Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
Water SA
versão On-line ISSN 1816-7950
versão impressa ISSN 0378-4738
Water SA vol.35 no.1 Pretoria Jan. 2009
Suitability of total coliform β-D-galactosidase activity and CFU counts in monitoring faecal contamination of environmental water samples
VC WutorI, *; CA TogoII; BI PletschkeII
IAgriculture and Agri-Food Canada, Lethbridge Research Center, PO Box 3000, Lethbridge, Alberta, Canada T1J 4B1
IIDepartment of Biochemistry, Microbiology and Biotechnology, Rhodes University, PO Box 94, Grahamstown 6140, South Africa
ABSTRACT
Total coliforms are a group of bacteria found in high numbers in mammalian intestines; hence their presence in water indicates the possible contamination with faecal material. Total and faecal coliform counts were monitored over a period of 18 months using mFC, m-Endo and CM1046 media together with enzymatic assays on 215 environmental water samples obtained from the Eastern Cape Province of South Africa. A positive correlation, with an R2 value of 0.9393 was observed between faecal and total coliform colony units employing mFc and m-Endo media, and 0.8818 using CM1046 media. Also, a positive correlation was observed between Escherichia coli colony-forming units and β-d-galactosidase (B-GAL) activity (R2=0.8542). Overall, this study indicated that faecal contamination of environmental water samples could be monitored by measuring total coliform β-galactosidase activity and total coliform colony-forming units.
Keywords: colony-forming units (CFUs), coliforms, E. coli, β-d-galactosidase (B-GAL), β-D-glucuronidase (GUD)
Introduction
Coliforms are a group of bacteria employed to assess the microbiological quality of water and/or food. Most faecal coliforms are present in large numbers among the intestinal flora of human and other warm-blooded animals, and are thus commonly found in faecal material. As a result, the presence of these non-pathogenic microorganisms in water indicates the potential presence of pathogenic microorganisms of faecal origin (Venter, 2000; Rompré et al., 2002) and consequently a health hazard. It is estimated that 80% of communicable diseases in the world are waterborne (Devi et al., 2008). Analysing for total coliform bacteria and Escherichia coli is the most common method used to test the hygienic quality of drinking water (Huertas et al., 2003; Pitkänen et al., 2007). Bacteria as diverse as Escherichia coli, Citrobacter, Klebsiella, Enterobacter, Serratia, Erwinia and Yersinia are all positive for the traditional coliform test (Standard Methods, 2005). Among the afore-mentioned bacteria, only Escherichia coli appear to be of faecal origin (Gofti et al., 1999; Rompré et al., 2002; Schraft and Watterworth, 2005) (Fig. 1).

However, there have been criticisms on the suitability of total coliforms, as markers of faecal contamination of water and/or food (Gofti et al., 1999; Leclerc et al., 2001; Stevens et al., 2001; Rose and Grimes, 2002; Hörman and Hänninen, 2006). Studies on total coliforms reveal that they can have an environmental origin besides faecal sources, thus these organisms may not be suitable indicators of faecal contamination (Leclerc et al., 2001). Escherichia coli and enterococci have been shown to be consistently associated with faecal pollution and thus the most reliable indicators of faecal contamination (Leclerc et al., 1999; 2001; Gofti et al., 1999; Rompré et al., 2002; Schraft and Watterworth, 2005). Furthermore, there have been occurrences of total coliforms in the absence of documented waterborne morbidity in the community, and outbreaks where coliforms have not been found (McFeters and Camper, 1983; McFeters et al., 1986). Although the latter could be due to the fact that the currently accepted methods may lead to underestimations of waterborne coliforms (Hudson et al., 1983; McFeters et al., 1986), this does not rule out the unreliability of coliforms as faecal indicators. Despite these setbacks, total coliforms are still being used in national and international guidelines for the microbiological quality assessment of water (Grabow et al., 1999; Noble et al., 2003; Horman et al., 2004; Byamukama et al., 2005).
The presence of total coliforms and E. coli (faecal coliform) could be detected by measuring the activity of β-d-galactosidase (B-GAL) and β-D-glucuronidase (GUD) enzymes respectively (Wutor et al., 2007a). These enzymes hydrolyse a wide range of chromogenic and fluorogenic substrates yielding products which can be monitored spectrophotometrically and fluorometrically, respectively. In this study, the chromogenic substrates chlorophenol red β-D-galactopyranoside (CPRG), and p-nitrophenyl-β-D-glucuronide (PNPG) were employed to measure B-GAL and GUD activities respectively, while mFc, m-Endo and CM 1046 media were used in CFU counts.
In this paper, we examine the relationship between faecal coliform (FC) and total coliform (TC), thus the potential of using TC β-d-galactosidase activity and CFU counts as a measure of faecal coliform contamination of environmental water samples.
Materials and methods
Chlorophenol red β-D-galactopyranoside (CPRG), disodium hydrogen phosphate, dihydrogen sodium phosphate were all obtained from Merck (Darmstadt, Germany), p-nitrophenyl-β-D-glucuronide (PNPG) (Merck, Darmstadt, Germany). The media, mFc, mEndo and CM1046 were obtained from Merck (Darmstadt, Germany), Fisher Scientific (Loughborough, UK) and Oxoid (Hampshire, UK) respectively. The buffers were prepared using water purified with a Milli-Q system (Millipore, Milford, CT, USA). All reagents were of the highest analytical grade available.
Sampling
Water collection was performed in accordance with the standard procedures outlined in Standard Methods, 2005. Water samples were collected weekly from two points along the Bloukrans River, Grahamstown in the Eastern Cape of South Africa as well as from other sources in the Eastern Cape Province of South Africa for a period spanning 18 months between 2005 and 2006. Samples were collected aseptically in 1 ℓ sterile Pyrex bottles (Schott Duran, Germany), placed on ice and transported immediately to the laboratory and analysed within an hour. Samples were collected in triplicates between 08:00 and 08:30 on each sampling day. In total, 215 samples were studied.
Enzyme Assays
The B-GAL assay was performed as described in Wutor et al. (2007b). Briefly, CPRG (80 µg in 20 µℓ water) was mixed with 90 µℓ 0.1 M sodium phosphate buffer (pH 7.8) in a 96-well, flat-bottomed micro-titre plate, and 90 µℓ of environmental (water) sample added to initiate the reaction. The change in absorbance was monitored at 575 nm over 24 h on a PowerWavex (Bio-Tek Instruments, USA).
Togo et al. (2006) described the GUD assay employed in the study. PNPG (10 mM) was prepared in 0.1 M Tris-HCl, pH 8.0 containing 0.6 mM CaCl2 buffer. Assay buffer (50 µℓ) and 90 µℓ enzyme solution/water sample were mixed in a 96-well, flat-bottomed micro-titre plate. The reaction was initiated by the addition of 110 µℓ PNPG. Kinetic readings (24 h at 1 h intervals) were performed at 405 nm and 25 ºC using a Power Wavex spectrophotometer (Bio-Tek Instruments, USA).
In each enzyme assay, 2 sets of controls were set up; an enzyme control and a substrate control. All readings were taken against milli-Q water (Millipore Corporation, USA). All assays were performed in triplicate, unless otherwise stated. All results are reported as means ± standard deviations.
Microbiological analyses
Water samples of 100 mℓ volumes were filtered through membrane filters (Whatman 0.45 µm pore size) and placed on mFc plates for the detection of faecal coliforms at 44.5°C over 24 h; CM 1046 media (selective E. coli/coliform chromogenic medium) and m-Endo-agar plates (total coliforms) and incubated at 37°C for 24 h, after which colonies were counted. All colonies which developed a pink colour were counted as other coliforms while those that developed a purple colour were counted as E. coli on the CM 1046 media (the sum of both pink and purple colony counts gave the total coliform load of the water sample under consideration) while red colonies with metallic reflections were counted as total coliforms (m-Endo agar) and colonies with light to dark blue colonies were counted as faecal coliforms (mFc agar). These were then expressed as coliform colony-forming units (CFU) per 100 mℓ (Wutor et al., 2007a).
Results and discussion
The source of B-GAL enzyme was validated as from E. coli by using SDS-PAGE and MALDI-ToF (data not shown). The MALDI-ToF technique has been used successfully in the analysis of proteins, peptides, glycoproteins, oligosaccharides, and oligonucleotides and has the capability of measuring masses to within 0.01% of the molecular mass of the sample (Takach et al., 1997; Whitelegge, 2003; Kammerera et al., 2005). Enzymatic assays were performed alongside growth on selected media to correlate enzymatic activity and colony counts.
Wutor et al. (2007a) in a previous study observed a positive correlation between total coliform CFUs and B-GAL activity; R2 of 0.941 for CM 1046 media, and 0.7051 for m-Endo media, while Togo et al. (2006) established a correlation of 0.8909 between GUD activity and E. coli CFUs.
Detection of TC and FC using media
Generally, we observed a significant (P<0.05) correlation between TC and FC on m-Endo and mFc media with a R2 value of 0.9392 and a R2 value of 0.8818 for TC and FC on CM 1046 media (Figs. 2 and 3). Though different media were employed in this analysis, the benefits of an early warning signal on the microbiological status of water from assessing only total coliforms cannot be underestimated.
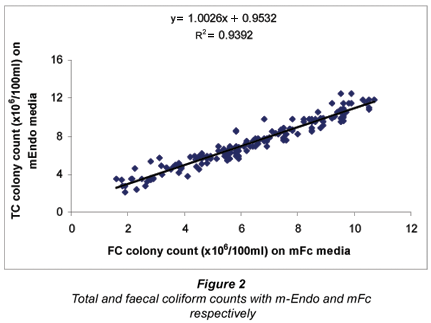
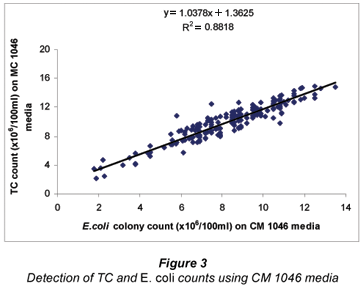
Detection of TC and FC
A positive correlation, with an R2 value of 0.8542 at 95% confidence interval was observed for E. coli colony counts and B-GAL activity (Fig. 4). It must be noted that this value, though lower than the previous statistically, yet still high, could be accounted for by the fact that not all E. coli cells will grow on the media, a major limitation in the use of the conventional plating procedure. However, those viable but non-culturable (VBNC) bacteria will still produce the B-GAL enzyme, thus making the enzyme assays better indicators than the plating on media (Pletschke et al., 2006; 2008). One major limitation in product development is the transferring of research from the laboratory to the field. In this work it is important to note that results were obtained from directly using the environmental samples to try easing the transfer.
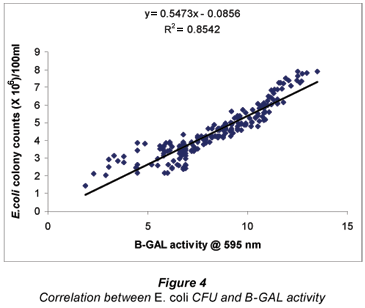
Farnleitner et al. (2001) and Garcia-Armisen et al. (2005) reported R2 values of 0.73 and 0.92 respectively, in correlation studies between GUD and E. coli. Variations in correlation coefficients for same enzyme can be attributed to differences in climatic conditions and the environments from which the samples were collected. Both factors dictate the nature and composition of interfering compounds in water samples. Furthermore, enzyme substrates are susceptible to interference from pollutants during analysis. For example, Farnleitner et al. (2002) employed 4-methylumbelliferyl-β-D-glucuronide, a fluorogenic substrate in their study, while the same substrate yielded unreliable results in our study (results not shown). Therefore, it is predicted that the future of water quality monitoring is likely to rely on a matrix approach placing less emphasis on any one single parameter (Ashbolt et al., 2001; Havelaar et al., 2001; Niemela et al., 2003; Van Heerden et al., 2005). Thus, the utilisation of the GUD and B-GAL assays remains relevant as complementary or presumptive tests for faecal pollution.
Conclusions
In conclusion, one could use total coliform β-D-galactosidase enzyme activity and CFU determination as a very good measure of microbial water quality assessment. Total coliform measurement may be considered as an imperfect but useful criterion of drinking water quality assessment and should find use at both small and large treatment facilities, as well as in routine analysis.
Acknowledgements
The authors would like to thank the Water Research Commission (WRC) of South Africa for financial assistance during the course of this study.
References
ASHBOLT NJ, GRABOW WOK and SNOZZI M (2001) Indicators of microbial water quality. In: Fewtrell L and Bartram J (eds.) Water Quality: Guidelines, Standards and Health. Risk Assessment and Management for Water-Related Infectious Disease. IWA Press, London, UK. 289-316. [ Links ]
BYAMUKAMA D, MACH RL, KANSIIME F, MANAFI M and FARNLEITNER AH (2005) Discrimination efficacy of fecal pollution detection in different aquatic habitats of a high-altitude tropical country, using presumptive coliforms, Escherichia coli, and Clostridium perfringens spores. Appl. Environ. Microbiol. 71 65-71. [ Links ]
DEVI R, ALEMAYEHU E, SINGH V, KUMAR A, and MENGISTIE E (2008) Removal of fluoride, arsenic and coliform bacteria by modified homemade filter media from drinking water. Bioresour. Technol. 99 2269-2274. [ Links ]
FARNLEITNER AH, HOCKE L, BEIML C, KAVKA GG and MACH RL (2002) Hydrolysis of 4-methylumbelliferyl-β-d-glucuronide in differing sample fractions of river waters and its implication for the detection of faecal pollution. Water Res. 36 975-981. [ Links ]
FARNLEITNER AH, HOCKE L, BEIWL C, KAVKA GG, ZEICHMEISTER T, KIRSCHNER AKT and MACH RL (2001) Rapid detection of Escherichia coli contamination in polluted water. Lett. Appl. Microbiol. 33 246-250. [ Links ]
GARCIA-ARMISEN T, LEBARON P and SERVAIS P (2005) β-D-glucuronidase activity assay to assess viable Escherichia coli abundance in fresh waters. Lett. Appl. Microbiol. 40 278-282. [ Links ]
GOFTI L, ZMIROU D, SEIGLE MF, HARTEMANN P and POTELON JL (1999) Waterborne microbiological risk assessment: a state of the art and perspectives. Epidemiol. Rev. 47 (1) 61-73. [ Links ]
GRABOW WOK, BOTMA KL, DE VILLIERS JC, CLAY CG and ERASMUS B (1999) Assessment of cell culture and polymerase chain reaction procedures for the detection of polioviruses in wastewater. Bull. World Health Organ. 77 973-980. [ Links ]
HAVELAAR A, BLUMENTHAL U, STRAUSS M, KAY D and BARTRAM J (2001) Guidelines: the current position. In: Fewtrell L and Bartram J (eds.) Water Quality: Guidelines, Standards and Health. World Health Organization and IWA Publishing, London, UK. [ Links ]
HÖRMAN A and HÄNNINEN M (2006) Evaluation of the lactose Tergitol-7, m-Endo LES, Colilert 18, Readycult Coliforms 100, Water-Check-100, 3M Petrifilm EC and DryCult Coliform test methods for detection of total coliforms and Escherichia coli in water samples. Water Res. 40 3249-3256. [ Links ]
HÖRMAN A, RIMHANEN-FINNE R, MAUNULA L, VON BONSDORFF CH, TORVELA N, HEIKINHEIMO A and HÄNNINEN ML (2004) Campylobacter spp., Giardia spp., Cryptosporidium spp., Noroviruses and indicator organisms in surface water in Southwestern Finland, 2000-2001. Appl. Environ. Microbiol. 70 (1) 87-95. [ Links ]
HUDSON LO, HARKLIN JW and BATTAGLIA M (1983) Coliforms in water distribution system a remedial approach. J. Am. Water Works Assoc. 75 564-568. [ Links ]
HUERTAS A, BARBEAU B, DESJARDINS C, GALARZA A, FIGUEROA MA and TORANZOS GA (2003) Evaluation of Bacillus subtilis and coliphage MS2 as indicators of advanced water treatment efficiency. Water Sci. Technol. 47 255-259. [ Links ]
KAMMERERA B, FRICKENSCHMIDT A, GLEITER CHM, LAUFER S and LIEBICH H (2005) MALDI-TOF MS Analysis of Urinary Nucleosides. J. Am. Soc. Mass Spectr. 16 (6) 940-947. [ Links ]
LECLERC H, DEVRIESE LA and MOSSEL DAA (1999) Taxonomical changes in intestinal (faecal) enterococci and streptococci: consequences on their use as indicators of faecal contamination in drinking water. J. Appl. Bacteriol. 81 459-466. [ Links ]
LECLERC H, MOSSEL DAA, EDBERG SC and STRUIJK CB (2001) Advances in the bacteriology of the coliform group: Their suitability as markers of microbial water safety. Ann. Rev. Microbiol. 55 201-234. [ Links ]
McFETERS GA and CAMPER AK (1983) Enumeration of indicator bacteria exposed to chlorine. Adv. Appl. Microbiol. 29 177-193. [ Links ]
McFETERS GA, KIPPIN JS and LECHEVALLIER MW (1986) Injured coliforms in drinking water. Appl. Environ. Microbiol. 51 (1) 1-5. [ Links ]
NIEMELA SI, LEE JV and FRICKER CR (2003) A comparison of the International Standards Organisation reference method for the detection of coliforms and Escherichia coli in water with a defined substrate procedure. J. Appl. Microbiol. 95 (6) 1285-1292. [ Links ]
NOBLE RT, MOORE DF, LEECASTER MK, McGEE CD and WEISBERG SB (2003) Comparison of total coliform, faecal coliform, and enterococcus bacterial indicator response for ocean recreational water quality testing. Water Res. 37 1637-1643. [ Links ]
PITKÄNEN T, PAAKKARI P, MIETTINEN IT, HEINONEN-TANSKI H, PAULIN L and HÄNNINEN M (2007) Comparison of media for enumeration of coliform bacteria and Escherichia coli in non-disinfected water. J. Microbiol. Methods. DOI: 10.1016/j.mimet.2006.10.007. [ Links ]
PLETSCHKE B, TOGO C and WUTOR V (2006) On-Line Real-Time Enzyme Diagnostic System for the Detection and Monitoring of Faecal Contamination of Water Intended for Drinking Purposes. WRC Report No. 1446/1/06. Water Research Commission, Pretoria, South Africa. [ Links ]
PLETSCHKE B, TOGO C, WUTOR V and LIMSON J (2008) On-Line Real-Time Enzymatic Biosensor System for the Rapid Detection of Faecal Contamination of Water Intended for Drinking Purposes. WRC Report No. 1603/1/08. Water Research Commission, Pretoria, South Africa. [ Links ]
ROMPRÉ A, SERVAIS P, BAUDART J, DE-RAUBIN MR and LAURENT P (2002) Detection and enumeration of coliforms in drinking water: current methods and emerging approaches. J. Microbiol. Methods 49 31-54. [ Links ]
ROSE JB and GRIMES DJ (2002) Reevaluation of Microbial Water Quality: Powerful New Tools for Detection and Risk Assessment. American Society of Microbiology, Washington, DC. [ Links ]
SCHRAFT H and WATTERWORTH LA (2005) Enumeration of heterotrophs, fecal coliforms and Escherichia coli in water: comparison of 3M™ Petrifilm™ plates with standard plating procedures. J. Microbiol. Methods 60 (3) 335-342. [ Links ]
STANDARD METHODS (2005) Standard Methods for the Examination of Water and Wastewater (20th edn.) Clesceri LS, Greenberg AE and Eaton AD (eds.). American Public Health Association, Washington DC. [ Links ]
STEVENS M, ASHBOLT N and CUNLIFFE D (2001) Microbial Indicators of Water Quality: An NHMRC Discussion Paper. National Health and Medical Council Act, Canberra. [ Links ]
TAKACH EJ, HINES WM, PATTERSON DH, JUHASZ P, FALICK AM, VESTAL ML and MARTIN SA (1997) Accurate Mass Measurements Using MALDI-TOF with Delayed Extraction. Communications from the 11th Int. Conf. on Methods in Protein Structure Analysis. J. Prot. Chem. 16 (5) 363-369(7). [ Links ]
TOGO C A, WUTOR VC and PLETSCHKE BI (2006) Properties of in situ Escherichia coli β-D-glucuronidase (GUS): evaluation of chemical interference on the direct enzyme assay for faecal pollution detection in water. Afr. J. Biotechnol. 5 (22) 2338-2344. [ Links ]
VAN HEERDEN J, EHLERS MM, HEIM A and GRABOW WOK (2005) Prevalence, quantification and typing of adenoviruses detected in river and treated drinking water in South Africa. J. Appl. Microbiol. 99 (2) 234-242. [ Links ]
VENTER SN (2000) Rapid Microbiological Methods: The Status Quo. Blue Pages, IWA, London, UK. [ Links ]
WHITELEGGE JP (2003) Thylakoid membrane proteomics. Photosynth. Res. 78 (3) 265-277. [ Links ]
WUTOR VC, TOGO CA and PLETSCHKE BI (2007A) Comparison of the direct enzyme assay method with the membrane filtration technique in the quantification and monitoring of microbial indicator organisms seasonal variations in the activities of coliforms and E. coli, temperature and pH. Water SA 33 (1) 107-110. http://www.wrc.org.za/downloads/watersa/2007/Jan%2007/2049.pdf [ Links ]
WUTOR VC, TOGO CA and PLETSCHKE BI (2007B) The effect of physico-chemical parameters and chemical compounds on the activity of β-d-galactosidase (B-GAL), a marker enzyme for indicator microorganisms in water. Chemos. 68 622-627. [ Links ]
Received 18 August 2008; accepted in revised form 8 January 2009.
* To whom all correspondence should be addressed
+1 (403) 3173358; fax: +1 (403) 3823156;
e-mail: WutorV@agr.gc.ca














