Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
Water SA
versión On-line ISSN 1816-7950
versión impresa ISSN 0378-4738
Water SA vol.40 no.1 Pretoria ene. 2014
TECHNICAL NOTE
Geographical differences in the relationship between total dissolved solids and electrical conductivity in South African rivers
H van Niekerk; MJ Silberbauer; M Maluleke
Resource Quality Services, Department of Water Affairs, Private Bag X313 Pretoria, South Africa
ABSTRACT
Electrical conductivity (EC) is a useful surrogate for total dissolved solids (TDS). EC is more rapidly and easily measurable with reasonably-priced equipment. However, as an indirect measure EC is subject to uncertainties that are not always apparent to the user. We set out to investigate the relationship between TDS and EC in 144 643 sample results available on the Department of Water Affairs water quality database. TDS is calculated as the sum of the major solutes determined by laboratory analysis and EC is a measurement in a flow cell. The median TDS:EC ratio for 332 high priority sites was 7 mg/ℓ: 1 mS/m. Regional differences ranged from 4.8 to 8.6. Investigation of 38 of these sites using Maucha diagrams suggested that the differences are related to the dominant major ions, with sodium chloride waters having a lower TDS:EC conversion factor than calcium bicarbonate waters. The practical application of these findings is that users of EC meters should not simply apply a blanket conversion factor, but need to select an applicable factor for the river system in which they are measuring.
Keywords: conversion factors, electrical conductivity, field instruments, rivers, total dissolved solids, water quality
INTRODUCTION
Many water sector analysts routinely use total dissolved solids or salts (TDS) as a measure of water salinity. User agencies include agriculture, industry, water supply, mining and resource management. Guidelines or resource objectives for TDS help to maintain optimum production in each sector.
The units of measurement for TDS are usually milligrams per litre (mg/ℓ), grams per litre (g/ℓ) parts per million (ppm) or parts per thousand (ppt or o/oo ). Direct methods for determining TDS concentration are gravimetric, for example evaporation at 180°C (APHA, 1999), flow densitometry or determination of the major individual solutes by laboratory analysis and their algebraic summation (APHA, 1998). These methods are expensive and time consuming, and a much cheaper, easier and quicker method to infer TDS concentration is by measuring electrical conductivity (EC) using an EC meter and converting the value to TDS with a constant conversion factor. The use of EC to determine TDS in water is based on the principle that pure water is a poor conductor of electricity and the ability to conduct electricity increases linearly with increasing ion concentration. The TDS:EC ratio for natural inland waters varies from 5.4 to 9.6 mg/ℓ per mS/m, depending on the ionic composition and strength of the solution being tested, so that water containing mainly NaCl will have a ratio closer to 5, while water with a high sulphate content may have a ratio as high as 9.6 (Hem, 1985). Groundwater TDS:EC ratios in South Africa vary from <6 in the south-west to >8 in the north-east (Simonic, 2000). The South African Water Quality Guidelines assume a general conversion factor (CF) of 6.5 for TDS:EC, although it is recommended by the Guidelines that site-specific CFs be used where more accurate TDS concentrations are required (DWAF, 1996).
Many EC meters and loggers have an option to perform the conversion internally, presenting the user with a TDS reading that is no more than an estimate derived from the EC. The TDS:EC conversion factor may be based on measurement of a standard solution of, for example, KCl, or it may be the average of a number of TDS:EC ratios in samples where both measurements are known. South African water quality practitioners use this method extensively and the reliability of the approach, which we will refer to as the TDS:EC method, is the subject of this paper.
Although the TDS:EC method is capable of providing very accurate TDS concentrations, the use of a universal conversion factor can result in over- or underestimates. Users of EC meters need to follow certain basic procedures to ensure good representative TDS values, namely, proper calibration and maintenance, correct instrument use and application of the correct TDS:EC conversion factor. Personal observation of the activities of field personnel in the Department of Water Affairs suggests that instrument maintenance is acceptable but that personnel tend to apply the built-in TDS:EC conversion factor without consideration of the implications. Users of EC meters may assume that the standard instrument setting is correct, or they may not even be aware that an indirect measurement is taking place. Standard instrument settings for well-known conductivity meters used in South Africa vary between 5 and 7, depending on the make and model. An EC reading of 70 mS/m could therefore imply a TDS of 350 to 490 mg/ℓ, depending on the built-in conversion factor setting.
METHODOLOGY
Monitoring sites
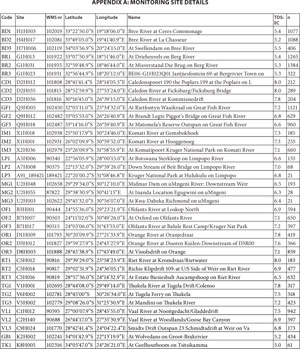
We calculated TDS:EC ratios for all 332 National Chemical Monitoring Programme (NCMP) priority river sites and examined in more detail a subset comprising 38 representative sites (Table 1, Fig. 1, Appendix A ). Selection criteria were data availability and spatial coverage of major river systems. The Tsitsikamma and Groot Brak rivers have only a single monitoring site each, because of their short length.
EC data and units
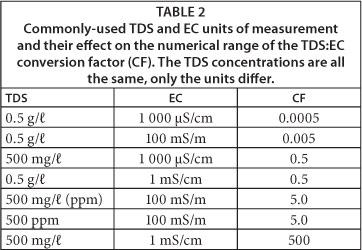
The SI unit of conductivity is siemens per metre (S/m), although this unit is not commonly used. In this paper, we use milli-siemens per metre (mS/m), although most field instruments report EC in millisiemens per centimetre (mS/cm) or micro-siemens per centimetre µS/cm). DWA uses mS/m as the standard EC unit of measurement (DWAF, 1996 and RQS, 2010). To avoid confusion when converting one unit to another the following rule can be applied: 1 mS/cm = 100 mS/m = 1 000 µS/cm. Using the wrong EC units can lead to confusion and the recording of erroneous results, with a potential error of several orders of magnitude (Table 2).
TDS data and units
All TDS data were sourced from the DWA water quality database and the measurement unit used in this paper is mg/l. TDS concentration is calculated as the sum of the concentrations for Na+, K+, Mg2+, Ca2+, Cl-, SO2-, TAL, NO3-, F-, PO43- and NH4+ in a sample filtered through a 0.45 µm filter (RQS, 2006).
Samples where the cation-anion balance deviated by more than 10% were ignored.
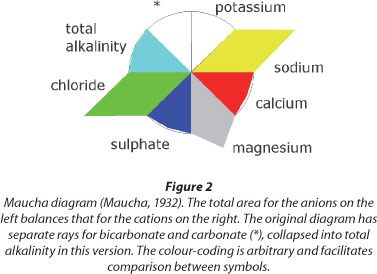
Maucha ionic diagrams are a useful method for comparing water chemistry types (Maucha, 1932; Broch and Yake, 1969; Day and King, 1995). The Maucha diagram summarises the ratios of the major ions present in a water sample (Fig. 2).
TDS:EC conversion factor calculation
The CFs were determined by calculating the TDS:EC ratios for individual samples and then calculating the median of all the individual ratios at that sight.
RESULTS AND DISCUSSION
The median TDS:EC conversion factor (CF) for 144 643 samples from the 332 high priority river monitoring sites was 7.0, ranging from 4.8 to 8.6 for individual sites. The results for the 332 monitoring sites imply that using a standard instrument setting CF of 6.5 to convert EC to TDS can underestimate TDS by as much as 25% or overestimate it by up to 35%.
The results and discussions of 38 sites in 14 river systems are tabulated and grouped into 4 geographical units, namely, the major west-flowing river systems, the major east-flowing rivers, south-eastern coastal rivers and south-western coastal rivers (Tables 3-6). The median CF for the 18 363 samples from the 38 sites was 6.9, ranging from 5.0 to 8.0.
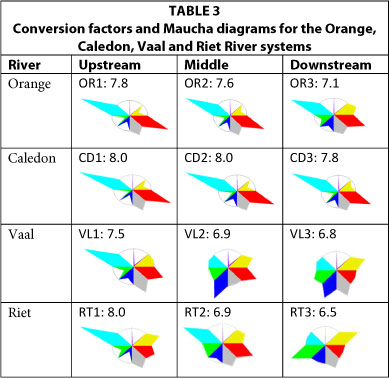
Major west-flowing rivers: Orange, Caledon, Vaal and Riet Rivers (Table 3)
The Orange-Caledon waters have a high total alkalinity, with an increase in the contribution of sodium, chloride and sulphate in the downstream reaches of the Orange River. Calculated CFs range from 7.1 to 7.8, so potential underestimates of TDS, when using a CF of 6.5, can vary from 9% in the lower Orange River to 17% in the upper Caledon River. The upper reaches of the Vaal and the Riet Rivers have higher CFs associated with the higher alkalinity. In both rivers, CFs decrease downstream, along with an increase in the proportion of sodium, chloride and sulphate. In the Riet River the underestimate in the derivation of TDS from EC varies from zero to 19%.
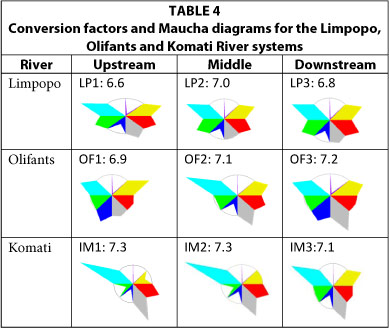
Major east-flowing rivers: Limpopo, Olifants and Komati Rivers (Table 4)
The CFs for Limpopo (6.6-7.0), Olifants (6.9-7.2) and the Komati (7.1-7.3) rivers remained fairly constant from the upper to the lower reaches. Note that the less alkaline Limpopo River water has a CF closer to 6.5, compared with the higher CFs of the Komati River where magnesium and bicarbonate are the major ions.
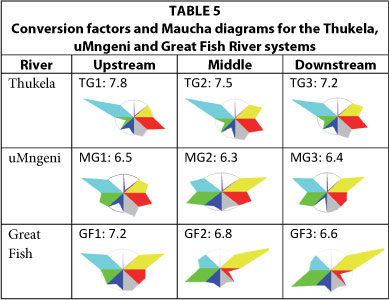
South-eastern coastal rivers: Thukela, uMngeni, and Great Fish Rivers (Table 5)
In the Thukela River, alkalinity is the major component of the anions and the CF ranges between 7.2 and 7.8. The uMngeni River contains a higher proportion of chloride and sulphate, with CFs ranging between 6.3 and 6.5. The Great Fish River's ionic composition differs markedly between the upstream section of the river and the two downstream reaches. Both downstream sites are sodium chloride dominated with CFs of 6.8 and 6.6. In contrast, the Maucha symbol for the upstream site shows alkalinity and sodium dominance, and a higher CF of 7.2. Converting EC to TDS using the recommended CF of 6.5 might yield acceptable results at the lower two sites, but will result in a consistent 10% underestimate at the upper site. The higher CFs in the upper reaches of the Great Fish River are very likely related to the transfer of alkaline waters from the Upper Orange River, cf. OR1 in Table 3, to the headwaters of the Great Fish River for irrigation purposes (Van Niekerk et al., 2009).
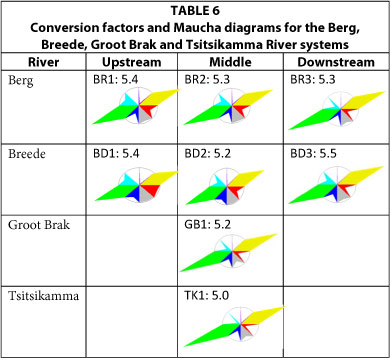
South-western coastal rivers: Berg, Breede, Groot Brak and Tsitsikamma Rivers (Table 6)
In contrast with the rivers discussed previously, the Berg, Breede, Groot Brak and Tsitsikamma river waters are all sodium chloride dominated with very low alkalinity. The CFs for these rivers vary between 5 and 5.5. Using a CF of 6.5 to determine the TDS from EC in the Tsitsikamma River will overestimate the actual TDS by 30%.
CONCLUSION
The DWA-recommended CF of 6.5 to derive TDS from EC is clearly a rough guideline to be used only when the actual applicable CF is not available or cannot be calculated. Blanket application of the 6.5 CF can lead to misleading results and possible errors during water quality assessments. Discussions with water quality practitioners have revealed that many were unaware of the magnitude of the variation and had assumed that a CF of 6.5 is applicable in all instances. It is therefore important that practitioners record the original EC value so that they can do a more accurate conversion when an applicable CF becomes available.
EC is often the variable of choice when comparing rivers or river reaches in terms of salinity. When using EC, it is important to consider the effect different TDS:EC ratios might have on the final results. For example, Site BR2 in the Berg River and Site RT1 in the Riet River both have median EC values of approximately 36 mS/m (WMS database); however, their median TDS concentrations are 194.5 mg/£ and 288.4 mg/ℓ, respectively. Using two popular EC meter brands with standard CFs of 5 (Brand A) and 7 (Brand B) the TDS readings from Brand A will be approximately 180 mg/ℓ for both rivers and the readings from Brand B will be approximately 252 mg/ℓ for both rivers. This means that Brand A will give you a 36% under-estimation of TDS at the Riet River site and Brand B will give you a 29% over-estimation at the Berg River site.
Users of EC meters or EC data should thus be aware of the potential errors and should use a site-specific conversion factor, where one is available. Both EC and TDS data for most of the South African rivers are available at www.dwa.gov.za/iwqs and can be used to calculate appropriate conversion factors for specific study areas.
REFERENCES
APHA (1998) 2520 Salinity. In: Standard Methods for the Examination of Water and Wastewater. American Public Health Association, Washington DC. [ Links ]
APHA (1999) 2540C Total Dissolved Solids dried at 180°C. In: Standard Methods for the Examination of Water and Wastewater. American Public Health Association, Washington DC. [ Links ]
BROCH ES and YAKE W (1969) A modification of Maucha's ionic diagram to include ionic concentrations. Limnol. Oceanogr. 14 933-935. [ Links ]
DAY JA and KING JM (1995) Geographical patterns, and their origins, in the dominance of major ions in South African rivers. S. Afr. J. Sci. 91 299-306. [ Links ]
DWAF (DEPARTMENT OF WATER AFFAIRS AND FORESTRY, SOUTH AFRICA) (1996) South African Water Quality Guidelines. Volume 1: Domestic Water Use (2nd edn.). Department of Water Affairs and Forestry, Pretoria. [ Links ]
DWAF (DEPARTMENT OF WATER AFFAIRS AND FORESTRY, SOUTH AFRICA) (2004) Strategic Framework for National Water Resource Quality Monitoring Programmes. Report No. N/0000/ REQ0204. Resource Quality Services, Department of Water Affairs and Forestry, Pretoria. [ Links ]
HEM JD (1985) Study and interpretation of the chemical characteristics of natural water. Water-Supply Paper 2254 (3rd edn.). US Geological Survey. United States Government Printing Office. [ Links ]
MAUCHA R (1932) Hydrochemische Metoden in der Limnologie. Binnengewasser 12173. [ Links ]
RQS (RESOURCE QUALITY SERVICES) (2006) TDS, Inorganic Chemistry Laboratory Methods Manual (Revision 6). Resource Quality Services, Department of Water Affairs and Forestry, Pretoria. [ Links ]
RQS (RESOURCE QUALITY SERVICES) (2010) EC, Inorganic Chemistry Laboratory Methods Manual (Revision 7). Resource Quality Services, Department of Water Affairs, Pretoria, South Africa. [ Links ]
SIMONIC M (2000) The assessment of ambient groundwater quality at a national scale in the Republic of South Africa - an electronic atlas. WRC Report No K5/841. Water Research Commission, Pretoria. [ Links ]
VAN NIEKERK H, SILBERBAUER MJ, and HOHLS BC (2009) Monitoring programme revision highlights long-term salinity changes in selected South African rivers and the value of comprehensive long-term data sets. Environ. Monit. Assess. 154(1-4) 401-411. [ Links ]
 Correspondence:
Correspondence:
H van Niekerk
+27 12 808 9502; fax: +27 12 808 0338
E-mail: VanNiekerkH2@dwa.gov.za
Received 29 June 2012
Accepted in revised form 9 January 2014