Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Water SA
On-line version ISSN 1816-7950
Print version ISSN 0378-4738
Water SA vol.36 n.3 Pretoria Apr. 2010
First report on the colony-forming freshwater ciliate Ophrydium versatile in an African river
PJ OberholsterI,*; PJ AshtonI; GB FritzII; A-M BothaIII
ICSIR Natural Resources and the Environment, PO Box 395, Pretoria 0001, South Africa
IIDepartment of Zoology; Biological Institute, Universität Stuttgart, Stuttgart, Germany
IIIDepartment of Genetics, Stellenbosch University, Matieland 7601, South Africa
ABSTRACT
Ophrydium versatile (Müller 1786) Ehrenberg 1830 – a symbiotic ciliate that forms gelatinous colonies – is widely distributed in temperate lakes in Europe and America, but has not previously been recorded from rivers. In this paper we report the first record of O. versatile in an African river, based on an identity confirmed by molecular taxonomic identification. The limnological conditions within the Lephalala River during the sampling period were characterised as oligotrophic with low DOC concentrations, similar to the conditions observed in temperate northern hemisphere lakes where these organisms have been recorded previously. The majority of O. versatile colonies occurred in areas where thin orange-coloured films containing high concentrations of iron (> 60% Fe) covered the substrate and bedrock of the river; this may be related to the abundant picophytoplankton that were associated with these films and oligotrophic conditions. The planktonic diatom Gomphonema venusta Passy was dominant in the water column throughout the study period and acted as an environmental indicator of low electrical conductivity (EC) conditions in the habitat where O. versatile colonies were recorded.
Keywords: Lapalala River, oligotrophic indicator, water quality
Introduction
Planktonic, green protozoan ciliates include a large variety of species that belong to different taxonomic groups, for example: holotrichs, peritrichs, oligotrichs and heterotrichs, all characterised by the presence of symbiotic algae inside their cytoplasm (Queimalinos et al., 1999). These green protozoan ciliates can occur in sufficiently large quantities in deep oligotrophic lakes, e.g. Lake Tanganyika, that they may lead to a notable overestimation of the phytoplankton biomass if this is evaluated on chlorophyll content alone (Hecky and Kling, 1981; Amblard et al., 1993). However, freshwater ciliated protozoa that form sessile colonies are relatively rare, especially species that possess photosynthetic cells. The species O. versatile (O.F. Muller, 1786), the subject of this paper, is widely distributed in transparent, temperate, oligotrophic lakes in Europe and North America (Winkler and Corliss, 1965; Sand-Jensen et al., 1994; 1997). To the authors' knowledge, this species has not previously been reported from Africa and little is known of its distribution in rivers or its relationship with the limnological features of rivers.
Both phytoplankton and protozoa include acellular organisms that live close to minimal conditions and are therefore strongly dependent on conditions that promote rapid growth, reduction, or subsidence of populations. The idea that the distribution of O. versatile communities is related to limnological features in lakes is not new. Data presented by Sand-Jensen et al. (1997) show that the preferred growth habitat of O. versatile is transparent temperate lakes with low concentrations of dissolved inorganic nutrients. The objectives of this paper are to:
- Report the first occurrence of O. versatile in an African river, confirmed by the use of molecular taxonomic identification tools
- Determine the limnological river features of the site where this species occurs.
Material and methods
Study site
The study site (23º 54′ 13.5′′ S; 28º 20′ 22.7′′ E) is located in the middle reaches of the Lephalala River and is one of several sampling sites within a larger study area that encompasses the adjacent catchments of the Mokolo and Lephalala Rivers (Fig. 1). These 2 rivers drain the mountainous Waterberg complex and flow north-westwards to join the Limpopo River, which forms the northern boundary between South Africa and Botswana. The geology of the upper reaches of the study area is characterized by fractured, horizontally bedded, metasedimentary rocks of the Waterberg Basin, dominated by iron-rich, feldspathic sandstones, quartzites, felsites and conglomerates. Further downstream towards the Limpopo River, silicified iron-rich sandstones overlie carbon-rich mudstones, shales and basalts of the Karoo Sequence (Barker et al., 2006). The soils are medium to deep sandy-clay loam soils in the upper reaches of the study area, grading to moderately deep sandy loam soils along the valley bottoms and the lower reaches of the river basins (Midgley et al., 1994).
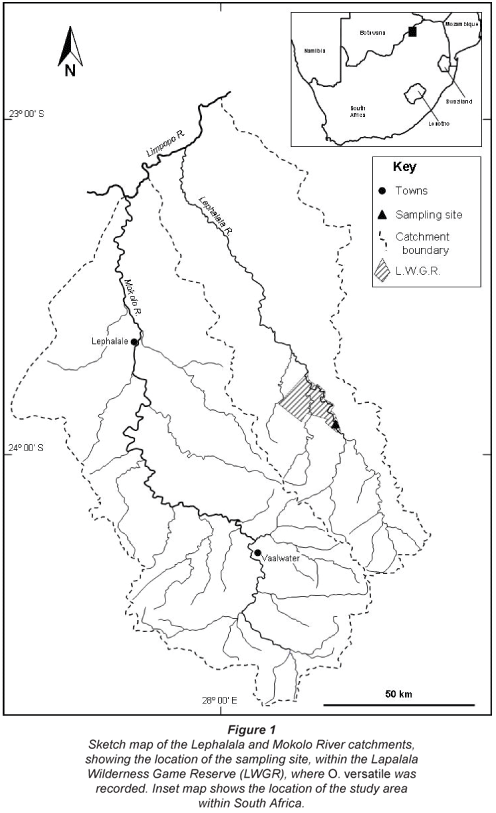
Mean annual rainfall is relatively low (560 mm/yr) and most rainfall is received as thunderstorms during the austral summer months (November to February); almost no rainfall is recorded during the dry winter months (May to August). High annual average rates of evaporation (1 900 mm/yr) exceed rainfall and the area is considered to be arid (Schulze, 1997). The natural vegetation of the Lephalala catchment is dominated by savannah vegetation forms, with Waterberg Mountain Bushveld in the upper reaches grading gradually into arid Limpopo Sweet Bushveld closer to the Limpopo River (Mucina and Rutherford, 2006). The dominant land use in the upper catchment is cattle and game ranching with small areas of irrigated agriculture (wheat, maize and lucerne). The lower reaches of the catchment support extensive areas of irrigated cotton and lucerne, as well as cattle and game ranching. The study site (Fig. 1) is situated inside the privately-owned Lapalala Wilderness Game Reserve (LWGR), and enjoys a high degree of protection from disturbance. Flows in the Lephalala River are perennial though there are wide seasonal variations that reflect the seasonality of rainfall; these seasonal variations are accentuated by the upstream abstraction of water for irrigation during the dry austral winter months. Water quality in the middle reaches of the Lephalala River is good, though return flows containing agro-chemicals from areas of irrigated agriculture located upstream of the study site pose potential risks to water quality.
Physical and chemical parameters
The dissolved oxygen, temperature, pH and electrical conductivity (EC) features of the surface water at the sampling site were measured in situ using a Hach sensionTM 156 portable multiparameter (Loveland, USA). Duplicate water samples (2 ℓ each) were collected for chemical analysis and chlorophyll-a (chl-a) analyses, while separate samples were collected for enumerating the phytoplankton assemblage. The water samples were preserved in pre-rinsed polyethylene bottles and placed in the dark on ice while in transit to the laboratory during the 3 sampling trips in August, October and December 2008. Chlorophyll a concentrations were measured with the ethanol extraction method of Sartory and Grobbelaar (1984) and results were expressed as µg/ℓ. The river water samples were filtered through 45 µm pore size GF/filters and stored in polyethylene bottles that had been pre-rinsed with dilute sulphuric acid (to pH 2.0) for analysis of dissolved nutrients. All analyses were carried out according to standard methods (Standard Methods, 1992) in the CSIR's accredited analytical laboratory. Concentrations of total nitrogen (TN) and total phosphorus (TP) were determined with the persulphate digestion technique. Nitrate concentrations were determined on an autoanalyser with the cadmium reduction method, while soluble reactive phosphorus concentrations were determined by the ascorbic acid method (Standard Methods, 1992). River flow velocity at the sampling site was determined using the method of Gore (2006).
Data analyses
Statistical differences were analysed by calculating the Pearson correlation and a t-test using the Sigma Plot (Jandel Scientific) program. Values of p < 0.05 were regarded as significant; correlations of r near zero were regarded as unrelated.
Phytoplankton sampling
Phytoplankton samples of the water column were collected at a depth of 50 cm with a surface water sampler, and each water sample (1 ℓ) was preserved in the field by addition of 2% formaldehyde. All identifications were made by using a compound microscope with 1 200 x magnification (Van Vuuren et al., 2006; Taylor et al., 2007). Sediment samples for benthic diatoms were collected by scraping one cm2 of the surface film on the river bedrock surface into a collecting basin using a scalpel. Mud and sediment samples were stored in the dark until preparation in the laboratory. The sediment was cleared of organic matter in a potassium dichromate and sulphuric acid solution and the cleared material was rinsed, diluted, and mounted in Pleurax medium for microscopic examination.
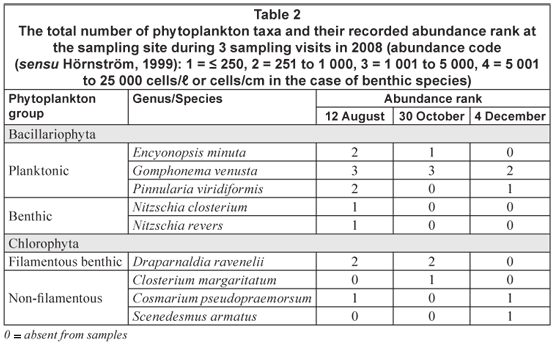
Diatom species were identified and counted, placing a drop of non-drying immersion oil on top of the cover slip just prior to microscope examination at 1 200 x magnification. The biovolumes of the more abundant taxa were estimated by measuring cell dimensions of at least 20 individuals and using the closest geometric formulae (Willen, 1976). Species identifications were performed according to Wehr and Sheath (2003) and Taylor et al. (2007). Strip counts were made until at least 300 individuals of each of the dominant phytoplankton species were counted. All counts were based on numbers of cells observed and the individual data were grouped into major algal groups (Lund et al., 1958; Willen, 1991). The total number of phytoplankton taxa and their frequency at each sampling site were categorized according to Hörnström (1999): 1 = ≤ 250, 2 = 251 to 1 000, 3 = 1 001 to 5 000, 4 = 5 001 to 25 000 cells/ℓ; the abundance of benthic species was expressed as cells/cm. The total number of taxa and their frequency were recorded after careful examination for at least 15 min and after not finding additional taxa. The summer near-surface average nutrient concentrations were used to classify the trophic state of the river sampling site (Dodds et al., 1997).
Protozoa sampling
Samples of free-swimming (ciliated) protozoa were collected with a 45 µm mesh plankton net and samples were transferred to bottles and stored in the dark during transit to the laboratory. The protozoa assemblage in each sample was determined and quantified in the laboratory using the live counting technique at 400 x or 1200 x magnification with a light microscope. Identification of ciliate protozoa was done according to Kahl (1930-1935), while the quantitative Protargol (QPS) method of Montagnes and Lynn (1987a; b) and Skibbe (1994) was used for staining and agar embedding.
Colonial ciliate protozoa (O. versatile) were collected mechanically from the Lephalala River (Fig. 2A) during the August and October field trips, where visible colonies were attached to the bedrock in shallow (0.25 to 0.5 m), slow-flowing (0.05 to 0.1 m/s) water (Figs. 2B and 2C). These samples were transferred from the field to the laboratory in a darkened cool-box. In the laboratory, colonies were studied in their entirety and were also macerated to release the contained zooids. The mean numbers and diameter of the endosymbiotic zoochlorellae algal cells of each Ophrydium colony were quantified in live ciliate specimens under direct microscopy. No Ophrydium colonies were recorded during the December sampling visit when water depths (0.75 m) and river flows (0.3 to 0.5 m/s) had increased as a result of early summer rains.
Molecular taxonomic identification was done according to Miao et al. (2004). Briefly, cells of the collected specimens were incubated in lysis buffers at 55ºC for 12 to 20 h and then extracted using a standard phenol:chloroform method (Sambrook et al., 1989). The DNA content was determined by spectrophotometry using a BioRad spectra-plus 20 Spectrophotometer. PCR amplification of the SSU rRNA genes was performed in a Perkin-Elmer GeneAmp PCR System 9600 (PE Applied Biosystem USA).
Two pairs of primers were used (Elwood et al., 1985); these were:
- Forward 5' aacctggttgatcctgccagt 3';
Reverse 5' aattgacggccatgcacc 3' - Forward 5' gacagtcagaggtgaaattct 3';
Reverse 5' tccttctgcaggttcacctac 3'
The fragment amplified by the 1st pair of primers was located approximately 1 100 bases from the 5' end of the 18S gene, while that amplified by the 2nd pair of primers was approximately 900 bases from the 3' end of the 18S gene. Both fragments were amplified in the test species using the following PCR procedure: an initial denaturation step of 94ºC for 10 min, followed by 35 cycles of 1 min at 94ºC, 0.5 min at 50ºC, and 1.5 min at 72ºC, and a final extension step at 72ºC for 10 min. The PCR products were purified using the Biostar Glassmilk DNA Purification Kit (BioStar International, Toronto, ON, Canada).
Ligation reactions were performed by overnight incubation at 4ºC of 10 µℓ mixtures containing approximately 50 ng pure DNA, 50 ng pGem-T Easy Vector (Promega Biotech, Madison, WI) and 3 units T4 DNA ligase. Ten-µℓ ligation reactions were used to transform 100 µℓ of HB 101 high-efficiency competent cells following the method of Sambrook et al. (1989). The cloned fragments were sequenced in both directions by Inqaba Biotech, using dye terminators and Taq FS with M13 forward and reverse primers. The sequence alignments and comparisons were performed using DNA tools, Ver. 5.1 (Rasmussen, 2001) and further refined by considering the secondary structure of the SSU rRNA molecule (Wuyts et al., 2001). The obtained sequences were compared to available sequences on GenBank, following the accession number AF401526 (1727 bp).
Results
Physical and chemical parameters
Electrical conductivity values at the sampling site increased from 50 µS/cm during August to 70 µS/cm during October as river flows declined (0.05 to 0.1 m/s), and then decreased to 43 µS/cm in December when river flows increased (Table 1). The low total dissolved salts (TDS) values calculated from the conductivity readings indicated that the water at this study site was moderately mineralised. The average water temperature for the sampling site increased from 14.9ºC in August to 26.6ºC in December, reflecting the seasonal increase in air temperatures with the onset of summer. Water pH values decreased from 8.3 in August to 7.4 in October and 6.8 in December. The average midday dissolved oxygen (DO) concentration increased from 5.2 mg/ℓ in August to 7.9 mg/ℓ in December.
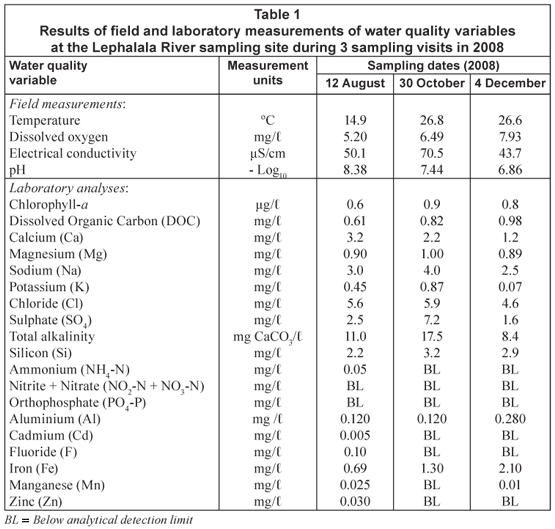
At the sampling site, a comparison of the sum of measured ions (in mg/ℓ) to the concentration of TDS derived from field measurements of electrical conductivity varied by less than 10%. This indicated that the laboratory analyses of macro- constituents were reliable and complete, and therefore provided an accurate representation of the prevailing water chemistry at each sampling visit.
The results of analyses for dissolved nutrients (nitrogen and phosphorus) were inconclusive, with most values being below the analytical detection limit (Table 1). Traces of aluminium, cadmium, fluoride, manganese and zinc were detected in samples taken during the fieldtrip in August 2008 but, with the exception of aluminium, declined to levels below their respective analytical detection limits in October and December. Aluminium concentrations remained constant during August and October but increased in December (Table 1).
The ionic dominance ratios for major cations and anions at each sampling remained approximately constant over the 3 sampling visits (Ca ≥ Na > Mg > K; CO3 > Cl > SO4), with slight variations in the proportions of calcium and sodium as the dominant cation. Carbonate remained the dominant anion in the water column during the 3 sampling visits.
Chemical analysis of the orange-coloured sediment found in association with the O. versatile colonies indicated that this material contained over 60% iron and was most probably an iron oxy-hydroxide precipitate.
Phytoplankton assemblage
The net phytoplankton assemblage at the study site consisted of 2 main groups: Bacillariophyta and Chlorophyta (Table 2). Bacillariophyta (diatoms) was the richest species group when the numerical abundance of species was considered. According to the trophic index of Dodds et al. (1997) the sampling site remained oligotrophic throughout the study. There was a positive correlation (r = 0.9413, p < 0.05) between the low average chl a concentration (0.8 µg/ℓ) and the low average nutrient concentration (< 0.20 mg/ℓ N and < 0.20 mg/ℓ P) recorded at the sampling site on all visits. The water column at the sampling site was dominated by large numbers (1 001 to 5 000 cells/ℓ) of the planktonic diatom Gomphonema venusta Passy. The benthic pinnate diatoms Nitzchia revers and Nitzchia closterium were observed in thin brown films covering the riverbed rock substrate but were only present in low numbers (< 250 cells/cm2). Cosmarium pseudopraemorsum was the dominant chlorophyta species (251 to 1 000 cells/ℓ) in the water column during August and October (Table 2).
Ciliate protozoa assemblage
Free-living protozoa were confined to the upper 1.5 cm of the surface water of the land/water contact zone at the sampling site. The only protozoan species observed in the water column was the periphytic ciliate Blepharisma lateritium (Kahl, 1930-1935), which is primarily bacteriovorous (Kalff, 2002; Oberholster et al., 2009). The average numerical abundance of these ciliates was low at the sampling site during August, with an average of 7 ciliates/mℓ. The higher average number of ciliates (11 ciliates/mℓ) during the October and December sampling visits correlated significantly (r = 0.9916; p < 0.05) with the higher average water temperature (26.8 ºC and 26.6 ºC, respectively).
Colonial protozoa assemblage
The molecular taxonomic identification confirmed that the colonial protozoan specimens were O. versatile Ehrenberg, a symbiotic ciliate that forms gelatinous colonies up to several centimetres in diameter. The small immature colonies (approximately 10 mm in diameter) were almost spherical while the larger colonies (up to 16 cm in diameter) become more obviously variable in their overall shape (Figs. 2B and 2C). Some of these larger colonies were flattened with a central cavity, giving them a doughnut-like appearance, with a width much greater than the height. Only green colonies were observed in our investigation.
Sections of living colonies, examined under various powers of a dissecting and inverted light microscope, revealed that there were 2 uneven layers of zooids. Each zooid was oriented so that its oral pole was towards the surface of the matrix. These zooids dwell within an independent sub-cylindrically shaped chamber of the gelatinous matrix. The ciliate zooids were characteristically flask shaped and covered with a highly sculptured pellicle (Fig. 2D). When fully extended, the mature zooids were between 350 to 450 µm long, while contracted individuals were between 150 and 210 µm long. The neck of each zooid was highly contractile and the feeding apparatus was pulled into the infundibulum in response to such stimuli as vibration and light. When fully extended, the buccal apparatus, composed of 2 spirally arranged rows of cilia. Abundant endosymbiotic zoochlorellae algal cells were present within each zooid, giving it its green colour. These algal cells are known as Zoochlorella parasitica Brandt, 1882, and were between 4.1 and 4.6 µm in diameter. Each zooid in this study contained between 450 and 620 algal cells. The algal cells underwent considerable movement due to the contraction and expansion of the ciliate.
The larval stage or telotroch of O. versatile occurred in much greater abundance during the August sampling trip compared to October, possibly due to lower water temperatures or lower river flows. The telotrochs were sub-cylindrical in shape, tapering towards the poles of the body, while the anterior end was blunt (Fig. 2E). The average size of the telotrochs was 110 x 60 µm; abundant zoochlorellae cells were present in each telotroch. The occurrence of colonial patches of O. versatile were positively correlated (r = 0.9083, p ≤ 0.05) with the presence of a thin orange-coloured film containing high concentrations of iron (> 60% Fe), which covered the substrate and bedrock at the sampling site (see Figs. 2B and 2C). There was a positive correlation (r = 0.9145, p ≤ 0.05) between the low average chl a concentrations (< 1 µg/ℓ) in the water column during August and December and the low average nutrient concentrations (< 0.20 mg/ℓ N and < 0.20 mg/ℓ P), and low concentrations of dissolved organic carbon (DOC = 0.61 to 0.98 mg/ℓ).
Discussion
The different net phytoplankton species recorded in this study do not, as far as is known, form any part of the food of colonial O. versatile, since this species is recorded to prey on bacteria and small eukaryotic organisms (fungi, protozoa and picophytoplankton; Goff and Stein, 1981). However, the presence of the different net phytoplankton species may indirectly act as indicators of the water quality where O. versatile occurs, because the diversity of phytoplankton communities can reflect an entire complex of ecological parameters at a particular site (Wehr and Sheath, 2003). Such indices are referred to as autecological, because they are based on the autecological characteristics of specific taxa. The planktonic diatom Gomphonema venusta Passy was the dominant phytoplankter in the water column throughout the study and acted as an environmental indicator of low electrical conductivity as measured during the study (Taylor et al., 2007). The average silica concentration at the Lephalala River site (2.8 mg/ℓ), was lower than the world average for rivers of 9 mg /ℓ (Horne and Goldman, 1995), but comparable to other South African river systems, such as the Vaal River (3 mg/ℓ). During the August sampling trip, silica concentrations (2.2 mg/ℓ) were lower and correlated positively (r = 0.8916, p ≤ 0.05) with increased algal growth (chl a = 1.7 μg/ℓ). It can be assumed from these data that the silica was incorporated into the cell walls of the abundant diatoms present at this site. The dissolved oxygen concentration at the sampling site (Table 1) increased steadily from August to December, reaching 98% saturation. The low average electrical conductivity (EC) values (Table 1) indicated that the river was unpolluted, because the typical upper limit of electrical conductivity for an unpolluted river is approximately 350 μS/cm (Koning and Roos, 1999).
Ophrydium versatile has been found in transparent oligotrophic lakes with low concentrations of DOC and nutrients (Sand-Jensen et al., 1997); these conditions are similar to the environmental conditions recorded for the Lephalala River observed in this study. The occurrence of O. versatile colonies in areas where thin orange films containing high concentrations of iron covered the substrate and bedrock of the river may possibly be related to the abundance of picophytoplankton that was associated with the oligotrophic water and these films (Kalff, 2002).
Conclusion
Data generated in this study indicate that colonies of the ciliate O. versatile can occur in slow-flowing rivers with low nutrient content and transparent (low turbidity) conditions. Furthermore, variables such as low electrical conductivity, relatively high iron concentrations and low concentrations of trace metals, may also play a role in favouring the distribution of this colonial ciliate in river environments. If this hypothesis is correct, the distribution of O. versatile can be used as an indicator of possible environmental changes in river systems, since they are in close contact with their immediate environment and only contain a plasma membrane that separates their living cytoplasm from changes in their external aquatic environment. Furthermore, these sessile specimens lack complex homeostatic systems – such as are found in fish – for dealing with environmental changes; this makes them ideal for monitoring rapid responses in river environments.
References
AMBLARD C, SIME-NGANDO T, RACHIQ S and BOURDIER G (1993) Importance of ciliated protozoa in relation to bacterial and phytoplanktonic biomass in an oligo-mesotrophic lake, during the spring diatom bloom. Aquat. Sci. 55 1-9. [ Links ]
BARKER OB, BRANDL G, CALLAGHAN CC, ERIKSSON PG and VAN DER NEUT M (2006) The Soutpansberg and Waterberg Groups and the Blouberg Formation. In: Johnson MR, Anhaeusser CR and Thomas RJ (eds.) The Geology of South Africa. Geological Society of South Africa, Johannesburg, and the Council for Geoscience, Pretoria, South Africa. 301-318. [ Links ]
DODDS WK, SMITH VH and ZANDER B (1997) Developing nutrient targets to control benthic chlorophyll levels in streams: A case study of the Clark Fork River. Water Res. 31 1738-1750. [ Links ]
ELWOOD HJ, OLSEN GJ and SOGIN ML (1985) The small subunit RNA gene sequences from the hypotrichous ciliates Oxytricha nova and Stylonychia pustulata. J. Mol. Biol. Eval. 2 399-410. [ Links ]
GOFF LG and STEIN JR (1981) Digestion in the peritrich ciliate Ophrydium versatile. Protoplasm 107 235-254. [ Links ]
GORE JA (2006) Discharge measurements and streamflow analysis. In: FR Hauer and GA Lamberti (eds.) Methods in Stream Ecology. Academic Press, San Diego, California, USA. 51-81. [ Links ]
HECKY RE and KLING HJ (1981) The phytoplankton and protozooplankton of the eutrophic zone of Lake Tanganyika: Species composition, biomass, chlorophyll content and spatio-temporal distribution. Limnol. Oceanog. 26 548-564. [ Links ]
HÖRNSTRÖM E (1999) Long-term phytoplankton changes in acid and limed lakes in SW Sweden. Hydrobiol. 394 93-102. [ Links ]
HORNE AJ and GOLDMAN CR (1995) Limnology. McGraw Hill, New York, USA. 576 pp. [ Links ]
KAHL A (1930-1935) Urtiere oder Protozoa. I: Wimpertiere oder Ciliata (Infusoria). In: F Dahl (ed.) Die Tierwelt Deutschland 18 (1930); 21 (1931); 30 (1935). Fischer-Verslag, Jena, Germany. 886 pp. [ Links ]
KALFF J (2002) Limnology: Inland Water Ecosystems. Prentice Hall, Upper Saddle River, New Jersey, USA. 535 pp. [ Links ]
KONING N and ROOS JC (1999) The continued influence of organic pollution on the water quality of the turbid Modder River. Water SA 25 285-292. [ Links ]
LUND JWG, KIPLING C and LE CREN EO (1958) The inverted microscope method of estimating algal numbers and the statistical basis of estimations by counting. Hydrobiol. 11 143-170. [ Links ]
MIAO W, FEN W-S, YU Y-H, ZHANG XY and SHEN YF (2004) Phylogenetic relationships of the subclass Peritrichia (Oligohymenophorea, Ciliophora) Interred from small subunit rRNA gene sequences. J. Eukary. Microbiol. 51 180-186. [ Links ]
MIDGLEY DC, PITMAN WV and MIDDLETON BJ (1994) Surface Water Resources of South Africa 1990 (1st edn.) 10 Volumes. WRC Report No. 298/1/94. Water Research Commission, Pretoria, South Africa. 1085 pp. [ Links ]
MONTAGNES DJS and LYNN DH (1987a) A quantitative protargol stain (QPS) for ciliates: a method description and test of its quantitative nature. Mar. Microbiol. Food Webs 2 83-93. [ Links ]
MONTAGNES DJS and LYNN DH (1987b) Agar embedding on cellulose filters: an improved method of mounting protists for protargol and Chatton-Lwoff staining. Trans. Am. Microscop. Soc. 106 183-186. [ Links ]
MUCINA L and RUTHERFORD MC (eds.) (2006) Savanna biome. Chapter 9. In: The Vegetation of South Africa, Lesotho and Swaziland. Strelitzia 16 439-538. [ Links ]
OBERHOLSTER PJ, ASHTON PJ and BOTHA A-M (2009) The appearance of new taxa: Invertebrates, phytoplankton and bacteria in an alkaline, saline, meteorite crater lake, South Africa. Arch. Hydrobiol. 174 (3) 271-282 [ Links ]
QUEIMALINOS CP, MODENUTTI BE and BALSEIRO EG (1999) Symbiotic association of the ciliate Ophrydium naumanni with Chlorella causing a deep chlorophyll a maximum in an oligotrophic South Andes lake. J. Plankt. Res. 21 167-178. [ Links ]
RASMUSSEN SW (2001) DNAtools, a Software Package for Sequence Analysis. The Carlsberg Laboratory, Copenhagen. URL: http://www.dnatools.dk (Accessed 10 April 2008). [ Links ]
SAMBROOK J, FRITSCH EF and MANIATIS T (1989) Molecular Cloning (2nd edn.). Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, USA. 333 pp. [ Links ]
SAND-JENSEN K, GEERTZ-HANSEN O, PEDERSEN O and NIELSEN HS (1994) Size dependence of composition, photosynthesis and growth in the colony-forming freshwater ciliate, Ophrydium versatile. Freshwater Biol. 31 (2) 121-130. [ Links ]
SAND-JENSEN K, PEDERSEN O and GEERTZ-HANSEN O (1997) Regulation and role of photosynthesis in the colony symbiotic ciliate Ophrydium versatile. Limnol. Oceanogr. 42 (5) 866-873. [ Links ]
SARTORY DP and GROBBELAAR JU (1984) Extraction of chlorophyll-a from freshwater phytoplankton for spectrophotometric analysis. Hydrobiol. 114 177-187. [ Links ]
SCHULZE RE (1997) South African Atlas of Agrohydrology and Agroclimatology. WRC Report No. TT 82/96. Water Research Commission, Pretoria, South Africa. 457 pp. [ Links ]
SKIBBE O (1994) An improved quantitative protargol stain for ciliates and other planktonic protists. Arch. Hydrobiol. 130 339-348. [ Links ]
STANDARD METHODS (1992) Standard Methods for the Examination of Water and Wastewater (19th edn.). APHA, AWWA, and WPCF, Washington, DC, USA. 624 pp. [ Links ]
TAYLOR JC, HARDING WR and ARCHIBALD CGM (2007) An Illustrated Guide to Some Common Diatom Species from South Africa. WRC Report No. TT 282/07. Water Research Commission, Pretoria, South Africa. 178 plates. [ Links ]
VAN VUUREN S, TAYLOR JC, GERBER A and VAN GINKEL C (2006) Easy Identification of the Most Common Freshwater Algae. North-West University and Department of Water Affairs and Forestry, Pretoria, South Africa. 200 pp. [ Links ]
WEHR JD and SHEATH RG (2003) Freshwater Algae of North America: Ecology and Classification. Academic Press, Massachusetts, USA. 834 pp. [ Links ]
WILLEN E (1976) A simplified method of phytoplankton counting. Brit. J. Phycol. 11 265-278. [ Links ]
WILLEN E (1991) Planktonic diatoms – an ecological review. Algol. Stud. 62 69-106. [ Links ]
WINKLER RH and CORLISS JO (1965) Notes on the rarely described, green colonial protozoon Ophrydium versatile (O.F.M.) (Ciliophora, Peritrichida) Trans. Am. Microsc. Soc. 84 (1) 127-137. [ Links ]
WUYTS J, VAN DE PEER Y, WINKELMANS T and DE WACHTER R (2001) The European database on small subunit ribosomal RNA. Nucleic Acid Res. 30 (1) 183-185. [ Links ]
Received 26 August 2009; accepted in revised form 23 February 2010.
* To whom all correspondence should be addressed.
+2721 888-2591 ; fax: +2721 808-5833;
e-mail: Anna.Oberholster@up.ac.za
















