Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
Water SA
versión On-line ISSN 1816-7950
versión impresa ISSN 0378-4738
Water SA vol.38 no.4 Pretoria ene. 2012
Assessment of microbial contamination of groundwater in upper Limphasa River catchment, located in a rural area of northern Malawi
Thokozani KanyerereI, IV; Jonathan LevyII; Yongxin XuI, *; John SakaIII
IDepartment of Earth Sciences, University of the Western Cape, Private Bag X17, Bellville 7535, South Africa
IIDepartment of Geology, Miami University, Oxford, OH 45056, USA
IIIDepartment of Chemistry, University of Malawi, Chancellor College, Box 280, Zomba, Malawi
IVDepartment of Geography and Earth Sciences, University of Malawi, Chancellor College, Box 280, Zomba, Malawi
ABSTRACT
In rural Africa, scientific evidence is often lacking to guide the scaling-up of groundwater as the safest source of potable water. An investigation was conducted in the Upper Limphasa Catchment in northern Malawi to determine the safety of groundwater sources and to explore factors influencing water quality. Water samples from 17 boreholes, 6 hand-dug wells and 90 households were analysed for selected parameters. Portable incubators, multi-parameter probe and colorimetric standard methods were used for field measurements, and standard methods were used for laboratory water analysis. Results were compared to specified guidelines of the World Health Organization and Malawi Bureau of Standards to establish the potability of water. Statistical results using non-parametric t-tests indicated that the wells were more contaminated with E. colibacteria than boreholes (p=6.2x10-6), suggesting non-consideration of local hydrogeologic factors in groundwater development. Water from boreholes that tested negative for pathogens at source tested positive at some households (total coliform:p=0.0042 and E. colip=7.8x107) suggesting the effect of handling practices. Water from wells that was not treated with chlorine showed higher levels of E. coli than treated water from the same sources, confirming the effectiveness of chlorine in reducing pathogenic bacteria in households' stored drinking-water, reinforcing the scientific basis for scaling up chlorine as effective disinfectant. However, this study demonstrated that chlorine failed to effectively eliminate all pathogens in drinking water. As a case study in tropical rural environments in Africa, these findings on the suitability of using chlorine as disinfectant and on factors explaining groundwater contamination, though provisional, provide a scientific basis for assessing cost-effectiveness and sustainability of scaling-up the use of chlorine as a curative remedy and of systematically investigating local hydrogeologic factors in order to implement measures to protect groundwater quality in poverty-prone rural communities.
Keywords: Chlorine treatment, drinking water; groundwater sources' vulnerability, pathogenic bacteria, social vulnerability, tropical rural communities
Please note corrigendum – see last page of PDF
Introduction
Access to safe and sufficient water and sanitation is a basic human need and is essential to human wellbeing (UN, 2006). One of the United Nations Millennium Development Goals (MDG) specifically addresses the problem of lack of access to safe drinking water. This MDG aims to halve the proportion of people without sustainable access to safe drinking water by 2015 (Cronin et al., 2006). Sub-Saharan Africa has the highest proportion of poor people in the world, 44% of the population in 2002 (World Bank, 2006). The region has the world's fastest growing population with a 2.2% annual growth rate leading to increased pressure on water resources (Bordalo and SavvaBordalo, 2007). About 22 African countries, including Malawi, fail to provide safe drinking water to half of their population (Pritchard et al., 2007).
Malawi's total population is 13 million, 11 million of which live in rural areas, and has an annual growth rate of 2.8%. The demand for natural resources, including water to sustain livelihoods, is therefore high (Malawi Government, 2008b; World Bank, 2005). Although water is generally abundant in Malawi, 60% of the population lacks safe drinking water and improved sanitation (Pritchard et al., 2007). Consequently, water-related diseases continue to be one of the major health problems in the country.
To contribute to achieving the UN MDGs, Malawi's specific water policy objectives were developed to ensure that at least 80% of the population have access to potable water, and to reduce non-functional water-points from 31°% to 25%, by 2011 (Malawi Government, 2008a). In Malawi, as in many developing countries, one of the major strategies to achieve these goals has been to install protected water sources, such as boreholes and protected shallow wells, to provide water of better quality (Wright et al., 2004). Most people in developing countries depend on untreated groundwater supplies as safe sources for their drinking water. The increasing demand to provide water through such sources has resulted in concerns that some groundwater sources might not be as safe as expected, thereby endangering people's lives. The major groundwater sources are:
- boreholes (tube wells) that are usually rig-drilled >20-m deep and potentially tap deeper aquifers; and
- shallow wells (hand-dug wells) that are usually dug <20-m deep and often tap unconfined aquifers (MacDonald and Davies, 2000).
In rural areas, shallow wells are the traditional means of accessing groundwater from shallow aquifers. A shallow well is considered protected when fitted with a proper lid on top (Malawi Government, 2005). Funds and services to provide groundwater wells in rural Malawi are provided by the Malawi Government agencies such as the water board division, water department and district assemblies, as well as non-governmental organisations (NGOs) that include UNICEF, Africare, ActionAid and religious groups (Malawi Government, 2008a).
There are a few potential problems that are typically associated with boreholes and protected shallow wells in Malawi. Firstly, their location downhill from village latrines makes them vulnerable to microbial contamination. Secondly, their locations with respect to the points-of-use (households) often require difficult transport from the source and then subsequent storage of water within the households. The microbial quality of water in storage facilities in households can be lower than that at the water source, suggesting that contamination is widespread during collection, transport, storage and drawing from containers before drinking (Wright et al., 2004). Thus contamination may reduce potential health benefits of water. Microbial contamination of groundwater supplies may also result from poor wellhead conditions, such as poor sanitary seals which can be risk factors for water quality rather than proximity of latrines (Cronin et al., 2006). A third potential problem is the lack of regular water-quality testing.
Groundwater sources are tested for microbial, physical and chemical quality during construction by drillers, which is a contractual requirement commonly practiced in Malawi. This is undertaken to establish if the water is fit for human consumption. The Central Laboratory of Malawi performs the analyses as soon as the water source has been developed and before people start using the source (Malawi Government, 2008a). However, this is not sustained on a regular basis. This is a common pattern in developing countries where more regular water sampling and analysis are considered too costly (MacDonald et al., 2005). Further, groundwater is often tested only when pollution from industrial effluents or commercial farming activities is suspected. Therefore, it is not surprising that in areas such as rural Malawi, where there are no or few industries or commercial farming operations, studies regarding microbial contamination of groundwater are seldom performed. Studies that focus on abundance and spatial variation of pathogens in groundwater sources, on contamination of water during transport to and storage in homes, are rare. However, such knowledge is vital for devising effective strategies to improve the quality of drinking water for rural populations, especially those who have low purchasing power for effectively treating drinking water at home.
The present study was undertaken to investigate indicators of pathogenic bacteria associated with boreholes and protected shallow wells in the Upper Limphasa Catchment, a rural area of northern Malawi. The area in question has never been previously studied in terms of its hydrogeology. This is despite the occurrence of a 2008 diarrhoea-related outbreak in the catchment, the worst in 10 years (Ndimbwa and Mpohah, 2008). Previous studies have concentrated in a similar environment in southern Malawi, where a database on water quality from protected shallow wells was obtained (Pritchard et al., 2007; Mkandawire, 2008). Until now, no similar studies have been conducted at all in northern Malawi.
River catchments and their geographical locations are usually unique and no single research site will provide a perfect analogue to guide water managers in understanding the hydrogeology of a different site. However, a study on pathogens in groundwater sources does provide a point of reference on the presence of pathogens in groundwater, enabling the suggestion of practical preventive measures around water points within a catchment. The purpose of this paper is to explore the nature of microbial contamination of drinking water in a rural catchment in Malawi and to use those findings to demystify general groundwater contamination and suggest a feasible guideline that would be suitable for poverty-prone rural communities within a tropical African environment, when developing groundwater sources for drinking.
Research activities on groundwater contamination in most developing countries, including Malawi, continue to focus on finding effective remedies to purify drinking water (Pritchard et al., 2007; 2009; 2010). Studies to assess aquifer vulnerability as a planning tool are numerous, with a few that focus on the village scale within a catchment (Robins, 2010). However, there has been limited work on demystifying groundwater contamination from the local hydrogeologic and social-spatial perspective that would enable practitioners to:
devise effective preventive measures to protect groundwa-ter from detected contamination; and
revise the approach to scale-up groundwater sources that are pathogenically infested.
This work was therefore conducted to:
determine the levels of pathogenic bacteria in groundwater sources and households to indicate the degree of potential pathogenic contamination in drinking water;
assess the changes in water quality between water sources and households that draw drinking water from the same sources, to detect changes that typically occur post-collection;
evaluate the effectiveness of home treatment of drinking water based on the turbidity and chlorine levels in household-stored drinking water; and
identify factors that contribute to contamination in ground-water sources, for possible intervention.
Site description
Location and administrative units
Malawi is a land-locked country located in Sub-Saharan Africa, lying in the southern end of one limb of the Great East African Rift Valley (Fig. 1). The country is about 901 km long and its width varies from 80 to 161 km. The country has a wide range of relief with elevations ranging from 37 m above sea level (m asl) on the southern border to over 2 400 m asl in the highlands (Malawi Government, 2008a). The relief strongly influences climate, drainage pattern, groundwater flow and population distribution (Sophocleous and Buchanan, 2003).
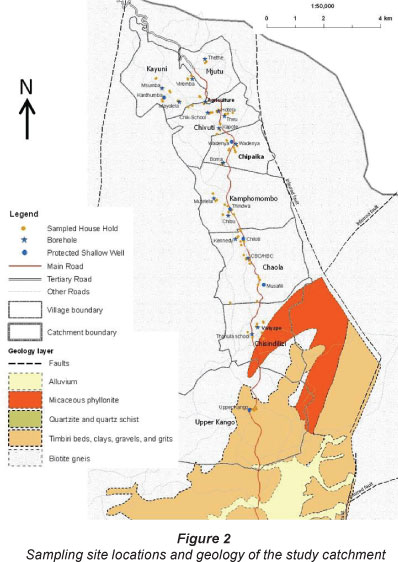
Administratively, Malawi is divided into 3 regions: North, Centre and South, which are further subdivided into 6, 9 and 13 districts, respectively. The districts, in turn, are administratively subdivided into traditional authorities presided over by local chiefs. Traditional authorities are further subdivided into villages which form the smallest administrative units, each presided over by a 'village head'. The study site is located in the Northern Region, which is home to about 13% of the total population (National Statistic Office, 2008). The study focused on 8 villages in the Nkhata Bay District within the Traditional Authorities Timbiri and Nyaluwanga. The villages were: Upper Kango, Chisindilizi, Chaola, Kamphomombo, Chipaika, Chivuti, Kayuni and Mjutu, all located within the Upper Limphasa River catchment, the study area (Fig. 2).
Hydrogeology and climate
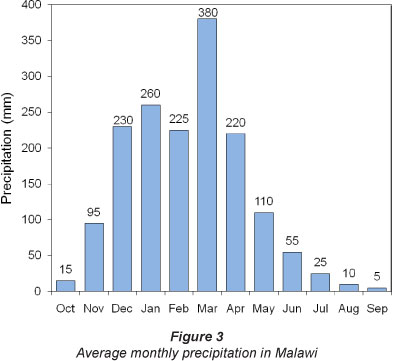
Malawi falls within the Basement Provinces, the Precambrian crystalline basement rock areas; groundwater only occurs at shallow depths in large parts of these regions. The country has almost 80% of its area underlain by such provinces (Malawi Government, 2008a). The study area encompasses escarpments associated with the Malawi Rift Valley System within the Basement Complex aquifer where the principal aquifers are contained within the fractured Basement Complex. The aquifers are poor and discontinuous because the weathering products are often stripped away by erosion, but there is considerable faulting in association with the development of East Africa's Great Rift Valley. At the southern extent of the study area there are some rocks of sedimentary origin, and unconsolidated quaternary alluvium such as clays, silts, sands and gravels derived from weathering of the escarpment area. The coarser-grained alluvium also serves as a local aquifer. No documented studies currently exist that delineate local groundwater flows or quantify aquifer parameters. If the groundwater flow follows the regional topography, then the flow direction is generally north to south. It is presumed that recharge occurs via direct infiltration of rainfall in the higher areas and discharge occurs to the valleys and Lake Malawi (Fig. 2). Malawi receives a total annual rainfall of over 1 600 mm, mostly occurring from November to May. This rainfall pattern is assumed to have implications for pathogen travel to groundwater. The highest and lowest monthly averages are 380 mm and 5 mm in March and September, respectively (Fig. 3; Malawi Government, 2008a).
Water resources
When compared with other countries in the Southern Africa Development Community (SADC), Malawi has plentiful freshwater resources, which comprise 5 lakes and many perennial rivers. The 5 lakes cover a total of about 29 796 km2 (Malawi Government, 2008a). Despite its abundance, the quality of surface water is often poor, typically carrying pathogens (UNICEF, 2005). Groundwater is less prone to contamination than surface water leading to the increasing demand for its exploitation, especially in rural areas (MacDonald et al., 2005). The Malawi Government and several NGOs have provided many boreholes and protected shallow wells. In 2008, the estimated coverage of water supplies in rural Malawi was 71%, relatively higher than in neighbouring Tanzania (46%), Zambia (41%) and Mozambique (26%) (Baumann and Danert, 2008). However, about 31°% of improved water points in rural Malawi were not functioning, reducing the effective coverage to 55% (Baumann and Danert, 2008). The quality of groundwater in rural areas is not regularly monitored hence a knowledge gap exists about groundwater quality. The quality determined during borehole/well construction and development cannot be guaranteed over time.
Methods
Water sampling and analysis
Water samples were taken from all 17 production boreholes (BHs), 6 protected shallow wells (PSWs) and 90 sampled households in the 8-village study area (Fig. 2). For each water source, 1 cluster of households that was closest to and 1 cluster that was farthest from the source were also selected for water sampling. Within each selected cluster, 2 households were randomly sampled. In total, 92 households were targeted, i.e., 4 households per water source in the study area. Two households were not included due to their inaccessibility.
Before sampling, water from BHs and PSWs was repeatedly pumped and measured for temperature, specific conductance and pH using an Oakton® multi-parameter probe. Water samples were collected only when all the readings stabilised. Conductivity measurements were converted to total dissolved solids (TDS in mg/l) values by multiplying the EC value (in mS/cm) by a factor of 6.4 (Weaver et al., 2007). Household water was sampled using the same utensils that the residents used to access their stored water. The pH, specific conductance and temperature were measured in the household water using an Oakton® multi-parameter probe.
All water was tested for contamination by total coliforms and E. coli bacteria. Bacteria analyses were performed with 3M Petrifilm® ChromoCult Coliform Agar together with a membrane-filtration unit and portable incubators. Analysis with the Petrifilm was done in one of two ways. In the case of the samples that were suspected to be highly contaminated, 1ml of sample water was applied directly to the agar film. When a lower level of contamination was suspected, 100 ml of sample water was passed through a 0.47-mm filter paper using a sterile membrane-filtration unit. After filtration, the filter paper was placed within the agar film which had been pre-wet with 1 ml of sterile water. Subsequently, the Petrifilm plates were incubated at 35ºC for 24 h and the colonies were counted, with blue colonies indicating E. coli bacteria and red colonies indicating total coliforms. Bacterial concentrations were recorded as colony-forming units per 100 ml of water (CFU/100 ml). The number of counted colonies for the 1-ml samples was multiplied by 100 to maintain consistency of units for concentrations. All bacterial analyses were performed in duplicate; the reported bacterial concentrations represent the average of the 2 analyses. In sampled households which reported that they treated their drinking water, the sampled water was tested for chlorine and turbidity levels, using colorimetric methods with a chlorine photometer and turbidity meter, respectively.
Water was also analysed for various elements, including the following major ions: HCO3-, CO32-, NO3-, PO43-, Cl-, F-, SO42-, Ca2+, Mg2+, K+ and Na+ and some trace metals including Al, As, Cd, Pb, Se and Zn. Water samples were collected according to standard procedures (Weaver et al., 2007; APHA, 1995). Ion analyses were performed by Bemlab (Pty) Ltd - Assay Laboratory in Stellenbosch, South Africa. Major and trace analyte concentrations were determined using an ICP-OES auto-analyser program as a standard method for the examination of water. A titration analysis method was utilised to analyse HCO3- and CO32- in triplicate and the mean of the results used (APHA, 1995). Bacteria and ion concentrations were compared to specified guidelines, 2008 World Health Organization (WHO) and 2005 Malawi Bureau of Standards (MBS), to provide:
insight concerning the general status of the quality of groundwater in the study area; and
a preliminary statement on how such water compares to the defined water quality objectives of the country.
Measurement of potential causal factors for groundwater contamination
The study used a specific-diagnostic-information technique to assess potential sources of E. coli contamination, including indirect and direct pathway factors. Each water source was evaluated in terms of its distance from potential sources of bacterial contamination such as latrines, animal corrals and streams/rivers. The physical condition of each water source was also assessed to determine potential risk of contamination from poor construction, condition or siting. Each source was assigned a risk-factor score calculated based on answers to a series of 10 yes-or-no questions that dealt with the proximity of the water source to potential sources of contamination, the existence and condition of a proper fence and cement apron and the condition of the hand pump (Howard et al., 2003). Each positive answer indicated increased threat of contamination. The risk-factor score was the number of 'yes' answers associated with the 10 questions.
The DRASTIC (Depth, Recharge, Aquifer, Soil, Topographic slope, Impact of vadose zone, Conductivity of aquifer, as shown in Table 7: Variable column) approach by Aller et al., (1987) is a widely-used approach for assessing aquifer vulnerability to contamination. The DRASTIC approach was used in this study to consider factors that a specific-diagnostic-information approach could not address. In addition, DRASTIC offers a rough management tool in lieu of more detailed hydrogeologic investigation. DRASTIC focuses on hydrogeologic factors that increase the potential for contaminants to reach a given borehole or shallow well, including depths of water sources, the recharge rate, aquifer geology and hydraulic conductivity, soil type, surface slope, and vadose zone properties. Depths of water sources were estimated from records that water-point committees kept when the water sources were drilled or installed. The net recharge of the area was approximated based on Malawi Government Reports (Malawi Government, 1986). Area geology and vadose zone characterisation was estimated based on a geologic map by Kim and Hamm (1999). Soil characteristics were approximated based on soil maps of the study area (Malawi Government, 1986). Aquifer hydraulic conductivity was estimated based on information from a study by Yu et al. (1992).
Statistical analyses
To test the effectiveness of household water treatment (i.e., the addition of Waterguard®), bacteria concentrations of treated water from 10 of the households were compared to those of untreated water from the other 80 households. Firstly, this analysis was performed so as to not introduce bias based on differences between water sources. Comparisons were only performed when there were both households treating water and households not treating water that was collected from the same source. For each water source, households with treatment and those without treatment were averaged. This allowed paired t-tests for bacterial concentrations of treated versus untreated water with each pair associated with a different water source. Water from only six of the water sources was used both by households that treated their water and households that did not treat their water.
The degree of bacterial contamination was used to evaluate differences between water sources, to assess the water-quality impact of water transport and home storage and to evaluate the effectiveness of home water treatment. To test for differences between populations, Student's t-tests (Snedecor and Cochran, 1980) were used according to the convention of Freedman et al. (1998), where p<0.05 was considered statistically significant, providing moderate evidence against the null hypothesis, and p<0.01 was considered highly significant, providing strong evidence against the null hypothesis. By extension, p-values between 0.05 and 0.1 were assumed to provide weak evidence against the null hypothesis. Most comparisons were performed using non-parametric methods (Wonnacott and Wonnacott, 1985) free of all assumptions regarding the distributions of the parameter values. Nonparametric methods, based on the relative ranks of the data rather than their absolute values, are preferred when the data exhibit non-normal distributions and there are large differences in variances among populations. With nonparametric analyses, p-values are approximate but satisfactory (Wonnacott and Wonnacott, 1985).
Correlation analysis was used to determine possible relationships between site characteristics and contamination levels, similar to the approach used by Conboy and Goss (2000). Such correlation analysis was thought to be potentially useful for identifying whether the placement of latrines and depths of the boreholes could explain some of the bacterial-concentration variability. If so, the relationships might help to establish proper guidelines to reduce a well's vulnerability to contamination. For these analyses, ranked data were also used. Correlations withp < 0.05 were considered to be statistically significant.
Results
Physical-chemical analyses of water from boreholes and protected shallow wells
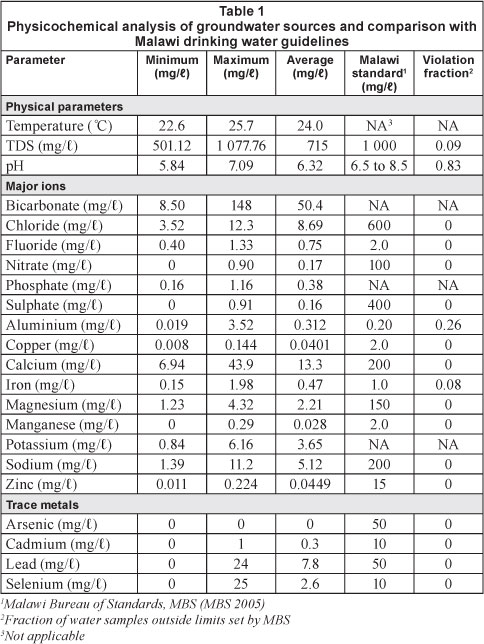
None of the BHs or PSWs had concentrations of any major ion that were of concern when compared with specified guidelines of WHO and MBS (Table 1). TDS concentrations were generally acceptable, being < 1 000 mg/l in 21 of the 23 sources. Water from 18 of the 23 sources had a pH less than 6.5. Pritchard et al. (2010) report that typical pH values of water sources in developing countries including Malawi fall between 5.5 and 8.0. The pH values in this study ranged between 5.84 and 7.09 with an average of 6.32 reflecting the siliceous nature of the underlying rocks.
Bacteriological analyses of water from boreholes and protected shallow wells
Of the 23 water sources that were sampled, 17 were BHs and 6 were PSWs with total coliform concentrations ranging from 0 to 50 CFU/100 ml in BHs and 0 to 2250 CFU/100 ml in PSWs (Table 2). Some of the total coliforms could be representative of naturally-occurring bacteria that do not necessarily reflect sewage contamination of the groundwater, so they were used only as a secondary indicator of contamination. Conversely, E. coli bacteria are indicators of contamination by human or animal faeces. In 3 of the BHs, 1 of the 2 duplicate samples had an E. coli concentration of 1 CFU/100 ml, resulting in an average concentration of 0.5 CFU/100 ml. Other than these 3 instances, no E. coli were detected in water from BHs. In the study area, counts as low as 1 CFU/100 ml may be due to user error in the field and are not necessarily an indication of contamination of the groundwater resources. Conversely, E. coli concentrations in the 6 PSWs ranged from 0 to 3 700 CFU/100 ml and 4 of the wells had concentrations that indicated sewage contamination.
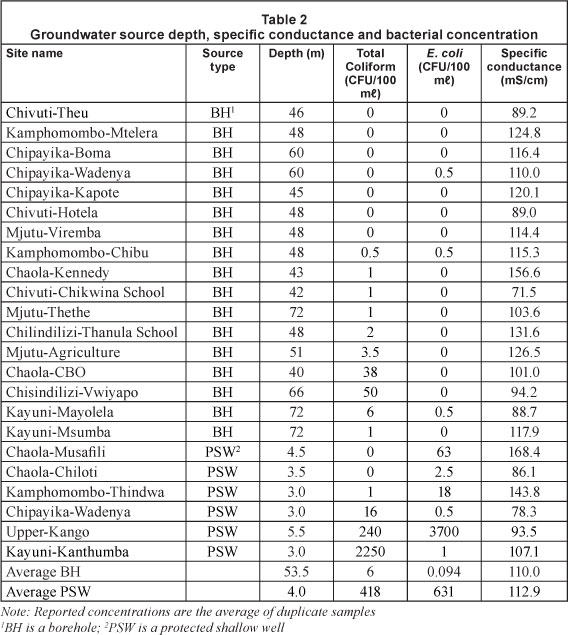
The hypothesis that PSWs were more contaminated than BHs was statistically assessed with a Student t-test. A summary of all t-test results is presented in Table 3; parametric t-tests provided weak evidence of a significant difference in bacterial concentrations. However, based on a nonparametric t-test, PSWs were significantly more contaminated with E. coli bacteria than boreholes (p = 6.2x10-6) (Table 3).
Effects of water handling on quality of water stored in the households
For each of the 23 water sources, water was sampled in 4 households (for 21 of the sources) or 3 households (for 2 of the sources) obtaining their water from that source. Practices for storing and treating drinking-water were also studied. Village residents typically collected and carried the water from each source in 20-l plastic buckets. Of the 90 sampled households, 45 stored their water in clay pots, 44 in plastic containers and 1 in metal pots. Seventy-seven of the households kept covers on their containers when they were not in use while 13 households left the containers uncovered. Only 10 of the households chlorinated their drinking water with Waterguard® treatment tablets (Table 4). Turbidity in stored household water ranged from 0 to 25.83 Formazine Nephelometric Units (FNU), averaging 2.17 FNU. Only 12% of the households had water with turbidity levels above the Malawi standard of 5 FNU (MBS, 2005).
Turbidity levels were not significantly correlated to either total coliform or E. coli concentrations.
Total colilform bacteria were detected in water from 57 of the households. Concentrations ranged from 1 to around 67 000 CFU/100 ml (i.e., 670 CFU in a 1-ml sample) and averaged around 5 150 CFU/100 ml for households with detections. E. coli bacteria were detected in 60 of the households with concentrations ranging from 1 to 14 500 CFU/100 ml and averaged around 300 CFU/100 ml for households with detections. E. coli bacteria were detected in 50 of the 66 households that obtained their water from boreholes despite the general lack of E. coli detections in water from the boreholes.
To test the effects of water collection, transport and storage on the quality of water, the bacteria concentrations at each source were compared with those in the households that obtained water from that source. For each water source, the concentrations found within the households using that source were averaged. Therefore, there were 46 observations: 23 represented the sources and 23 represented the average of the households associated with each of those sources. Only households that provided no water treatment were used in this analysis so that only the effects of post-collection practices could be evaluated. Nonparametric t-tests were applied using the relative ranks of the 46 observations to detect significant differences between the mean ranks of the water sources versus the household averages. Both total coliform (p=0.0042) and E. coli ( p=7.8x10-7) concentrations were higher, on average, in the households than at the source, indicating that there was bacterial contamination associated with the methods used to collect, transport and/or store the water in the home (Table 3).
A paired t-test was used to more directly compare bacteria concentrations at the BH sources with the average concentrations in the households using those boreholes. There were 17 paired observations, one for each of the boreholes. The paired t-tests indicated no significant difference with respect to total coliform, but, as with the nonparametric test, E. coli concentrations were significantly higher in the households (mean = 0.088 CFU/100 ml) than at BHs (mean = 14.5 CFU/100 ml) (p=1.1x10-4) (Table 3). In the case of the data from protected shallow wells and households using the protected shallow wells, there were large and uneven variances thus precluding a paired t-test to directly compare bacterial concentrations.
Effects of point-of-use treatment on quality of water stored in the households
The mean bacteria concentrations of the untreated water (7 540 CFU/100 ml for total coliform and 74 CFU/100 ml for E. coli) were much higher than the mean concentrations of the treated water (5.5 CFU/100 ml for total coliform and 4.0 CFU/100 ml for E. coli). However, the associated variances were so large that the differences were not significant using this parametric analysis. So, a nonparametric approach was applied in which all the treated and untreated waters were combined into 1 data set and ranked. Then t-tests were used to detect significant differences between the mean ranks of the treated versus the untreated water. The nonparametric tests were performed for the 6 pairs of samples used in the parametric analysis and indicated that water treatment did significantly lower E. coli concentrations (p=0.0049) (Table 3).
In the nonparametric approach, there was no pairing according to water source. Without the pairing, there was no reason to limit the analysis to just those 6 sources serving both households with and without water treatment. Therefore, the same nonparametric analysis was repeated using all of the households, 80 with untreated water and 10 with treated water. Based on this nonparametric analysis, there was only weak evidence (p=0.075) that water treatment lowered the total coliform concentration, but strong evidence (p=0.011) that water treatment with chlorine effectively lowered E. coli concentrations (Table 3).
Chlorination improved water quality but did not eliminate all of the bacteria. This is probably because low chlorine levels were applied thereby killing too little bacteria. However, except in one case, the free and total chlorine concentrations were equal to each other within the margin of error. WHO (2009) reports that chlorine levels can be affected by water temperature, turbidity and electrical conductivity. However, chlorine concentrations were not significantly correlated to bacterial concentrations, turbidity or temperature (Table 4). Temperatures of the stored water ranged from 22°C to 29°C and averaged 25°C. The WHO (2009) recommends that water should be at around 18°C for chlorine to effectively work within 30 min of being added to drinking water. The colder the water is the more time is needed for chlorine to be effective.
Potential causal factors for microbial contamination in groundwater
An analysis of the relationship between the degree of contamination and site characteristics, such as depths of BHs and PSWs, distance to potential sources of contamination such as latrines and streams/rivers, including slope degrees and percentages, is provided in Tables 5 and 6. The results seem to suggest that depth possibly explains the difference in the degree of microbial contamination. However, when the 2 populations were examined individually, the degree of microbial contamination was not significantly related to either depths of BHs or PSWs. The depths for BHs ranged from 42 m to 72 m while depths of the PSWs ranged from 3.0 m to 5.5 m.

If the microbial contamination of groundwater emanated from latrines and/or animal corrals/kraals, then there might be a relationship between BHs and PSWs and the potential sources of contamination. Latrines were usually located downhill from villages, but uphill of the water sources (Fig. 4). There was no significant general relationship between the distance from latrines and/or animal corrals and the degree of contamination. However, the water source with the highest measured E. coli concentration, the Upper Kango PSW, (3 700 CFU/100 ml) was also the water source that was nearest to a latrine (10 m) and the only water source located in an alluvial local aquifer in the study area (Fig. 2). A further detailed assessment addressing this hypothesis is therefore required.

Comparatively, PSWs were located nearer surface-water drainage (rivers/streams) than BHs (Figs. 5 and 6 and Table 6). The mixing of surface water and groundwater in such areas is unavoidable. The groundwater in PSWs appeared to exist under direct influence of the surface water (Fig. 5). In some cases PSWs were located in-stream (Fig. 5). The slopes to PSWs were steeper than those to BHs (Table 6). The location of PSWs in low-lying areas, in-stream/swampy areas or closer to surface waters is associated with large differences in bacterial concentrations between PSWs and BHs. Possibly, location of PSWs explains the presence of pathogenic contamination in PSWs. However, differences in the condition of sanitary seals and plain casings in BHs and PSWs were not assessed. Nevertheless, field measurements and observations made on distances between potential sources of contamination and water sources, location sites of water sources, potential sources of contamination (surface-water interaction), and steepness in terms of slope degrees and percentages, provide insights useful for reconsidering guidelines on water service delivery in rural areas (Tables 5 and 6).

In addition to the difference in slopeposition between BHs and PSWs, field observations on the undulating topographic structure (Figs. 6 and 7) in the study area helped to visualise sources of contaminants to water sources. Such supporting evidence was in agreement with Sugden (2010), who reported that the greater the hydraulic gradient towards the water source, the higher the risk of water point contamination. These field observations provided insights on potential factors contributing to groundwater contamination observed in some water sources.


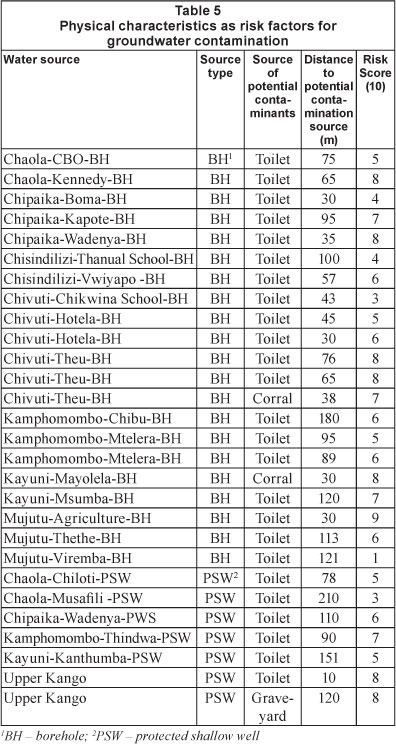
Each water source was assigned a risk-factor score (0 to 10 with 10 indicating conditions most prone to contamination) taking into account its physical condition, degree of protection and proximity to potential sources of contamination (Howard et al., 2003). Risk-factor scores for the 23 water sources ranged from 1 (for Mjutu-Viremba borehole) to 9 (for Mjutu-Agriculture borehole) and averaged 5.8 (Table 5). However, there was no significant relationship between the risk-factor score and the degree of microbial contamination, suggesting that vital controlling factors were missing from the analyses; hence the application of the DRASTIC model.
Application of the DRASTIC approach to explain bacterial contamination
The DRASTIC approach was applied using a combination of field measurements and previously-created geologic and pedologic maps (Table 7). The rating associated with the depth-to-water of the water source was 9 for the PSWs and 1 for BHs (Table 7). Theoretically, depth-to-water is important because there is a greater chance for contaminant attenuation as the depth-to-water increases, due to longer travel times and more contact with potential sorbents (Aller et al., 1987; Xu and Braune, 2010).
The most predominant soils in the study area are lithosols which are shallow and stony soils formed from granite and gneiss rocks (Malawi Government, 2008a). These soils consist of imperfectly weathered rock fragments and are mostly sand and gravel with low runoff potential. The DRASTIC system of Aller et al. (1987) gives this soil group a ranking of 6. Rahman (2008) cautions that, in addition to soil type, soil cover, which was not included in the analysis, also influences the surface and downward movement of contaminants.
Based on geological maps (Fig. 2), the major aquifers are gneiss (mainly biotite gneiss) in most parts of the study area followed by micaceous phyllonite and Timbiri beds, along with clays, gravels and grits in the extreme south. Yu et al., (1992) reported that typical hydraulic conductivities for these units, depending on the degree of weathering, are about 4.6 x 10-5 to 3.2 x 10-4 m/s for the granite, 1 x 10-4 to 4.6 x 10-4 m/s for the gneiss and schist and about 1x 10-3 m/s for the alluvium. Borrowing from the work of Yu et al. (1992) on hydraulic conductivities in basement and alluvial aquifers, similar to those in the study area, hydraulic conductivity was assigned a rating of 4 with a weight of 3 on the DRASTIC model by Aller et al. (1987); aquifer media (gneiss) was given a rating of 5 and a weight of 3; impact of vadose was given a rating of 5 with a weight of 4. Net recharge from report (Malawi Government, 1986) was given a rating of 4 with a weight of 3. An assessment based on these estimations demonstrates that these factors are no threat to groundwater contamination. Theoretically, factors with high scores on the DRASTIC Model, as shown in Table 6, highlight significant variables that would explain contamination in groundwater sources in the study area. Practically, such factors rarely work in isolation but in combination, based on basic hydrogeologic knowledge of groundwater contamination (Roberts et al., 2001; Kim and Hamm, 1999).
Discussion
Implication of potential factors for groundwater contamination for water service provision
The importance of providing scientific evidence for contamination of groundwater sources,in order to inform the revision of guidelines on rural water service provision, cannot be overemphasized. Site characteristics that were assessed included: location of latrines upslope from water sources (slope aspect/ slope effect); location of water sources where groundwater and surface water interacts in low-lying areas; distance between water points and sources of contamination (latrines and surface waters); depths of water sources and topography (Tables 5 and 6; Figs. 4, 5, 6 and 7). Different approaches were employed to examine the effects of these site characteristics on groundwater source contamination. The analysis has identified possible factors that contaminate water at specific sites, and particularly in protected shallow wells. In line with the application of precautionary and differentiated principles in groundwater protection (Xu and Reynders, 1995; Xu and Braune, 1995), these findings are adequate to guide the appropriate location of PSWs.
The DRASTIC approach (Table 7) provides a well-established means of assessing the potential of groundwater to become contaminated. Applying this approach, parameters used and described in this field study should provide a measure of the risk of contamination, as well as indicators that can provide a basis for more robust field measurements of all of the DRASTIC parameters. Downscaling such an approach to the catchment level poses its own challenges, particularly as DRASTIC was developed for regional-scale application and is data intensive (Robins, 2010). Nevertheless, using this technique provides a starting point for exploring more robust catchment-fitting methodologies for assessing groundwater contamination.
A simple vulnerability assessment scorecard technique developed by Robins et al. (2003) and applied in Mangochi, Southern Malawi, is more suitable for assessing the effect of site characteristics on groundwater contamination. The technique is based on the DRASTIC principles, but, instead of being quantitative and data intensive, it is qualitative, subjective and site-specific which makes it applicable at catchment, sub-catchment and village level (Robins, 2010). However, the scorecard also relies on data derived from a comprehensive and well-documented drilling programme.
Theoretically, areas with low slope tend to be more vulnerable to groundwater contamination as these are areas where water can pool for a longer period of time, thereby allowing a greater infiltration and hence a greater potential for contamination migration (Rahman, 2008). The location of water sources in low-lying wet areas (Fig. 5) poses a threat of groundwater contamination, partly because aquifers in such areas are likely to be in close hydraulic connection with surface water. Rahman (2008), working on the Great North Indian Plain, observed that the PSWs that were more contaminated were located in relatively flat areas. This is similar to our findings.
Factors responsible for the contamination of drinking-water sources cannot be deduced based on specific diagnostic information assessments and use of the DRASTIC approach alone. Field observations on location of water sources versus latrines (Fig.4), low-lying wet areas (Fig.5) and slope aspects (Table 6) provided significant additional information to guide proper location of water sources and sanitation services, in addition to knowledge of the local hydrogeologic environment. Robins et al. (2007) emphasize that factors observed in the field, such as topography and slope, vegetation and land use, are vital in assessing potential factors explaining contamination of ground-water sources at the catchment level. Robins et al. (2007) further give the example of how local surface water pools, resulting from rainfall, create a concentrated and prolonged zone of potential infiltration of contaminants, and of how cracks or fractures can offer direct and rapid pathways from groundwater to the water table. This justifies the use of both field observation and measurement techniques to explain possible factors for observed contamination.
Attempt to assess groundwater contamination in basement complex aquifer
Discussion of groundwater quality protection needs to be conducted within the context of set criteria for water quality, with an emphasis on the hydrogeological condition of the area where such water resides. For example, flow patterns of groundwater in the aquifer need to be explored to explain general contamination in the aquifer. In this study, the groundwater flow pattern was assumed to follow the topographic structure. Our findings provided adequate preliminary evidence to caution water service providers on the siting of PSWs. Our assumption on groundwater flow patterns was based on basic principles of groundwater flow: that water moves from a higher to lower hydraulic head through the most permeable parts of the geologic structure; and that recharge depends on the characteristics of the uppermost geologic and soil layers and on slope.
As the study area is a basement complex aquifer (Fig. 2), flow processes were assumed to take place via 2 scenarios:
fractured hard rock aquifer with preferential flow pattern along faults and fractures; and
primary unconfined aquifer on top with secondary confined aquifer at the bottom.
The groundwater flow pattern in a fractured rock aquifer can follow lineaments such as faults. Preferential flow along lineaments is common during recharge in this type of aquifer with almost no natural protective layer to help attenuate contamination along the faults. Although no pathogenic bacteria were detected in boreholes in this study area, the need to avoid siting water sources in such lineament environments exists, in line with precautionary and differentiated principles (Fig. 2).
A primary unconfined aquifer has alluvium material which usually filters contaminants originating on the surface through its particles and pore spaces. The alluvium materials act as a natural protective layer. Uniform recharge occurs in the unsaturated zone and contaminants travel freely down to the water table. The difference in pathogen concentration levels in water sources in such environments will depend on the thickness of the alluvial material, which attenuates, adsorbs and filters microbes as they travel through it to the aquifer, and on the slope between the water source and source of contaminants, among other factors. MacDonald et al. (2005) point out that shallow soil layers between aquifers and the surface result in high vulnerability of contamination. With depths of PSWs in this study ranging from 3.0 to5.5 m, with average of 4 m (Table 2), higher concentrations of allogeneic bacteria are expected. Upper Kango PSW was the only water point sampled that is located in an alluvial aquifer and displayed the highest concentration levels of both allogeneic and pathogenic bacteria. This is likely due to the effect of slope and thin alluvium material, as observed by MacDonald et al., (2005).
General physicochemical and microbial safety of groundwater sources
Findings regarding the concentrations of inorganic solutes in this study agree with those of Pritchard et al. (2007; 2009; 2010), working in Malawi, who concluded that the quality of drinking water from groundwater sources, both boreholes and shallow wells, in terms of physicochemical status, is within acceptable limits as set by both WHO (2008) and MBS (2005). However, the pH of most of the sampled water was < 6.5, and therefore outside the MBS (2005) and WHO (2008) recommended limits, with a violation fraction of 83% (Table 1). Cantor et al. (2000) and Hoke (2005) discussed the potential negative effects of low pH values on drinking water when stored in metal containers. The low pH values can lead to health concerns associated with corrosion of the metal containers. Fortunately, 99% of the sampled households keep their drinking water in ceramic (45%) and plastic containers (44%). These findings have wider application to households in this study area and other areas with similar geology. There is a need to encourage communities in such areas to keeping their drinking water in ceramic or plastic containers.
Social vulnerability: Contamination of water sources and household practices in water handling
Theoretically, groundwater provides safe drinking water (Xu and Braune, 2010), but some groundwater sources clearly do not provide potable water. This study has demonstrated how communities in northern Malawi are vulnerable to contracting waterborne diseases when they depend on certain groundwater sources as their sole source of drinking water. Field measurements of high levels of E. coli in PSWs confirmed that these sources remain risky. Hence, guidelines and regulations about such sources require revision in order for these sources to provide communities with potable water. Renewed reflection on PSWs as safe sources for potable water in rural areas requires commitment of both key governmental and private-sector agencies for rural water and sanitation services. Scaling up PSWs as a means of providing potable water to rural communities seems risky and counter-productive. With only 40% of Malawi's people having access to safe drinking water (Pritchard et al., 2007), the country faces difficulties in achieving the drinking-water MDG by 2015. Data from this study indicate that reliance on PSWs will not help Malawi to reach this goal. In addition, it is clear that there are deficiencies in hygiene practices associated with collection, transport and storage of water in households. Even when water at the source was free of bacterial contamination, water tested in the households was often contaminated with E.coli. Such household contamination indicates the necessity to disinfect drinking water at point-of-use. A possible door-to-door health education campaign might enable communities to improve hygiene practices.
Scientific versus socio-economic appropriateness of chlorinating drinking water
A review by Nath et al. (2006) on hygiene practices of households showed that improving the microbial quality of household-stored water using point-of-use treatment and safe storage practices reduces the incidence of waterborne disease in communities and households by up to 50%, even in the absence of other programmes. This study indicated that the use of Waterguard® chlorination significantly reduced contamination by E.coli bacteria in household-stored water, and similar health benefits to those observed by Nath et al. (2006) were therefore expected, despite the fact that chlorine failed to eliminate all bacteria in drinking water. Similar results were observed in rural south India where Firth et al. (2010) studied point-of-use treatment to decrease contamination and found that use of chlorine reduced coliform counts to potable levels but did not eliminate all of the bacteria.
Turbidity, temperature and pH have been identified as crucial physical or chemical parameters that affect the effectiveness of chlorine in drinking water (WHO, 2008; 2009). Theoretically, chlorine treatment in drinking water does not effectively penetrate suspended silt and organic particles where bacteria may reside (WHO, 2009). The higher the turbidity levels, the higher the risk of gastrointestinal diseases (WHO, 2008). Turbid materials can shield pathogens thereby interfering with effectiveness of both chlorine and ultraviolet sterilisation of water (WHO, 2008; 2009). For effective chlorination treatment, water should be at turbidities < 30 FNU. The overall average water turbidity in sampled households was low, around 2.7 FNU, at which level it should not impede sterilisation with chlorine. In fact, the highest recorded turbidity was 25.8 FNU.
In water with a temperature above 18ºC, chlorine should be in contact with the water for at least 30 min. If the water is colder the contact time should be increased (WHO, 2009). The average temperature for water stored in sampled households was 25ºC. Therefore, the temperature should not have had negative impacts on chlorination effectiveness. Health workers teach residents to wait for 30 minutes after pouring chlorine in their drinking water. However, investigation of compliance to this instruction was beyond the scope of this study.
Vanderslice and Briscoe (1993) observed that drinking water from contaminated groundwater sources is more risky than drinking water that has been contaminated in one's own household, due to the fact that people build up resistance to pathogens to which they are commonly exposed at household level. The need to chlorinate drinking water from PSWs cannot be overemphasized; yet the current rate of usage of chlorine is discouraging. The use of chlorine in water from PSWs confirms that it is at least a partially-effective solution but it remains a socio-economically unsuitable answer for the majority of rural residents. In rural south India, Firth et al. (2010) found the addition of chlorine to be widely unaccepted among the population sampled. Conversely, participants for this study were willing to use chlorine but they could not afford to purchase it on a regular basis. The observed poverty levels in northern Malawi in terms of income and earnings suggest difficulties in the affordability, sustainability and feasibility of scaling up such an intervention for households that derive their water from PSWs. Tumwine (2005) observed similar barriers among East African communities. In this study, only 11% of the sampled households were using chlorine and only 1 household out of the 23 that drew water from PSWs used chlorine effectively. The lack of widespread use of water treatment and especially the low use among people using PSWs raises fundamental research questions regarding the ability of communities to adopt chlorination as a widespread practice. As a case study, these findings have wider implications in poverty-prone rural areas where chlorine use is encouraged.
Conclusions
The study has demonstrated the difficulty in applying popular methods such as DRASTIC in assessing vulnerability to groundwater contamination at village level within a sub-catchment, and to guide development of rural water supplies. However, this study has illustrated how feasible and useful such approaches can be to rural water service providers if used in combination with field observations and measurements. The need exists to further refine the recently-developed scorecard methodologies for assessing vulnerability of small-scale areas such as villages, especially in terms of the data required and its subjectivity.
This study has shown that even with a basic knowledge of local hydrogeologic environments, using the geology of the area and simple field measurements and observations, it is possible to provide adequate preliminary evidence on ground-water contamination in order to guide developers of rural water supplies on appropriate siting of water sources, and especially protected shallow wells.
It has been revealed that people's preferred traditional practices (local knowledge) in relation to containers for drinking water storage has potential to protect them from potential dangers of their low pH drinking water. There is a need to further explore existing local knowledge practices on water utilisation and management.
The discussion has revealed how human health in rural communities becomes vulnerable to water-related diseases through drinking water from protected shallow wells. It has been shown that scaling up such water sources remains risky to people's health and counterproductive to achieving the MDG on widening access to safe and clean water. A proactive review on guidelines for PSWs is unavoidable.
The study has shown the effectiveness of chlorine in reducing pathogenic bacteria in drinking water, as well as possible socio-economic factors that affect the effectiveness of such intervention. Despite being a scientifically-proven effective solution to water purification, its low adoption remains a risk factor for water-related disease. Hence, preventing contamination of water sources seems more appropriate than curative intervention at household levels in poverty-prone rural communities. These findings suggest the need to apply precautionary and differentiated principles to protect the quality of ground-water when providing water services to rural communities.
Acknowledgements
The authors gratefully acknowledge the Norwegian Programme for Development, Research and Higher Education (NUFU) for funding research expenses under a NUFU project entitled 'Capacity building in Water Sciences for improved assessment and management of water resources'. We also thank the Fulbright U.S. Scholar Program and the Fulbright Regional Sharing Program, both sponsored by the U.S. Department of State and the Bureau of Educational and Cultural Affairs. We are especially grateful to the people in the communities of the Limphasa catchment for allowing sampling of their water sources and household water and for all their help and hospitality. Special appreciation is expressed to all of the people who took part in the field activities. The reviewers are thanked for their comments on the manuscript.
Corrigendum
Please note corrigendum – see last page of PDF.
References
APHA (AMERICAN PUBLIC HEALTH ASSOCIATION) (1995) Standard Methods for the Examination of Water and Wastewater (19th edn.). United Book Press Inc., Baltimore. [ Links ]
ALLER LJH, LEHR JH and PETTY R (1987) DRASTIC: Standardized System to Evaluate Groundwater Pollution Potential using Hydrogeologic Settings. National Water Well Association, Worthington, Ohio, United States of America. [ Links ]
BAUMANN E and DANERT K (2008) Operation and Maintenance of Rural Water Supplies in Malawi. SKAT Study Findings. UNICEF, Lilongwe, Malawi. [ Links ]
BORDALO AA and SAVVA-BORDALO J (2007) The quest for safe drinking water: An example from Guinea-Bissau (West Africa). Water Res. 41 (13) 2978-2986. [ Links ]
CANTOR AF, JAE PE, PARK K and VAIYAVATJAMAI P (2000) The effect of chlorine on corrosion in drinking water systems in chlorine's effect on corrosion in drinking water system. Midwest Technology Assistance Center, Urbana-Champaign, University of Illinois, USA. [ Links ]
CONBOY MJ and GOSS MJ (2000) Natural Protection of Groundwater against Bacteria of Fecal Origin J. Contam. Hydrol. 43 1-24. [ Links ]
CRONIN AA, BRESLIN N, GIBSON J, and PEDLEY S (2006) Monitoring source and domestic water quality in parallel with sanitation risk identification in Northern Mozambique to prioritize protection interventions. J. Water Health 4 (3) 333-345. [ Links ]
FIRTH J, BALRA JV, MULIYIL J, ROY S, RANI LM, CHANDRE-SEKHAR R and KANG G (2010) Point-of-use interventions to decrease contamination of drinking water: A randomized, controlled pilot study on efficacy, effectiveness and acceptability of closed containers, Moringa oleifera and in-home chlorination in rural South India. Am. J. Trop. Med. Hyg. 82 (5) 759-765. [ Links ]
FREEDMAN D, PISANI R and PURVES R (1998) Statistics (3rd edn.). WW Norton Publishing, New York. [ Links ]
HOKO Z (2005) An assessment of the water quality of drinking water in rural districts in Zimbabwe: The case of Gokwe South Nkayi, Lupane and Mwenezi Districts. Phys. Chem. Earth 30 859-866. [ Links ]
HOWARD G, PEDLEY S, BARRETT M, NALUNBENGA M and JOHAL K (2003) Risk factors contributing to microbiological contamination of shallow groundwater in Kampala, Uganda. Water Res. 37 3421-3429. [ Links ]
KIM YJ and HAMM SY (1999) Assessment of the potential for groundwater contamination using the DRASTIC technique, Chongju Area, South Korea. Hydrogeol. J. 7 227-235. [ Links ]
MALAWI BUREAU OF STANDARDS (2005) Malawi Standards: Borehole and Shallow Well Water Quality-Specification. Malawi Standards Board, Blantyre, Malawi, URL: mbs@malwi.net. [ Links ]
MALAWI GOVERNMENT (2008a) The Second National Communication of the Republic of Malawi to The United Nations Framework Convention on Climate Change. Climate Change Project Office; Department of Environmental Affairs, Lilongwe, Malawi. [ Links ]
MALAWI GOVERNMENT (2008b) Population and Housing Census 2008, Main Report. Malawi Government, National Statistics Office, Zomba, Malawi. [ Links ]
MALAWI GOVERNMENT (2005) The 2005 National Water Policy. Ministry of Irrigation and Water Development, Malawi Government, Lilongwe, Malawi. [ Links ]
MALAWI GOVERNMENT (1986) National Water Resources Master Plan, Report and Appendices. Department of Water Resources, Ministry of Works and Supplies, Lilongwe, Malawi. [ Links ]
MACDONALD AM and DAVIES J (2000) A Brief Review of Groundwater For Rural Water Supply in Sub-Saharan Africa. Technical Report WC/00/33. Overseas Geology Series. British Geology Survey and Department for International Development, Nottingham. [ Links ]
MACDONALD A, DAVIES J, CALOW R and CHILTON J (2005) Developing groundwater: A guide for rural water supply. ITDG Publishing, Warwickshire. [ Links ]
MKANDAWIRE T (2008) Quality of groundwater from shallow well in selected villages in Blantyre District, Malawi. Phys. Chem. Earth 807-811. [ Links ]
NATH KJ, BLOOMFIELD SF and JONES M (2006) Household water storage, handling and point-of-use treatment. A review commissioned by the International Scientific Forum on Home Hygiene. URL: http://www.ifh-homehygiene.org (Accessed June 2010). [ Links ]
NDIMBWA S and MPOHA OS (2008) Personal communication 5-10 July, 2008. Steven Ndimbwa, Health Surveillance Assistant, and Oscar Stalin Mpoha, District Environmental Health Officer, PO Box 63, Nkhata Bay, Malawi. [ Links ]
PRITCHARD M, CRAVEN T, MKANDAWIRE T, EDMONDSON AS and O'NEILL JG (2010) A study of the parameters affecting the effectiveness of Moringa oleifera in drinking water purification in Malawi. Phys. Chem. Earth 35 791-797. [ Links ]
PRITCHARD M, MKANDAWIRE T, EDMONDSON A, O'NEILL JG and KULULANGA G (2009) Potential of using plant extract for purification of shallow well water in Malawi. Phys. Chem. Earth 34 799-805. [ Links ]
PRITCHARD M, MKANDAWIRE T and O'NEILL JG (2007) Biological, chemical and physical drinking water quality from shallow well in Malawi: Case study of Blantyre, Chiradzulu and Mulanje. Phys. Chem. Earth 32 1167-1177. [ Links ]
RAHMAN A (2008) A GIS based DRASTIC model for assessing groundwater vulnerability in shallow aquifer in Aligarh, India. Appl. Geogr. J. 28 32-53. [ Links ]
ROBERTS L, CHARTIER Y, CHARTIER O, MALENGA G, TOOLE M and RODKA H (2001) Keeping clean water clean in a Malawi Refugee Camp: A randomized intervention trial. Bull. World Health Organ. 79 (4) 280-287. [ Links ]
ROBINS NS (2010) A low tech approach to evaluating vulnerability to pollution of basement aquifers in sub-Saharan aquifer. In: Xu Y and Braune E (eds.) (2010) Sustainable Groundwater Resources in Africa: Water Supply and Sanitation Environment. UNESCO International Hydrological Programme, Paris, France. [ Links ]
ROBINS NS, DAVIES J, HANKIN P and SAUER D (2003) Groundwater and data: An African Experience. Waterlines 21 (4) 19-21. [ Links ]
ROBINS NS, CHILTON PJ and COBBING JE (2007) Adapting existing experience with aquifer vulnerability and groundwater protection in Africa. J. Afr. Earth Sci. 47 30-38. [ Links ]
SNEDECOR GW and COCHRAN WG (1980) Statistical Methods (7th edn.). Iowa State University Press, Ames, Iowa. [ Links ]
SOPHOCLEOUS M and BUCHANAN RC (2003) Groundwater recharge in Kansas. URL: http://www.kgs.ukans.edu/Publications/ pic22/pic22_1.html (Accessed December, 2010) [ Links ]
TUMWINE JK (2005) Clean Drinking Water for Homes in Africa and other Less Developed Countries. Br. Med. J. 331 468-469. [ Links ]
UNICEF (UNITED NATIONS CHILDREN'S FUND) (2005) Water, Environment and Sanitation; Malawi Country Profile. UNICEF Malawi, Lilongwe, Malawi. [ Links ]
UN (UNITED NATIONS) (2006) The Millennium Development Goals Report 2006. United Nations, New York. [ Links ]
VANDERSLICE J and BRISCOE J (1993) All coliforms are not created equal: a comparison of the effects of water sources and in-house contamination on infantile disease. Water Resour. Res. 29 (7) 1983-1995. [ Links ]
WEAVER JMC, CAVE L and TALMA AS (2007) Groundwater Sampling: A Comprehensive Guide for Sampling Methods. WRC Report No. TT 303/07. Water Research Commission, Pretoria. [ Links ]
WONNOCOTT RJ and WONNACOTT TH (1985) Introductory Statistics (4th edn.). John Wiley and Sons, New York. [ Links ]
WORLD BANK (2006) The World Bank Annual Report 2006: Operational Summary, Fiscal 2006. World Bank, Washington DC. [ Links ]
WORLD BANK (2005) Global Monitoring Report: Millennium Development Goals from Consensus to Momentum. World Bank, Washington DC. [ Links ]
WHO (WORLD HEALTH ORGANIZATION) (2009) Measuring Chlorine levels in Water Supplies: Technical Note for Emergencies. Final Draft-2009. WHO, Leicestershire. [ Links ]
WHO (WORLD HEALTH ORGANIZATION) (2008) Guidelines for Drinking-Water Quality (3rd edn.), Volume 1. Recommendations. WHO, Geneva (web version). [ Links ]
WRIGHT J, GUNDRY S and CONROY R (2004) Household drinking water in developing countries: a systematic review of microbiological contamination between source and point-of-use. Trop. Med. Int. Health 9 (1) 106-117. [ Links ]
XU Y and BRAUNE E (eds.) (2010) Sustainable Groundwater Resources in Africa: Water Supply and Sanitation Environment. UNESCO International Hydrological Programme, Paris, France. [ Links ]
XU Y and BRAUNE E (1995) A guideline for groundwater protection for the Community Water Supply and Sanitation Programme. Department of Water Affairs and Forestry, Pretoria, South Africa. [ Links ]
XU Y and REYNDERS AG (1995) A three-tier approach to protect groundwater resources in South Africa. Water SA 21 (3) 177-186. [ Links ]
YU IH, KIM YJ, KIM WY, LEE DY, KIM JY, LEE CW and LEE SR (1992) Advanced technology for Environmental Geologic Mapping (Pt.I). KR-92(T)-1, Korea Institute of Geology, Mining and Minerals, Ministry of Science and Technology, Taejon, South Korea. [ Links ]
Received 21 May 2011; accepted in revised form 25 June 2012.
* To whom all correspondence should be addressed. ffi +27 21 959-2439; fax: +27 21 959-3118; e-mail: yxu@uwc.ac.za














