Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
Water SA
versión On-line ISSN 1816-7950
versión impresa ISSN 0378-4738
Water SA vol.38 no.4 Pretoria ene. 2012
Nitrate-induced changes and effect of varying total nitrogen to total phosphorus ratio on the phytoplankton community in Lake Chivero, Zimbabwe: microcosm experiments
Lindah MhlangaI, *; Wilson MhlangaII
IUniversity of Zimbabwe, Department of Biological Sciences, PO Box MP 167, Mt. Pleasant, Harare, Zimbabwe
IIBindura University of Science Education, Department of Environmental Science, Private Bag 1020, Bindura, Zimbabwe
ABSTRACT
Microcosm experiments were conducted to assess nitrate-induced changes and the effect of varying the ratio of total nitrogen to total phosphorus (TN:TP) on the winter and late summer phytoplankton communities in Lake Chivero, Zimbabwe. In both winter and summer, nitrate addition altered species composition from a cryptophyte-dominated to a chlorophytedominated assemblage and increased chlorophyll a concentration and total algal biomass. The shift in algal dominance and increase in chlorophyll a concentration and total algal biomass also occurred in the control showing that isolation from allogenic processes like turbulence had an effect on phytoplankton structure. Microcystis aeruginosa, a common cyanobacterium in Lake Chivero, did not assume dominance in any of the treatments. From an application perspective it is interesting that varying nitrate loadings in microcosms favoured chlorophytes rather than increasing undesirable cyanobacteria. Nutrient manipulation can be used as a management option to avoid cyanobacteria occurrence, by maintaining either cryptophytes or chlorophytes. However, the practicality of such an intervention needs to be assessed.
Keywords: chlorophytes, cryptophytes, Lake Chivero, microcosms, phytoplankton, TN:TP ratio
Introduction
Lake Chivero, Zimbabwe, is a man-made reservoir that was created in 1956 and is located approximately 40 km southwest of the city of Harare. It is the city's primary water supply reservoir. The lake is hyper-eutrophic (Magadza, 2003), owing to the discharge of nutrient-rich sewage and industrial effluent from Harare and satellite towns into its tributary streams. This has led to a predominance of cyanobacteria (Munro, 1966; Marshall, 1997), the detrimental effects of which have included gastroenteritis outbreaks in children correlated with decay of Microcystis aeruginosa blooms (Zilberg, 1966; Marshall, 1991), periodic fish kills due to depletion of oxygen in the hypolimnion (Moyo, 1997; Mhlanga et al., 2006a), transient influenza-like reactions upon inhalation of aerosols from tap water linked to high endotoxin levels (Annadotter et al., 2005) and objectionable tastes and odours in the drinking water (Marshall, 1997).
Despite the ecological and potential health challenges posed by the predominance of cyanobacteria there has been limited work done in Lake Chivero on phytoplankton ecology, with the exception of the unpublished work by Falconer (1973). Development of effective lake and watershed management strategies for Lake Chivero will be determined by assessing which nutrient limits phytoplankton growth, especially the predominance of cyanobacteria. Phosphorus has been considered to be the primary nutrient limiting phytoplankton growth in freshwater ecosystems and management efforts have focused on controlling phosphorus loading (Dzialowski et al., 2005).
Lake Chivero, which receives high loadings of nitrogen and phosphorus (Nhapi, 2004), has a relatively low total nitrogen to total phosphorus (TN:TP) ratio. This is attributed to the low TN:TP ratio of sewage effluent (Robarts, 1981), which is the main source of nutrients in the lake. The nutrient-loading ratio of the sewage effluent in Lake Chivero is in the order of 1:2 TN:TP and has produced a system that is nitrogen-limited (Robarts, 1981). It has been observed that nitrogen limitation occurs more commonly than previously thought (Dzialowski et al., 2005). Using bioassay cultures with Selenastrum capricornutum, Robarts and Southall (1975; 1977) established that nitrogen was potentially the primary growth-limiting nutrient in Lake Chivero.
This study was conducted to determine how varying nitrate loadings would influence phytoplankton species composition and biomass in Lake Chivero. It is of management interest to be able to maintain optimal TN:TP ratios that can support socially-and ecologically-preferable chlorophytes, baccillariophytes and cryptophytes rather than undesirable cyanobacteria. The objectives of this study were:
to determine the effect of increasing nitrate concentration and the associated increase of TN:TP ratio on phytoplankton species composition, biomass, succession, and dominance patterns; and
to determine species-specific differences in phytoplankton responses to the changes of TN:TP ratio in microcosms and compare these to changes in the lake.
Materials and methods
Microcosm experiments
Two in-situ microcosm experiments of 9 days duration (Experiment 1: end of winter, August 2005 and Experiment 2: end of the rainy season, April 2006) were run in a small sheltered bay where the depth was about 3 m. There were 5 treatments each replicated 12 times. Water from the lake (surface to 0.5 m) with natural populations of phytoplankton was collected into five 25-litre plastic buckets. The first bucket was used as a control with no treatment added. The other 4 buckets were spiked with nitrate (NaNO3) at 4 different concentrations. The ambient nitrate concentration was increased 1, 20, 50 and 100 times in treatments designated as 1NP, 20NP, 50NP and 100NP respectively. The ambient phosphate (K2HPO4) concentration was maintained in each treatment. In the control, no nutrient adjustments were made. The spiked water was thoroughly mixed with a stirrer. From each bucket a sample of 1 l was collected for analysis of physical and chemical variables and phytoplankton. The remaining water in each respective treatment was then put in twelve 2-1 cylindrical polyethylene bottles, making a total of 60 microcosms. The bottles were filled to 1.5 l, closed at the top with caps and then suspended on a floater in the lake and incubated for 10 days. The floater enabled the bottles to be suspended in water which avoided direct exposure to ambient solar radiation at the lake surface thereby limiting the effect of photo-inhibition and possible ultraviolet damage to phytoplankton. Photo-inhibition, however, could not be totally excluded because it is known to occur in in-situ incubated bottles in shallow waters during warm seasons (Goldman and Dennett, 1984). The natural zooplankton community was included in all treatments.
Sampling and sample analysis
Nutrient concentrations in the microcosms were measured at 2-day intervals. Three replicate bottles from each series were removed every other day for analysis. Sampling of surface water in the lake was also carried out every other day. Nutrients were measured by the methods described by Golterman, Clymo and Ohnstad (1978) and chlorophyll a (ng-l-1) by the acetone extraction method (Golterman et al., 1978).
After collection, phytoplankton samples were immediately preserved in Lugol's solution. Cell counts were made with an inverted microscope according to Utermõhl (1958) and Cronberg (1982). Algae were enumerated until approximately 100 cells of the dominant species were recorded, giving a precision of ±20% within a 95% confidence limit. The bio-volume of each species was calculated from linear dimensions and corresponding formulae (Rott, 1981), and used to estimate algal biomass as wet weight.
Statistical analysis
Within each experiment, differences among treatments in nutrient concentrations and phytoplankton biomass were tested using Repeated Measures Analysis of Variance (RM-ANOVA) for the 9 sampling days. For each experimental run a separate RM-ANOVA was conducted using the program Statistica 7.
Results
The average water temperature in the lake was similar to that in microcosms during the first experiment (F = 2.0;p > 0.05) being 19.8ºC and 23ºC at the beginning and end of the experiment, respectively. The average water temperature was significantly different between the lake and microcosms during the second experiment (F = 4.3;p < 0.05). The average temperature in the lake was 21.6ºC and 26.7ºC at the beginning and end of the experiment, respectively, while in microcosms the average temperature was 22.4ºC at the beginning and 28ºC at the end of the experiment.
After spiking, nitrate and total nitrogen concentrations in treatments ranged as follows: NP100 > NP50 > NP20, while the concentrations in the lake, control and NP1 were similar. Nitrate concentration was significantly different between NP100, NP50 and NP20 during either experiment (Experiment 1: F = 107.6;p < 0.05; Experiment 2: F = 241.4;p < 0.05) but was not significantly different between the lake, control and NP1 during either experiment. Nitrate levels remained high throughout the experimental period during both experiments. Total nitrogen concentration was significantly different between NP100, NP50 and NP20 during either experiment (Experiment 1: F = 50.8;p < 0.05; Experiment 2: F = 1792.2; p < 0.05) but was not significantly different between the lake, control and NP1 during either experiment.
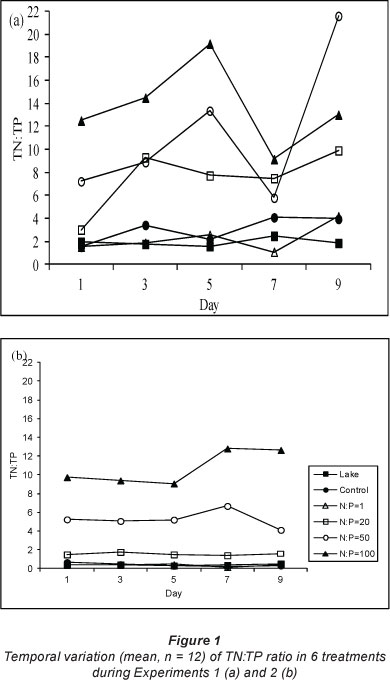
During Experiment 1, orthophosphate was depleted in all treatments by Day 9 whereas, although concentrations decreased, orthosphosphate was not depleted during Experiment 2. During Experiment 1, lake orthophosphate concentration ranged between 0.9 and 1.2 mg-L1 while the average concentration in microcosms at the beginning of the experiment was 1.3 mg-L1 and had declined to 0.01 mg-L1 by Day 9. Lake orthophosphate concentration ranged between 2.8 and 3.3 mg-l-1 during Experiment 2 while average concentrations in microcosms were 4.08 mg-l-1 and 2.5 mg-l-1 at the beginning and end of the experiment, respectively. There were significant differences in temporal changes in orthophosphate concentrations within treatments during both experiments (Experiment 1: F = 4.6; p < 0.05; Experiment 2: F = 3.1;p < 0.05). There were no significant differences in temporal changes within treatments for total phosphorus during Experiment 1 (F = 0.9; p > 0.05), while during Experiment 2 differences in temporal changes within treatments were significant (Fig. 1) (F = 12.5; p < 0.05). As with nitrate and total nitrogen the lake TN:TP ratio was similar to the control and NP1 but significantly different from other treatments (Fig. 1) (Experiment 1: F = 32.5; p < 0.05; Experiment 2: F = 214.6; p < 0.05).
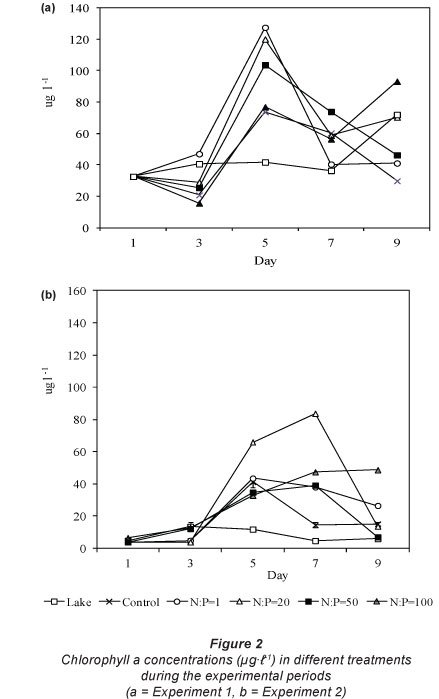
Chlorophyll a concentration was similar in all treatments at the beginning of both experiments. It declined with rising TN:TP on Days 3 and 5 (Fig. 2). During Experiment 1 the concentrations at Day 5 were NP1 > NP20 > NP50 > NP100. Chlorophyll a concentration also declined with rising TN:TP during Experiment 2 where at Day 5 chlorophyll concentrations were NP20 > NP1 > Control > 50 > NP100. Chlorophyll a concentration was very variable in the lake during Experiment 1, fluctuating between 40.5 ng-l-1 and 71.9 ng-l-1. During Experiment 2 the concentration was relatively stable, fluctuating between 4.7 ng-l-1 and 13.5 ng-l-1.
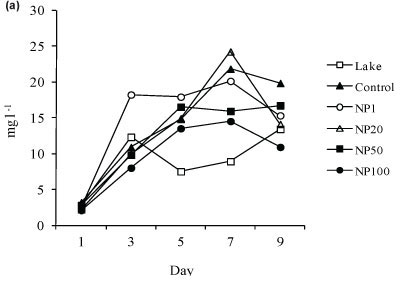
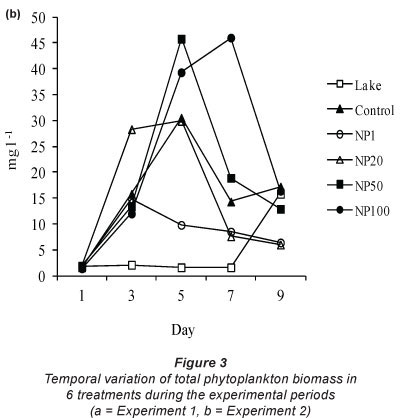
The variation in total algal biomass as the biological response of the phytoplankton assemblage to nitrate addition is shown in Fig. 3. Total algal biomass in the lake fluctuated between 2.8 and 13.4 mg-l-1 wet weight and between 1.6 and 15.8 mg-l-1 wet weight during Experiment 1 and Experiment 2, respectively. An increase in algal biomass occurred in all treatments. During Experiment 1 the highest biomasses were attained at Day 7 in all treatments with a maximum of 24.2 mg-l-1 wet weight in NP20. The trend in total biomass increase was generally similar in all treatments including the control. Only the control and NP1 had significantly higher biomasses than the lake while the rest had biomasses similar to those in the lake (Fig. 3a). Total biomass in NP100 was significantly lower than in the control and in NP1 (Fig. 3a).
During Experiment 2 highest biomasses were attained at Day 5 in all treatments except NP100, where highest biomass occurred at Day 7 (Fig. 3b). Only in NP50 and NP100 was total biomass significantly (p < 0.05) higher than in the lake while in the remaining treatments biomasses were similar to those in the lake (Fig. 3b). The total biomass in NP100 was significantly (p < 0.05) higher than in NP1.
During Experiment 1 the phytoplankton assemblage in the lake over the 9-day period was dominated by Cryptomonas which occurred with Pediastrum sp. and Scenedesmus sp. (Fig. 4). Addition of nitrate and consequent increase in TN:TP ratio resulted in a shift to an assemblage dominated by Coelastrum occurring with Scenedesmus and Pediastrum in all treatments including the control (Fig. 4). By Day 9 Coelastrum sp. was dominant in all treatments. Cryptomonas markedly declined with increase in TN:TP ratio and totally disappeared at Day 7 and Day 5 in NP50 and NP100, respectively (Fig. 4). All the treatments showed the same pattern and, regardless of the lack of added nitrate in the control, a shift to dominance by chlorophytes also occurred. The conditions within the microcosms favoured the development of chlorophytes (Scenedesmus, Coelastrum and Pediastrum).
A similar response was observed during Experiment 2 (Fig. 5). The lake and the inoculum in all treatments were dominated by Cryptomonas and 2 species of diatoms, Cyclotella and Aulacoseira (= Melosira), and to a lesser extent chlorophytes (Pediastrum, Coelastrum, Scenedesmus) and Microcystis. Cryptomonas markedly declined, especially in NP20, NP50 and NP100 where by Day 3 it was negligible in the culture. Cryptomonas also declined in the control and NP1 but remained in the culture up to Day 9. The diatoms Cyclotella and Aulacoseira (= Melosira) also decreased and were very low in all treatments by Day 9. Chlorophytes Coelastrum,, Pediastrum and Scenedesmus became dominant from Day 5 in all treatments, including the control. Favourable growth for Pediastrum occurred in NP1 and NP20. Microcystis increased only slightly in NP1.
Discussion
The environment in Lake Chivero during microcosm experiments selectively favoured Cryptomonas sp. A species is favoured when both absolute concentrations of nitrogen and phosphorus and their ratio in the environment conforms to that particular species' requirements (Levich et al., 1993). The patterns of temporal change exhibited by Cryptomonas in all the treatments showed that its favourable TN:TP ratio during the experiments ranged between 1 and10, in contrast to previous findings. For example, Healy and Henzel (1980) found an optimum TN:TP ratio of 39 for Cryptomonas erosa, while Schollhorn and Granelli (1997) observed an increase of Cryptomonas spp. in TN:TP ratios of 16, 40 and 100 during the first half of their experiment and a decrease thereafter. In their experiment, high cell densities were maintained longer in the TN:TP ratios of 40 and 100 than in 16, indicating that the optimum ratio might be above 16. The patterns observed during the experiments conversely showed that the species in Lake Chivero tolerated a TN:TP ratio of up to 10.
Shift to a dominance by chlorophytes, mainly Scenedesmus spp., has also been reported from enclosure experiments (Pick, 1989; Tilman et al., 1986). The genus Scenedesmus comprises of fast-growing opportunistic species which, according to the resource-competition theories, are favoured over larger phyto-plankton under the conditions found in the microcosms because they have a higher nutrient affinity resulting from their small size and higher surface area to volume ratios (Raven, 1998).
The increase of Scenedesmus was most notable during Experiment 2 in treatments NP50 and NP100. Levich and Bulgakov (1993) observed a replacement of all species by the chlorophyte Scenedesmus quadricauda at a TN:TP ratio of 20. High ratios (20-50) in nutrient media stimulated growth of chlorophytes, but the favourable ratios were near 20 (Levich et al., 1993). Levich et al. (1993) observed that although chlorophytes remain dominant at higher ratios (50 and 100) their biomass does not increase. In this study it was observed that increasing the TN:TP ratio above 10 did not markedly increase total biomass but favoured dominance by chlorophytes. After enrichment chlorophytes benefited from Cryptomonas decrease and become established in the microcosms. Cryptophytes are strongly susceptible to zooplankton consumption, unlike chlorophytes which are not particularly good quality food (Barone and Naselli-Flores, 2003; Sterner, 1989), so the changes observed in microcosms may be partly explained by depredation by zooplankton.
Influence of light needs to be established further, although in the microcosms the light conditions of the phytoplankton are better than those in the lake, where the phytoplankton species are circulated in the light for a shorter period and remain down in dim light for most of the day (Knapp et al., 2003; Olrik, 1994). Consequently, in the microcosms small fast-growing diatoms and chlorophytes will dominate and the more shade-adapted cryptophytes will decline, whereas in the lake the more shade-adapted cryptophytes will dominate (Reynolds et al., 2002; Reynolds, 1988; Reynolds, 1984).
During Experiment 2, the diatom Cyclotella sp. initially responded fast to added nitrate and became the dominant species, assuming 77% of the total biomass in all treatments by Day 3, after which it declined. In contrast, Aulacoseira granulata immediately declined upon nitrate addition. This was most apparent in Treatments NP50 and NP100 where it had disappeared in culture by Day 5. The decline of A. granulata, a functionally ruderal species (Reynolds et al., 2002), could have also been due to negligible circulation in microcosms. Lagus et al., (2004) reported fast growth response to nutrient enrichment by centric diatoms in enclosure experiments, as exhibited by Cyclotella. Its later decline could have been due to limitation by availability of silica (Conley et al., 1993) or lack of mixing. The initial high growth rate of Cyclotella indicates that silica may not have limited phytoplankton growth. Cyclotella was competitively superior to Aulacoseira under nitrate addition. The favourable TN: TP ratio for diatoms lies between 5 and 20 (Levich et al., 1993), a condition that prevailed in Lake Chivero during the study period (Mhlanga, 2007).
Tilman's resource competition theory states that under nutrient limitation in equilibrium conditions those species that have either the lowest requirements for the limited resources, or the highest ability to utilise it, will succeed in competition (Tilman et al., 1982). Observations from these experiments support this theory. Under nitrogen limitation, smaller species with the greatest abilities to utilise nitrogen over M. aeruginosa seem to now be competitively successful in Lake Chivero (Mhlanga, 2007). The resource-ratio hypothesis predicts that the relative abundances of coexisting species depend on the ratio of the limiting resources, not on the absolute concentrations (Tilman et al., 1982). Although both nitrogen and phosphorus are high in Lake Chivero, their ratio seems to have a marked influence on algal assemblage dynamics. Cryptophytes seemed to have been superior competitors at the prevailing TN:TP ratio, favouring a range of 1 to 10 in particular, which should be their optimal resource ratio in Lake Chivero. Increases in nitrate concentrations, as observed during this experiment, would competitively exclude them, leading to dominance by chlorophytes favoured by elevated nitrate concentrations. This is in accordance with the ideas of Tilman (1982), who stated that superior competitors are expected to be dominant at their optimal resource ratios and to be succeeded by others with different optimal resource ratios, if the resource ratios change in the environment.
It was not possible in either experiment to separate the effect of nitrate-induced change from that of isolation, since similar changes occurred in both the nitrate-added treatments and the control. The effect of nitrate can be illustrated by grouping the treatments into 3 groups: (i) lake (ii) control + NP1 + NP20 and (iii) NP50 + NP100. The pattern in the lake was markedly different from group (ii) and (iii) since Cryptomonas dominated throughout. In the second group Cryptomonas occurred only in low concentrations until Day 9 while in the third group it declined drastically and was absent in culture by Day 5. It can be inferred that at > 50 x nitrate addition the decline was accelerated by nitrate addition while below that the observed changes could be linked to isolation only.
Increasing nitrate concentration for the phytoplankton assemblage of Lake Chivero increased phytoplankton biomass and chlorophyll a concentration. Chlorophyll analysis is accepted as an indirect measure of phytoplankton biomass (Võrõs and Padisak, 1991). Phytoplankton biomass and chlorophyll a concentration are supposed to be correlated. Disparities observed during this study have elsewhere been attributed to variations in chlorophyll a content of the phytoplankton assemblage and some external and internal factors (Võrõs and Padisak, 1991). Light limitation for instance can result in increased cellular chlorophyll a (Desortova, 1981; Hunter and Laws, 1981). In Lake Chivero light is a major limiting factor due to algal self-shading (Robarts, 1979), which could have caused disparities but could also be attributed to uncounted species (Talling, 1986; Kebede and Belay, 1994), mainly picophytoplankton, as observed in Zeekoevlei by Harding (1996). Aphanocapsa sp. and Pseudoanabaena mucicola were not enumerated during this study and since, according to Padisák and Dokulil (1994), they can constitute a significant portion of total phytoplankton biomass their exclusion can underestimate biomass. While chlorophyll a could be the most accurate measurement for this study, both chlorophyll a concentration and biomass estimates fall within the ranges previously reported for Lake Chivero (Falconer, 1973; Thornton, 1980; Ndebele, 2003; Annadotter et al., 2005).
Phosphorus concentration is high in Lake Chivero indicating hyper-eutrophic conditions (Mhlanga et al., 2006b). Nitrogen limitation of phytoplankton growth is known to occur when phosphorus concentrations are high (Dzialowski et al., 2005). However increase in total biomass in the control indicates that, other than nitrate addition, isolation and, most likely, differences in lake and microcosm temperature during Experiment 2 also enhanced biomass accumulation, or that ambient nutrient concentration in the confined assemblage was adequate to induce change, particularly as nutrients and physical conditions in lake, control and NP1 were similar. The phytoplankton assemblage was N-limited up to the period between Day 5 and 7. After attaining maximum biomass other factors became limiting. The most likely limiting factor then could have been light. Phosphorus could have also been limiting because further additions of nitrate above 20 x for Experiment 1 were not accompanied by an increase in biomass. However, for Experiment 2, only additions of nitrate above 50 x (treatments NP50 and N100) enhanced biomass to levels significantly higher than those in the lake. Phosphorus became limiting at different levels of nitrate addition probably due to differences in ambient nitrogen concentrations during the two experimental periods.
Microcystis aeruginosa, a dominant species in Lake Chivero (Munro, 1966; Marshall, 1997), did not assume dominance either in August 2005 or April 2006, most probably because Cryptomonas dominated the lake phytoplankton assemblage. Microcystis was not observed in August 2005 but occurred at very low levels in April 2006. Dominance of Microcystis, even though the nutrient levels were high in microcosms, could have been limited by the small quantity of 'seed' in the inoculum. This study showed that it may be possible to manage the composition of phytoplankton in Lake Chivero by varying nitrate concentrations and nutrient ratios in order to optimise the dominance of cryptophytes, bacillariophytes and chlorophytes and to decrease cyanobacteria. However, other physical factors like turbulence also have a marked effect on species dominance. Maintaining a cryptomonad, diatom or chlorophyte dominance would lead to a socially and ecologically acceptable lake provided that this is not precluded by selective grazing impacts of zooplankton -a factor neither controlled nor examined in this study.
Acknowledgements
This study was supported by a research grant received from Water Research Fund for Southern Africa and technical support from the University of Zimbabwe's Lake Kariba Research Station.
References
ANNADOTTER H, CRONBERG G, NYSTRAND R and RYLANDER R (2005) Endotoxins from cyanobacteria and gram-negative bacteria as the cause of an acute influenza-like reaction after inhalation of aerosols. Ecohealth 2 1-14. [ Links ]
BARONE R and NASELLI-FLORES L (2003) Distribution and seasonal dynamics of cryptomonads in Sicilian water bodies. Hydrobiologia 502 (1-3) 325-329. [ Links ]
CONLEY DJ, SCHELSKE CL and STOERMER EF (1993) Modification of the biogeochemical cycle of silica with eutrophication. Mar. Ecol. Prog. Ser. 101 179-192. [ Links ]
CRONBERG G (1982) Phytoplankton changes in Lake Trummen induced by restoration. Folia Limnol. Scand. 18 1-119. [ Links ]
DESORTOVA B (1981) Relationship between chlorophyll a concentration and phytoplankton biomass in several reservoirs in Czechoslvakia. Int. Rev. Gesamten Hydrobiol. 66 153-169. [ Links ]
DZIALOWSKI AR, WANG S, LIM N, SPOTTS WW and HUGGINS DG (2005) Nutrient limitation of phytoplankton growth in central plains reservoirs, USA. J. Plankton Res. 27 (6) 587-595. [ Links ]
FALCONER AC (1973) The Phytoplankton Ecology of Lake Mcllwaine, Rhodesia. M. Phil. thesis, University of London. [ Links ]
GOLDMAN JC and DENNETT MR (1984) Effect of photoinhibition during bottle incubations on the measurements of seasonal primary production in a shallow coastal water. Mar. Ecol. Prog. Ser. 15 169-184. [ Links ]
GOLTERMAN HL, CLYMO RL and OHNSTAD MAM (1978) Methods for the Physical and Chemical Analysis of Freshwater IBP Handbook No. 8 (2nd edn.). Blackwell Scientific Publications, London. 214 pp. [ Links ]
HARDING WR (1996) The Phytoplankton Ecology of a Hypertrophic, Shallow Lake, with Particular Reference to Primary Production, Periodicity and Diversity. D. Phil. Thesis, University of Cape Town, South Africa. [ Links ]
HEALY FP and HENZEL LL (1980) Physiological indicators of nutrient deficiency in lake phytoplankton. Can. J. Fish. Aquat. Sci. 37 442-453. [ Links ]
HUNTER BL and LAWS EA (1981) ATP and chlorophyll as estimators of phytoplankton carbon biomass. Limnol. Oceanogr. 26 944-956. [ Links ]
KEBEDE E and BELAY A (1994) Species composition and phyto-plankton biomass in a tropical African lake (Lake Awasa, Ethiopia). Hydrobiologia 288 13-32. [ Links ]
KNAPP CW, DeNOYELLES (Jr.) F, GRAHAM DW and BERGIN S (2003) Physical and chemical conditions surrounding the diurnal vertical migration of Cryptomonas spp. (Cryptophyceae) in a seasonally stratified mid-western reservoir (USA). J. Phycol. 39 (5) 855-861. [ Links ]
LAGUS A, SUOMELA J, WEITHOFF G, HEIKKILA K, HELMINEN H and SIPURA J (2004) Species-specific differences in phytoplankton responses to N and P enrichments and the N:P ratio in the Archipelago Sea, northern Baltic Sea. J. Plankton Res. 26 (7) 779-798. [ Links ]
LEVICH AP and BULKAKOV NG (1993) Possibility of controlling the algal community structure in the laboratory. Biol. Bull. Russ. Acad. Sci. 20 457-464. [ Links ]
LEVICH AP, CHUDOYAN AA, BULGAKOV NG and ARTYU-CHOVA VT (1993) The regulation of phytoplankton structure in vitro by N:P ratios. Russ. J. Aquat. Ecol. 1 149-158. [ Links ]
MAGADZA CHD (2003) Lake Chivero, A management case study. Lakes Reservoirs: Res. Manage. 8 69-81. [ Links ]
MARSHALL BE (1991) Toxic cyanobacteria in Lake Chivero: a possible health hazard? Trans. Zimbabwe Sci. Assoc. 65 16-19. [ Links ]
MARSHALL BE (1997) Lake Chivero after forty years: the impact of eutrophication. In: Moyo NAG (ed.) Lake Chivero: A Polluted Lake. University of Zimbabwe Publications, Harare. 134 pp. [ Links ]
MHLANGA L (2007) Environmental Variables and the Development of Phytoplankton Assemblages in a Hypereutrophic Reservoir. D. Phil. thesis, University of Cape Town, South Africa. [ Links ]
MHLANGA L, DAY J, CHIMBARI M, SIZIBA N and CRONBERG G (2006a) Observations on limnological conditions associated with a fish kill of Oreochromis niloticus in Lake Chivero following collapse of a bloom. Afr. J. Ecol. 44 199-208. [ Links ]
MHLANGA L, DAY J, CRONBERG G, CHIMBARI M, SIZIBA N and ANNADOTTER H (2006b) Cyanobacteria and cyanotoxins in the source water from Lake Chiveri, Harare, Zimbabwe, and the presence of cyanotoxins in drinking water. Afr. J. Aquat. Sci. 31 (2) 165-173. [ Links ]
MOYO NAG (1997) Causes of massive fish deaths in Lake Chivero. In: Moyo NAG (ed.) Lake Chivero: A Polluted Lake. University of Zimbabwe Publications, Harare. [ Links ]
MUNRO JL (1966) A limnological survey of Lake Mcllwaine, Rhodesia. Hydrobiologia 28 281-308. [ Links ]
NDEBELE MR (2003) Some Aspects of the Ecology of Phytoplankton in Lake Chivero, Harare, Zimbabwe. MSc. Thesis, University of Zimbabwe. [ Links ]
NHAPI I (2004) Options for Wastewater Management in Harare, Zimbabwe. D. Phil. thesis, Wagenigen University, The Netherlands. [ Links ]
OLRIK K (1994) Phytoplankton-Ecology. Determining factors for distribution of phytoplankton in freshwater and the sea. Ministry of the Environmental Protection Agency. Nofo Print, Elsimore. [ Links ]
PADISAK J and DOKULIL MW (1994) Meroplankton dynamics in a saline, turbulent, turbid shallow lake (Neusiedlersee, Austria and Hungry). Hydrobiologia 289 23-42. [ Links ]
PICK FR (1989) Species-specific phytoplankton responses to nutrient enrichment in limnetic enclosures. Arch. Hydrobiol. Beith. Ergebr. Limnol. 32 177-187. [ Links ]
RAVEN JA (1998) The twelfth Tansley Lecture:Small is beautiful: the picophytoplankton. Funct. Ecol. 12 503-513. [ Links ]
REYNOLD CS, HUSZAR V, KRUK C, NASELLI-FLORES L and MELO S (2002) Towards a functional classification of freshwater phytoplankton. J. Plankton Res. 24 (5) 417-428. [ Links ]
REYNOLDS CS (1984) Phytoplankton periodicity: the interactions of form, function and environment variability. Freshwater Biol. 14 111-142. [ Links ]
REYNOLDS CS (1988) Functional morphology and the adaptive strategies of freshwater phytoplankton. In: Sandgren CD (ed.) Growth and Reproductive Strategies of Freshwater Phytoplankton. Cambridge University Press, Cambridge. 388-433. [ Links ]
ROBARTS RD (1979) Underwater light penetration, chlorophyll a and primary production in a tropical African lake (Lake Mcllwaine, Rhodesia). Arch. Hydrobiol. 86 423-444. [ Links ]
ROBARTS RD (1981) The phytoplankton. The Zimbabwe Science News 15 95-96. [ Links ]
ROBARTS RD and SOUTHALL GC (1975) Algal bioassays of two tropical Rhodesians reservoirs. Acta Hydrochim. Hydrobiol. 3 369-377. [ Links ]
ROBARTS RD and SOUTHALL GC (1977) Nutrient limitation of phytoplankton growth in seven tropical man-made lakes, with special reference to Lake Mcllwaine, Rhodesia. Arch. Hydrobiol. 79 (10) 1-35. [ Links ]
ROTT E (1981) Some results from phytoplankton counting inter-calibrations. Schweiz. Z. Hydrol. 43 34-62. [ Links ]
SCHÖLLHORN E and GRANELLI W (1997) The importance of increasing N/P ratios for phytoplankton biomass and species composition in an oligotrophic lake. Verh. Internat. Verein. Limnol. 26 615-620. [ Links ]
STERNER RW (1989) The role of grazers in phytoplankton succession. In: Sommer U (ed.) Plankton Ecology: Succession in Phytoplankton Communities. Springer-Verlag, Berlin. 107-170. [ Links ]
TALLING JF (1987) The phytoplankton of Lake Victoria (East Africa) Ergebn. Limnol. 25 229-256. [ Links ]
THORNTON JA (1980) Factors Influencing the Distribution of Reactive Phosphorus in Lake Mcllwaine, Zimbabwe. D.Phil. dissertation, University of Zimbabwe. [ Links ]
TILMAN D (1982) Tests of resource competition theory using four species of Lake Michigan algae. Ecology 58 338-348. [ Links ]
TILMAN D, KIESLING R, STERNER RW, KILHAM SS and JOHNSON FA (1986) Green, blue-green and diatom algae: taxo-nomic differences in competitive ability for phosphorus, silicon and nitrogen. Arch. Hydrobiol. 106 473-485. [ Links ]
TILMAN D, KILHARN SS and KILHAM P (1982) Phytoplankton community ecology: the role of limiting nutrients. Ann. Rev. Ecol. Syst. 13 349-372. [ Links ]
UTERMÖHL H (1958) Zur Vervollkommung der quantitativen. Phytoplankton methodik. Mitt. Int. Verein. Theor. Angev. Limnol. 9 1-38. [ Links ]
VÖRÖS L and PADISÁK J (1991) Phytoplankton biomass and chlo-rophyll-a in some shallow lakes in central Europe. Hydrobiologia 215 (2) 119-119. [ Links ]
ZILBERG B (1966) Gastro-enteritis in Salisbury European children -A five-year study. Centr. Afr. J. Med. 12 (9) 164-168. [ Links ]
Received 25 October 2011; accepted in revised form 11 July 2012.
* To whom all correspondence should be addressed. ffi +263 4 480303; fax: +263 4 480303; e-mail: wmhlanga@africaonline.co.zw