Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Water SA
On-line version ISSN 1816-7950
Print version ISSN 0378-4738
Water SA vol.39 n.2 Pretoria Jan. 2013
Real-time PCR quantitative assessment of hepatitis A virus, rotaviruses and enteroviruses in the Tyume River located in the Eastern Cape Province, South Africa
Timothy Sibanda; Anthony I Okoh*
Applied and Environmental Microbiology Research Group (AEMREG), University of Fort Hare, P Bag X1314, Alice 5700, South Africa
ABSTRACT
We applied real-time RT-PCR (reverse transcription-polymerase chain reaction) to assess the incidence of hepatitis A virus, rotaviruses and enteroviruses in the Tyume River, an important water resource in the impoverished Eastern Cape Province of South Africa. Detection of noroviruses was done using conventional semi-nested RT-PCR. Water samples were collected once monthly from 6 sampling sites over a 12-month period starting in August 2010 and ending in July 2011. Hepatitis A virus was detected in 13% of the samples in concentrations ranging between 1.67x103 genome copies/ℓ and 1.64x104 genome copies/ℓ while rotaviruses were detected in 4% of the samples with concentrations ranging from 9x10' genome copies/l to 5.64X103 genome copies/ℓ. Enteroviruses were not detected in any of the samples, while noroviruses were detected in 4% of the samples. All hepatitis A and rotaviruses positive samples were from the upstream sections of Tyume River while noroviruses were detected in samples from downstream sections only. Statistical analysis showed that occurrence of the viruses in Tyume River was sporadic. Risk analysis showed that hepatitis A virus posed greater risk than rotaviruses for both recreational and domestic water uses. Because of the low infectious dose of enteric viruses, the detection of even low concentrations of hepatitis A virus, rotaviruses and noroviruses in surface water poses a significant risk to public health.
Keywords: Hepatitis A virus, rotaviruses, noroviruses, TaqMan real-time PCR, reverse transcription, Tyume River, public health
Introduction
Ground and surface water sources may be subjected to faecal contamination from a variety of sources, including sewage treatment plant effluents, on-site septic waste treatment discharges, land runoff from urban, agricultural and natural areas, and leachates from sanitary landfills (Abbaszadegan, 2001). Consequently, millions of people throughout the world do not have access to microbiologically safe water for domestic, recreational and other general purpose uses (Gibson et al., 2011). Health risks associated with surface water use, either raw or treated, include infectious diseases predominantly caused by human and animal enteric pathogens, most notably viruses. RNA viruses constitute the most abundant group of pathogens in man, animals and plants (Bustin and Mueller, 2005). Surveillance of source waters for viral pathogens is therefore necessary to protect public health. The culture-propagation procedure is still the best method to enumerate viruses and demonstrate their infectivity. However, for the detection of noroviruses (NoVs), hepatitis A virus (HAV) and other enteric viruses like enteroviruses (EVs) for which appropriate cell cultures are not available, slow or limited (Hong et al., 2011), molecular techniques become the most viable option. Molecular techniques have been successfully applied on environmental samples, allowing a rapid and specific detection of human enteric viruses (Bosch et al., 2008; De Paula et al., 2007; Costafreda et al., 2006). The ability of real-time reverse transcription polymerase chain reaction (qRT-PCR) to generate accurate quantitative data has had a huge impact on the study of viral agents of infectious disease (Schutten and Niesters, 2001).
Hepatitis A virus has a worldwide distribution and is the aetiological agent of hepatitis, an acute, usually self-limiting infection of the liver. On average, about 1.5 million cases of clinical hepatitis are recorded world-wide each year (WHO, 2003). Hepatitis A virus belongs to the genus Hepatovirus (Feinstone, 1996) and has a positive-sense, single-stranded RNA genome which is 7.5 kb in length (Murray et al., 2005). The virus is shed in the faeces of persons with both asymptomatic and symptomatic infection and under favourable conditions HAV may survive in the environment for months (CDC, 1999). Hepatitis A disease is rarely fatal but may represent a substantial economic burden, particularly in countries with low and intermediate incidence rates where a larger portion of the adult population do not have immunity against the disease (Grabow, 1997). Infected persons are infective from 14 to 21 days before the onset of jaundice and up to 7 to 8 days after jaundice has resolved (Tong et al., 1995). Food and water have been identified as the main vehicles for HAV transmission (Koopmans et al., 2002). Possible routes of infection with HAV include close personal contact with infected persons (Adhami and Carey, 2010), recreational exposure to faecally polluted surface water (Hunter, 1997), as well as consumption of contaminated food and water (Koff, 1995).
On the other hand rotaviruses (RVs) are responsible for the majority of acute gastroenteritis infections occurring in young children worldwide (Jothikumar et al., 2009). The genus Rotavirus belongs to the family Reoviridae and can be divided into 7 groups, A-G, with the majority of human infections being caused by viruses of Group A (Adlhoch et al., 2011). Rotaviruses are non-enveloped RNA viruses (Fritzinger et al., 2011) with a segmented double-stranded RNA genome (Bustin and Mueller, 2005). Parashar et al. (2006) estimated that infection by Group A rotaviruses results in about 600 000 fatalities annually, most of them in developing countries, while Freeman et al. (2008) postulated that by the age of 5 years, nearly all children in both industrialised and developing countries would have been infected with Group A rotaviruses. While RV infections in young children can be severe, with life-threatening diarrhoea, Kang et al. (2004) report that infections in older individuals may be asymptomatic or be associated with mild enteric symptoms, which the authors speculated to be due to increasing cross-protective immunity as a result of repeated infections.
Noroviruses (NoVs) belong to the genus Norovirus. They comprise of non-enveloped, single-stranded RNA viruses which have been found in humans, pigs, cattle, sheep and mice (Wolf et al., 2010). Human NoVs (HuNoVs) are the most common etiological agent for gastroenteritis outbreaks as well as the leading cause of non-bacterial gastroenteritis in all age groups (Gentry et al., 2009; Siebenga et al., 2009). Human noroviruses GI and GII have been detected in both freshwaters and estuarine waters worldwide. Aw and Gin (2010) reported the detection of both NoV genogroups GI and GII in 100% of sewage and secondary effluent samples in Singapore while Lee and Kim (2008) reported genetic diversity of HuNoVs detected in river water in Korea. Numerous studies have also detected NoVs in environmental waters in Europe (Lysen et al., 2009; Lodder and de Roda Husman, 2005), in the United States of America (Gentry et al., 2009) and in South America (Victoria et al., 2010). A recent report on the detection of enteric viruses in selected urban and rural river water and sewage in Kenya (Kiulia et al., 2010) revealed that HuNoVs GI and GII were detected in 90% of samples collected from urban rivers and streams. Though the first documented NoV outbreaks in South Africa were described as early as 1993, the current NoV prevalence and circulating genotypes are unknown due to lack of NoV outbreak reporting systems (Mans et al., 2010). However, work done by Mans et al. (2013) shows that NoVs are still widely distributed in sewage-polluted river water in South Africa.
Enteroviruses (EVs) are RNA viruses belonging to the Picornaviridae family and are classified into 5 human pathogenic species including poliovirus and human enteroviruses (HEVs) A, B, C and D (Dierssen et al., 2008). Echoviruses and coxsackieviruses are the most commonly identified causes of viral meningitis in the paediatric population (Archimbaud et al., 2004) and in adults (Hong et al., 2010). Enteroviruses are associated with diverse clinical syndromes, ranging from mild febrile illness to severe central nervous system diseases, such as aseptic meningitis and encephalitis, potentially leading to paralysis (Hong et al., 2011). These viruses are faecally shed in extremely high numbers from infected individuals, and stable in the environment for extended time periods (Connell et al., 2012). They have a worldwide distribution and have previously been detected in surface water in the USA (Donaldson et al., 2002), Switzerland (Gilgen et al., 1995), Japan (Haramoto et al., 2005) and in treated drinking water in South Africa (Vivier et al., 2004).
Contamination of surface waters with enteric viruses is a concern for public health, especially if these surface waters are used for recreational purposes and production of drinking water (Mans et al., 2013; Rutjes et al., 2005). Pathogen monitoring should be a part of the future characterisation of microbiological hazards in water in order to address risk and effect prevention strategies (Rose and Molloy, 2007). Many areas in developing countries lack access to safe drinking water with around 70% of the global population without improved drinking water sources residing in rural areas (WHO and UNICEF, 2010). Settlements in rural areas are situated far apart and divided by rough terrain, dramatically reducing the capacity to provide a centralised drinking water system. Thus, rural populations commonly obtain water on an individual or household basis from nearby surface and groundwater sources where the microbial quality is often unknown (Peter-Varbanets et al., 2009). Tyume River water is used for the production of drinking water, irrigation and recreational purposes. Faecal contamination of Tyume River has previously been linked to the presence of bacterial pathogens (Momba et al., 2006a). However, traditional bacterial indicators and/or pathogens often fail to predict or correlate with the occurrence of pathogenic water-borne viruses of public health concern (Brooks et al., 2005). To the best of our knowledge, no research has previously been done to detect the presence of viral pathogens in Tyume River. There is therefore a need to cover this knowledge gap. In this paper we report on incidences of some HAV, RVs, EVs and NoVs in Tyume River as well as the risk associated with accidental consumption of water contaminated with these viruses.
MATERIALS AND METHODS
Description of study site
The Tyume River is located in the Nkonkobe Local Municipality, under the Amathole District Municipality, in the Eastern Cape Province of South Africa. It flows from the upper part of the Amathole Mountains in Hogsback, passing through the lower coastal escarpment down to Alice, through several rural settlements, and finally joins the Keiskamma River at Manqulweni community. Close proximity of the river to its host communities makes it an ideal water source for domestic activities where piped potable water is not available. The Tyume River also feeds the Binfield Park Dam which serves as a source of raw water for a drinking water treatment plant in the area. The selected sampling sites, for this study were, from upstream to downstream: Hala, Khayalethu, Sinakanaka, Alice, Drayini and Manqulweni.
Hala is a community immediately downstream from the source of the Tyume River in Hogsback. With the river source at Hogsback considered 'pristine' and inaccessible, a sampling point in this community located at 32°36'39"S 26°54'34"E was chosen as the first sampling site. Khayalethu is a rural community upstream of the Binfield Park Dam. The Khayalethu sampling site is located at 32°38'22"S 26°56'10"E. The inhabitants of this settlement use the river water for irrigation, recreation, stock watering and domestic purposes. Sinakanaka is also a rural settlement further downstream from Khayalethu. The sampling site in this community is located at 32°45'37"S 26°51'27"E. The Tyume River is very important to the inhabitants of this settlement as it is used for drinking, fishing, irrigation, livestock watering, recreation and other domestic purposes.
Alice is a small town which has several suburbs. The sampling site is located close to a bridge of a commercial road (R63) linking Alice Town to East London at 32°47'17"S 26°50'31"E. The river is extensively used for irrigation, fishing and domestic purposes, as well as a source of drinking water for livestock. Drayini is a rural town downstream of Alice on the banks of the Tyume River. The sampling site is located at 32°48'37"S 26°52'20"E just downstream of the University of Fort Hare farmlands and wastewater treatment plant effluent discharge point. Here the river water is mainly used for livestock watering. Manqulweni is located further downstream from Drayini near the confluence of Tyume River and the Keiskamma River, at 32°54'50"S 26°56'13"E.
Sampling
Water samples were collected once monthly at each sampling site over a 12 month period from August 2010 to July 2011, giving a total of 72 samples for the duration of the study. Samples were transported in portable ice chests (cooler boxes) to the Applied and Environmental Microbiology Research Group (AEMREG) Laboratory at the University of Fort Hare, Alice, for analysis within 6 h of sample collection, following the procedure recommended by APHA (2005).
Concentration of viruses in water
The adsorption-elution method of Haramoto et al. (2005), with a recovery efficiency of 56%, was used to concentrate viruses in water samples, with some modifications. Under neutral pH conditions viruses are negatively charged and are positively charged under acidic conditions. Multivalent cations (Mg2+, Al3+) can change the surface charge of viruses thereby allowing adsorption to negatively charged membranes. Aliquots of 5 mℓ of 250 mM AlCl3 were passed through HA filters (0.45 µm pore size and 47 mm diameter, Millipore) to form cation (Al3+)-coated filters. Subsequently, 1ℓ of the water samples was passed through the filters. The cations were then washed by passing 200 ml volumes of 0.5 mM H2SO4 through the membranes. Viral particles were eluted with 10 mℓ of 1 mM NaOH. Eluates were placed in tubes containing 0.1 mℓ of 50 mM H2SO4 and 0.1 ml of 100x Tris-EDTA (TE) buffer for neutralisation before ultraconcentration. Ultraconcentration was done using Centriprep YM-50 ultrafiltration devices (Millipore) to obtain final volumes of approximately 700 µℓ. The sample concentrates were stored at -80°C until ready for use. Storage of viruses at temperatures below -60°C has been shown to result in insignificant loss of both titre and infectivity for periods longer than a decade (Merrill et al., 2012; Gould, 1999).
Extraction of viral nucleic acids
RNA was extracted using 200 µℓ of the final concentrated sample using commercial RNA purification kits (Quick-RNATM MiniPrep (Zymo Research, USA)) following the method of Boom et al. (1990). This method is based on the lysing and nuclease-inactivating properties of the chaotropic agent guanidinium thiocyanate (GSCN) together with the nucleic acid-binding properties of silica particles in the presence of this agent. To remove contaminating DNA, the eluates were treated with RNase-free DNase followed by heating at 70ºC to inactivate the DNase. However, the RNA extraction efficiency of this method was not monitored but was assumed to be 100%. Purified viral RNA was eluted in 60 µℓ of RNase-free water.
Quantification of HAV, RV and EV genomes by qRT-PCR
Hepatitis A virus and RVs were quantified in a 2-step protocol where RNA was first reverse transcribed into cDNA in a separate reverse-transcription step. Briefly, 10 µℓ of template RNA, 1 µℓ of Random Hexamer Primer, 1 µℓ dNTP mix, 2.5 µℓ DEPC-treated water, 4 µℓ 5X RT buffer, 0.5 µℓ Ribolock RNase inhibitor and 1 µℓ RevertAid Premium Reverse Transcriptase (Fermentas Life Sciences) were added in the indicated order into a 0.5 ml PCR tube on ice. The mixture was briefly vortexed to ensure total mixing and thereafter centrifuged. The tubes were then incubated at 25°C for 10 min followed by 30 min at 60°C. The reaction was terminated by heating at 85°C for 5 min. For RVs, prior to the reverse transcription reaction the RNA was denatured by heating at 95°C for 5 min followed by incubation on ice for 2 min to separate its double-stranded RNA.
To quantify the viruses, the resultant cDNA was used as template in a quantitative TaqMan real-time PCR (StepOnePlus PCR system; OPTIPLEX 755, Applied Biosystems) with TaqMan probes using a 96-well plate. The wells were loaded with 20 µℓ of a reaction buffer containing 12.5 µℓ of 2x TaqMan universal PCR MasterMix [Applied Biosystems], 400 nM sense primer, 400 nM antisense primer, and 250 nM TaqMan probe and PCR grade water (Haramoto et al., 2008). Subsequently, 5 µℓ aliquots of sample cDNA were added with mixing to give 25-µℓ total reaction mixtures. The plate was sealed and loaded in the thermocycler. The assays were performed under the following cycling conditions: HAV (10 min at 95°C for Taq activation, and 45 cycles of denaturation at 95°C for 15 s, annealing at 60°C for 1 min, and extension at 70°C for 1 min); RVs (Taq activation at 95°C for 15 min; 45 cycles of denaturation at 95°C for 15 s, annealing at 55°C for 30 s, and extension at 72°C for 30 s) and EVs (Taq activation at 95°C for 10 min; 45 cycles of denaturation at 94°C for 15 s, annealing at 58°C for 1 min, and extension at 72°C for 20 s). Fluorescence data were collected at the end of the annealing step. The probes and primer pairs used for quantification are shown in Table 1.
To determine the qRT-PCR limit of detection, tenfold serial dilutions of cDNA (for each virus) with quantities ranging from 100 000 genomic equivalents to 0.1 genomic equivalents were run under the same conditions as those of the individual viruses. Primer specificity and possible cross-reactivity was determined by substituting target cDNA with non-target DNA. The standard curve for each virus was formulated as described by Brooks et al. (2005) and Haramoto et al. (2008). Briefly, RNA was extracted from positive ATCC strains (HAV: ATCC VR-1357; strain PA21 and RVs: ATCC VR-2274; strain 248) using commercially available extraction kits (Quick-RNATM MiniPrep (Zymo Research, USA)). The RNA extracts were then reverse transcribed into cDNA using random primers. The resultant cDNA was subsequently quantified using a Qubit® fluorometer (probes.invitrogen.com/qubit) and diluted by serial tenfold dilution. The sample extracts and standards were then subjected to real-time PCR simultaneously, followed by analysis using SDS software (Applied Biosystems") to obtain quantitative data on the titre of viral cDNA in a well. Two wells were used for each of the standards, negative controls (no template controls) and samples, and the average used for subsequent calculations. The total number of viruses in the viral suspensions and eluted samples were estimated by multiplying the titre of viruses per millilitre by the volumes of the samples.
Detection of NoV genogroups by conventional semi-nested PCR
Norovirus genogroups GI and GII were detected by conventional semi-nested PCR as described by Victoria et al. (2010). NoV genogroup GI and GII specific primer sets which target the viral RNA-dependent RNA polymerase gene (Boxman et al., 2006) were used. These primer sets have been used in previous studies for detecting NoV GI and GII in environmental samples (Victoria et al., 2010). The primer sets are shown in Table 2.
PCR cycling conditions for both norovirus GI and GII were as follows: 1st round PCR; 3 min at 94°C to activate the Taq DNA polymerase followed by 40 cycles of 1 min at 94°C, 1.5 min at 37°C, 1 min at 72°C, and a final extension of 72°C for 7 min. The 2nd round PCR was run under the same conditions as the first round, except that initial Taq activation temperature time was increased from 3 min at 94°C to 5 min at 94°C. The composition of the reaction mixtures was as follows: 12.5 µℓ PCR Master Mix (Fermentas), 1 µM of each of the forward and reverse primers, 5 µℓ of cDNA (and for 2nd round PCR, 2.5 µℓ of 1st round PCR amplicon), and nuclease-free water to give a total reaction volume of 25 µℓ. Amplified products were analysed on ethidium bromide-stained 2% agarose gels.
Risk assessment
A major limitation of the real-time PCR assay used in this study is its inability to determine the viability and infectivity of viruses detected, as the presence of viral nucleic acid does not necessarily indicate the presence of infectious viruses (Bofill-Mas et al., 2010; Hamza et al., 2009). To circumvent this limitation, ratios of infectious viruses to total virus particles based on outcomes of previous studies (Pinto et al., 2009; Grabow et al., 1992) were used to estimate the infectious virus doses for the viruses in this work. In the case of rotaviruses grown in the MA104 cell line, the ratio of infectious virus particles to total detected virus particles was 1:40 000 (Rodriguez et al., 2009; Ward et al., 1984) while for hepatitis A virus the ratio was 1:60 (Pinto et al., 2009; Deng et al., 1994;). Calculations for the microbial risk assessment were done to assess the fitness-of-use of the water for domestic and recreational purposes based on assuming accidental consumption of 10 mℓ (Steyn et al., 2004) and 100 mℓ (Venter et al., 2007) of the river water, respectively. The total number of infectious viruses was estimated first by 'correcting' the virus concentrations obtained directly from the qRT-PCR assay. 'Correcting' was done by multiplying the virus concentrations by the inverse of the mean recovery efficiency of the filtration method, which in this case was 56% (Haramoto et al., 2005). The corrected virus concentrations were then multiplied by the ratio of infectious virus particles to total virus particles for each virus in order to obtain the infectious dose of each virus. Estimates of risks of daily infection for the enteric viruses were determined using the models (WHO, 2001; Haas, 1996) shown below:
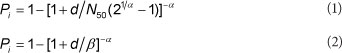
Equations (1) and (2) were used for HAV and RVs, respectively. The parameters are described in Table 3.
TABLE 3
Parameters used in estimating the risks of daily infection using Eqs. (1) and (2)
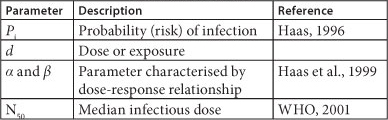
Values of α and β were 0.2531 and 0.4265, respectively, for rotaviruses (Haas et al., 1993), while for HAV, N50 and α assumed values of 100 and 0.2, respectively.
Statistical analysis
Results were analysed using the Statistical Package for the Social Sciences (IBM SPSS Statistics release 19; IBM, USA). To test if viral detection depended on season or sampling site, a one-way ANOVA and Tukey's studentized range (HSD) Test were used. All tests were carried out at a 5% level of significance.
Results
The detection limit of the real-time PCR assays was determined to be 10 genomic equivalents achieved, with a Ct value <35. Samples with Ct values >35 were therefore considered negative. When non-target cDNA was used, there was no amplification, confirming the specificity of the primer-probe combinations for the target cDNA. Hepatitis A virus was detected in 13% of the samples in concentrations ranging between 1.67x103 genome copies/ℓ and 1.64x104 genome copies/ℓ. Of these positive samples, 56% were collected between October 2010 and January 2011 while the other 44% were collected between June and July 2011. Hepatitis A virus detection was not affected by season (P>0.05). The amplification plot and standard curve and the log10genome copies of HAV per litre obtained after the real-time PCR assay are displayed in Figs. 1 and 2.
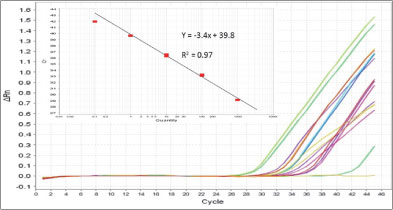
Figure 1
Standard curve and amplification plot for HAV quantitation in Tyume River
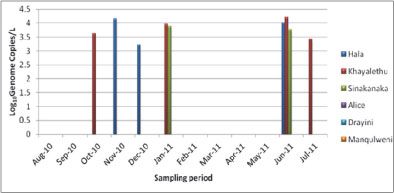
Figure 2
log10genome copies/ℓ of HAV at selected sites along Tyume River
Rotavirus RNA was detected in 4% of the samples in concentrations ranging between 9x101 genome copies/ℓ and 5.64x103 genome copies/ℓ. Of the positive samples about 67% were collected during the winter months (June and July 2011) while 33% were collected in September 2010 (spring). Rotavirus detection did not follow any seasonal pattern (P<0.05). Enteroviruses were not detected in any of the samples, while noroviruses were detected in 4% of the samples (gel picture not shown). All NoV positive samples were collected from 2 downstream sites. Norovirus GI was detected in samples from Alice and Drayini sampling sites in August and September 2010 respectively while NoV GII was detected in a sample from the Alice sampling site in October 2010. Detection of NoVs was sporadic (P<0.05).
Risk assessment
The uncorrected and corrected mean concentrations of viruses as well as the calculated mean concentration of infectious viruses in Tyume River are shown in Table 4.
Assuming accidental ingestion of 10 ml and 100 ml for domestic and recreational water usage, respectively, the calculated doses were 2.4x100 and 2.4x101 for HAV and 8.43x10-4 and 8.43x10-3 for RVs, respectively. The daily risk of infection results are displayed in Fig. 3.
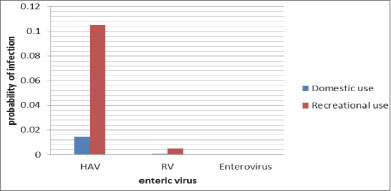
Figure 3
Risk of infection from enteric viruses in Tyume River
DISCUSSION
Hepatitis A infection is the leading cause of acute viral hepatitis throughout the world (Costafreda et al., 2006) while RV gastroenteritis continues to be the single most important cause of dehydration in young children (Kang et al., 2004). In this study, both viruses were detected only in samples collected from the upstream sampling sites where population pressure is less compared to the downstream stretch of Tyume River. This observation is in tandem with documented findings that the distribution patterns of HAV and RVs in different geographical areas of the world are closely related to socioeconomic development, with endemicity being high in less-developed regions (Jothikumar et al., 2009; Fernandez-Molina et al., 2004). Additionally and most crucially, Kang et al., (2004) suggested that exposure to an environment contaminated with human and animal faeces, and close contact with animals in the domestic environment, are factors that are likely to promote mixed RV infections and inter-species transmission of RV strains, enabling viral reassortment and the emergence of new strains. Such scenarios are most commonly observed in rural areas, especially of developing countries. In this study, communities along the upstream stretch of Tyume River are rural, with cattle pens situated close to houses, possibly for the security of the animals. Homes are also situated further apart than in the downstream stretch, which is characterised by urban to semi-urban settlements. The sanitary infrastructure in the upper Tyume catchment may therefore not be as established as it is in the lower Tyume with the possible consequence that the majority of the population still use the 'bush-toilet' system. In the event of flash storms, raw human waste is washed down into the rivers possibly contaminating surface waters with enteric pathogens. Also, the use of pools found in the river for recreational activities like swimming is common practice among rural children, which raises both the chances of surface water contamination with enteric viruses and also the risk of swimmers getting infected. Waterborne RVs have been detected elsewhere (Kittigul et al., 2005b) and the first large waterborne epidemic in an adult population was caused by Group B rota-viruses in China (Logan et al., 2006). Faecally-polluted natural surface water used for recreational activity could therefore pose a potential health risk to the public (Venter et al., 2007). Such swimming activities are also likely to cause localised distributions of enteric pathogens along stretches of the river course. It is a widely acknowledged fact that by the age of 10, nearly 100% of children in low socio-economic groupings would have suffered from and hence acquired immunity against, HA infections (Taylor et al., 2001). Such statistics could be attributed to a tendency by the children in these low socio-economic groupings to 'play-in-the-water' thereby exposing themselves to HAV infection at that early age.
Untreated or insufficiently treated wastewater has been noted to play an important role in the transmission of both HAV and RVs (Lodder and de Roda Husman, 2005; Mara, 2000). In a study by Lodder and de Roda Husman (2005) they found that the concentrations of RV RNA in naturally contaminated raw and treated sewage were similar, suggesting inefficient removal of rotaviruses by the treatment processes used in the involved sewage treatment plant. Viral contamination of water sources has been frequently reported as a primary source of gastroenteritis or hepatitis outbreaks (Brassard et al., 2005). The structural characteristics of HAV make it a very stable virus, largely resistant to physical-chemical agents (De Paula et al., 2007). Consequently, HAV can survive in water for long periods of time (Soule et al., 1999). Several studies (Espinosa et al., 2008; Rutjes et al., 2005; Caballero et al., 2004) have also demonstrated the stability of RVs in water, and therefore the potential risk for the human population to acquire RV infections associated with environmental contamination by water-borne transmission. No detection of both HAV and RVs in samples from middle to downstream Tyume may have a twofold interpretation. It could mean that these viruses were not in circulation in the host population; hence their absence also from the environment. Alternatively, these findings may also be an indication of the higher concentrations of PCR inhibitors from downstream samples which could also have caused false negative results to be obtained. However, no dilution of samples was carried out in order to ascertain if indeed the negative results were a consequence of inhibition.
The detection of HAV in this study is in tandem with the findings of Taylor et al. (2001), who also detected HAV in surface river and dam water used for recreational and domestic purposes in South Africa. Because only a few viral particles are needed to cause disease (De Paula et al., 2007), detection of low concentrations of the virus in water becomes significant. The occurrence of HAV in raw water sources has been tied to epidemiological features such as an outbreak in a particular community, and it is thought that the minimal infectious dose is extremely low, possibly as low as a single infectious particle (Zhi-Yi et al., 1992). HAV has also been estimated to impose a large economic burden throughout the world, with adult sufferers estimated to miss 30 days of work per annum (Berge et al., 2000). Berge et al. (2000) also estimated that medical treatment and work loss account for an estimated ZAR4 billion annually in the United States. While epidemiological studies have shown that HAV is endemic in South Africa (Venter et al., 2007) the burden of HAV infection in South Africa is however, still unknown. This is because HAV can cause asymptomatic infections that go unrecognised until secondary person-to-person spread finally leads to overt disease in hardly traceable pockets of the population (Bosch, 1998).
Occurrence of infectious enteric viruses in environmental samples is mostly determined using cell culture techniques (Fong and Lipp, 2005). However, cell culture detection of HAV tends to be lengthy, with reduced specificity (Li et al., 2002) and sensitivity (Kittigul et al., 2005a), especially when applied to environmental samples where the virus titre could be low. The real-time PCR technique is an efficient tool in detecting HAV in environmental samples because it combines PCR amplification with the use of a probe to confirm the identity of the PCR product (De Paula et al., 2007). Results of a study carried out by Villar et al. (2006) to evaluate methods used to concentrate and detect HAV in water samples also confirmed that compared to qualitative PCR, real-time PCR detects low concentrations of genome per millilitre and is more suitable than qualitative PCR for the detection of HAV RNA in environmental samples.
In the case of RVs, combinations of techniques have been used for its detection, which includes cell culture (Rutjes et al., 2009), electron microscopy (EM) (Bishop et al., 1973), latex agglutination (LA) and enzyme immunoassay (EIA) (Pang et al., 2004). However, molecular techniques have also proven to be a technological advancement in the detection of RVs, both in clinical and environmental samples. Gunson et al. (2003) reported a 48% increase in the detection rate of Group A rotaviruses by reverse transcription-PCR compared to EIA or EM. Logan et al. (2006) however, reported a 110% and 186% increase in the detection of RVs by real-time PCR compared to LA and EM, respectively. Interestingly, Pang et al. (2004) observed, using the same primers, that the real-time RT-PCR assay for RVs detection in clinical samples was 1 000 times more sensitive than conventional RT-PCR. However, PCR methods are susceptible to inhibitors widely existing in water samples and may consequently yield false negative results (Yang et al., 2011).
Group A rotaviruses have been detected in untreated and treated drinking-water samples in Southern Africa (Van Zyl et al., 2006). While detection of both HAV and RVs was observed to be confined to the upstream stretch of the river compared to the downstream (P<0.05), occurrence of both viruses tended to be sporadic (P>0.05).
In general, seasonality of virus infections is difficult to detect in surface water samples because, whereas circulation within the human population is mainly during the summer season, their detection in environmental water samples is mainly during winter (Rutjes et al., 2009), chiefly because of the low temperatures which significantly reduce the deactivation rates. Previous findings note that, whereas RV infections are common all-year round in tropical climates (Cook et al., 1990), RV levels in the environment are generally higher during winter and spring (Hejkal et al., 1984), corresponding to seasonal variations of rotaviral diarrhoea in the population (Mehnert and Stewien, 1993).
Unlike other RNA viruses detected in this study, all NoV detections were in samples collected from the downstream stretch of Tyume River which is more impacted by wastewater effluents than the upper stretch of the river. Noroviruses have previously been detected in wastewater and surface water (Haramoto et al., 2005; Lodder and De Roda Husman, 2005). It has also been noted that NoVs in the urban environment may be transported by stormwater runoff, combined and sanitary sewer overflows, and discharge of wastewater treatment plant effluents (Arnone and Walling, 2007). Norovirus detection in samples from the downstream sampling sites might have been influenced by their relatively close proximities to sewage outfall points. The Alice sampling site is immediately downstream of the effluent discharge points of the Victoria Hospital and Alice Town wastewater treatment plants. The Drayini sampling site, while located downstream of the Alice sampling site, is also located immediately downstream of the University of Fort Hare wastewater treatment plant effluent discharge point. Similar results have been reported by Aw et al. (2009), who detected human NoVs in downstream waters of urban rivers and the receiving estuarine bay, suggesting urban runoff as a source of viral contamination. Considering that viral RNA was concentrated from only 1 ℓ of sample, it is possible that more positive samples would have been detected had larger volumes been used, since previous findings have shown that there are low concentrations of viruses in environmental waters (Aw et al., 2009), necessitating the concentration of NoVs from larger volumes of water. Noroviruses, like most other enteric viruses, are able to survive treatment processes if there is inadequate chlorination. While it is reported that NoVs can be destroyed by 'adequate chlorination' (Shin and Sobsey, 2008), their physico-chemical stability helps them to pass through sewage treatment without inactivation and reach many kinds of environmental waters (Victoria et al., 2010).
Naked RNA has been found to have limited survival in the environment (Tsai et al., 1995). Also, viral capsids and not naked RNA has been observed to bind to the membrane used for the selective recovery of viruses (Katayama et al., 2002). This may imply that the RNA found in the water is most likely accompanied by virus particles and would most probably cause infection. Viruses have also been reported to survive and remain infective for up to 130 days in seawater, and for up to 120 days in freshwater and sewage (Fong and Lipp, 2005). Rotaviruses in particular have been found to be stable in environmental conditions. Fischer et al. (2002) have reported that in tropical temperatures (30ºC), RV particles can survive for more than 2 months and can maintain infectivity for more than 32 months at <10ºC. While molecular detection methods cannot differentiate between infective and damaged viruses, both these classes of viruses are nevertheless detected by PCR and any such detections therefore could pose a public health risk to the consumer. In a rural setting with no potable water sources, the risk is made all the more real not only by the dependence (of rural people) on surface water for domestic and general purpose uses, but also by the possibility that all contaminating faecal matter, in which viruses could be present in large quantities, would have undergone little or no treatment at all.
Risk assessment
For both HAV and RVs, the calculated risk of infection was higher for recreational water use compared to domestic water use, most likely because of the larger volume (100 mℓ) involved which has the consequence of also increasing the dosage. Even though EVs were not detected in this study, they are nevertheless very widespread in environmental waters and the risk they pose to public health can therefore not be underestimated. It is possible that the results could have been different had sample volumes larger than 1 ℓ been used, considering that these enteric viruses are found in low concentrations in natural water environments and also the possibility of PCR inhibition which could have led to false negative results being obtained.
Exposure to a minimal volume of 10 mℓ Tyume River water would lead to a 1:2 000 risk of infection from RVs which is 5 times more than the acceptable risk level of 1:10 000. Still, this assumes accidental consumption in a domestic set-up as opposed to complete reliance on such water for all domestic uses including drinking. Rotaviruses are the leading cause of gastrointestinal morbidity and mortality among young children and are of much greater public health concern to young children and immunocompromised persons and populations than the general population (USEPA, 2010). Hepatitis A virus presented significantly higher risk of infection figures compared to RVs in the case of ingestion of 10 m£ of river water. The risk of infection with HAV in faecally-polluted water has been found to increase with increased immersion in contaminated water (Gammie and Wyn-Jones, 1997; Taylor et al., 1995). This implies that water from Tyume River may not be suitable for full-contact recreational activities. This risk is significantly higher in children under 10 years of age as well as in immunocompromised individuals (Venter et al., 2007). This risk of infection calculation presents a number of uncertainties: The volume of water that was used to assess the fitness-for-use of the water for domestic purposes is far below what an individual human being could consume per day. Even then, the amount of water that samples of individuals consume per day may differ between individuals depending on their levels of physical activity and the state of their health. In this regard, if a volume larger than 10 mℓ were used, the estimated risk could only be larger. However, since environmental samples usually yield a much higher ratio of infectious viruses to total PCR detectable viruses than those viruses that have been adapted to cell culture (Reynolds et al., 1996), the calculated risk of infection values in this study could still be an overestimation of the actual risk. A combination of characteristics cause human enteric viruses to be of great public health concern and these are: increased stability in the environment when compared with faecal bacteria (Fujioka and Yoneyama, 2002), resistance to some water treatment processes (WHO, 2008) and ability to cause infection at low doses (Fong and Lipp, 2005). Jiang et al. (2001) established a link between urban stormwater run-off and the virological contamination of receiving waters making it needful to study the virological water quality of streams and rivers, especially those relied upon as source waters for drinking and recreation. Since the detection of NoVs and HAV on the basis of infectivity is complicated by the absence of reliable cell culture methods (Blaise-Boisseau et al., 2010), this real-time PCR detection of these and other viruses in a river of strategic importance to its host community, as Tyume River is, is enough to constitute a public health risk. Because access to potable water in some pockets of the population within the Eastern Cape Province remains a pipe-dream (Momba et al., 2006b) the findings of this study point to a possible public health concern which needs urgent interventions from the responsible authorities.
Conclusion
Even though the proportion of infective viruses was only estimated in this study, the fact remains that there is considerable risk of infection posed by the use of raw surface water for either domestic or recreational purposes. The findings of this study also suggest that the presence of enteric viruses in the environmental surface waters is related less to general faecal pollution than it is to the presence of infected individuals in the host population. Future research work in this field may include cell culture to verify the proportion of infectious viruses to total virus particles in environmental water samples. Questionnaire surveys may also be conducted in communities within river catchments in the wider Eastern Cape Province so that risk assessment profiling is aligned to water-use patterns specific for communities in those catchments.
Acknowledgements
We are grateful to the Water Research Commission (WRC) of South Africa for funding this research (WRC Project No. K5/1968).
References
ABBASZADEGAN M (2001) Advanced detection of viruses and protozoan parasites in water. Rev. Biol. Biotechnol. 1 (2) 21-26. [ Links ]
ADHAMI T and CAREY WD (2010) Disease management project. The Cleveland Clinic Foundation: Centre for Continuing Education. URL: http://www.clevelandclinicmeded.com/medicalpubs/disease-management/hepatology/viral-hepatitis/ (Accessed 4 June 2012). [ Links ]
ADLHOCH C, KAISER M, HOEHNE M, MARQUES AM, STEFAS I, VEAS F and ELLERBROK H (2011) Highly sensitive detection of the group A Rotaviruses using Apolipoprotein H-coated ELISA plates compared to quantitative real-time PCR. Virol. J. 8 (63) 1-7. [ Links ]
APHA (AMERICAN PUBLIC HEALTH ASSOCIATION) (2005) Standard Methods for the Examination of Water and Wastewater (20th edn.) American Public Health Association (APHA), Washington DC, USA. [ Links ]
ARCHIMBAUD C, MIRAND A, CHAMBON M, REGAGNON C, BAILLY J-L, PEIGUE-LAFEUILLE H and HENQUELL C (2004) Improved diagnosis on a daily basis of enterovirus meningitis using a one-step real-time RT-PCR assay. J Med. Virol. 74 604-611. [ Links ]
ARNONE RD and WALLING JP (2007) Waterborne pathogens in urban watersheds. J. Water Health 5 149-162. [ Links ] AW TG and GIN KY-H (2010) Environmental surveillance and molecular characterization of human enteric viruses in tropical urban wastewaters. J. Appl. Microbiol. 109 716-730. [ Links ]
AW TG, GIN KY, OON LLE, CHEN EX and WOO CH (2009) Prevalence and genotypes of human noroviruses in tropical urban surface waters and clinical samples in Singapore. Appl. Environ. Microbiol. 75 4984-4992. [ Links ]
BERGE JJ, DRENNAN DP, JACOBS RJ, JAKINS A, MEYERHOFF AS and STUBBLEFIELD W (2000) The cost of hepatitis A infections in American adolescents and adults in 1997. Hepatology 31 469-473. [ Links ]
BISHOP RF, DAVIDSON GP, HOLMES IH and RUCK BJ (1973) Virus particles in epithelial cells of duodenal mucosa from children with acute nonbacterial gastroenteritis. Lancet 2 1281-1283. [ Links ]
BLAISE-BOISSEAU S, HENNECHART-COLLETTE C, GUILLIER L and PERELLE S (2010) Duplex real-time qRT-PCR for the detection of hepatitis A virus in water and raspberries using the MS2 bacteriophage as a process control. J. Virol. Methods 166 48-53. [ Links ]
BOFILL-MAS S, CALGUA B, CLEMENTE-CASARES P, LA ROSA G, LACONELLI M, MUSCILLO M, RUTJES S, DE RODA HUSMAN AM, GRUNERT A, GRÀBER I, VERANI M, CARDUCCI A, CALVO M, WYN-JONES P and GIRONES R (2010) Quantification of human adenoviruses in European recreational waters. Food Environ. Virol. 2 101-109. [ Links ]
BOOM R, SOL CJA, SALIMANS MMM, JANSEN CL, WERTHEIN-VAN DILLEN PME and NOORDAA J (1990) Rapid simple method for purification of nucleic acids. J. Clin. Microbiol. 28 495-503. [ Links ]
BOSCH A (1998) Human enteric viruses in the water environment: a minireview. Int. Microbiol. 1 191-196. [ Links ]
BOSCH A, GUIX S, SANO D and PINTO RM (2008) New tools for the study and direct surveillance of viral pathogens in water. Curr. Opin. Biotechnol. 19 1-7. [ Links ]
BOXMAN IL, TILBURG JJ, TE LOEKE NA, VENNEMA H, JONKER K, DE BOER E and KOOPMANS M (2006) Detection of noro- viruses in shellfish in The Netherlands. Int. J. Food Microbiol. 108 391-396. [ Links ]
BRASSARD J, SEYER K, HOUDE A, SIMARD C and TROTTIER Y-L (2005) Concentration and detection of hepatitis A virus and rotaviruses in spring water samples by reverse transcription-PCR. J. Virol. Methods 123 163-169. [ Links ]
BROOKS HA, GERSBERG RM and DHAR AK (2005) Detection and quantification of hepatitis A virus in seawater via real-time RT-PCR. J. Virol. Methods 127 109-118. [ Links ]
BUSTIN SA and MUELLER R (2005) Real-time reverse transcription PCR (qRT-PCR) and its potential use in clinical diagnosis. Clin. Sci. 109 365-379.doi:10.1042/CS20050086 [ Links ]
CABALLERO S, ABAD FX, LOISY F, LE GUYADER FS, COHEN J, PINTO RM and BOSCH A (2004) Rotaviruses virus-like particles as surrogates in environmental persistence and inactivation studies. Appl. Environ. Microbiol. 70 3904-3909. [ Links ]
CDC (CENTERS FOR DISEASE CONTROL AND PREVENTION) (1999) Prevention of hepatitis A through active and passive immunisation: recommendations of the Advisory Committee on Immunisation Practices (ACIP). Morb. Mortal. Wkly. Rep. (MMWR) 48 1-34. [ Links ]
CONNELL C, TONG H-I, WANG Z, ALLMANN E and LU Y (2012) New approaches for enhanced detection of enteroviruses from Hawaiian environmental waters. PLoS ONE 7 (5) e32442. DOI:10.1371/journal.pone.0032442. [ Links ]
COOK SM, GLASS RI, LEBARON CW and HO MS (1990) Global seasonality of rotaviruses infections. Bull. WHO 68 171-177. [ Links ]
COSTAFREDA MI, BOSCH A and PINTO RM (2006) Development, evaluation, and standardization of a real-time TaqMan reverse transcription-PCR assay for quantification of hepatitis A virus in clinical and shellfish samples. Appl. Environ. Microbiol. 72 3846-3855. [ Links ]
DE PAULA VS, DINIZ-MENDES L, VILLAR LM, LUZ SLB, SILVA LA, JESUS MS, DA SILVA MNVSS and GASPAR AS (2007) Hepatitis A in environmental water samples from the Amazon Basin. Water Res. 41 1169-1176. [ Links ]
DENG MY, DAY SP and CLIVER DO (1994) Detection of hepatitis A virus in environmental samples by antigen-capture PCR. Appl. Environ. Microbiol. 60 1927-1933. [ Links ]
DIERSSEN U, REHREN F, HENKE-GENDO C, HARSTE G and HEIM A (2008) Rapid routine detection of enterovirus RNA in cerebrospinal fluid by a one-step real-time RT-PCR assay. J. Clin. Virol. 42 58-64. [ Links ]
DONALDSON KA, GRIFFIN DW and PAUL JH (2002) Detection, quantitation and identification of enteroviruses from surface waters and sponge tissue from the Florida Keys using real-time RT- PCR. Water Res. 36 2505-2514. [ Links ]
ESPINOSA AC, MAZARI-HIRIART M, ESPINOSA R, MARURI-AVIDAL L, ME'NDEZ E and ARIAS CF (2008) Infectivity and genome persistance of rotaviruses and astrovirus in groundwater and surface water. Water Res. 42 2618-2628. [ Links ]
FEINSTONE SM (1996) Hepatitis A: Epidemiology and prevention. Eur. J. Gastroenterol. Hepatol. 8 300-305. [ Links ]
FISCHER TK, STEINSLAND H and VALENTINER-BRANTH P (2002) Rotaviruses particles can survive storage in tropical temperatures for more than 2 months. J. Clin. Microbiol. 40 4763-4764. [ Links ]
FONG TT and LIPP EK (2005) Enteric viruses of humans and animals in aquatic environments: health risks, detection, and potential water quality assessment tools. Microbiol. Mol. Biol. Rev. 69 357-371. [ Links ]
FREEMAN MM, KERIN T, HULL J, McCAUSTLAND K and GENTSCH J (2008) Enhancement of detection and quantification of rotaviruses in stool using a modified real-time RT-PCR assay. J. Med. Virol. 80 1489-1496. [ Links ]
FRITZINGER AE, WALTERS CC, KELLY ST and TONEY DM (2011) Viral gastroenteritis: Pathogenesis and laboratory detection and characterization in the Commonwealth of Virginia. Clin. Microbiol. Newsletter 33 (4) 1-7. [ Links ]
FUJIOKA RS and YONEYAMA BS (2002) Sunlight inactivation of human enteric viruses and faecal bacteria. Water Sci. Technol. 46 291-295. [ Links ]
GAMMIE AJ and WYN-JONES AP (1997) Does hepatitis A pose a significant health risk to recreational water users? Water Res. Technol. 35 55-68. [ Links ]
GENTRY J, VINJE J, GUADAGNOLI D and LIPP EK (2009) Noroviruses distribution within an estuarine environment. Appl. Environ. Microbiol. 75 5474-5480. [ Links ]
GIBSON KE, OPRYSZKO MC, SCHISSLER JT, GUO Y and SCHWAB KJ (2011) Evaluation of human enteric viruses in surface water and drinking water resources in southern Ghana. Am. J. Trop. Med. Hyg. 84 (1) 20-29. [ Links ]
GILGEN M, WEGMÜLLER B, BURKHALTER P, BÜHLER H-P, MÜLLER U, LÜTHY J and CANDRIAN U (1995) Reverse transcription PCR to detect enteroviruses in surface water. Appl. Environ. Microbiol. 61 (4) 1226-1231. [ Links ]
GOULD EA (1999) Methods for long-term virus preservation. Mol. Biotechnol. 13 (1) 57-66. DOI: 10.1385/MB:13:1:57. [ Links ]
GRABOW WOK (1997) Hepatitis viruses in water: Update on risk and control. Water SA 23 379-386. [ Links ]
GRABOW WOK, PUTTERGILL DL and BOSCH A (1992) Propagation of adenovirus types 40 and 41 in the PLC/PRF/5 primary liver carcinoma cell line. J. Virol. Methods 37 201-207. [ Links ]
GREGORY JB, WAYNE LT and NOBLE RT (2006) Rapid one-step quantitative reverse transcriptase PCR assay with competitive internal positive control for detection of enteroviruses in environmental samples. Appl. Environ. Microbiol. 72 3960-3967. [ Links ]
GUNSON RN, MILLER J, LEONARD A and CARMAN WF (2003) Importance of PCR in the diagnosis and understanding of rota-viruses illness in the community. Commun. Dis. Public Health 6 63-65. [ Links ]
HAAS CN (1996) How to average microbial densities to characterise risk. Water Res. 30 (4) 1036-1038. [ Links ]
HAAS CN, ROSE JB and GERBA CP (1999) Quantitative Microbial Risk Assessment. Wiley, New York. [ Links ]
HAAS CN, ROSE JB, GERBA C and REGLI S (1993) Risk assessment of virus in drinking water. Risk Anal. 13 (5) 545-552. [ Links ]
HAMZA IA, JURZIK L, STANG A, SURE K, ÜBERLA K and WILHELM M (2009) Detection of human viruses in rivers of a densely-populated area in Germany using a virus adsorption elution method optimized for PCR analyses. Water Res. 43 2657-2668. [ Links ]
HARAMOTO E, KATAYAMA H, OGUMA K and OHGAKI S (2005) Application of cation-coated filter method to detection of noro-viruses, enteroviruses, adenoviruses, and Torque teno viruses in the Tamagawa River in Japan. Appl. Environ. Microbiol. 71 (5) 2403-2411. [ Links ]
HARAMOTO E, KATAYAMA H, UTAGAWA E and OHGAKI S (2008) Development of sample storage methods for detecting enteric viruses in environmental water. J. Virol. Methods 151 1-6. [ Links ]
HEJKAL TW, SMITH EM and GERBA CP (1984) Seasonal occurrence of rotaviruses in sewage. Appl. Environ. Microbiol. 47 588-590. [ Links ]
HONG J, KANG B, KIM A, HWANG S, AHN J, LEE S, KIM J, PARK J-K and CHEON D-S (2011) Development of a highly sensitive realtime one step RT-PCR combined complementary locked primer technology and conjugated minor groove binder probe. Virol. J. 8 330. [ Links ]
HONG J, KANG B, KIM A, HWANG S, LEE S, KIM J, LEE H-Y, KANG S-H and CHEON D-S (2010) Enhanced detection of enteroviruses in clinical samples by reverse transcription-PCR using complementary locked primer technology. J. Clin. Microbiol. 48 (2) 615-616. [ Links ]
HUNTER PR (1997) Viral hepatitis. In: HUNTER PR (ed.) Waterborne Disease: Epidemiology and Ecology. John Wiley & Sons Ltd., Chichester. 207-221. [ Links ]
JIANG S, NOBLE R and CHUI WP (2001) Human adenoviruses and coliphages in urban runoff-impacted coastal waters of Southern California. Appl. Environ. Microbiol. 67 179-184. [ Links ]
JOTHIKUMAR N, KANG G and HILL VR (2009) Broadly reactive TaqMan® assay for real-time RT-PCR detection of rotaviruses in clinical and environmental samples. J. Virol. Methods 155 126-131. [ Links ]
KANG G, ITURRIZA-GOMARA M, WHEELER JG, CRYSTAL P, MONICA B, RAMANI S, PRIMROSE B, MOSES PD, GALLI-MORE CI, BROWN DW and GRAY J (2004) Quantitation of Group A rotaviruses by real-time reverse-transcription-polymerase chain reaction: Correlation with clinical severity in children in South India. J. Med. Virol. 73 118-122. [ Links ]
KATAYAMA H, SHIMASAKI A and OHGAKI S (2002) Development of a virus concentration method and its application to detection of enterovirus and Norwalk virus from coastal seawater. Appl. Environ. Microbiol. 68 1033-1039. [ Links ]
KITTIGUL L, EKCHALOEMKIET S, UTRARACHKIJ F, SIRIPA-NICHGON K, SUJIRARAT D, PUNGCHITTON S and BOON-THUM A (2005a) An efficient virus concentration method and RT-nested PCR for detection of rotaviruses in environmental water samples. J. Virol. Methods 124 117-122. [ Links ]
KITTIGUL L, UTHAISIN A, EKCHALOEMKIET S, UTRARACHKIJ F and LUKSAMIJARULKUL P (2005b) Detection and characterization of hepatitis A virus in water samples in Thailand. J. Appl. Microbiol. 100 1318-1323. [ Links ]
KIULIA NM, NETSHIKWETA R, VAN ZYL WB, KIRAITHE MM, NYACHIEO A, MWENDA JM and TAYLOR MB (2010) The detection of enteric viruses in selected urban and rural river water and sewage in Kenya, with special reference to rotaviruses. J. Appl. Microbiol. DOI:10.1111/j.1365-2672.2010.04710.x [ Links ]
KOFF RS (1995) Preventing hepatitis A infections in travellers to endemic areas. Am. J. Trop. Med. Hyg. 53 586-590. [ Links ]
KOOPMANS M, VON BONSDORFF C, VINJE' J, DE MEDICI D and MONROE S (2002) Foodborne viruses. FEMS Microbiol. Rev. 26 187-205. [ Links ]
LEE C and KIM S-J (2008) The genetic diversity of human noroviruses detected in river water in Korea Water Res. 4 (2) 4477-4484. [ Links ]
LI J-W, WANG X-W, YUAN C-Q, ZHENG J-L, JIN M, SONG N, SHI X-Q and CHAO F-H (2002) Detection of enteroviruses and hepatitis a virus in water by consensus primer multiplex RT-PCR. World J. Gastroenterol. 8 (4) 699-702. [ Links ]
LODDER WJ and DE RODA HUSMAN AMD (2005) Presence of noroviruses and other enteric viruses in sewage and surface waters in The Netherlands. Appl. Environ. Microbiol. 71 1453-1461. [ Links ]
LOGAN C, O'LEARY JJ and O'SULLIVAN N (2006) Real-time reverse transcription-PCR for detection of rotaviruses and adenovirus as causative agents of acute viral gastroenteritis in children. J. Clin. Microbiol. 44 (9) 3189-3195. [ Links ]
LYSEN M, THORHAGEN M, BRYTTING M, HJERTQVIST M, ANDERSSON Y and HEDLUND K-O (2009) Genetic diversity among food-borne and waterborne noroviruses strains causing outbreaks in Sweden. J. Clin. Microbiol. 47 2411-2418. [ Links ]
MANS JC, DE VILLIERS J, DU PLESSIS NM, AVENANT T and TAYLOR MB (2010). Emerging noroviruses GII.4 2008 variant detected in hospitalised paediatric patients in South Africa. J. Clin. Virol. 49 258-264. [ Links ]
MANS J, NETSHIKWETA R, MAGWALIVHA M, VAN ZYL WB and TAYLOR MB (2013) Diverse norovirus genotypes identified in sewage-polluted river water in South Africa. Epidemiol. Infect. 141 (2) 303-313. [ Links ]
MARA DD (2000) The production of microbiologically safe effluents for wastewater reuse in the Middle East and North Africa. Water Air Soil Pollut. 123 595-603. [ Links ]
MEHNERT DU and STEWIEN KE (1993) Detection and distribution of rotaviruses in raw sewage and creeks in São Paulo, Brazil. Appl. Environ. Microbiol. 59 140-143. [ Links ]
MERRILL DR, WADE CD, FAHNESTOCK P and BAKER RO (2012) Long-term and short-term stability of viruses depend on storage temperature and preservation methods. URL: http://www.beiresources.org/Portals/2/PDFS/Long-Term%20and%20Short-Term%20Stability%20of%20Viruses.pdf (Accessed 24 February 2012). [ Links ]
MOMBA MNB, OSODE AN and SIBEWU M (2006a) The impact of inadequate wastewater treatment on the receiving water bodies - Case study: Buffalo City and Nkonkobe Municipalities of the Eastern Cape Province. Water SA 32 (5) 687-692. [ Links ]
MOMBA MNB, TYAFA Z, MAKALA N, BROUCKAERT MB and OBI CL (2006b) Safe drinking water still a dream in rural areas of South Africa. Case Study: The Eastern Cape Province. Water SA 32 (5) 715-720. [ Links ]
MURRAY PR, ROSENTHAL EJ and PFALLER MA (2005) Medical Microbiology Vol. 2. (5th edn.). Elsevier Inc., Philadelphia U.S.A. [ Links ]
PANG XL, LEE B, BOROUMAND N, LEBLANC B, PREIKSAITIS JK and IP CCY (2004) Increased detection of rotaviruses using a real time reverse transcription-polymerase chain reaction (RT-PCR) assay in stool specimens from children with diarrhea. J. Med. Virol. 72 496-501. [ Links ]
PARASHAR UD, GIBSON CJ, BRESEE JS and GLASS RI (2006) Rotaviruses and severe childhood diarrhea. Emerg. Infect. Dis. 12 304-306. [ Links ]
PETER-VARBANETS M, ZURBRÜGG C, SWARTZ C and PRONK W (2009) Decentralized systems for potable water and the potential of membrane technology. Water Res. 43 245-265. [ Links ]
PINTO RM, COSTAFREDA MI and BOSCH A (2009) Risk assessment in shellfish-borne outbreaks of hepatitis A. J. Appl. Microbiol. 75 7350-7355. [ Links ]
REYNOLDS KA, GERBA CP and PEPPER IL (1996) Detection of infectious enteroviruses by an integrated cell culture-PCR procedure. Appl. Environ. Microbiol. 62 1424-1427. [ Links ]
RODRIGUEZ RA, PEPPER IL and GERBA CP (2009) Application of PCR-based methods to assess the infectivity of enteric viruses in environmental samples. Appl. Environ. Microbiol. 75 (2) 297-307. [ Links ]
ROSE JB and MOLLOY SL (2007) Globalization effects on water quality: monitoring the impact on and control of waterborne disease. In: Taylor WW, Schechter MG, Wolfson LG (eds.) Globalization: Effects on Fisheries Resources. Cambridge University Press, New York. 92-119. [ Links ]
RUTJES SA, ITALIAANDER R, VAN DEN BERG HHJL, LODDER WJ and DE RODA DE RODA HUSMAN AM (2005) Isolation and detection of enterovirus RNA from large-volume water samples by using the NucliSens miniMAG system and real-time nucleic acid sequence-based amplification. Appl. Environ. Microbiol. 71 (7) 3734-3740. [ Links ]
RUTJES SA, LODDER WJ, VAN LEEUWEN AD and DE RODA DE RODA HUSMAN AM (2009) Detection of infectious rotaviruses in naturally contaminated source waters for drinking water production. J. Appl. Microbiol. 107 97-105. DOI:10.1111/j.1365-2672.2009.04184.x. [ Links ]
SCHUTTEN M and NIESTERS HG (2001) Clinical utility of viral quantification as a tool for disease monitoring. Expert. Rev. Mol. Diagn. 1 153-162. [ Links ]
SHIN GA and SOBSEY MD (2008) Inactivation of noroviruses by chlorine disinfection of water. Water Res. 42 4562-4568. [ Links ]
SIEBENGA JJ, VENNEMA H, ZHENG DP, VINJÉ J, LEE BE, PANG XL, HO EC, LIM W, CHOUDEKAR A, BROOR S, HALPERIN T, RASOOL NB, HEWITT J, GREENING GE, JIN M, DUAN ZJ, LUCERO Y, O'RYAN M, HOEHNE M, SCHREIER E, RATCLIFF, RM, WHITE PA, IRITANI N, REUTER G and KOOPMANS M (2009) Noroviruses illness is a global problem: emergence and spread of noroviruses GII.4 variants, 2001-2007. J. Infect. Dis. 200 802-812. [ Links ]
SOULE H, GENOULAZ O, GRATACAP-CAVALLIER B, MALLARET MR, MORAND P, FRANCOIS P, LUU DUC BIN D, CHARVSIER A, BOST-BRU C and SEIGNEURIN JM (1999) Monitoring rota-viruses environmental contamination in a pediatric unit using polymerase chain reaction. Infect. Control Hosp. Epidemiol. 20 (6) 432-434. [ Links ]
STEYN M, JAGALS P and GENTHE B (2004) Assessment of microbial infection risks posed ingestion of water during domestic water use and full-contact recreation in a mid-southern African region. Water Sci. Technol. 50 301-308. [ Links ]
TAYLOR MB, BECKER PJ, JANSE VAN RENSBURG E, HARRIS BN, BAILEY IW and GRABOW WOK (1995) A serosurvey of water-borne pathogens amongst canoeists in South Africa. Epidemiol. Infect. 115 229-307. [ Links ]
TAYLOR MB, COX N, VREY MA and GRABOW WOK (2001) The occurrence of hepatitis A and astrovirus in selected river and dam waters in South Africa. Water Res. 35 2653-2660. [ Links ]
TONG MJ, EL-FARRA NS and GREW MI (1995) Clinical manifestations of hepatitis A: Recent experience in a community teaching hospital. J. Infect. Dis. 171 (1) S15-S18. [ Links ]
TSAI YL, TRAN B and PALMER CJ (1995) Analysis of viral RNA persistence in seawater by reverse transcriptase-PCR. Appl. Environ. Microbiol. 61 (1) 363-366. [ Links ]
USEPA (2010) Quantitative microbial risk assessment to estimate illness in freshwater impacted by agricultural animal sources of faecal contamination. U.S. Environmental Protection Agency Office of Water, December 2010. EPA 822-R-10-005.USEPA, Washington. 456 pp. [ Links ]
VAN ZYL WB, PAGE NA, GRABOW WOK, STEELE AD and TAYLOR MB (2006) Molecular epidemiology of group A rota-viruses in water sources and selected raw vegetables in Southern Africa. Appl. Environ. Microbiol. 72 4554-4560. [ Links ]
VENTER JME, VAN HEERDEN J, VIVIER JC, GRABOW WOK and TAYLOR MB (2007) Hepatitis A virus in surface water in South Africa: what are the risks? J. Water Health 5 (2) 229-240. [ Links ]
VICTORIA M, GUIMARÃES FR, FUMIAN TM, FERREIRA FFM, VIEIRA CB, SHUBO T, LEITE JPG and MIAGOSTOVICH MP (2010) One year monitoring of noroviruses in a sewage treatment plant in Rio de Janeiro, Brazil. J. Water Health 8 (1) 158-165. DOI:10.2166/wh.2009.012. [ Links ]
VILLAR LM, DE PAULA VS, DINIZ-MENDES L, LAMPE E and GASPAR AMC (2006) Evaluation of methods used to concentrate and detect hepatitis A virus in water samples. J. Virol. Methods 137 169-176. [ Links ]
VIVIER JC, EHLERS MM and GRABOW WOK (2004) Detection of enteroviruses in treated drinking water. Water Res. 38 2699-2705. [ Links ]
WARD RL, KNOWLTON DR and PIERCE MJ (1984) Efficiency of human rotaviruses propagation in cell culture. J. Clin. Microbiol. 19 748-753. [ Links ]
WHO (2001) Water Quality: Guidelines, Standards and Health. IWA Publishing, London, UK. [ Links ]
WHO (2003) Immunizations, vaccines and biological: hepatitis A vaccine. URL: http://www.who.int/vaccines/en/hepatitisa.shtml (Accessed 4 June 2012). [ Links ]
WHO (2008) Microbial aspects. In: Guidelines for Drinking water Quality (3rd edn.). World Health Organization, Geneva. 121-144. [ Links ]
WOLF S, HEWITT J and GREENING GE (2010) Viral multiplex quantitative PCR assays for tracking sources of faecal contamination. Appl. Environ. Microbiol. 76 1388-1394. [ Links ]
WHO and UNICEF (2010) Progress on Sanitation and Drinking Water: 2010 Update. URL: http://www.who.int/watersanitation health/publications/9789241563956/en/index.html (Accessed 1 June 2012). [ Links ]
YANG W, GU AZ, ZENG S-Y, LI D, HE M and SHI H-C (2011) Development of a combined immunomagnetic separation and quantitative reverse transcription-PCR assay for sensitive detection of infectious rotaviruses in water samples. J. Microbiol. Methods 84 447-453. [ Links ]
ZHI-YI X, ZI-HUA L, JIAN-XIANG W, ZAI-PING X and DE-XIANG D (1992) Ecology and prevention of a shellfish-associated hepatitis A epidemic in Shanghai, China. Vaccine 10 (1) S67-S68. [ Links ]
Received 9 July 2012
Accepted in revised form 6 March 2013.
* To whom all correspondence should be addressed. +27 40 602-2365; e-mail: aokoh@ufh.ac.za














