Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
Water SA
versión On-line ISSN 1816-7950
versión impresa ISSN 0378-4738
Water SA vol.39 no.3 Pretoria 2013
Characterisation of wastewater for modelling of wastewater treatment plants receiving industrial effluent
FT Mhlanga*; CJ Brouckaert
Pollution Research Group, School of Chemical Engineering, University of KwaZulu-Natal, Durban, 4041, South Africa
ABSTRACT
Bio-process modelling is increasingly used in design, modification and troubleshooting of wastewater treatment plants (WWTPs). Characterisation of the influent wastewater to a WWTP is an important part of developing such a model. The characterisation required for modelling is more detailed than that routinely employed for monitoring WWTP operation. Wastewater characteristics depend on the sources within the catchment served by a particular WWTP, and the presence of industrial effluents can cause the wastewater to be significantly different from purely domestic effluent. eThekwini's wastewater treatment system is one of those most affected by industrial effluents in South Africa. Where industrial pollutants cause particular problems, additional measurements, beyond those standardly applied in sewage treatment, are required. Since influent characteristics vary from one catchment to another, this paper presents and compares results of influent wastewater characterisation from three municipal WWTPs, two of which are operated by eThekwini Water Services, which receive a combination of industrial and domestic wastewater. The paper also presents efforts to fractionate the influent COD of another WWTP run by eThekwini municipality and receiving a complex mixture of industrial and domestic effluent. The influent characterisation involves the determination of the volumes and concentrations of the carbonaceous, nitrogenous and phosphorus fractions in the wastewater, as well as other constituents present in the wastewater. This paper focuses on the carbonaceous fraction in the wastewater.
Keywords: influent characterisation, industrial effluent, process modelling
LIST OF ACRONYMS
ASM1 Activated sludge model number 1
ASM2 Activated sludge model number 2
ASM2d Activated sludge model number 2d
ASM3 Activated sludge model number 3
BOD Biological oxygen demand
COD Chemical oxygen demand
DO Dissolved oxygen
F/M Food to mass ratio
IAWPRC International Association on Water Pollution Research and Control
IAWQ International Association on Water Quality
IWA International Water Association
MLVSS Mixed liquor volatile suspended solids
OUR Oxygen uptake rate
RBCOD Readily biodegradable COD
TSS Total suspended solids
VSS Volatile suspended solids
WWTP Wastewater treatment plant
WWTW Wastewater treatment works
WRC Water Research Commission
LIST OF SYMBOLS AND UNITS
CTCOD Total chemical oxygen demand (mgO2.ℓ-1)
S1 Inert soluble substrate (mgO2.ℓ-1)
SS Readily biodegradable substrate (mgO2.ℓ-1)
XAUT Autotrophic biomass (mgO2.ℓ-1)
XH Heterotrophic biomass (mgO2.ℓ-1)
XS Slowly biodegradable biomass (mgO2.ℓ-1)
X1 Particulate inert organics (mgO2.ℓ-1)
YH Yield of heterotrophic biomass (mgCOD-mgCOD-1)
INTRODUCTION
Bio-process modelling is fundamental in designing and managing wastewater treatment plants (WWTPs). Process models for WWTPs find application in forecasting, fault detection, monitoring plant operations and research (Lindberg, 1997). At a fundamental level a model may be a conceptual image of how a system functions, which alone cannot provide sufficient information about the behaviour of the actual system. To learn more about the system a pilot plant can be constructed. However, the pilot plants may have limitations of time and resources which prevent exploration of all potentially feasible solutions of the system, hence the turn to mathematical models which allow relatively more exploration of the feasibility space (Henze et al., 1987).
The modelling of biological wastewater treatment systems has developed from fundamental concepts to mathematical models. The IAWPRC, later IAWQ and now IWA (International Water Association) Task Group (Henze et al., 1987, Henze et al., 2000) has introduced an activated sludge model suite (ASM1, ASM2, ASM2d, ASM3 and other models), which provides researchers and practitioners with a standard set of basic models for biological wastewater treatment processes. These standard models may be used as building blocks for more complex biological processes. Wastewater characterisation generates input data for the bioprocess models.
Wastewater characterisation is crucial in process modelling because the quality of the model's predictions depends on the wastewater characterisation (Henze et al., 1995). The composition of wastewater has a significant impact on the performance of the activated sludge unit, particularly for nutrient removal systems (Melcer, 2003). A single characteristic may have such impact as determining whether or not a system designed for excess phosphorus removal will in fact remove phosphorus (Melcer, 2003). Hence a detailed knowledge of the composition of influent going into the wastewater treatment system is essential for the development of a reliable model. In the context of modelling WWTPs, wastewater characteristics are not only important for activated sludge system modelling, simulating the performance of other unit operations also depends on appropriate wastewater characterisation (Melcer, 2003).
Wastewater characteristics depend on the sources within the catchment served by a particular WWTP. The presence of industrial effluents can cause the wastewater to be significantly different from purely domestic effluent. Domestic effluent is discharged from sanitary conveniences in factories and residential, commercial, and various institutional properties. Domestic effluent constitutes of primarily water (approximately 99%) together with organic and inorganic constituents (Wun, 2006). Variations in domestic effluent characteristics across a given community tend to be relatively small, although variation between communities can be more readily detected.
Industrial effluent, on the other hand, varies in composition depending on the type of industry and materials processed in that catchment area. The composition of industrial effluent includes a range of constituents, such as high concentrations of readily biodegradable and non-biodegradable organic matter, inorganic matter, and potentially inhibitory substances. Total suspended solids (TSS), biological oxygen demand (BOD5) and COD values may be in the tens of thousands of milligrams per litre (Wun, 2006). Industrial effluents may also be severely nutrient deficient and contain high concentrations of heavy metals. In addition to the complex composition of industrial effluent, the flow pattern of industrial effluent streams can be very different from that of domestic effluent since the former would be influenced by the nature of the operations within a factory, such as shifts, whether a batch or continuous process is used, and other factors.
Chemical characterisation of municipal wastewater is concerned with three major constituents: organic, nitrogenous and phosphorus compounds (Wentzel et al., 2006). However, this paper focuses on the organic fraction of the wastewater.
The COD of municipal wastewater is divided into 3 main fractions: non-biodegradable, biodegradable and active biomass. The non-biodegradable COD has 2 fractions: the nonbiodegradable particulate, X1, and non-biodegradable soluble, S1 The biodegradable COD also has 2 fractions: the slowly biodegradable, XS, and readily biodegradable, SS, fractions. The active biomass consists of heterotrophic, XH, and autotrophic organisms, XAUT (Wentzel et al., 1995). The concentration of autotrophic biomass, XAUT, in the influent is negligible; hence the total COD can be expressed as the sum of 5 fractions (Wentzel et al., 1995):
CTCOD = S1+SS+X1+XS+XH
EXPERIMENTAL
Source of wastewater samples
Effluent samples were collected from 5 WWTPs. Four of these WWTPs; Marianridge, Darvill, Hammarsdale and Verulam
WWTPs receive a significant portion of industrial effluent to treat. The fifth WWTP, Shallcross WWTP, receives only domestic effluent.
Marianridge and Shallcross WWTP make up uMhlatu-zana Works, which is situated about 25 km from Durban. Marianridge WWTP receives a nominal average inflow of 8 Mℓ.d-1 of which about 30% is industrial wastewater and 70% domestic wastewater. The physical unit operations consist of screens and grit chambers. The biological reactor used is operated as an extended aeration basin, which is followed by secondary treatment in secondary settling tanks. The process configuration of Shallcross WWTP is similar to that of Marianridge WWTP. Shallcross WWTP handles about 2 Mℓ.d-1 of purely domestic wastewater. The final effluent from the two WWTPs is combined and dosed with chlorine before it is released to Umhlatuzana River (Mhlanga, 2008).
Hammarsdale WWTP is located in a sub-catchment of the Umlaas River known as the Sterkspruit River. Hammarsdale is located in KwaZulu-Natal between Durban and Pietermaritzburg. The Hammarsdale WWTP is a modified Bardenpho process with a design capacity of 27 Mℓ.d-1. Alum is added to the effluent after secondary treatment, which then enters a maturation channel before discharge into the Sterkspruit River (Fennemore et al., 2006).
Darvill Wastewater Treatment Works is situated on the eastern boundary of Pietermaritzburg. The wastewater treatment works receives all domestic and industrial effluent from the Pietermaritzburg-Msunduzi Transitional Local Council area (Hunt, 1998). The works were upgraded in 1992-1994 to a capacity of 60 Mℓ.d-1 and the process was upgraded from activated sludge to biological nutrient removal (Hunt, 1998).
Verulam WWTP is located about 27 km north of Durban. The WWTP consists of a Bardenpho-type activated sludge unit with a design capacity of 8 Mℓ.d-1 and a 4 Mℓ.d-1 circular activated sludge plant. The 4 Mℓ.d-1 activated sludge plant operates when the influent wastewater exceeds 8 Mℓ.d-1. The excess influent is diverted from the 8 Mℓ.d-1 activated sludge plant to the 4 Mℓ.d-1 activated sludge plant. The treated effluent is chlorinated and pumped to a pond which discharges into the nearby river.
Hourly samples of influent were drawn from the inlet of the WWTP for 24 h to make up a flow-weighted composite sample. The total COD of the composite sample was determined using the procedure outlined in Standard Methods (1995) and then the COD fractionation was carried out using procedures presented under 'methods'.
METHODS
In order to determine the amount of readily biodegradable fraction, SS, and heterotrophic active biomass, XH, the batch test procedure outlined in Wentzel et al. (1995) was followed. 1.5 ℓ of wastewater collected from the WWTP was poured into a bioreactor. The stirrer was switched on before an aliquot of the uniformly-mixed wastewater was drawn to determine the initial total COD. The total COD of wastewater was determined using the standard test presented in Standard Methods (1995). The bioreactor used had a cooling jacket which kept the contents of the reactor at 20 ± 1°C during the course of the experiment. Agitation was provided by a magnetic stirrer and the contents of the beaker were assumed to be completely mixed. The sample was aerated intermittently using an air pump and an aeration stone. The pH in the respirometer was maintained at 7.0 ± 0.2 by addition of hydrochloric acid and sodium hydroxide. The dissolved oxygen concentration (DO) was measured with an oxygen sensor (YSI DO probe) connected to the UCT OUR meter. The oxygen uptake rate (OUR) was calculated and stored inside the UCT OUR meter, which also controlled the aeration in the bioreactor. The aeration intervals were set at 4.5 to 6 mgO2.ℓ-1. Surface aeration of the contents of the bioreactor was prevented by introducing a predetermined nitrogen and air mixture on the surface of the contents of the bioreactor. A simplified version of the UCT Model (Dold et al., 1991) was used to interpret the respirogram.
The inert soluble substrate, SI, was determined by a floc-culation-filtration procedure on the wastewater collected at the end of the batch respirometric test (Wentzel et al., 1999). After running the batch test for 24 h or more, the only soluble COD remaining should be non-biodegradable soluble COD (Wentzel et al., 1999). Therefore, at the end of the batch test, 1 ℓ of the bioreactor contents was drawn as sample, flocculated and filtered using a 0.45 µm filter. The COD of the filtrate gives the inert soluble fraction of the total COD of the wastewater.
To determine the inert particulate COD fraction, XI, and slowly biodegradable fraction, XS, a model of the biological processes taking place in the bioreactor was used. The modified UCT model (Dold et al., 1991) was used to model the biological processes taking place inside the bioreactor during the experiment. The model output (OUR) was then compared to the measured OUR (respirogram) before XI and XS were tuned to fit the model-predicted respirogram to the one obtained from experiment.
RESULTS AND DISCUSSION
The shape of the OUR profiles obtained from the aerobic batch test on composite samples from Marianridge, Darvill, and Hammarsdale WWTPs were similar to Fig. 1. The profile shows an increase of the OUR from the start of the test when the amount of readily biodegradable substrate present is not limiting the growth of biomass. This is followed by a sharp decrease in OUR, when the readily biodegradable substrate is depleted. After this period the readily biodegradable substrate is only available due to the hydrolysis of slowly biodegradable substrate. However the OUR profiles obtained from the composite samples collected from Verulam WWTP vary significantly from Fig. 1. Figures 2 to 6 show some of the OUR profiles obtained from the aerobic batch test for Verulam wastewater samples.
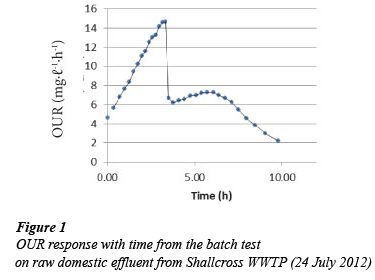

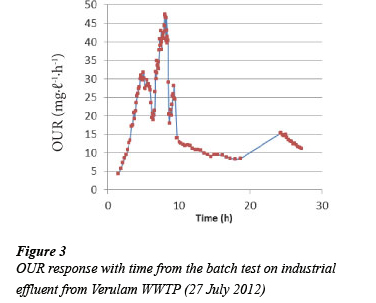


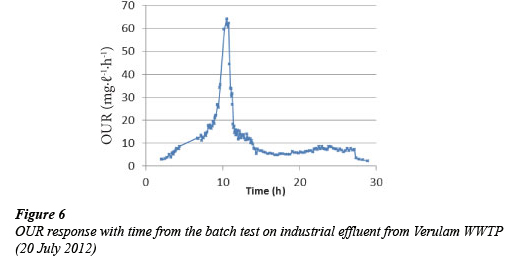
The significant variations observed in the OUR profiles for industrial effluent collected from Verulam WWTP when compared with OUR profiles from domestic effluent from uMhlatu-zana Works were as follows:
While Figs. 2 to 6 show the expected increase in OUR at the beginning of the test until the OUR reaches a maximum value when the readily biodegradable substrate runs out, it takes a longer time to utilise the readily biodegradable substrate in industrial effluent (up to 10 h) as compared to domestic effluent, which takes about 4 h. This might be due to the higher average total COD of the industrial effluent (1 600 mgCOD.ℓ-1) than domestic effluent (715 mgCOD.ℓ-1), and a slower rate of utilising the readily biodegradable substrate and other components present in industrial effluent.
After the readily biodegradable substrate runs out in industrial effluent, the expected precipitous drop seen in Fig. 1 (domestic effluent) does not occur. Instead a slower decline of the OUR is observed. The profile of the decline in OUR has a 'shoulder' before the OUR gets to the endogenous phase as seen in Fig.3. The cause for this 'shoulder' cannot be explained with certainty at this point. A possible explanation can be the presence of another biodegradable substrate which remains unutilised by the biomass until after the readily biodegradable substrate is utilised. The delay, from the start of the test, in utilising this suspected substrate might be the time it takes the biomass to acclimatise and be able to utilise this suspected substrate. Further exploration of this idea is being considered.
The area under the OUR profile between the start of the test and the beginning of the endogenous phase for the industrial effluent is much larger than that for the domestic effluent. This corresponds to the average initial total CODs of the two different wastewaters. The average total COD of the domestic effluent was lower (715 mgCOD.ℓ-1), while it was higher (1 600 mgCOD.ℓ-1 for industrial effluent.
The absence of a clearly defined precipitous drop of the OUR after utilising the available readily biodegradable substrate for industrial effluent collected from Verulam WWTP makes it difficult to interpret the OUR results using the modified UCT model. The modified UCT model does not take into account the degradation of substrate during the time period immediately after the precipitous drop and before the start of the endogenous phase; hence reliable COD fractionation for the industrial effluent from Verulam WWTP cannot be obtained using the current modified UCT model at this point. OUR results from Marianridge, Darvill, and Hammarsdale were interpreted using the modified UCT Model, since their batch test results showed the expected OUR profiles (Fig. 1). The results obtained from the COD fractionation of industrial effluent from the three WWTPs are summarised in Table 1.

The Marianridge influent contains about 62.3% biodegradable organic matter, of which 44.2% is slowly biodegradable particulate XS and 18.1 % is readily biodegradable substrate SS. Wentzel and Ekama (2006) gives the proportion of XS in typical South African wastewater as 60% and the readily biodegradable fraction SS as 20%. This shows that the influent from Marianridge WWTP contains less biodegradable substrate.
However it should be noted that the COD fractions given by Wentzel and Ekama (2006) are based on municipal wastewater of mainly domestic origin. The lower biodegradable component and higher non-biodegradable components in the influent to the Marianridge WWTP is clearly due to the presence of industrial wastewater from factories discharging into the plant, with textile effluent making up a significant portion of the industrial influent wastewater to the plant.
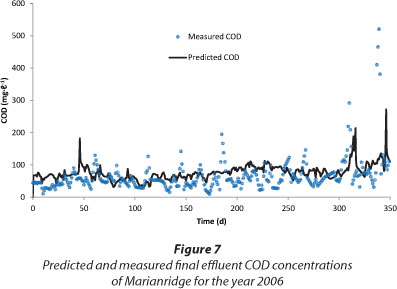
The COD fractionation of the influent wastewater from Marianridge was used to calibrate and validate the model for Marianridge WWTP. Reliable predictions were obtained from the model when the COD fractionation results were used as input representing the influent wastewater in the model. The influent characterisation was assessed to be satisfactory for modelling, because of the modelling response achieved even before calibration. Figure 7 shows simulations from the Marianridge WWTP model with the fractionation results as input.
The COD fractionation results for Darvill WWTP suggested a very high fraction of nonbiodegradable organic matter, 36.7% particulate inerts and 7.3% soluble inerts. The number of samples that were available from the COD fractionation of wastewater from Darvill WWTP was limited to a few samples, resulting in a data set which was unreliable, even for simulations in the Darvill WWTP model. The simulations that were carried out on the Darvill WWTP model showed that there was significant variation between historical measured values and predicted values of variables such as the final effluent COD. A notable observation was relatively higher final effluent COD values predicted by the model when compared to measured values. A possible cause for such high predicted final effluent COD values would be the high non-biodegradable COD fraction in the influent characterisation results. Furthermore the results for Hammersdale WWTP presented a similar challenge of having limited samples to test, thus limiting the discussion of the results to the conclusion that more samples need to be analysed to produce more reliable results.
CONCLUSION
COD fractionation of influent wastewater for a given WWTP can be done using the above presented method. A very detailed or a less detailed procedure, depending on the intended use of the process model, can be carried out. A more detailed procedure will give relatively more reliable results, as seen in the case of Marianridge WWTP. The reliability of the COD fractionation of the influent wastewater can further be investigated by assessing how well the simulation results of the plant model match the measured values. The two cases of Darvill and Hammarsdale WWTP show that more tests are required to have reliable COD fractionation of the influent wastewater. The batch test results of the industrial effluent from Verulam WWTP confirm the complexity of industrial effluent and its significant variation when compared to domestic effluent. A modified model needs to be used to interpret the respirogram for industrial effluent, such as the effluent from Verulam WWTP.
Complete and reliable COD fractionation results will complement regular influent characterisation that is routinely employed for monitoring WWTP operations, in order to produce the detailed representation of the influent wastewater that is required for the development of calibrated plant-specific models. The application of the plant-specific models includes process optimisation, simulations for design purposes, and scenario analysis, in order to gain information to make decisions on how best to run the WWTP, or to regulate the discharge of industrial effluents.
ACKNOWLEDGEMENTS
The authors would like to thank the South African Water Research Commission (WRC) and eThekwini Municipality for supporting this research through funding, providing information and laboratory services.
REFERENCES
DOLD PL, BAGG WK and MARAIS GvR (1980) Measurement of readily biodegradable COD fraction in municipality wastewater by ultra-filtration. UCT Report No. W57, Department of Civil Engineering, University of Cape Town, South Africa. [ Links ]
HENZE M, GRADY CPL, GUJER W, MARAIS GvR and MATSUO T (1987) Activated Sludge Model No.1. IAWPRC Scientific and Technical Report No.1. IAWPRC, London. [ Links ]
HENZE M, GUJER W, MINO T and VAN LOOSDRECHT MCM (2000) Activated Sludge Models ASM1, ASM2, ASM2d, and ASM3. IWA Scientific and Technical Report No. 9. IWA, London. [ Links ]
HENZE M, GUJER W, MINO T, MATSUO T, WENTZEL CM and MARAIS GvR (1995) Activated Sludge Model No.2. IAWQ Scientific and Technical Report No.3. IAWQ, London. [ Links ]
HUNT RP (1998) Strategic assessment for the disposal of sewage sludge Darvill wastewater treatment works, Pietermaritzburg. Proc. WISA 1998 Biennial Conference & Exhibition, 4-7 May 1998, Cape Town. URL: http://www.ewisa.co.za/misc/WISAConf/default1998.htm [ Links ]
EKAMA GA, DOLD PL and MARAIS GvR (1986) Procedures for determining influent COD fractions and the maximum specific growth rate of heterotrophs in activated sludge systems. Water Sci. Technol. 18(6) 91-114. [ Links ]
FENNEMORE CD, GALLAGHER D and NAICKER D (2006) The Hammarsdale experience - An exercise in cooperative governance. Proc. WISA 2006 Biennial Conference & Exhibition, 21-25 May 2006, Durban. URL: http://www.ewisa.co.za/misc/WISAConf/default2006.htm. [ Links ]
KAPPELER J and GUJER W (1992) Estimation of kinetic parameters of heterotrophic biomass under aerobic conditions and characterization of wastewater for activated sludge modelling. Water Sci. Technol. 25(6) 105-124. [ Links ]
LINDBERG CF (1997) Control and estimation strategies applied to the activated sludge unit. PhD thesis, Uppsala University, Sweden. [ Links ]
MELCER H (2003) Methods of Wastewater Characterization in Activated Sludge Modelling. Water Environment Research Foundation, Alexandria. IWA Publishing, London. [ Links ]
MHLANGA FT (2008) Modelling the Marianridge wastewater treatment plant. MSc Eng thesis, University of KwaZulu-Natal. [ Links ]
ORHON D and CORGNOR EU (1996) COD fractionation in wastewater characterization: The state of the art. Chem. Tech. Biotechnol. 68(1) 283-293. [ Links ]
STANDARD METHODS (1995) Standard Methods for the Examination of Water and Wastewater (19th edn.). American Public Health Association, Washington DC, USA. [ Links ]
WENTZEL MC and EKAMA GA (2006) Characterization of municipal wastewater. Department of Civil Engineering, University of Cape Town, South Africa. [ Links ]
WENTZEL MC, MBEWE A and EKAMA GA (1995) Batch measurements of readily biodegradable COD and active organism concentration in municipal wastewater. Water SA 21 (2) 117-124. [ Links ]
WUN JERN NG (2006) Industrial Wastewater Treatment.Technological University, Singapore. 1-11. ISBN 978-1-86094-580-9. [ Links ]
This paper was originally presented at the 2012 Water Institute of Southern Africa (WISA) Biennial Conference, Cape Town, 6-10 May 2012.
* To whom all correspondence should be addressed. +27 31 260-1129; Fax: +27 31 260-1118; e-mail: 207528092@stu.ukzn.ac.za














