Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
Water SA
versión On-line ISSN 1816-7950
versión impresa ISSN 0378-4738
Water SA vol.38 no.1 Pretoria ene. 2012
Removal and transformation of hexavalent chromium in sequencing batch reactor
Siew-Leng Loo; Kwok-Yii Leong; Poh-Eng Lim*
School of Chemical Sciences, Universiti Sains Malaysia, 11800 Penang, Malaysia
ABSTRACT
The objectives of this study are to evaluate the efficiency of removal of hexavalent chromium (Cr(VI)) in a sequencing batch reactor (SBR) and to ascertain the fate of Cr(VI) in the treatment process. An SBR was operated with the FILL, REACT, SETTLE, DRAW and IDLE periods in the time ratio of 2:12:2:1.5:6.5 for a cycle time of 24 h. The study was divided into 5 phases with the addition of 0.5, 2.0, 3.0 and 5.0 mg/ℓ of Cr(VI) in Phases II, III, IV and V for a duration of 46, 75, 43 and 16 operational cycles, respectively. The Cr(VI) removal efficiencies for SBR were found to be 79.8, 88.4 and 99.8% in Phases III, IV and V, respectively. The results revealed that Cr(VI) removal efficiency improved with acclimated activated sludge. Determination of Cr in the suspended sludge showed that around 95% of the Cr species were Cr(III). Determination of Cr concentration profiles during the FILL and REACT periods showed that the predominant species was Cr(III) as Cr(VI) was bio-reduced. The proposed Cr(VI) removal mechanism involves bioreduction to Cr(III) which was subsequently precipitated and adsorbed by activated sludge. Precipitation rather than sorption is envisaged to be the main path of removal of Cr(III) from the solution.
Keywords: Sequencing batch reactor, hexavalent chromium, removal, transformation, mechanism
Introduction
Chromium species in the +3 and +6 oxidation states are more commonly found in the environment. Cr(VI) is widely used in industries such as electroplating, metal finishing, wood preservation and leather tanning. It exists as different forms of oxyanions depending on the pH of the solution. Under strongly acidic conditions (pH<1), Cr(VI) exists as Cr2O7 2- , whereas between pH 2 and 6 it exists as HCrO4 -. Under neutral or alkaline conditions, Cr(VI) is present as CrO42- (Kimbrough et al., 1999). These compounds are highly toxic due to their strong oxidising nature. Owing to the toxicity, exposure to Cr(VI) presents an acute health risk and chronic exposure may lead to mutagenesis and carcinogenesis. In contrast, Cr(III) is much less toxic and is essential for mammals in trace quantities (Orozco et al., 2008).
Conventional methods of removing Cr(VI) include chemical reduction to Cr(III) followed by precipitation under alkaline conditions, removal by ion exchange, or adsorption by various materials (Kumar et al., 2007; Orozco et al., 2008). However, most of the conventional methods are costly and involve the generation of a large amount of secondary wastes which preclude an efficient and cost-effective technology in treating chromium-containing wastewater. Biological treatment methods such as the conventional activated sludge process and sequencing batch reactor (SBR) may be a better alternative due to their lower operational cost, steady performance and easy recovery of some valuable metals. Several researchers have shown that the activated sludge process is effective in treating chromium-containing wastewater (Imai and Golyna, 1990; Stasinakis et al., 2004; Chen and Gu, 2005). The SBR is a modified version of the activated sludge process which carries out functions of equalisation, treatment and sedimentation in the same reactor in a temporal rather than spatial sequence. It offers the advantages of better tolerance to shock load, good settleability, simplicity and ease in operation as well as compact layout. Sirianuntapiboon and Hongsrisuwan (2007) have shown that SBR was effective in removing Cu and Zn in industrial and synthetic wastewaters.
Studies on Cr(VI) removal mechanisms using various biosorbents in batch systems under acidic conditions have been conducted (Reddad et al., 2003; Park et al., 2005a; Park et al., 2005b; Xu et al., 2007; Park et al., 2008; Wang et al., 2009). The mechanisms proposed by these researchers were similar and generally involve the adsorption of the anionic Cr(VI) on protonated adsorption sites, followed by the reduction to Cr(III) by electrons donated by adjacent functional groups. However, the removal mechanism of Cr(VI) in a bioreactor may be different due to the difference in pH which affects the surface charge of the biomass and the complexity of the solution chemistry. Several researchers have investigated the Cr(VI) removal mechanism in the activated sludge process (Imai and Gloyna, 1990; Stasinakis et al., 2004; Chen and Gu, 2005). Among them, Stasinakis et al. (2004) proposed a 2-step mechanism involving the bioreduction of Cr(VI) to Cr(III) and subsequent adsorption of Cr(III) onto suspended solids. A relatively small portion of Cr(III) (< 14%) could be precipitated (Stasinakis et al., 2003). The conclusion was drawn solely based on the findings from the speciation study conducted on the suspended solids and the effluent, but the transformation undergone by Cr(VI) during the treatment process was not discussed. In light of the above observations, the objectives of this study are: (i) to evaluate the removal efficiency of Cr(VI) in SBR under relatively low Cr(VI) loading rates and (ii) to ascertain the fate of Cr(VI) in the treatment process.
Experimental
Experimental set-up and operation
A plexiglass reactor with dimensions of 19.5 x 25 x 20 cm (width x length x height) and a working volume of 10 ℓ was operated on a 24-h cycle with the FILL, REACT, SETTLE, DRAW and IDLE periods in the time ratio of 2:12:2:1.5:6.5. Mixing was provided during the FILL period by using an ejector. During each cycle, 7 ℓ of influent and treated effluent were introduced and drawn, respectively. The sludge age was controlled at around 10 days. The base mix for the reactor was of the following composition (concentration in mg/ℓ): bactopeptone (188), sucrose (563), KH2PO4(35), KH2PO4(180), (NH4)2SO4(226), NaHCO(576), MgSO4(49), FeCl3.6H2O (10), CaCl(42). The seed sludge for the reactor was obtained from a local municipal wastewater treatment plant. Ethanol solution (≈ 4.6 % v/v) with a theoretical COD of 75.8 mg O2/mℓ was added as a carbon source on the commencement of the anoxic period. The study was divided into 5 phases. In Phase I, the reactor was operated without the addition of Cr(VI) for a period of 60 operational cycles. The reactor was then loaded with 0.5, 2, 3 and 5 mg/ℓ of Cr(VI) during the FILL period in Phases II, III, IV and V, respectively, for a duration of 46, 75, 43 and 16 operational cycles, respectively.
Determination of performance parameters
The mixed liquor suspended solids (MLSS) concentration and sludge volume index (SVI) were determined regularly in every phase following the Standard Methods (APHA, 1998). The Cr(VI) removal efficiency was evaluated by determining the concentrations of Cr(VI) and total Cr in the effluent for every operational cycle. Wasted sludge for every operational cycle was obtained to determine the concentration of total Cr accumulated in the sludge. During each phase, concentration profiles of Cr(VI), Cr(III) and total Cr in the aqueous and particulate phases during the FILL and REACT periods were determined. Mixed liquor samples were obtained from the reactor at a predetermined time interval and separated into the aqueous and particulate phases via filtration. The dissolved oxygen (DO) concentration and pH profiles were determined in situ by using a DO meter (YSI 550A) and a pH meter (Cyber Scan 500), respectively.
Total Cr and Cr(VI) concentrations in the aqueous phase were determined following Method No. 3111 B/3113 B and Method No. 3500-Cr B, respectively (APHA, 1998). Total Cr concentration was analysed by direct analysis of the filtrate using a flame atomic absorption spectrophotometer (FAAS) (Perkin Elmer AAS 3100) or a graphite furnace-AAS (Perkin Elmer Analyst 100), depending on the concentration ranges, while Cr(VI) was measured colourimetrically using an UV-Vis spectrophotometer (Hitachi U-2000). The Cr(VI) concentration in the suspended solids was determined by the selective leaching of Cr(VI) using sodium carbonate followed by final detection by graphite furnace-AAS (Perkin Elmer Analyst 100) as described in Elci et al. (2010). Total Cr concentration in the suspended sludge was determined by open digestion with concentrated nitric acid followed by FAAS analysis (APHA, 1998). For the aqueous and solid phases, Cr(III) concentrations were calculated from the difference between the total Cr and Cr(VI) concentrations.
FTIR analysis
Samples of the suspended sludge before and after exposure to different concentrations of Cr(VI) were obtained and dried in the oven (Memmert 400) at 105ºC. A minute amount of dried sludge was ground with dried KBr into fine particles by using mortar and pestle. The mixture was made into a translucent disk by compressing the mixture in a die using a manual hydraulic press. The resultant KBr disk was analysed using a FTIR spectrophotometer (Perkin Elmer 2000).
Adsorption studies
Equilibrium adsorption studies were conducted to determine the adsorption capacity of the suspended activated sludge for Cr(VI) and Cr(III) at pH 6, 7 and 8. Dried activated sludge of 0.100 g in weight and <1 mm in size was shaken at 250 r/ min with 20.0 mℓ of Cr(VI) and Cr(III) solutions of concentrations ranging from 1.0 to 10.0 mg/ℓ, for a duration based on the preliminary contact time study. After shaking, the sample was filtered and the filtrate was analysed for Cr(VI) and Cr(III) concentrations using the analytical methods described in the previous section. The point of zero charge (pHpzc) of the activated sludge was also determined (Ofomaja et al., 2009).
Results and discussion
MLSS concentrations and SVI values
The trends of MLSS concentrations and SVI values of the suspended sludge during the operational period are shown in Fig. 1. The decrease in MLSS concentration in Phase I (before Cr(VI) addition) was primarily due to sludge age control. At the beginning of Phase II, the MLSS concentration decreased drastically with the addition of 0.5 mg/ℓ of Cr(VI) due to the inhibitory effects of Cr(VI) on the growth of the biomass. Once the biomass had been acclimated to Cr(VI), the MLSS concentration increased, even at higher Cr(VI) dosages, in Phases IV and V. These results agree with those of Yetis et al. (1999), who reported that Cr(VI) exerted stimulatory effects on biomass yield up to 25 mg/ℓ of Cr(VI) in a continuous activated sludge system with acclimated cultures.
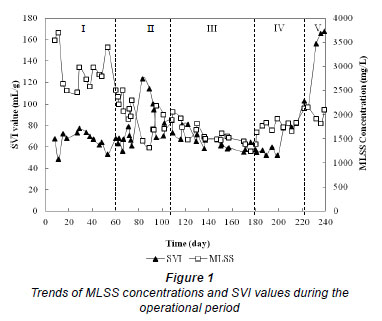
Figure 1 shows that the SVI values effectively varied between the values of 60 to 100 mℓ/g throughout Phases I, II, III and IV, which is indicative of good settleability and compressibility (Fig. 1). However, the SVI values increased drastically in Phase V indicating that the sludge had yet to be acclimated to higher Cr(VI) concentrations in this phase, which was of relatively short duration.
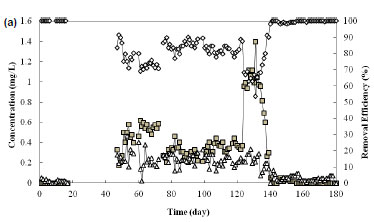
Cr removal
The Cr(VI) removal efficiency was determined by monitoring the concentration of Cr(VI) in the effluent. Figure 2a depicts the effluent Cr(III) and Cr(VI) concentrations and the Cr(VI) removal efficiencies in the SBR. During Phase II, the reactor was very efficient in Cr(VI) removal as practically no Cr(VI) was detected in the effluent. When the influent Cr(VI) was increased to 2.0 mg/ℓ in Phase III, the mean removal efficiency decreased briefly but recovered quickly to 80±6%. A similar trend was observed in Phase IV in which an increase of Cr(VI) to 3.0 mg/ℓ resulted in a drastic decrease in the removal efficiency for a brief period before recovering to more than 99%. In Phase V, the removal efficiency remained above 99% despite an increase of Cr(VI) to 5.0 mg/ℓ. The recovery in removal efficiency may be attributed to biomass acclimation to Cr(VI)containing feed. Chen and Gu (2005) also found that acclimated activated sludge had a higher Cr(VI) removal efficiency.
In comparison to other treatment systems, the SBR in this study was able to achieve a relatively high Cr(VI) removal efficiency. In comparison, Imai and Gloyna (1990) reported that the Cr(VI) removal efficiency for an aerated fed-batch activated sludge reactor operated at pH 7 and 8 was less than 25%. Stasinakis et al. (2004) reported a mean Cr(VI) removal of 42±11% using an aerobic activated sludge system. The higher Cr(VI) removal efficiency achieved in this study might be due to the employment of agitated FILL period without aeration. At such a low-DO environment, the reduction of Cr(VI) to Cr(III) was highly favourable thereby decreasing the concentration of Cr(VI). Stasinakis et al. (2004) studied the effect of aerobic and anoxic reactors in the reduction of Cr(VI) and found that the inclusion of an anoxic reactor prior to the aerobic reactor had improved the mean Cr(VI) removal to 84±9% when the influent Cr(VI) concentration was at 1 mg/ℓ.
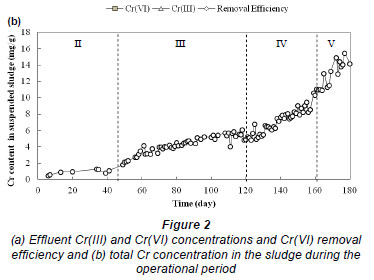
Since Cr introduced into the reactor was almost completely removed during most phases, the removed Cr species should be accumulated in the sludge. Therefore, samples of suspended sludge for each operational cycle were obtained for the determination of total Cr. Suspended sludge samples collected in Phases IV and V were also analysed for the content of Cr species and it was found that Cr(III) constituted 95±1% of Cr in the sludge. Figure 2b depicts the trend of total Cr concentrations in the suspended sludge with the operational period, which showed a general increase of Cr content in the sludge with time. The Cr species removed from the bulk solution could accumulate in the suspended sludge through processes such as adsorption, precipitation and intracellular uptake of Cr species by living cells (Imai and Golyna, 1996). The rates of increase in the Cr content in Phases II, III, IV and V were estimated to be 0.015, 0.044, 0.136 and 0.199 mg/g per day, respectively, based on a linear fit to the data for each phase shown in Fig. 2b. The higher rate of increase in Phases IV and V was attributed to the higher concentrations of Cr(VI) being added and higher Cr(VI) removal efficiencies.
FTIR analysis
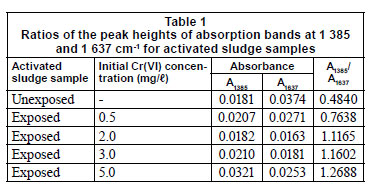
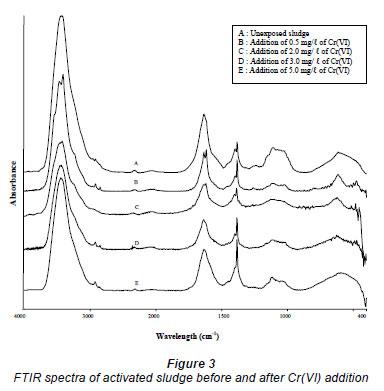
The adsorption sites on the activated sludge surface can be identified by analysing the FTIR spectra of the sludge before and after Cr(VI) addition (Fig. 3). The FTIR spectrum of the unexposed sludge displayed a broad absorption peak at 3 430 cm-1 which may be assigned to the presence of -O-H and -N-H functionalities. The peaks at 1 637 and 1 385 cm-1 were probably due to the C=O and the C-O stretching of carboxylate group. A shoulder at 1 107 cm-1 could be assigned to cellulosic -O-H. The high functionalities of the activated sludge indicate that adsorption may occur through surface complexation in addition to electrostatic attraction and ion exchange mechanisms. Two changes were observed in the FTIR spectra of the sludge after Cr(VI) addition. The first change was the weaker appearance of the shoulder at 1 107 cm-1 indicating the involvement of cellulosic -O-H in sequestering Cr(III) and to a lesser extent Cr(VI). This showed that extracellular polymer (EP) produced by the activated sludge played a role in the adsorption process because its main composition is polysaccharide (Brown and Lester,1979). The disappearance of the cellulosic -O-H after the adsorption of Cd by EP was also observed by Zheng et al. (2008). The second change in the FTIR spectra of the sludge after Cr(VI) addition was the relative height of the peaks at 1 637 and 1 385 cm-1. The maximum peak height of the 2 peaks was obtained by using a spectral calculator. Table 1 shows that the ratio of the peak height of adsorption due to C-O stretching to that of adsorption due to C=O stretching increased with initial Cr(VI) concentration. Pagnanelli et al. (2009) also observed a shift in the relative intensities of the carboxylate peaks at 1 621 and 1 418 cm-1, indicative of interaction between metal ions with carboxylic adsorbent sites.
Equilibrium adsorption studies
The adsorption data were fitted to the Freundlich and Langmuir models to determine the adsorption capacities of the dried suspended sludge for Cr(VI) and Cr(III). The linearised Freundlich and Langmuir isotherms are expressed, respectively, by the following equations:
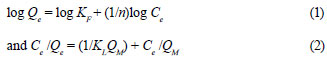
where:
Qe is the amount of Cr adsorbed per unit mass of the adsorbent at equilibrium (mg/g),
KF is a constant related to the adsorption capacity of the adsorbent,
n is a constant related to the affinity of the activated sludge for Cr,
Ce is the residual concentration of Cr in the dissolved phase after adsorption (mg/ℓ),
QM is the maximum adsorption capacity (mg/g) and
KL is a constant related to the affinity of adsorption (ℓ/mg).
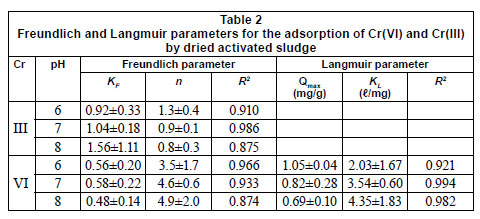
The Freundlich and Langmuir parameters are shown in Table 2. The Freundlich model adequately described (R2 > 0.87) the adsorption data for Cr(VI) and Cr(III). However, the Cr(III) adsorption data could not fit the Langmuir model. The KF values for Cr(III) were higher than those of Cr(VI) indicating that the sludge had a higher adsorption capacity for Cr(III). Table 2 shows a decreasing QM value for Cr(VI) adsorption with the increase in pH from 6 to 8. The pHpzc of the sludge was found to be 6.4 indicating that the net surface charge of the sludge was negative at pH values above 6.4. Thus, the electrostatic repulsion between the anionic Cr(VI) species and the negative surface charge of the sludge resulted in a relatively lower adsorption of Cr(VI) at higher pH values. This finding suggests that the accumulation of total Cr in the suspended sludge may in part be due to the adsorption of Cr(III) rather than Cr(VI).
Transformation of Cr(VI) during the FILL and REACT periods in SBR
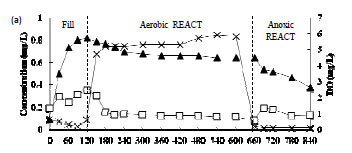
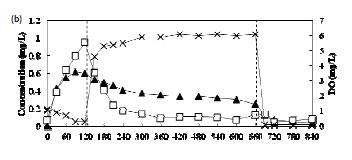
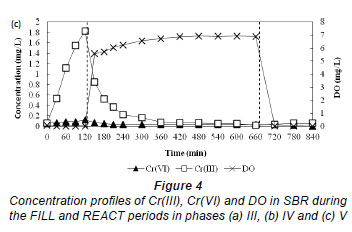
Figure 4a-c shows the concentration profiles of Cr(III), Cr(VI) and DO during the FILL and REACT periods on the 73rd, 43rd and 14th cycles in Phases III, IV and V, respectively. The pH of the reactor varied between pH 7 and 8. Figure 4a shows a build-up of Cr(VI) and relatively low Cr(III) concentrations during the FILL period in Phase III. This could be explained by the fact that acclimation had not yet been achieved. It was also observed that Cr(VI) removal during the anoxic REACT period was faster than the aerobic REACT period. The addition of ethanol solution into the reactor at the beginning of the anoxic period could have aided the reduction of Cr(VI) by acting as an electron donor. It has been shown that Cr(VI) reduction efficiency is dependent on the organic substrate concentration (Stasinakis et al., 2003). In Phase IV, there was more Cr(III) than Cr(VI) found during the FILL period (Fig. 4b) and in Phase V, a near-complete removal of Cr(VI) accompanied by a build-up of Cr(III) was observed. This result indicates that the activated sludge was acclimated to Cr(VI) and most of the Cr(VI) removed was reduced to Cr(III).
Figure 4c also provides the evidence that the removal of Cr(VI) via reduction to Cr(III) during the FILL period mainly occurred in the aqueous phase as intracellular reduction of Cr(VI) would be unlikely due to electrostatic repulsion between the net negative surface charge on the sludge at the operating pH range of 7-8 in the reactor and the anionic Cr(VI). The hypothesis is also supported by the observation of a relatively high Cr(III) concentration in the solution during the FILL period. If Cr(VI) reduction were to take place on the sludge, the resulting Cr(III) species would not be released easily to the solution as it would be attracted to the negatively-charged activated sludge. The relatively high Cr(III) concentration that was not removed during the FILL period in Phases IV and V might be due to complexation with the added organic substrates. It has been shown that metal precipitation ceased with increasing organic concentration (Remoundaki et al., 2007).
Cr(III) was observed to be removed from the solution very rapidly at the beginning of the aerobic REACT period in Phases IV and V (Fig. 4b and 4c). Precipitation is envisaged to be the main path of removal as the adsorption capacity of the biomass for Cr(III) has long been fulfilled at this stage. Steritt et al. (1981) found that the main Cr(III) removal mechanism in an activated sludge system operated at pH 7.4-7.7 was precipitation. The enhanced precipitation of Cr(III) upon the start of the aerobic REACT period in Phases IV and V may be the result of a combination of several factors including (i) increase in the pH of the mixed liquor, (ii) decreasing organic content due to microbial metabolism of organics in the presence of O2 and (iii) high Cr(III) concentration which had exceeded the solubility product of Cr(OH)3. The precipitated Cr(III) may then be physically entrapped in the activated sludge flocs or it may settle independently from the solution forming a part of the sludge.
Oxidation of Cr(III) to Cr(VI) was not observed during the aerobic REACT period even when the dissolved oxygen concentration was relatively high (Fig. 4b-c). This can be explained by the relatively slow rate of Cr(III) oxidation as compared to sorption and precipitation (Schroeder and Lee, 1975).
Conclusions
The Cr(VI) removal efficiencies for SBR were found to be 80, 89 and more than 99% when the influent Cr(VI) concentrations were 2, 3 and 5 mg/ℓ (loading rates of 1.4 x 10-3, 2.1 x 10-3, 3.5 x 10-3 kg/m3 day), respectively. The results revealed that Cr(VI) removal efficiency improved with acclimated activated sludge The adsorption data of Cr(VI) and Cr(III) fitted the Freundlich model fairly well. The adsorption study showed that the activated sludge had a relatively higher adsorption capacity for Cr(III) than for Cr(VI). The adsorption sites were identified as the carboxyl and cellulosic alcohol groups via the FTIR analysis. Determination of Cr in the suspended sludge showed that most of the Cr species were Cr(III). Determination of the Cr concentration profiles during the FILL and REACT periods showed that Cr(III) was the dominant species in the solution. Precipitation is envisaged to be the main path of removal for Cr(III).
References
APHA (1998) Standard Methods for the Examination of Water and Wastewater (20th edn.). American Public Health Association, Washington, DC. [ Links ]
BROWN MJ and LESTER JN (1979) Metal removal in activated sludge: The role of bacterial extracellular polymers. Water Res. 13 817-837. [ Links ]
CHEN Y and GU G (2005) Preliminary studies on continuous chromium(VI) biological removal from wastewater by anaerobic-aerobic activated sludge process. Bioresour. Technol. 96 1713-1721. [ Links ]
ELCI L, DIVRIKLI U, AKDOGAN A, HOL A, CETIN A and SOYLAK M (2010) Selective extraction of chromium(VI) using a leaching procedure with sodium carbonate from some plant leaves, soil and sediment samples. J. Hazard. Mater. 173 778-782. [ Links ]
IMAI A and GOLYNA EF (1990) Effects of pH and the oxidation state of chromium on the behaviour of chromium in the activated sludge process. Water Res. 24 1143-1150. [ Links ]
IMAI A and GOLYNA EF (1996) Speciation of chromium(III) in activated sludge. Water Environ. Res. 68 301-310. [ Links ]
KIMBROUGH DE, COHEN Y, WINER AM, CREELMAN L and MABUNI C (1999) A critical assessment of chromium in the environment. Crit. Rev. Env. Sci. Technol. 29 1-46. [ Links ]
KUMAR PA, RAY M and CHAKRABORTY S (2007) Hexavalent chromium removal from wastewater using aniline formaldehyde condensate coated silica gel. J. Hazard. Mater. 143 24-32. [ Links ]
OFOMAJA AE, NAIDOO EB and MODISE SJ (2009) Removal of copper(II) from aqueous solution by pine and base modified pine cone powder as biosorbent. J. Hazard. Mater. 168 909-917. [ Links ]
OROZCO AMF, CONTRERAS EM and ZARITZKY NE (2008) Modelling Cr(VI) removal by a combined carbon-activated sludge system. J. Hazard. Mater. 150 46-52. [ Links ]
PAGNANELLI F, MAINELLI S, BORNORONI L, DIONISI D and TORO L (2009) Mechanisms of heavy-metal removal by activated sludge. Chemosphere 75 1028-1034. [ Links ]
PARK D, YUN YS, JO JH and PARK JM (2005a) Mechanism of hexavalent chromium removal by dead fungal biomass of Aspergillus niger. Water Res. 39 533-540. [ Links ]
PARK D, YUN YS and PARK JM (2005b) Studies on hexavalent chromium biosorption by chemically-treated biomass of Ecklonia sp. Chemosphere 60 1356-1364. [ Links ]
PARK D, YUN YS, LEE HW and PARK JM (2008) Advanced kinetic model of the Cr(VI) removal by biomaterials at various pHs and temperatures. Bioresour. Technol. 99 1141-1147. [ Links ]
REDDAD Z, GERENTE C, ANDRES Y and LE CLOIREC P (2003) Mechanism of Cr(III) and Cr(VI) removal from aqueous solutions by sugar beet pulp. Environ. Technol. 24 257-264. [ Links ]
REMOUNDAKI E, HATZIKIOSEYIAN A and TSEZOS M (2007) A systematic study of chromium solubility in the presence of organic matter: consequences for the treatment of chromium-containing wastewater. J. Chem. Technol. Biotechnol. 82 802-808. [ Links ]
SCHROEDER DC and LEE GF (1975) Potential transformations of chromium in natural waters. Water Air Soil Pollut.4 355-365. SIRIANUNTAPIBOON S and HONGSRISUWAN T (2007) Removal of Zn2+ and Cu2+ by a sequencing batch reactor. Bioresour. Technol. 98 808-818. [ Links ]
STASINAKIS AS, THOMAIDIS NS, MAMAIS D, KARIVALI M and LEKKAS TD (2003) Chromium species behavior in the activated sludge process. Chemosphere 52 1059-1067. [ Links ]
STASINAKIS AS, THOMAIDIS NS, MAMAIS D and LEKKAS TD (2004) Investigation of Cr(VI) reduction in continuous-flow activated sludge systems. Chemosphere 57 1069-1077. [ Links ]
STERRITT RM, BROWN MJ and LESTER JN (1981) Metal removal by adsorption and precipitation in the activated sludge process. Environ. Pollut. Series A Ecol. Biol. A 24 313-323. [ Links ]
WANG XS, CHEN LF, LI FY, CHEN KL, WAN WY and TANG YJ (2009) Removal of Cr(VI) with wheat-residue derived from black carbon: Reaction mechanism and performance. J. Hazard. Mater. 175 816-822. [ Links ]
XU H, WONG YS, WONG MF and FUNG NYT (2007) Biosorption and bioreduction of Cr(VI) by a microalgal isolate, Chlorella miniata. J. Hazard. Mater. 146 65-72. [ Links ]
YETIS U, DEMIRER GN and GOKCAY CF (1999) Effect of chromium(VI) on the biomass yield of activated sludge. Enzyme Microb. Technol. 25 48-54. [ Links ]
ZHENG L, TIAN Y, DING AZ and WANG JS (2008) Adsorption of Cd(II), Zn(II) by extracellular substances extracted from waste activated sludge. Water Sci. Technol. 58 195-200. [ Links ]
Received 7 April 2011; accepted in revised form 14 December 2011.
* To whom all correspondence should be addressed. +604 653-3550; fax: +604 657-4854; e-mail: pelim@usm.my