metall. 104 517-527. [ Links ]
DHAL B, DAS NN, THATOI HN and PANDEY BD (2013) Characterizing toxic Cr(VI) contamination in chromite mine overburden dump and its bacterial remediation. J. Hazardous Mater. 260 141-149. https://doi.org/10.1016/j.jhazmat.2013.04.050 [ Links ]
DING Y, SONG Z, FENG R and GUO J (2014) Interaction of organic acids and pH on multi-heavy metal extraction from alkaline and acid mine soils. Int. J. Environ. Sci. Technol. 11 33-42. https://doi.org/10.1007/s13762-013-0433-7 [ Links ]
DIONEX CORPORATION (2003) Determination of hexavalent chromium in drinking water using ion chromatography. Application Update 144, LPN 1495. Dionex Corporation, Sunnyvale, CA. [ Links ]
DIONEX CORPORATION (2011) Sensitive determination of hexavalent chromium in drinking water. Application Update 179. Dionex Corporation, Sunnyvale, CA. [ Links ]
DU PREEZ SP, BEUKES JP and VAN ZYL PG (2015) Cr (VI) Generation during flaring of CO-rich off-gas from closed ferrochromium submerged arc furnaces. Metall. Mater. Trans. B. 46 1002-1010. https://doi.org/10.1007/s11663-014-0244-3 [ Links ]
FENDORF SE and LI G (1996) Kinetics of chromate reduction by ferrous iron. Environ. Sci. Technol. 30 1614-1617. https://doi.org/10.1021/es950618m [ Links ]
GASIK M (2013) Handbook of ferroalloys: theory and technology, Butterworth-Heinemann. [ Links ]
GERICKE WA (1998) Environmental solutions to waste products from ferrochrome production. In: Proceedings of the 8th International Ferroalloys, Beijing, China. 51-58. [ Links ]
GERICKE WA (1995) Environmental aspects of ferrochrome production. In: Proceedings of the 7th International Ferroalloys Congress, Trondheim, Norway. 131-140. [ Links ]
HE YT, CHEN CC and TRAINA SJ (2004) Inhibited Cr(VI) reduction by aqueous Fe(II) under hyper-alkaline conditions. Environ. Sci. Technol. 38 5535-5539. https://doi.org/10.1021/es049809s [ Links ]
HININGER I, BENARABA R, OSMAN M, FAURE H, ROUSSEL AM and ANDERSON RA (2007) Safety of trivalent chromium complexes: no evidence for DNA damage in human HaCaT keratinocytes. Free Radic. Biol. Med. 42 1759-1765. https://doi.org/10.1016/j.freeradbiomed.2007.02.034 [ Links ]
ICDA (INTERNATIONAL CHROMIUM DEVELOPMENT ASSOCIATION) (2012) Statistical Bulletin, 2012 edition. ICDA, Paris. 46 pp. [ Links ]
KASSEM TS (2010) Kinetics and thermodynamic treatments of the reduction of hexavalent to trivalent chromium in presence of organic sulphide compounds. Desalination. 258 206-218. https://doi.org/10.1016/j.desal.2010.03.011 [ Links ]
LEE PK, KANG MJ, JO HY and CHOI SH (2012) Sequential extraction and leaching characteristics of heavy metals in abandoned tungsten mine tailings sediments. Environ. Earth Sci. 66 1909-1923. https://doi.org/10.1007/s12665-011-1415-z [ Links ]
LIND BB, FÄLLMAN, AM and LARSSON LB (2001) Environmental impact of ferrochrome slag in road construction. Waste Manage. 21 255-264. https://doi.org/10.1016/S0956-053X(00)00098-2 [ Links ]
LOOCK-HATTINGH MM (2016) Cr(VI) contamination of aqueous systems. PhD thesis, North-West University, South Africa. [ Links ]
LOOCK-HATTINGH MM, BEUKES JP, VAN ZYL PG and TIEDT LR (2015) Cr(VI) and conductivity as indicators of surface water pollution from ferrochrome production in South Africa: Four Case studies. Metall. Mater. Trans. B. 46 2315-2325. https://doi.org/10.1007/s11663-015-0395-x [ Links ]
LOOCK MM, BEUKES JP and VAN ZYL PG (2014) A survey of Cr(VI) contamination of surface and drinking water in the proximity of ferrochromium smelters in South Africa. Water SA 40 709-716. https://doi.org/10.4314/wsa.v40i4.16 [ Links ]
MAINE C, SMIT, J and GIESEKKE E (2005) The solid stabilization of soluble wastes generated in the South African ferrochrome industry. Water Research Commission, Pretoria. [ Links ]
MARCH J (1992) Advanced Organic Chemistry: Reactions, Mechanisms, and Structure. John Wiley & Sons, New York. [ Links ]
MOLOKWANE PE, MELI KC and NKHALAMBAYAUSI-CHIRWA EM (2008) Chromium (VI) reduction in activated sludge bacteria exposed to high chromium loading: Brits culture (South Africa). Water Res. 42 4538-4548. https://doi.org/10.1016/j.watres.2008.07.040 [ Links ]
MURTHY YR, TRIPATHY SK and KUMAR CR (2011) Chrome ore beneficiation challenges & opportunities - a review. Miner. Eng. 24 (5) 375-380. https://doi.org/10.1016/j.mineng.2010.12.001 [ Links ]
NIEMELÄ P and KAUPPI M (2007) Production, characteristics and use of ferrochromium slags. In: Proceedings of the 11th International Ferro Alloys Conference, New Delhi, India. 171-179. [ Links ]
NIEMELÄ P, KROGERUS H and OIKARINEN P (2004) Formation, characterization and utilization of CO-gas formed in ferrochrome smelting. In: Proceedings of the 12th International Ferroalloys Congress, Cape Town, South Africa. 68-77. [ Links ]
NOISH 7605 (2003) Chromium, Hexavalent by Ion Chromatography Method 7605. NIOSH Manual of Analytical Methods (NMAM). NOISH, Cincinnati. [ Links ]
PAKTUNC D (2004) A computer program for analyzing complex bulk XAFS spectra and for performing significance tests. J. Synchrotron. Radiat. 11 295-298. https://doi.org/10.1107/S0909049504003681 [ Links ]
PROCTOR DM, OTANI JM, FINLEY BL, PAUSTENBACH DJ, BLAND JA, SPEIZER N and SARGENT EV (2002) Is hexavalent chromium carcinogenic via ingestion? A weight-of-evidence review. J. Toxicol. Env. Health A 65 701-746. https://doi.org/10.1080/00984100290071018 [ Links ]
RAVEL B and NEWVILLE M (2005) ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron. Radiat. 12 537-541. https://doi.org/10.1107/S0909049505012719 [ Links ]
RIEKKOLA-VANHANEN M (1999) Finnish expert report on best available techniques in ferrochrome production. Finnish Environmental Institute, Helsinki. 51 pp. [ Links ]
SEAMAN JC, BERTSCH PM and SCHWALLIE L (1999) In situ Cr (VI) reduction within coarse-textured, oxide-coated soil and aquifer systems using Fe (II) solutions. Environ. Sci. Technol. 33 938-944. https://doi.org/10.1021/es980546+ [ Links ]
SKOOG DA, WEST DM, HOLLER FJ and CROUCH SR (2013) Fundamentals of Analytical Chemistry, Nelson Education, Belmont. [ Links ]
THOMAS DH, ROHRER JS, JACKSON PE, PAK T and SCOTT JN (2002) Determination of hexavalent chromium at the level of the California Public Health Goal by ion chromatography. J. Chromatogr. A. 956 255-259. https://doi.org/10.1016/S0021-9673(01)01506-0 [ Links ]
VAN STADEN Y, BEUKES JP, VAN ZYL PG, DU TOIT JS and DAWSON NF (2014) Characterization and liberation of chromium from fine ferrochrome waste materials. Miner. Eng. 56 112-120. https://doi.org/10.1016/j.mineng.2013.11.004 [ Links ]
VENTER AD, BEUKES JP, VAN ZYL PG, JOSIPOVIC M, JAARS K and VAKKARI V (2016) Regional atmospheric Cr (VI) pollution from the Bushveld Complex, South Africa. Atmos. Pollut. Res. 7 762-767. https://doi.org/10.1016/j.apr.2016.03.009 [ Links ]
ZELIĆ J (2005) Properties of concrete pavements prepared with ferrochromium slag as concrete aggregate. Cement Concrete Res. 35 2340-2349. https://doi.org/10.1016/j.cemconres.2004.11.019 [ Links ]
Received 7 September 2016
Accepted in revised form 27 March 2017
* To whom all correspondence should be addressed. Tel.: +27 (0) 18 299 2337; fax: +27 18 (0) 299 2350; e-mail: paul.beukes@nwu.ac.za














