Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
Water SA
versión On-line ISSN 1816-7950
versión impresa ISSN 0378-4738
Water SA vol.36 no.3 Pretoria abr. 2010
Life-history traits of Streptocephalus purcelli Sars, 1898 (Branchiopoda, Anostraca) from temporary waters with different phenology
ER De RoeckI,II; A WaterkeynII; L BrendonckII
IRoyal Belgian Institute of Natural Sciences, Freshwater Biology, Vautierstraat 29, 1000 Brussel, Belgium
IILaboratory of Aquatic Ecology and Evolutionary Biology, K.U.Leuven, Charles Deberiotstraat 32, 3000 Leuven, Belgium
ABSTRACT
Rock-pools have specific environmental characteristics, usually with multiple short and unpredictable hydroperiods in 1 season. This requires a specific set of life-history adaptations of their permanent inhabitants, which affects species occurrence. In Southern African rock-pools, large branchiopods are usually represented by Branchipodopsis species. During an intensive sampling campaign in 2004 in the Western Cape of South Africa, a population of Streptocephalus purcelli Sars, 1898 was discovered for the first time in a clear rock-pool with a short hydroperiod. Its usual habitat is a more turbid mud-pool with a longer hydroperiod. We investigated under standard laboratory conditions the differences in early life-history traits between the rock-pool population and a conspecific mud-pool population. Dormant eggs of the rock-pool population were smaller than those of the mud-pool population. The smaller larvae also hatched earlier. These characteristics are in accordance with what is expected for populations living in ephemeral systems. The rock-pool population, on the other hand, also exhibited a higher total hatching percentage, a longer hatching duration, a longer maturation time and a lower mortality. Although, deduced from the last mentioned life-history traits, S. purcelli seemed not well-adapted to unpredictable and ephemeral rock-pool habitats, they could probably occur due to the absence of the competitively stronger species in the region or due to dispersal and mass effects.
Keywords: Streptocephalus, temporary rock-pools, Anostraca, South Africa
Introduction
Local pool characteristics, such as hydroperiod, conductivity and temperature, are main factors determining species occurrence. In temporary aquatic habitats, the duration of time that pools contain water (hydroperiod) is one of the most important variables impacting population and community characteristics (Bilton et al., 2001, Eitam et al., 2004; Tavernini et al., 2005; Williams, 2006; Waterkeyn et al., 2008). Despite being subjected to time pressure and unpredictability, temporary pools often contain a rather diverse assemblage of permanent and temporary residents (Eitam et al., 2004; Williams, 2006). Some of the most striking permanent inhabitants are anostracans (fairy shrimps, Crustacea: Branchiopoda). To persist in temporary wetlands that have a short hydroperiod and are often irregularly filled, anostracans are characterised by a specific set of life-history traits (Wiggins et al., 1980; Lahr et al., 1999). Fairy shrimps survive the dry period by forming a resting egg bank that buffers the population from catastrophic events. Especially their early life-history traits (such as hatching fraction, maturation time and size of first brood) experience a strong time pressure in systems with a short hydroperiod (Hildrew, 1985; Brendonck et al., 2000; Mura et al., 2003). To counter the unpredictability of their habitat, anostracans exhibit a risk-spreading strategy, as not all eggs of 1 generation hatch within the next inundation. As such, the long-term fitness of a genotype is increased by spreading the high risk of mortality (Philippi and Seger, 1989; Simovich and Hathaway, 1997). This bet-hedging strategy is expected to occur in highly variable habitats, where there is a high possibility of abortive hatching (reproductive failure of a cohort) (Simovich and Hathaway, 1997).
Rock-pools in arid and semi-arid areas are very specific temporary habitats, because of their often extremely short hydroperiod and irregular hydrological regimes, which depend on their size and the local climate (Ranta, 1982; Jocqué et al., 2006; Hulsmans et al., 2008). Some examples of such rock-pool habitats are the outcrops in arid and semi-arid Southern Africa (Hamer and Martens, 1998), the gnammas of Australia (Bayly, 2001) and the potholes in the USA (Chan et al., 2005). On account of their specific environmental characteristics, rock-pools usually contain a small set of highly specialised (usually congeneric) anostracan species in a certain region. In North Africa and Europe these are Tanymastix species, in North America Branchinecta species, while in Southern Africa Branchipodopsis species are known as the rock-pool specialists (Thiéry, 1986; Brendonck et al., 2000). Branchipodids cope with the time pressure of their habitat by reaching maturity within the 1st week after inundation and producing almost daily small broods of resting eggs (Brendonck et al., 2000).
In many parts of the world Streptocephalus species are not generally known to occur in unpredictable rock-pool habitats with a short hydroperiod. In Texas, in a region with both lowland- and rock-pools, S. texanus Packard, 1871 did not occur in the rock-pools, which were inhabited by Branchinecta packardi Pearse, 1912 (Belk, 1991). According to Belk (1991), this streptocephalid could not mature and reproduce fast enough to survive the short and variable hydroperiod of rock-pools. In general, Streptocephalus species usually need at least 10 days to mature (Hamer and Brendonck, 1997). A similar pattern was discovered in Branchinecta sandiegonensis Fugate, 1993 and Streptocephalus woottoni Eng, Belk and Eriksen, 1990 in California (Hathaway and Simovich, 1996). S. woottoni was never found in shallow, potentially short-lived wetlands. The 2 species only co-occurred (but never simultaneously) in deeper wetlands, as B. sandiegonensis reaches maturity quickly (1 to 2 weeks) and dies before S. woottoni matures. In north-eastern Natal (South Africa), Streptocephalus species inhabit the central, deeper, unvegetated regions of pools, while Triops, conchostracans and Branchipodopsis, adopting a typical r-selected life-history, live in the early drying littoral zone of the pools (Hamer and Appleton, 1991).
In this paper, the presence of S. purcelli in a rock-pool is documented for the first time. To explore the characteristics of this unusual population, the early life-history traits of this relatively short-lived South African rock-pool population (hydroperiod of about 1 month) are explored and compared with a S. purcelli population occurring in a nearby mud-pool with a longer hydroperiod (about 3 months).
Study area
The studied rock-pool (33° 25' 12.9" S; 18° 17' 46.5" E) and mud-pool (33° 27' 14.7" S; 18° 49' 55.3" E) are situated, respectively, in and close to Rondeberg Private Nature Reserve, along the West Coast of the Western Cape, South Africa (Fig. 1). The rock-pool is situated on top of a granite outcrop. In the rainy season of 2004, the rock-pool contained water for about 1 month, while the water in the mud-pool lasted for about 3 months. The study area lies within the winter rainfall region, where the climate is Mediterranean (Fuggle and Ashton, 1979) and most of the rain falls from May to August. In these months, the average rainfall over the last 7 years was 56.3 mm per month. The mean annual rainfall was 362 mm at Rondeberg Nature Reserve (M Duckitt, 2006).
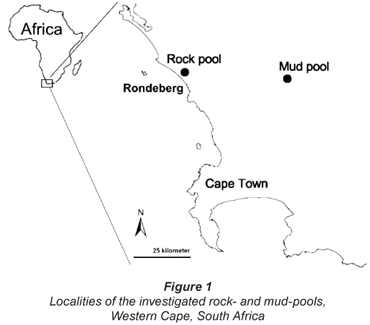
No other rock-pools containing large branchiopods were found in the area, but the region does contain many scattered mud-pools, usually inhabited by Streptocephalus dendyi Barnard, 1929 and S. purcelli (De Roeck et al., 2007).
Materials and methods
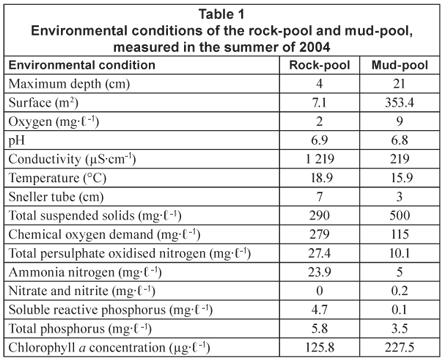
Several environmental conditions that were measured in the rock and mud-pool in the rainy season of 2004 are summarised in Table 1. For materials and methods used for these measurements refer to De Roeck et al. (2007). Additionally, ammonium, nitrite and nitrate were analysed at the Scientific Services Branch of the Cape Town Unicity. These samples were taken in a similar way as the other nutrients (see De Roeck et al., 2007).
The 2 wetlands were sampled for large branchiopods at the beginning of September 2004. To make sure that no other large branchiopod species were missed in the active communities, sediment from the studied pools was collected and incubated under laboratory conditions. For materials and methods refer to De Roeck et al. (2007). For both wetlands, hatching of the resting egg bank did not yield large branchiopod species that were not detected in the active population.
About 30 S. purcelli females with filled brood pouches were collected in the field at both study sites as material for the life history experiment. They were kept in separate vials until egg deposition. Afterwards, the eggs were kept wet for about a week before drying at room temperature. After drying, these resting eggs were stored for 20 weeks in darkness at 30°C. Three sets of 180 resting eggs·pool-1 were incubated in 2 ℓ aquaria filled with 1.5 ℓ EPA medium. They were inundated at predetermined optimal conditions (for both populations, at a conductivity of 100 µS·cm-1, temperature of 13°C, and under constant illumination and aeration). The optimal conditions were deducted from unpublished data comparing the hatching frequency of S. purcelli larvae at 4 different conductivities (20, 100, 200 and 400 µS·cm-1) and 2 different temperatures (13°C and 20°C), which are representative for the situation in the field after first filling.
The investigated life-history traits included: diameter of resting eggs, cumulative hatching percentage, day of first hatching, hatching period, size of the freshly hatched nauplii, day when half of the population was mature and mortality after 27 d. The resting egg size of randomly chosen eggs was determined for 30 eggs·pool-1. The diameter of these eggs was measured after 1 d of inundation, with a calibrated eyepiece under a set of binoculars. After first hatching, the light regime was switched to 12 h of darkness and 12 h of illumination. The animals were fed 105 algae cells per mℓ (50% S. obliquus and 50% R. subcapitata) daily. Life-history traits were assessed daily, together with the removal of any dead individuals. For both populations, 3 aquaria containing 180 resting eggs each were inundated to assess the hatching variables. For each wetland 54 nauplii from the same developmental stage, which were freshly hatched in the laboratory, were randomly selected and their size was measured on a digital microscopic image taken by means of the program Das (Cam2Disk). The length from the eye to the beginning of the telson was calculated by the programme Image Pro Plus 5.0 (Media Cybernetics). Per population, all nauplii were afterwards randomly divided over 6 aquaria to determine total mortality and the day when half of the female population was mature. Mortality was determined 27 d after the start of the experiment.
Results
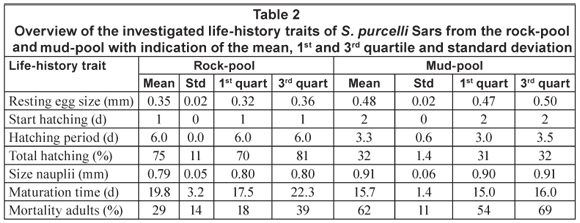
Results of the life-history trait comparison between the rock- and mud-pool populations are presented in Table 2. The rock-pool population had smaller eggs, started hatching earlier (1 d) and revealed a longer (about 3 d) hatching period. This population, moreover, had a higher total hatching percentage (about 40%) and smaller larvae. The rock-pool population also matured later and had a lower mortality. Rock-pool organisms finally had a strikingly darker colour, in juveniles as well as in the adult stage.
Discussion
The occurrence of S. purcelli in a rock-pool in Rondeberg (Western Cape, South Africa) is peculiar, since to our knowledge no other streptocephalids have been found in rock-pools in Southern Africa. The dominant anostracan genus usually encountered in Southern African rock-pools is Branchipodopsis (Hamer and Martens, 1998; Brendonck et al., 2000). They exhibit peculiar life-history traits, making them well-adapted to such ephemeral and highly diluted habitats. They hatch and mature fast and deposit an almost daily brood of relatively small resting eggs (Brendonck et al., 2000). In other parts of the world, streptocephalids were found in rock-pools with longer hydrocycles. In Algeria, Streptocephalus torvicornis (Waga, 1842) and Streptocephalus rubricaudatus (Klunzinger, 1867) were found in deep rock-pools with a long hydroperiod (Beladjal and Mertens, 2003). In North America and Australia, however, there were also some reports of Stretocephalidae occurring in rock-pools with a shorter hydroperiod, as exemplified by Streptocephalus texanus Packard, 1871 and Streptocephalus sp., respectively (Eng et al., 1990; Graham, 1994; Bayly, 2001). These occurrences prove that Streptocephalus species have the potential to adapt to ephemeral rock-pool environments.
None of the three other known rock-pools in the area around Rondeberg contained anostracans (De Roeck, personal observation). Other S. purcelli populations occurred in many mud-pools in the area, all with a longer hydroperiod than the studied rock-pool (personal observation). No other large branchiopod species hatched from the resting egg bank, which indicates that S. purcelli was the only species occurring in the rock- and the mud-pool.
The boundaries of the geographic distribution of the Southern African rock-pool specialist Branchipodopsis are situated more than 150 km to the east of the studied rock-pool and lie mostly within semi-arid and arid Southern Africa (Hamer and Brendonck, 1997; De Roeck et al., 2007). Possibly they could not adapt to the local Mediterranean climate. In Southern Africa, climate appears to be an important determinant for the distribution of anostracans (Hamer and Brendonck, 1997). The rock-pool in Rondeberg is very small and isolated, which furthermore minimises the chance for successful colonisation by these obligatory sexual and bet-hedging Branchipodopsis species. Field experiments with Branchipodopsis wolfi Daday, 1910 indicate that long-distance wind dispersal by means of resting stages is limited (Brendonck et al., 2000). This was confirmed indirectly by the patterns of genetic variation in the rock-pool populations; gene flow between metapopulations was seriously constrained by distances of 2 km or even less (Hulsmans et al., 2007).
The resting eggs of the Rondeberg rock-pool population were smaller than those of the mud-pool population. Other authors have also discovered notable differences in resting egg size of conspecific streptocephalid populations: Brendonck et al. (1993) for Streptocephalus proboscideus (Frauenfeld, 1873), Hildrew (1985) for Streptocephalus vitreus (Brauer, 1877) and Belk (1977) for Streptocephalus sealii Ryder, 1879. These authors state that different selection pressures amongst habitats, such as food availability, competition, predation pressure or environmental certainty, could explain these size differences. In the investigated rock-pool, the relatively short hydroperiod (about 1 month) is likely the dominant selection factor, although more extensive research is necessary to confirm this. S. dendyi occurring in mud-pools in the same region also revealed a positive correlation between the hydroperiod and the resting egg size of its inhabitants (Waterkeyn, 2005). To maximise the amount of offspring in an unpredictable habitat, the best strategy for a female is to quickly produce many, small resting eggs instead of a few large ones. An extensive egg bank is also needed to increase its buffering capacity through bet-hedging under a regime with a high risk of subsequent abortive hatchings. Linked to the smaller egg size, hatched larvae of the investigated rock-pool population were also smaller than those from the mud-pool.
The rock-pool anostracans started hatching earlier (1 d after inundation). Early hatching is characteristic for Anostraca from ephemeral rock-pools as it reduces the chance for abortive hatching (Brendonck et al., 2000). Surprisingly, the total hatching period was longer for the rock-pool population (6 d) than for the mud-pool population (3.3 d). This may seem maladaptive because the individuals that hatch late have less time to complete their life cycle if the rock-pool dries up quickly. It remains to be tested whether 'late hatching eggs' and their nauplii were also larger, therefore being more resistant to invertebrate predation, which becomes important later during the hydrocycle of pools.
In total, more eggs hatched from the rock-pool (75%) than from the mud-pool population (32%). In case of an abortive hatching this would result in a serious depletion of the egg bank, which again seems to be disadvantageous for inhabitants of short-lived pools. Theoretically, the hatching fraction should correspond with the chance for growth and reproduction (principle of bet-hedging) (Cohen, 1966; Brown and Venable, 1986). The hatching percentage of the Rondeberg rock-pool population is similar to the hatching fraction (80%) of B. wolfi eggs from rock-pools in the semi-arid southeast of Botswana, but only when inundated under laboratory conditions that usually do not occur in the field (Brendonck et al., 1998). When B. wolfi eggs were hatched under circumstances imitating the natural conditions of temperature, the hatching fraction was indeed much lower (less than 20%) (Brendonck and Riddoch, 2001). As hatching seems to be conditional, it is possible that the experimental hatching conditions were more favourable for the rock-pool individuals. In any case, although absolute values derived under artificial conditions should be interpreted with care, the data gathered under common garden conditions point at differences in triggers and hatching patterns between the studied S. purcelli populations.
The rock-pool population showed a slower maturation time than the mud-pool population, which is in contrast to expectations for organisms living under time stress. In contrast to streptocephalids, most Branchipodopis species reach maturity within less than a week, making them well adapted to short living pools (Brendonck et al., 2000). Streptocephalus species on the other hand usually need more than 10 d to mature (Belk, 1991; Hamer and Brendonck, 1997), although laboratory experiments also revealed some very fast maturing (within 5 d after inundation) streptocephalids (Ali and Dumont, 1995). The absence of S. texanus in rock-pools, which were instead inhabited by B. packardi, was explained by the slow maturation of the streptocephalid (Belk, 1991). Analogously, Beladjal et al. (2003) concluded that Branchipus schaefferi Fischer, 1834 was better adapted to short living pools than S. torvicornis, due to its shorter maturation time. A Chirocephalus diaphanus Prévost, 1820 population occurring in a predictable semi-permanent pool, was also characterised by slower growth than a population from a shorter-living, irregularly-filled pool (Mura et al., 2003). Hathaway and Simovich (1996) also indicated that a slower maturation rate helps to explain the absence of S. woottoni Eng, Belk and Eriksen, 1990 from pools with a short hydroperiod. On the other hand, Eulimnadia texana (Packard, 1871), a conchostracan from North America, showed a later age at maturity in ponds with a shorter duration (Marcus and Weeks, 1997), which, as in the case of S. purcelli, seems to be maladaptive. Although the rock-pool in Rondeberg had a relatively short (1 month in the rainy season of 2004) and variable hydroperiod, the hydroperiod was still longer than in many other rock-pools in arid and semi-arid regions of Southern Africa (a few days to a few weeks). The life-history patterns observed in the Rondeberg population are probably caused by this relatively longer hydroperiod, which may often exceed maturation time, or by other selection pressures such as predation, competition and food availability. The lower mortality of the rock-pool population was probably associated with its late maturation time. A similar association was demonstrated for the conchostracan E. texana (Marcus and Weeks, 1997).
The rock-pool anostracans had a darker colour than those of the mud-pool. Like most rock-pools on a granite substrate, the studied rock-pool contained clear water and was rather shallow. Transparency and UV-permeability of aquatic systems are positively correlated (Sommaruga, 2001) and a high UV-radiation can be disadvantageous for zooplankton (Tollrian and Heibl, 2004). The darker colour may therefore have a protective function (Williams, 1985; Tollrian and Heibl, 2004).
Our results suggest that there are differences between both studied populations, which are possibly genetic or related to an impact of maternal effects on the hatching patterns of the offspring. The food availability of the mother may, for instance, influence the hatching pattern, size or maturation time of the offspring. To test the cause of the life-history differences, research on more populations and generations is necessary, which was not possible since only one S. purcelli rock-pool population is presently known and since we have experienced that it was difficult to breed these animals in the laboratory. In general, S. purcelli seemed less adapted to the rock-pool hydrological regime than real rock-pool specialists such as many branchipodids. Despite this, S. purcelli was still sufficiently adapted to survive the harsh conditions. Another possibility is that the rock-pool population is sustained by dispersal from neighbouring (mud-) pools through mass effects (a population living in a habitat with unsuitable conditions for reproduction can survive due to frequent dispersal of individuals from another habitat with suitable conditions for reproduction). This explanation is, however, less plausible since the rock-pool site is situated on a rather isolated mountain top. Due to the absence of any branchipodids in the area, S. purcelli was probably safe from competition by better-adapted rock-pool specialists.
Acknowledgments
This project was supported by the VLIR-project 'Sustainable utilisation of subterranean water resources for improvement of quality of life' (VLIR IUC University of the Western Cape, South Africa) and by the FWO grant G.0443.05 'Impact of hydrology on diversity of aquatic organisms in temporary wetlands in the Cape Region (South Africa)'. This research was also funded by a Ph.D. grant of the Institute for the Promotion of Innovation through Science and Technology in Flanders (IWT Vlaanderen). Special thanks go to Tom Van Engeland, Tom Callens and Bram Vanschoenwinkel for helping with the fieldwork and to Professor Breugelmans for his grammatical comments during the preparation of this work. We thank Scientific Services (Cape Town) for analysing the water samples. We also thank the Western Cape Nature Conservation and Mr. and Mrs. Duckitt of Rondeberg Private Nature Reserve for giving permission to sample large branchiopods and for the supply of information about the region.
References
ALI JA and DUMONT HJ (1995) Larviculture of the fairy shrimp, Streptocephalus proboscideus (Crustacea: Anostraca): effect of food concentration and physical and chemical properties of the culture medium. Hydrobiol. 298 159-165. [ Links ]
BAYLY I (2001) Invertebrate occurrence and succession after episodic flooding of a central Australian rock-hole. J. R. Soc. West. Aust. 84 29-32. [ Links ]
BELADJAL L and MERTENS J (2003) Interstitial remains for fauna reconstruction of desert pools using fairy shrimps as example (Anostraca). J. Crustac. Biol. 23 60-68. [ Links ]
BELADJAL L, PEIREN N, VANDEKERCKHOVE TTM and MERTENS J (2003) Different life histories of the cohabiting fairy shrimps Branchipus schaefferi and Streptocephalus torvicornis (Anostraca: Crustacea). J. Crustac. Biol. 23 300-307. [ Links ]
BELK D (1977) Evolution of egg size strategies in fairy shrimps. Southwest. Nat. 22 99-105. [ Links ]
BELK D (1991) Why only one of two common Central Texas Anostraca atop enchanted rock. Hydrobiol. 212 83-86. [ Links ]
BILTON D, FOGGO A and RUNDLE S (2001) Size, permanence and the proportion of predators in ponds. Arch. Hydrobiol. 154 451-458. [ Links ]
BRENDONCK L, CENTENO DM and PERSOONE G (1993) Fecundity and resting egg characteristics of some subtropical fairy shrimp and clam shrimp species (Crustacea, Branchiopoda), reared under laboratory conditions. Arch. Hydrobiol. 126 445-459. [ Links ]
BRENDONCK L, HAMER M, RIDDOCH B and SEAMAN M (2000) Branchipodopsis species – specialists of ephemeral rock pools. Afr. J. Aquat. Sci. 25 98-104. [ Links ]
BRENDONCK L and RIDDOCH B (2001) Hatching characteristics of the fairy shrimp Branchipodopsis wolfi in relation to the stochastic nature of its habitat, desert rock pools. Verh. – Int. Ver. Theor. Angew. Limnol. 27 3931-3935. [ Links ]
BRENDONCK L, RIDDOCH B, VAN DE WEGHE V and VAN DOOREN T (1998). The maintenance of egg banks in very short-lived pools – a case study with anostracans (Branchiopoda). Arch. Hydrobiol. Spec. Issues Adv. Limnol. 52 141-161. [ Links ]
BROWN JS and VENABLE DL (1986) Evolutionary ecology of seedbank annuals in temporally varying environments. Am. Nat. 127 31-47. [ Links ]
CHAN MA, MOSER K, DAVIS JM, SOUTHAM G, HUGHES K and GRAHAM T (2005) Desert potholes: ephemeral aquatic microsystems. Aquat. Geochem. 11 279-302. [ Links ]
COHEN D (1966) Optimizing reproduction in a randomly varying environment. J. Theor. Biol. 12 119-129. [ Links ]
DE ROECK E, VANSCHOENWINKEL BJ, DAY JA, XU Y, RAITT L and BRENDONCK L (2007) Conservation status of large branchiopods in the Western Cape, South Africa. Wetlands 27 (1) 162-173. [ Links ]
DUCKITT M (2006) Personal communication, 6 January 2006. Rondeberg Private Nature Reserve, PO Box 70, Darling, 7345, South Africa. [ Links ]
EITAM A, BLAUSTEIN L, VAN DAMME K, DUMONT H and MARTENS K (2004) Crustacean species richness in temporary pools: relationships with habitat traits. Hydrobiol. 525 125-130. [ Links ]
ENG LL, BELK D and ERIKSEN CH (1990) Californian Anostraca – distribution, habitat, and status. J. Crustac. Biol. 10 247-277. [ Links ]
FUGGLE RF and ASHTON ER (1979) Climate. In: Day J, Siegfried WR, Louw GN and Jarman ML (eds.) Fynbos Ecology: A Preliminary Synthesis. South African National Scientific Programmes Report No. 40, CSIR, Pretoria, South Africa. [ Links ]
GRAHAM TB (1994) Predation by dipteran larvae on fairy shrimp (Crustacea, Anostraca) in Utah rock-pools. Southwest. Nat. 39 206-207. [ Links ]
HAMER M and APPLETON C (1991) Life-history adaptations of phyllopods in response to predators, vegetation, and habitat duration in North-Eastern Natal. Hydrobiol. 212 105-116. [ Links ]
HAMER M and BRENDONCK L (1997) Distribution, diversity and conservation of Anostraca (Crustacea: Branchiopoda) in Southern Africa. Hydrobiol. 359 1-12. [ Links ]
HAMER M and MARTENS K (1998) The large branchiopoda (Crustacea) from temporary habitats of the Drakensberg region, South Africa. Hydrobiol. 384 151-165. [ Links ]
HATHAWAY SA and SIMOVICH MA (1996) Factors affecting the distribution and co-occurrence of two southern californian anostracans (branchiopoda), Branchinecta sandiegonensis and Streptocephalus woottoni. J. Crustac. Biol. 16 669-677. [ Links ]
HILDREW AG (1985) A quantitative study of the life history of a fairy shrimp (Branchiopoda: Anostraca) in relation to the temporary nature of its habitat, a Kenyan rainpool. J. Anim. Ecol. 54 99-110. [ Links ]
HULSMANS A, MOREAU K, DE MEESTER L, RIDDOCH BJ and BRENDONCK L (2007) Direct and indirect measures of dispersal in the fairy shrimp Branchipodopsis wolfi indicate a small scale isolation-by-distance pattern. Limnol. Oceanogr. 52 676-684. [ Links ]
HULSMANS A, VANSCHOENWINKEL B, PYKE C, RIDDOCH BJ and BRENDONCK L (2008) Quantifying the hydroregime of a temporary pool habitat: A modelling approach for ephemeral rock pools in SE Botswana. Ecosyst. 11 89-100. [ Links ]
JOCQUÉ M, MARTENS K, RIDDOCH B and BRENDONCK L (2006) Faunistics of ephemeral rock pools in southeastern Botswana. Arch. Hydrobiol. 165 415-431. [ Links ]
LAHR J, DIALLO AO, NDOUR KB, BADJI A and DIOUF PS (1999) Phenology of invertebrates living in a Sahelian temporary pond. Hydrobiol. 405 189-205. [ Links ]
MARCUS V and WEEKS SC (1997) The effects of pond duration on the life history traits of an ephemeral pond crustacean, Eulimnadia texana. Hydrobiol. 359 213-221. [ Links ]
MURA G, FANCELLO G and DI GUISEPPE S (2003) Adaptive strategies in populations of Chirocephalus diaphanus (Crustacea, Anostraca) from temporary waters in the Reatine Apennines (Central Italy). J. Limnol. 62 35-40. [ Links ]
PHILIPPI T and SEGER J (1989) Hedging ones evolutionary bets, revisited. TREE 4 41-44. [ Links ]
RANTA E (1982) Animal communities in rock pools. Annales Zoologici Fennici 19 337-347. [ Links ]
SIMOVICH MA and HATHAWAY SA (1997) Diversified bet-hedging as a reproductive strategy of some ephemeral pool anostracans (Branchiopoda). J. Crustac. Biol. 17 38-44. [ Links ]
SOMMARUGA R (2001) The role of solar UV radiation in the ecology of alpine lakes. Photochem. Photobiol. 62 35-42. [ Links ]
TAVERNINI S, MURA G and ROSSETTI G (2005) Factors influencing the seasonal phenology and composition of zooplankton communities in mountain temporary pools. Int. Rev. Hydrobiol. 90 358-375. [ Links ]
THIÉRY A (1986) Les crustacés branchiopods (Anostraca, Notostraca et Conchostraca) du Maroc occidental. 1. Inventaire et repartition. Bull. Soc. Hist. Nat. Toulouse 122 145-155. [ Links ]
TOLLRIAN R and HEIBL C (2004) Phenotypic plasticity in pigmentation in Daphnia induced by UV radiation and fish kairomones. Funct. Ecol. 18 497-502. [ Links ]
WATERKEYN A (2005) Levensgeschiedeniskenmerken van grote branchiopoden in relatie tot de hydrologie van tijdelijke wetlands in de Kaapstreek. M. Thesis., K.U. Leuven. [ Links ]
WATERKEYN A, GRILLAS P, VANSCHOENWINKEL B and BRENDONCK L (2008) Invertebrate community patterns in Mediterranean temporary wetlands along hydroperiod and salinity gradients. Freshwater Biol. 53 1808-1822. [ Links ]
WIGGINS GB, MACKAY RJ and SMITH IM (1980) Evolutionary and ecological strategies of animals in annual temporary pools. Arch. Hydrobiol. Suppl. 58 97-206. [ Links ]
WILLIAMS DD (1985) Biotic adaptations in temporary lentic waters, with special reference to those in semi-arid and arid regions. Hydrobiol. 125 85-110. [ Links ]
WILLIAMS DD (2006) The Biology of Temporary Waters. Oxford University Press, Oxford. 337 pp. [ Links ]
Received 17 July 2009; accepted in revised form 23 February 2010.
* To whom all correspondence should be addressed.
+32 26 274139; fax: +32 26 274113;
e-mail: els.deroeck@naturalsciences.be














