Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Water SA
On-line version ISSN 1816-7950
Print version ISSN 0378-4738
Water SA vol.40 n.1 Pretoria Jan. 2014
Characteristics of a bioflocculant produced by a consortium of Cobetia and Bacillus species and its application in the treatment of wastewaters
AM Ugbenyen; AI Okoh
Applied and Environmental Microbiology Research Group (AEMREG), Department of Biochemistry and Microbiology, University of Fort Hare, Private Bag X1314, Alice 5700, South Africa
ABSTRACT
The characteristics of a bioflocculant produced by a consortium of 2 bacteria belonging to the genera Cobetia and Bacillus was investigated. The extracellular bioflocculant was composed of 66% uronic acid and 31% protein and showed an optimum flocculation (90% flocculating activity) of kaolin suspension at a dosage of 0.8 mg/mℓ, pH of 8, and with Ca2+ as a coagulant aid. The bioflocculant is thermally stable, with a high residual flocculoccating activity of 86.7%, 89.3% and 87% after heating at 50oC, 80oC and 100oC, respectively, for 25 min. FTIR analysis of the bioflulant indicated the presence of hydroxyl, amino, carbonyl and carboxyl functional groups. Scanning electron microscopy (SEM) revealed a crystal-linear sponge-like bioflocculant structure and EDX analysis of purified bioflocculant indicated an elemental composition in mass proportions of C:N:O:S:P of 6.67:6.23:37.55:0.38:4.42 (% w/w). The produced bioflocculant was highly efficient in removing turbidity and reducing chemical oxygen demand (COD) in brewery wastewater, dairy wastewater and river water. The bioflocculant could flocculate kaolin clay more efficiently than traditional flocculants; alum and polyethylenimine.
Keywords: Extracellular, bioflocculant, consortium, characteristic, Cobetia sp., Bacillus sp.
INTRODUCTION
Flocculants are chemicals that stimulate flocculation by aggregation of colloids and other suspended particles, forming a floc (IUPAC, 1997). Flocculants are widely applied in various industrial processes, including wastewater treatment, downstream processing, and food and fermentation processes (Mabinya et al., 2011; Nakata and Kurane, 1999; Salehizadeh and Shojaosadati, 2001). Although chemical flocculants have been widely used due to their effective flocculating activity and low cost, some synthetic flocculants are known to be hazardous to the environment (Master et al., 1985; Kowall et al., 1989; Dearfield and Abermathy, 1988). Some examples of chemical flocculants include: aluminium chlorohydrate, aluminium sulphate, calcium hydroxide, iron sulphide, iron(III)chloride, polyacrylamide, chitosan, guar gum, etc.
In recent years, utilisation of microbial flocculants has been promoted due to their biodegradability and their environmentally inert nature (Li et al., 2009; Liu et al., 2010). Currently, bioflocculants are attracting considerable attention as a promising substitute for chemical flocculants. In nature, microorganisms do not live in isolation; they coexist with many other microorganisms forming relationships that have an effect on the biological adequacy of all interacting species. Nevertheless, over the past decades, emphasis with regard to the bioflocculant field has mainly been placed on pure cultures. However, Zhu et al. (2004) and Zhang et al. (2007) have reported that the combination of strains of microorganisms in consortia produced bioflocculants that possessed better flocculating activity and higher bioflocculant yield than pure strains.
In this study we assessed the characteristics of a bioflocculant produced by a consortium of 2 bacteria, viz. Cobetia sp. OAUIFE and Bacillus sp. MAYA, which were previously isolated from the sediment of Algoa Bay in the Eastern Cape Province of South Africa, as a result of efforts to discover and explore new bioflocculants offering potential as alternatives to chemical or synthetic flocculants.
EXPERIMENTAL
Bacteria and culture conditions
The bacteria were isolated from sediment samples of Algoa Bay in the Eastern Cape Province of South Africa and maintained in 20% glycerol at -80°C in the culture collection of the Applied and Environmental Microbiology Research Group (AEMREG), University of Fort Hare, Alice, South Africa after affirmation of their bioflocculant production potential.
The culture medium consisted of 20 g glucose, 0.5 g urea, 0.5 g yeast extract, 0.2 g (NH4)2SO4, 2 g KH2PO4, 5 g K2HPO4, 0.1 g NaCl and 0.2 g MgSO4 7H2O in a litre of natural seawater filtered using Whatman filter paper (Zhang et al., 2007). A loopful of each bacterial colony was inoculated separately into 50 mℓ of the medium and incubated for 72 h at 28oC with shaking at 160 r/min, and was used as a pre-culture for subsequent inoculations. For the bulk fermentation 10 ml of each pre-culture was inoculated into 1 ℓ of the culture medium, indicating a 2% (v/v) inoculum size, incubated with shaking at 160 r/min for 72 h at 28oC.
Purification of bioflocculant
Isolation and purification of the bioflocculant was done according to the method described by previous reports (Chang et al., 1998; Chen et al., 2002; Ugbenyen et al., 2012). After 72 h of fermentation, the culture broth was centrifuged (8 000 g, 30 min) to remove bacterial cells. One volume of distilled water was added to the supernatant phase and centrifuged (8 000 g, 15 min) to remove insoluble substances. To the supernatant, 2 volumes of ethanol were added, stirred, and left to stand for 12 h at 4°C. The precipitate was vacuum-dried to obtain crude bioflocculant. The crude product was dissolved in distilled water to yield a solution, to which one volume of a mixed solution of chloroform and n-butylalcohol (5:2, v/v) was added, stirred and allowed to stand for 12 h at room temperature. Two volumes of ethanol were again added to recover the precipitate, which was then lyophilised.
Flocculation test of bioflocculant
Flocculating activity was measured as described elsewhere (Kurane et al., 1986), with modifications. Briefly, 3 ml of 1% CaCl2 and 2 ml of bioflocculant solution were added to 100 ml kaolin suspended solution (4 g/ℓ) in a 250 ml flask. The mixture was vigorously stirred and poured into a 100 mℓ measuring cylinder and allowed to stand for 5 min. The optical density (OD) of the clarifying solution was measured with a spectrophotometer at 550 nm. A control experiment was prepared using the same method, except that the bioflocculant solution was replaced with distilled water (B). The flocculating activity was measured using the equation:
Flocculating activity (%) = [(B - A)/B] × 100
where:
A is the absorbance of the sample experiment at 550 nm
B is the absorbance of the control at 550 nm
For application with real wastewater, kaolin suspension was replaced with the various wastewater types, i.e., brewery waste-water, dairy wastewater and river water.
Effect of dosage
Different concentrations of the purified bioflocculant were dissolved in distilled water to make bioflocculant solutions of 0.2, 0.4, 0.6, 0.8 and 1.0 mg/ml. The flocculation assay of the different bioflocculant solutions was done as described above.
Effect of pH and metal ions on bioflocculant activity
The effects of pH and metal ion on flocculating activity of the bioflocculant were assessed in accordance with the description of Liu et al. (2010). The pH of the bioflocculant solutions were varied across the range of 3-12 using either 0.1 M HCl or NaOH, while the metal ion candidates included Na+, K+, Li+, Mg2+, Mn2+, Al3+ and Fe3+ as their chloride salts. With regard to the effects of metal ion assays, flocculating activity assays were conducted as described above, but CaCl2 solution was replaced by a solution of the above metal ion candidates.
Thermal stabilty of the purified bioflocculant
The thermal stability of the purified bioflocculant was also assessed by incubating solutions of the bioflocculant at 50°C, 80°C and 100°C for 25 min (Gong et al., 2008) after which residual flocculating activity was determined.
Analysis of purified bioflocculant
Total protein content of bioflocculants was determined by Lowry's method using bovine serum albumin (BSA) as a standard (Lowry et al., 1951). Total sugar content of bioflocculant was determined by the phenol-sulphuric acid method using glucose as standard solution (Dubios et al., 1956) and uronic acid was quantified by the carbazole method (Jayaraman, 1981).
FTIR analysis of the purified bioflocculant was done using a Fourier-transform infrared spectrophotometer (Perkin Elmer System 2000, England) over a wave number range of 4 000 to 370 cm-1.
A scanning electron microscopy (SEM) image of the purified bioflocculant was taken using JEOL (JSM-6390LV, Japan) and energy dispersive X- ray analysis (EDX) was conducted using a Thermo Super Dry II X-ray Detector using a Noran System Six Software package.
Analysis of wastewaters
The chemical oxygen demand (COD) and turbidity of the wastewaters were measured using a spectrophotometer (Spectroquant Pharo 100 M, EU) and turbidimeter (HACH, USA), respectively, according to the manufacturer's instructions. For flocculation efficiency assays the wastewaters were used instead of a kaolin suspension.
The residual COD and turbidity were determined according to the method of Gong et al. (2008) and the removal efficiency was calculated as follows:
Removal efficiency (%) = [C0 - C/C0] × 100
where:
C0 is the initial value and C is the value after the flocculation treatment
Statistical analysis
Data were analysed by one-way analysis of variance (ANOVA) using MINITAB Student Release 12 statistical package. A significance level of p< 0.05 was used. The mean values are from 3 replicates.
RESULTS
Yield and composition of the bioflocculant
Purification of the fermentation product resulted in a bioflocculant yield of 0.256 g/ℓ. The biochemical analysis of the purified bioflocculant showed that it was composed of uronic acid (66.2%), protein (31.5%) and neutral sugar (0.5%) (Table 1).
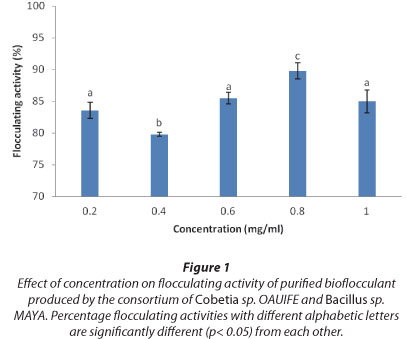
Effect of bioflocculant dose on the flocculation of kaolin
The effect of bioflocculant dose on flocculating activity of the bioflocculant from the consortium is as shown in Fig. 1. The concentration range was within 0.2-1.0 mg/mℓ. The flocculating activities of the bioflocculant from the consortium were above 80% in the range of 0.2-1.0 mg/mℓ and the maximum was observed at 0.8 mg/mℓ, with a flocculating activity of 90.0%.
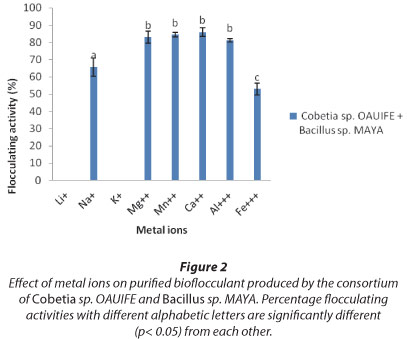
Effects of metal ion
The effect of various metal ions on flocculating activity of the bioflocculant produced by the consortium of Cobetia sp. OAUIFE and Bacillus sp. MAYA is shown in Fig. 2. Virtually all of the metal ions stimulate flocculating activity of the bioflocculant from the consortium to a level above 50%, except Li+ and K+ which completely inhibit flocculating activity of the bioflocculant. Divalent cations were observed to show better stimulation of the flocculating activity of the bioflocculant produced by the consortium of Cobetia sp. OAUIFE and Bacillus sp. MAYA. The bioflocculant showed optimum flocculating activity with Ca2+ when compared with trivalent cations like Al3+ and Fe3+.
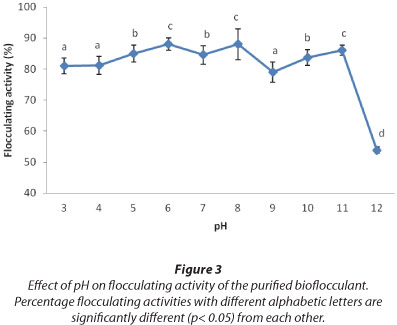
Effects of pH and heat
The effect of pH on flocculating activity of the purified bioflocculant from the consortium was assessed at concentration of 0.8 mg/mℓ with the pH of the solution ranging from 3-12 (Fig. 3). The bioflocculant flocculated a kaolin suspension with over 70% flocculating activity over a wide range of pH, 3-11, with maximum flocculating activity of 88% attained at a weak alkaline pH of 8.
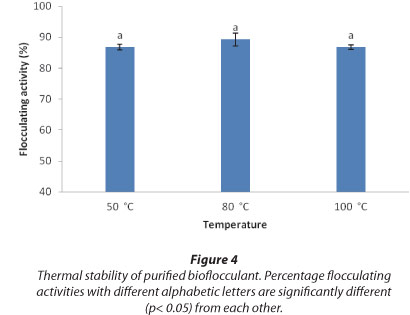
With regards to the effect of heat on the purified bioflocculant, heating the bioflocculant solution at 50oC, 80oC and 100oC over a period of 25 min revealed a fairly constant residual flocculating activity of about 87% (Fig. 4).
FTIR
The result of the FTIR spectral analysis is shown in Fig. 5. The spectrum shows important peaks indicating the presence of certain functional groups. Peaks at 3 412 cm-1 suggest the presence of hydroxyl and amino groups in the purified bioflocculant. A weak C-H asymmetric stretching vibration band was observed at 2 925 cm-1. The peaks between 1 456 cm-1 and 1 729 cm-1 are indicative of the presence of a carbonyl group; the former is characteristic of C=O in an amide group (Shriner et al., 1998, Wang et al., 2011) while the latter is assigned to the asymmetric C=O stretching in carboxylate. The peak at 1 053 cm-1 is indicative of C-O stretching vibration. The absorption peak at 913 cm-1 is characteristic of all sugar derivatives.
SEM observation
With respect to SEM analysis, Fig. 6a, b and c shows the scanning electron micrograph of the purified bioflocculant, kaolin powder, and flocculation of kaolin suspension by the purified bioflocculant, respectively. The morphology of the purified bioflocculant shown in Fig. 6a is a crystal linear sponge-like structure, while Fig. 6c shows the formation of a large floc as a result of the interaction between bioflocculant and suspended kaolin particles.
The EDX analysis of the purified bioflocculant revealed its elemental composition (% w/w), as C:N:O:S:P in the ratio 6.67:6.23:37.55:0.38:4.42 (Table 2).
Wastewater flocculation assay
The flocculation of some wastewaters by the purified bioflocculant revealed that the bioflocculant flocculated brewery wastewater, dairy wastewater and river water, at flocculating efficiencies of 90.2%, 78.8% and 98.7%, respectively (Table 3).
A comparison of the flocculating activities of the purified bioflocculant with commercially available inorganic and synthetic flocculants is shown in Table 4. The bioflocculant revealed a higher flocculation of kaolin clay, of 90%, compared with alum and polyethylenimine, which recorded a flocculating activity of 66.82% and 42.85%, respectively, for a kaolin suspension. Polyacrylamide displayed the highest flocculating activity, of 93.19%.
DISCUSSION
Many studies have reported optimum dosage of different bioflocculants from sole microorganisms (Yokoi et al., 1997; Yim et al., 2007; Lu et al., 2005; Suh et al., 1997; Salehizadeh et al., 2000; Nakata and Kurane, 1999; Fujita et al., 2000) but reports on consortia of microorganisms are still very scarce in the literature. From this study we observed a good flocculating activity in the bioflocculant produced by the consortium of Cobetia sp. OAUIFE and Bacillus sp. MAYA, at different doses ranging from 0.2 mg/mℓ to 1.0 mg/mℓ. The optimum bioflocculant dose for the purified bioflocculant was 0.8 mg/mℓ with a resultant flocculating activity of 90%. Wang et al. (2011) similarly reported that the bioflocculant CBF-F26 produced from a mixed culture of Rhizobium radiobacter F2 and Bacillus sphaeicus F6 at a dose concentration of 12 mg/ℓ showed a maximum flocculating activity of 96%.
It has been well documented that to achieving high flocculating activity, metal ions are usually required to aid bioflocculants (Salehizadel and Shojaosadati, 2002; Gong et al., 2008; Elkady et al., 2011). This is because metal ions help to neutralise negative charges on most bioflocculants and the suspended kaolin particles with which they react, thereby increasing the adsorption of the bioflocculant onto the kaolin particle (Wu and Ye, 2007).
From Fig. 2, nearly all of the metal ions stimulate flocculation of the bioflocculant produced by the consortium of Cobetia sp. and Bacillius sp., with flocculating activities of above 50%, with the exception of Li+ and K+, which completely inhibited flocculation by the bioflocculant. Divalent cations (Mg2+, Mn2+, Ca2+) exhibited better stimulation of flocculation, with Ca2+ showing the highest stimulation. This is similar to the observations of Wang et al. (2011) that monovalent cations (K+ and Na+) had little effect on the flocculating activity of CBF-F26 but divalent cations like Ca2+, Zn2+, Fe2+ enhanced the flocculation by the bioflocculant appreciably. The high flocculating activities with divalent ions may be due to the presence of a large number of carboxylate groups on the bioflocculant, that can serve as binding sites for divalent cations (Prasertsan et al., 2006). However, a monovalent cation like Na+ could not stimulate the flocculating activity of the bioflocculant from the consortium effectively, likely due to the weak electro-static force between the monovalent cation and the bioflocculant (Li et al., 2008). Salehizadel and Shojaosadati (2002) and Elkady et al. (2011) reported similar findings where monovalent cations showed weak stimulation of flocculation by their respective bioflocculants.
One of the key factors influencing flocculating activity of bioflocculants is the pH of the reaction mixture (Yokoi et al., 1996). One of the ways in which pH influences flocculating activity is by affecting the stability of suspended particles and the formation of floccules. From Fig. 3, the bioflocculant from the consortium flocculated a kaolin suspension over a wide range of pH, from 3-11. Highest flocculating activity was observed at pH 8. This is similar to results reported by Liu et al. (2010), in which the bioflocculant MBF-W6 showed flocculating activity over a wide range of pH, with the highest flocculating activity of 96.8% at pH 5.6.
From Fig. 4, heating the purified bioflocculant from the consortium at 100oC for 25 min did not significantly affect its residual flocculating activity (87%), when compared with the other two temperature regimes of 50oC and 80oC, thus confirming that the bioflocculant is stable to heat. The bioflocculant compared favourably with others reported in literature to be thermally stable. Gong et al. (2008) reported that bioflocculant SF-1 when heated at 100oC for 15 min retained 85% residual flocculating activity, while bioflocculant CBF-F26 retained more than 90% residual flocculating activity after heating to 100oC (Wang et al., 2011).
The bioflocculant was more stable than bioflocculant As-101, which lost 50% residual flocculating activity after heating for 15 min at 100oC (Salehizadel et al., 2000). The stability of the bioflocculant to heat could be as a result of the presence of uronic acid in the compound bioflocculant, contributing hydrogen bonding to the bioflocculant structure.
Presence of hydroxyl and carboxyl groups within the bioflocculant molecule, as indicated by the FTIR spectrum (Fig. 5), enhances the formation of hydrogen bonds which might be responsible for thermal stability of the bioflocculant. These groups, together with the amino group, could serve as a binding site for metal ions which enhance flocculating activity of the bioflocculant by bridging between it and the suspended kaolin particles in solution, and are likely to be the preferred groups for the process of adsorption (Comte et al., 2006).
Comparing the surface morphology of the purified bioflocculant from the consortium of Cobetia sp. OAUIFE and Bacillus sp. MAYA, as revealed by the SEM image in Fig. 6a, showed a similarity with the bioflocculant TJ-1 produced by Proteus mirabilis (Xia et al., 2008), which also had a crystalline linear morphology. In Fig. 6c, there is the formation of large floc as a result of the interaction of the bioflocculant with kaolin clay, which makes for easy settling of the floc due to gravity.
As seen in Table 3, the flocculating properties of the bioflocculant in wastewater were investigated. The different source waters used - river water, brewery wastewater and dairy wastewater - were selected to incorporate a range of soluble content (COD) and particulate substances (turbidity). River water represented a typical surface water with low COD and turbidity. The bioflocculant showed almost 99% flocculating efficiency for the river water. A similar study by Gong et al. (2008) showed that bioflocculant SF-1 from Serratia ficaria flocculated river water by up to 90.4%.
The brewery and dairy wastewaters were flocculated by the bioflocculant with flocculating efficiencies of 90.2% and 78.8%, respectively, which suggests that the bioflocculant has potential for use in wastewater treatment and other downstream processes. Many microorganisms' flocculants have been reported to have potential in treatment of wastewater. For example, Shimiziu (1985) reported that A. faecalis showed good efficiency in removal of organic material, while Kurane and Nohata (1991) reported the treatment of a blue wastewater from a paper manufacturing company with a mixture of A. latus and chitosan which showed 94% decolourisation.
When the bioflocculant from the consortium is compared with the synthetic flocculant (Table 4) with respect to flocculation of kaolin clay, the bioflocculant had a flocculation efficiency of 90% compared to 66.8% and 42.9% for alum and polyethylenimine, respectively, while the flocculating efficiency of polyacrylamide (93.2%) was not significantly different from that of the bioflocculant. Nakamura et al. (1976) also reported that the bioflocculant from A. sojae AJ-7002 compared favourably with synthetic flocculant like ferric chloride, sodium alginate and polyacrylamide.
CONCLUSIONS
The bioflocculant from the consortium of Cobetia sp. OAUIFE and Bacillus sp. MAYA was a glycoprotein. Functional groups that favour flocculation processes, such as hydroxyl, carboxyl and amino groups, were revealed to be present in the bioflocculant, which was also shown to be thermostable. The bioflocculant significantly flocculated some wastewaters better than some conventional inorganic and synthetic flocculants, suggesting that it is an attractive candidate in water treatment and other biotechnology applications.
ACKNOWLEDGEMENTS
We are grateful to the University of Fort Hare and the National Research Foundation (NRF) of South Africa for financial support.
REFERENCES
COMTE S, GUIBAUD G and BAUDU M (2006) Biosorption properties of extracellular polymeric substances (EPS) resulting from activated sludge according to their type: soluble or bound. Process Biochem. 41 815-823. [ Links ]
CHANG WC, SOON AY, IN HO and SANG HP (1998) Characterization of an extracellular flocculating substance produced by a planktonic cyanobacterium, Anabaena sp. Biotechnol. Lett. 20 643-646. [ Links ]
CHEN H, ZHANG JF, JIANG PJ, YANG SL and LIU ZL (2002) Composition and characterization of microbiological flocculant SC06. Environ. Chem. 21 360-364. [ Links ]
DEARFIELD KL and ABERMATHY CO (1988) Acrylamide: its metabolism developmental and reproductive effects, genotoxicity and carcinogenicity. Mutat. Res. 195 45-77. [ Links ]
DUBOIS M, GILLES KA, HAMILTON JK, REBERS PA and SMITH F (1956) Colorimetric method for determination of sugars and related substances. Anal. Chem. 28 350-356. [ Links ]
ELKADY MF, SOHA F, SAHAR Z, GADALLAH A and DESOUKY A (2011) Bacillus mojavensis strain 32A, a bioflocculant-producing bacterium isolated from an Egyptian salt production pond. Bioresour. Technol. 1028143-8151. [ Links ]
FUJITA M, IKE M, TACHIBANA S, KITADA G, KIM SM and INOUE Z (2000) Characterization of a bioflocculant produced by Citrobacter sp. TKF04 from acetic and propionic acids. J. Biosci. Bioeng. 89 40-46. [ Links ]
GONG W, WANG S, SUN X, LIU X, YUE Q and GAO B (2008) Bioflocculant production by culture of Serratia ficaria and its application in wastewater treatment. Bioresour. Technol. 99 4668-4674. [ Links ]
IUPAC (1997) Compendium of Chemical Terminology (2nd edn.) Blackwell Scientific Publications, Oxford. [ Links ]
JAYARAMAN J (1981) Laboratory Manual in Biochemistry. Wiley Eastern, Delhi, India. 75-76. [ Links ]
KOWALL NW, PENDLEURY WW and KESSLER JB (1989) Aluminium-induced neurofibrillary degeneration affects a subset of neurons in rabbit cerebral cortex, basal forebrain and upper brain stem. Neuroscience 29 329-337. [ Links ]
KURANE R, TAKEDA K, and SUZUKI T (1986) Screening for characteristics of microbial flocculants. Agric. Biol. Chem. 50 2301-2307. [ Links ]
KURANE R, NOHATA Y (1991) Microbial flocculation of waste liquids and oil emulsion by a bioflocculant from Alcaligenes latus. Agric. Biol. Chem. 551127-1129. [ Links ]
LI WW, ZHOU WZ, ZHANG YZ, WANG J and ZHU XB (2008) Flocculation behavior and mechanism of an exopolysaccharide from the deep-sea psychrophilic bacterium Pseudoalteromonas sp. SM9913. Bioresour. Technol. 996893 -6899. [ Links ]
LI Z, ZHONG S, LEI H, CHEN R, YU Q and LI HL (2009) Production of a novel bioflocculant by Bacillus licheniformis X14 and its application to low temperature drinking water treatment. Bioresour. Technol. 100 3650-3656. [ Links ]
LIU W, WANG K, LI B, YUAN H and YANG J (2010) Production and characterization of an intracellular biofloculant by Chryseobacterium daeguense W6 cultured in lownutrition medium. Bioresour. Technol. 101 1044-1048. [ Links ]
LOWRY OH, ROSEBROUGH NJ, FARR AL and RANDALL RJ (1951) Protein measurement with Folin Phenol reagent. J. Biol. Chem. 196 256-275. [ Links ]
LU WY, ZHANG T, ZHANG DY, LI CH, WEN JP and DU LX (2005) A novel bioflocculant produced by Enterobacter aerogenes and its use in defecating the trona suspension. Biochem. Eng. J. 271-7. [ Links ]
MABINYA LV, COSA S, MKWETSHANA N and OKOH AI (2011) Halomonas sp. OKOH-A marine bacterium isolated from the bottom sediment of Algoa Bay-produces a polysaccharide bioflocculant: partial characterization and biochemical analysis of its properties. Molecules 164358-4370. [ Links ]
MASTER C, MULTHAUP G and SIMMS G (1985) Neuronal origin of a cerebral amyloid: neurofibrallary tangles of Alzheimer's disease contain the same protein as the amyloid of plaque cores and blood vessels. EMBO J. 42757-2763. [ Links ]
NAKAMURA J, MIYASHIRO S and HIROSE Y (1976) Screening, isolation and some properties of microbial cell flocculants. Agric. Biol. Chem. 40 377-383. [ Links ]
NAKATA K and KURANE R (1999) Production of an extracellular polysaccharide bioflocculant by Klebsiella pneumonia. Biosci. Biotechnol. Biochem. 63 2064-2068. [ Links ]
PRASERTSAN P, DERMLIM W, DOELLE H and KENNEDY JF (2006) Screening, characterization and flocculating property of carbohydrate polymer from newly isolated Enterobacter cloacae WD7. Carbohydr. Polym. 66 289-297. [ Links ]
SALEHIZADEH H and SHOJAOSADATI SA (2001) Extracellular biopolymeric flocculants: Recent trends and biotechnology importance. Biotechnol. Adv. 19371-385. [ Links ]
SALEHIZADEH H and SHOJAOSADATI SA (2002) Isolation and characterisation of a bioflocculant produced by Bacillus firmus. Biotechnol. Lett. 24 35-40. [ Links ]
SALEHIZADEH H, VOSSOUGHI M and ALEMZADEH I (2000) Some investigations on bioflocculant producing bacteria. Biochem. Eng. J. 539-44. [ Links ]
SHIMIZIU N (1985) Floc-forming bacteria isolated from activated sludge in high BOD loading treatment. J Ferment. Technol. 6367-71. [ Links ]
SHRINER RL, HERMANN CKF, MORRILL TC, CURTIN DY and FUSON RC (1998) The systematic identification of organic compounds. Wiley, New York. [ Links ]
SUH HH, KWON GS, LEE CH, KIM HS, OH HM and YOON BD (1997) Characterization of bioflocculant produced by Bacillus sp. DP-152. J. Ferment. Bioeng. 84 (2) 108-112. [ Links ]
UGBENYEN A, COSA S, MABINYA L, BABALOLA OO, AGHDASI F and OKOH A (2012) Thermostable bacterial bioflocculant produced by Cobetia spp. isolated from Algoa Bay (South Africa). Int. J. Environ. Res. Public Health 9 (6) 2108-2120. [ Links ]
WANG L, MA F, QU Y, SUN D, LI A, GUO J and YU B (2011) Characterization of a compound bioflocculant produced by mixed culture of Rhizobium radiobacter F2 and Bacillus sphaeicus F6. World J. Microbiol. Biotechnol. 27 2559-2565. [ Links ]
WU JY and YE HF (2007) Characterization and flocculating properties of an extracellularbiopolymer produced from a Bicillus subtilis DYU1 isolate. Process Biochem. 421114-1123. [ Links ]
XIA S, ZHANG Z, WANG X, YANG A, CHEN L, ZHAO J, LEONARD D and JAFFREZIC-RENAULT N (2008) Production and characterization of bioflocculant by Proteus mirabilis TJ-1. Bioresour. Technol. 99 6520-6527. [ Links ]
YIM JH, KIM SJ, AHN SH and LEE HK (2007) Characterization of a novel bioflocculant, p-KG03, from a marine dinoflagellate, Gyrodinium impudicum KG03. Bioresour. Technol. 98361-367. [ Links ]
YOKOI H, ARIMA T, HIROSE J, HAYASHI S and TAKASAKI Y (1996) Flocculation properties of poly(y-glutamic acid) produced by Bacillus subtilis. J. Ferment. Bioeng. 82 84-87. [ Links ]
YOKOI H, YOSHIDA T, MORI S, HIROSE J, HAYASHI S and TAKASAKI Y (1997) Biopolymer flocculant produced by an Enterobacter sp. Biotechnol. Lett. 19 569-573. [ Links ]
ZHANG Z, LIN B, XIA S, WANG X and YANG A (2007) Production and application of a novel bioflocculant by multiple-microorganism consortia using brewery wastewater as carbon source. J. Environ. Sci. 19667-673. [ Links ]
ZHU YB, FENG M and YANG JX (2004) Screening of complex bioflocculant producing bacterium and their flocculating mechanism. J. Harbin Inst. Technol. 36(6) 759-762. [ Links ]
 Correspondence:
Correspondence:
AI Okoh
+27 40 602 2365; fax: +27 866 286824
E-mail: aokoh@ufh.ac.za
Received 29 November 2012
Accepted in revised form 17 December 2013














