Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Water SA
On-line version ISSN 1816-7950
Print version ISSN 0378-4738
Water SA vol.34 n.5 Pretoria Oct. 2008
Distribution of the pill clam Pisidium langleyanum Melvill & Ponsonby, 1891 (Bivalvia: Sphaeriidae) in South Africa
KN de KockI, *; CT WolmaransII
IUnit for Environmental Sciences and Management, Potchefstroom Campus of the North-West University, Private Bag X6001, Potchefstroom 2520, South Africa
IISchool of Environmental Sciences and Development, Zoology, Potchefstroom Campus of the North-West University, Private Bag X6001, Potchefstroom 2520, South Africa
ABSTRACT
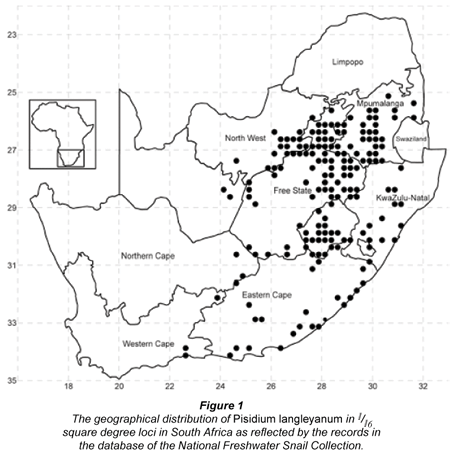
This article focuses on the geographical distribution and habitats of the pill clam, Pisidium langleyanum as reflected by 632 samples on record in the database of the National Freshwater Snail Collection (NFSC). The 177 different loci (1/16 -degree squares) from which these samples were collected display a relatively continuous distribution in southern Gauteng, north-eastern Free State, the central area of Mpumulanga and the western part of Lesotho. However, it is discontinuously spread through KwaZulu-Natal and the Eastern Cape, poorly represented in North West, nearly absent in the Northern and Western Cape and completely absent from Limpopo. Details of each habitat as described by collectors during surveys were statistically analysed, as well as altitude and mean annual air temperatures and rainfall for each locality. This species was reported from 10 of the 14 water-body types represented in the database, but the largest number of samples was recovered from rivers, swamps and streams. Chi-square and effect-size values were calculated and an integrated decision tree constructed from the data which indicated that temperature, altitude, types of water-body and substrata were the important factors that significantly influenced the distribution of P. langleyanum in South Africa. In view of the many agents reported for this genus elsewhere in the world that could facilitate its passive dispersal and the fact that this species was already recorded in 1891 from South Africa, it is suggested that its absence in large areas of this country could most probably be attributed to unsuitable environmental conditions. The possible effect of climatic changes on the geographical distribution of P. langleyanum and the conservation status of Pisidium species in South Africa is briefly discussed. The feasibility to exploit this species for monitoring heavy metal pollution in freshwater biotopes and its ability to act as intermediate host for economically important trematode parasites should be investigated.
Keywords: Pisidium langleyanum, pill clam, freshwater Bivalvia, geographical distribution, habitat analysis, South Africa
Introduction
The family Spheariidae includes the smallest freshwater bivalves and is distributed worldwide. According to Kuiper (1983) the origin of Pisidium is certainly Mesozoic and their centre of evolution lies without doubt in the Holarctic Region. Biodiversity of freshwater ecosystems is being lost at an alarming rate and still little is known of the functional role of most of this biodiversity (Vaughn and Hakenkamp, 2001). Although freshwater burrowing bivalves have the potential to strongly influence ecosystem processes these authors point to the fact that the ecosystem roles of burrowing freshwater bivalves have been particularly understudied. Unfortunately this also holds true for the bivalves of South Africa. To our knowledge research on the Spheariidae of South Africa, up till the present, has been limited largely to a few taxonomic studies conducted and published many years ago. In an account of the freshwater molluscs of the Belgian Congo Pilsbry and Bequaert (1927) reported 9 species of Pisidium described from Africa at that stage, but predicted that Africa would doubtless prove to have many more species of this genus when special methods for collecting aquatic minutiae are employed. It is therefore not surprising that a total of 20 species were reported for Africa by Mandahl-Barth (1988). With regard to South Africa Connolly (1939) reported the following three species of the genus Pisidium: P. langleyanum Melvill & Ponsonby, 1891, P. ovampicum Ancey, 1890 and P. costulosum Connolly, 1931. Two and a half decades later seven species were reported by Kuiper (1964) in his contribution towards knowledge of the South African species of the genus Pisidium. The four additional species added to the list of Connolly (1939) by this author comprised P. pirothi Jickeli, 1881, P. viridarium Kuiper, 1956, P. harrisoni Kuiper, 1964 and P. casertanum (Poli, 1791). According to Kuiper (1964), P. langleyanum, P. costulosum and P. harrisoni seem to be Southern African endemics, while the others are also known to occur elsewhere in Africa. Another species, P. (Parapisidium) reticulatum was reported from the Okavango Delta, Botswana (Appleton et al., 2003). In his account of the Mollusca of Southern Africa, Appleton (2002) mentions that 8 species of Pisidium have been reported from South Africa of which 6 are represented in the database of the National Freshwater Snail Collection (NFSC) of South Africa. We have no records of either P. harrisoni or P. reticulatum in our database.
In view of the fact that they can utilise habitats such as springs, small creeks and peat bogs where no other bivalves can survive, Korniushin (2000) is of the opinion that investigation of Pisidium species is important not only for understanding the structure and history of the fauna, but also because they could be used for monitoring environmental conditions. Their investigation is also important in view of reports from elsewhere in the world that representatives of this genus can serve as intermediate hosts for trematode parasites (Cannon, 1972; Rantanen et al., 1998). To our knowledge the role of local representatives of this genus in the epidemiology of mollusc-borne parasitic diseases has not yet been investigated. Little is also known of the conservation status of the South African Sphaeriidae, but Herbert (1998) is of the opinion that P. harrisoni may meet the criteria for Red listing. From elsewhere in the world it is reported that native burrowing bivalves are declining at a catastrophic rate (Vaughn and Hakenkamp, 2001). A significant decrease of species richness and density of gastropods and bivalves due to climatic warming could also be observed in a river system in France (Mouthon and Daufresne, 2006).
This is an account of the habitats and geographical distribution of P. langleyanum currently the Pisidium species with the largest number of records in the database of the NFSC of South Africa. The possible effect of climatic changes on the geographical distribution of P. langleyanum and the conservation status of Pisidium species in South Africa is briefly discussed.
Methods
Details of the habitats of all samples of P. langleyanum that could be located on the 1:250 000 topo-cadastral map series of South Africa, dating from 1956 until 2007, were extracted from the database of the NFSC. The majority of these samples were collected by staff of government and local health authorities and staff of the former Snail Research Unit at the Potchefstroom University. In the instructions supplied, collectors were requested to carefully screen also the sediment of water-bodies for burrowing molluscs. Guidelines were made available for the construction of scoops with a fine mesh which could be used as a spade to sample the upper layers of the substratum even for the smaller burrowing bivalve species. The number of loci (1/16 square degrees) in which the collection sites were located, was distributed in intervals of mean annual air temperature and rainfall, as well as intervals of mean altitude to illustrate the frequency of occurrence within specific intervals. Rainfall, temperature and altitude data were obtained in 2001 from the Computing Centre for Water Research, University of KwaZulu-Natal (disbanded since). A temperature index was calculated for all mollusc species in the database from their frequencies of occurrence within the selected temperature intervals and the results were used to rank them in order of association with low to high climatic temperatures. The method of calculation is discussed in detail in our earlier publications (De Kock and Wolmarans, 2005a; 2005b). Chi-square values (Statistica, Release 7, Nonparametrics, 2X2 Tables, McNemar, Fischer exact) were calculated to determine the significance in difference between the frequency of occurrence in, on, or at the range of options for each variable, such as type of water-body, type of substratum or temperature interval. In addition an effect size (Cohen, 1977) was calculated for all the different variables discussed in this paper. The effect size is an index which measures the degree of discrepancy between the frequency distribution of a given species in the set of alternatives of a given variable such as water-bodies, as compared to the frequency distribution of all other mollusc species in the database in the set of alternatives for the same variable (Cohen, 1977). According to this author values for this index in the order of 0.1 and 0.3 indicate small and moderate effects respectively, while values in the order of 0.5 and higher indicate practical significant large effects. More details of the significance and interpretation of specific values calculated for this statistic in a given situation, is discussed in our earlier publications (De Kock and Wolmarans, 2005a; 2005b).
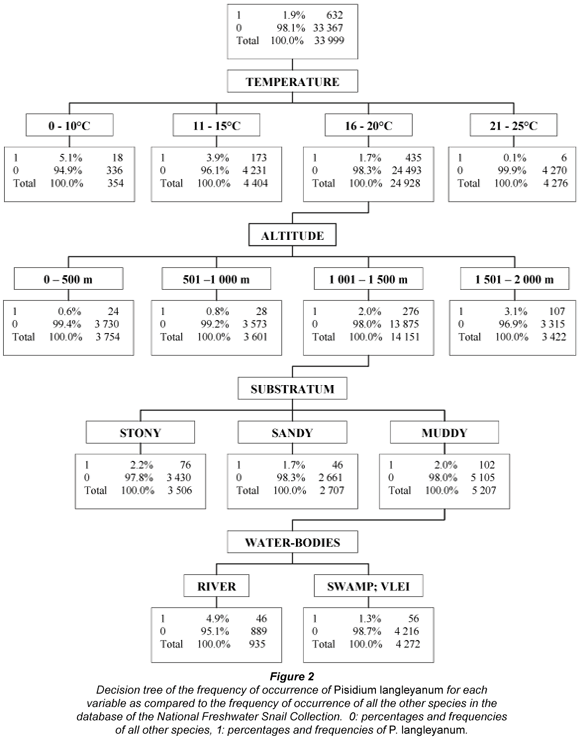
An integrated decision tree (Breiman et al., 1984) was also constructed from the data. This statistical model enables the selection and ranking of those variables that can maximally discriminate between the frequency of occurrence of a given species under specific conditions as compared to all other species in the database. This was accomplished by making use of the SAS Enterprise Miner for Windows NT Release 4.0, April 19, 2000 programme and Decision Tree Modelling Course Notes (Potts, 1999).
Results
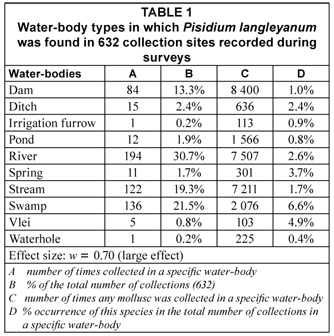
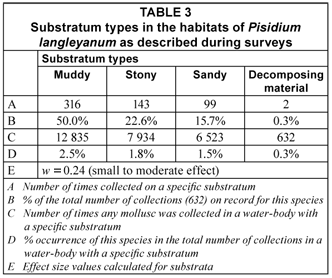
The 632 samples of P. langleyanum were recovered from 177 loci (Fig. 1). This species was collected in 10 of the 14 different water-body types represented in the database. Although the largest number of samples came from rivers (194), the 136 samples from swamps represented a higher percentage (6.6%) of the total number of times molluscs were reported from this type of water-body in the database (Table 1). The frequency of occurrence in swamps differed significantly (p < 0.05) from all the other water-body types except from vleis (χ2 = 0.47, df = 1; p > 0.05). The majority of samples were reported from perennial habitats (79.9%) of which the water was described as clear (79.4%), fresh (84.0%) and standing (47.9%). There were, however, no significant differences in the frequency of occurrence in water-bodies with fast running, slow running or standing water. Fifty percent of the samples were recovered from water-bodies with a predominantly muddy substratum (Table 3) and this differed significantly (p < 0.05) from the occurrence on all other substrata types on record in the database. The 316 samples of P. langleyanum recorded from habitats with a muddy substratum represented 2.5% of the 12 835 samples on record in the database for all mollusc species reported from this type of substratum (Table 3).
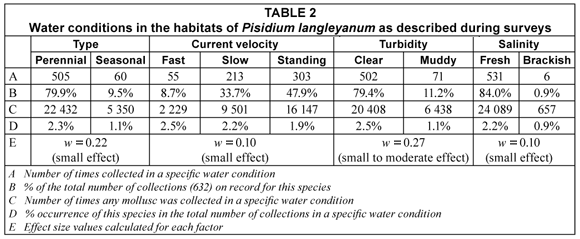
The majority of samples came from sites which fell within the temperature interval ranging from 16 to 20°C; however, the 18 and 173 samples from sites falling within the 0 to 10°C and 11 to 15°C intervals respectively, both represented higher percentages of the total number of times that molluscs were recovered from sites falling within a specific interval (Table 4). The frequency of occurrence in sites falling within the 16 to 20°C interval differed significantly (p < 0.05) from the other three intervals. The 452 samples reported from sites falling within the rainfall interval ranging from 601 to 900 mm/a represented more than 70% of the total number of samples on record for this species and differed significantly (p < 0.05) from the frequency of occurrence within any of the other rainfall intervals in the database. Although the 316 samples recovered from sites falling within the 1 001 to 1 500 altitude interval accounted for exactly 50% of the total number of samples on record for this species, the 118 samples falling within the 2 001 to 2 500 altitude interval represented a much higher percentage (20.1%) of the total number of times that molluscs were recovered from sites falling within a specific interval (Table 4). The frequency of occurrence from sites falling within the 2 001 to 2 500 altitude interval consequently differed significantly (p < 0.05) in this respect from all the other altitude intervals.
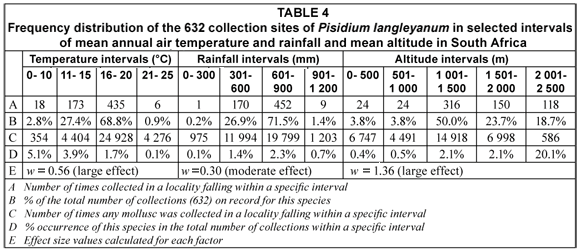
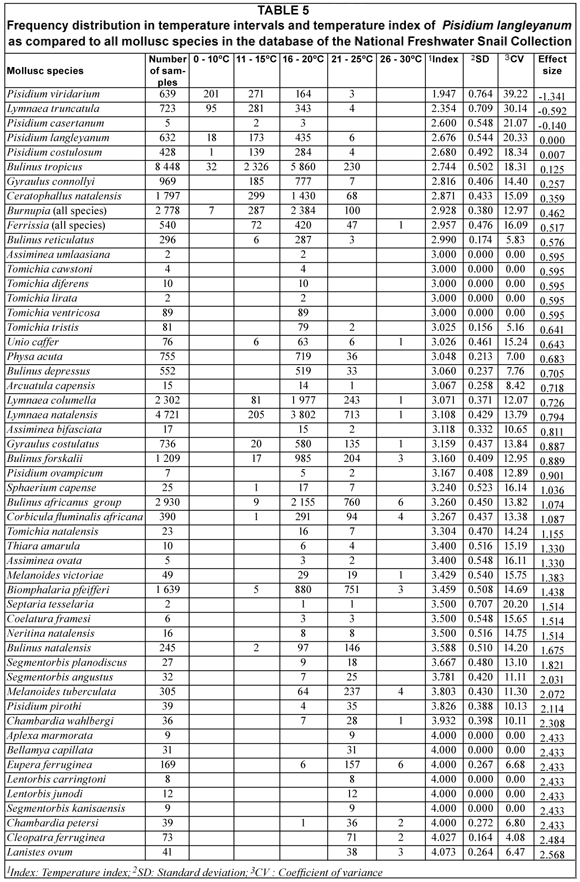
From the effect size values calculated for the various parameters investigated it can be deduced that temperature, altitude and water-bodies as such, had a large effect and substratum a small to moderate effect on the distribution of P. langleyanum as reflected by the samples on record in the NFSC (Tables 1 to 4). These parameters were likewise selected by the decision tree analysis as being important in establishing the documented geographical distribution of this species in South Africa. The temperature index calculated for P. langleyanum ranked it in the fourth position in respect of its association with low temperatures as compared to all other mollusc species represented in the database of the NFSC (Table 5). It can furthermore be deduced from the effect size values calculated for the temperature indices that this species differed significantly (w > 0.5) from 47 of the other 53 mollusc species on record in the database (Table 5).
Discussion
Only 3 Pisidium species, namely P. langleyanum, P. viridarium and P. costulosum are well-represented in the database of the NFSC. Identification of these species was determined exclusively on shell characters based on guidelines of Kuiper (1964) and the revised key published by Appleton (2002).
Pisidium langleyanum which is the most widespread species of this genus in South Africa, as reflected by records of the NFSC, can easily be distinguished from the other two species by the relatively larger umbones, which are placed way to the back (Mandahl-Barth, 1988) and the sculpture of regular dense striae of the shell (Appleton, 2002). The prominent umbones of this species were already mentioned as diagnostic feature by Pilsbry and Bequart (1927). This species was originally described in 1891 from Port Elizabeth by Melvill and Ponsonby (1891); however, the oldest record in the database of the NFSC only dates back to 1958 and was collected in a dam in the Boshof district of the Free State.
The 177 loci from which the 632 samples of P. langleyanum were recovered are located largely in the eastern parts of South Africa (Fig. 2). This species is well-represented in Gauteng, south eastern Mpumulanga, Lesotho and north eastern Free State. It is discontinuously spread through a large area of the Eastern Cape and KwaZulu-Natal, completely absent from Limpopo and poorly represented in Northern and Western Cape.
Sphaeriids have many and frequent possibilities of passive dispersal from one water-body to another, even from one river system to another over mountains and seas (Kuiper, 1983). According to this author they are carried by insects, birds, amphibians and fish. A resting Pisidium keeps its valves slightly gaping and quickly closes its valves when touched by the antenna or tarsus of a beetle or a feather of a diving bird (Kuiper, 1983). Attached in this way by means of its closed valves specimens are able to withstand incidental environmental dryness rather well (Kuiper , 1983) and can consequently be transported over long distances. The important role played by active and passive dispersal in establishing the geographical distribution of Pisidium in Michigan, U.S.A. is discussed in detail by Heard (1962). As mentioned earlier, the presence of P. langleyanum in South Africa was established as early as 1891. The absence of this species from large areas in this country should therefore rather be attributed to unfavourable environmental conditions than to the lack of opportunity to disperse. From the effect sizes calculated for the parameters investigated (Table 4) it is clear that temperature and altitude had been important factors in establishing the documented geographical distribution of this species in South Africa. Its close association with low climatic temperatures as reflected by its temperature index (Table 5), supplies a plausible explanation for the complete absence of any records of this species from Limpopo and this supports the supposition of Brown (1978) that P. langleyanum could possibly be restricted to the cooler areas of South Africa. In view of the above it seems justifiable to assume that global warming could have a limiting effect on the extent of the geographical distribution of this species in South Africa. A significant decrease of species diversity and density of gastropods due to climatic warming were reported for the Saône River and its two main tributaries (France) (Mouthon and Daufresne, 2006). These authors are of opinion that an increase in the frequency of hot summers could lead to the extinction of more than half the mollusc species currently inhabiting the potamic area of the River Saône. Most of the sites on record for P. langleyanaum in the database of the NFSC were sampled many years ago and have not been revisited since, but specimens of this species, often sharing the same habitat with P. costulosum, are still recovered from sites in the Mooi River catchment area recorded earlier by De Kock and Van Eeden (1969). However, the related species P. pirothi reported from the Letaba River in the Kruger National Park in 1967 (Oberholzer and Van Eeden, 1967) could not be recovered during extensive surveys conducted in 1995, 2001 and 2006 (De Kock and Wolmarans, 1998; De Kock et al., 2002; Wolmarans and de Kock, 2006). Whether this could be attributed to warmer climatic temperatures is debatable because the temperature index calculated for P. pirothi ranks it eleventh out of 53 mollusc species due to its association with high climatic temperatures (Table 5).
As reported for Corbicula fluminalis africana (De Kock and Wolmarans, 2007a), P. langleyanaum is widespread in a variety of freshwater habitats including marshes across Southern Africa except for the arid western parts. Although a considerable number of records of various other mollusc species were reported from these western parts, the diversity is significantly lower as compared to the situation in the eastern parts of this country (De Kock et al., 1989; De Kock et al., 2001; De Kock et al. 2002; De Kock and Wolmarans, 2005c; De Kock and Wolmarans, 2007b). The obvious explanation for this phenomenon according to Brown (1978) is that water in itself is the primary requisite for an aquatic mollusc and that there can be no doubt that the virtually arid zone extending from the Western Cape through Namibia, the Northern Cape and Botswana is of overriding importance in that region.
Being a bottom dweller the possibility exists that this species could be more widespread than reported in this paper due to the fact that it could easily be overlooked during a casual survey. The majority of samples on record in the database of the NFSC have, however, been collected by specially trained staff. Most of these surveys were done in shallow water near the margin of water-bodies which might not have been the most appropriate procedure for collecting bivalves. However, in a quantitative survey of molluscs in the Mooi River (North-West Province) with various appropriate devices for the quantitative sampling of benthic organisms, it was established that the largest populations of P. langleyanum were to be found either in or on the sediments in shallow runs near the margins of water-bodies (De Kock and Van Eeden, 1969).
As mentioned earlier, it was suggested by Korniushin (2000) that Sphaeriidae could be used in monitoring environmental conditions in some countries. The lifespan of Pisidium spp. is typically 1 to 3 years (Heard, 1965; Holopainen, 1979; Vincent et al., 1981; Rantanen et al., 1998) and in view of the fact that P. langleyanum can exploit a large variety of water-bodies, are a bottom dweller and filter feeder and are already present in many river systems in South Africa, it could be worthwhile to investigate the feasibility of utilising it as an indicator of heavy metal pollution. It is a known fact that bivalves can accumulate heavy metals from superficial sediments (Pourang, 1996). Due to the fact that bivalves yearly produce an incremental layer of their shells, consisting mainly of calcium carbonate and a small fraction of organic substance, Carell et al. (1987) maintain that environmental history can be revealed by analysing elements in mussel shells along the annual growth increments. According to these authors live animals and museum material, as well as sub-fossil material could be used for these analyses. The collection of preserved samples of bivalves from all over South Africa in the NFSC span a period of more than four decades and could prove to be a most valuable asset for similar investigations.
As mentioned earlier the functional role of freshwater burrowing bivalves have been particularly understudied, however, they filter phytoplankton, bacteria and particulate organic matter from the water column and they affect nutrient dynamics in freshwater systems through excretion and biodeposition of faeces and pseudofaeces (Vaughn and Hakenkamp, 2001). James (1987) also came to the conclusion that excretory products from bivalves should be an important and readily available source of nutrients to be utilised by phytoplankton. Although one might consider the possible impacts of populations of P. langleyanum on the freshwater biotope as unimportant due to the relatively small size of individuals, the large number of specimens often encountered in suitable habitats could compensate for their lack in size. In a quantitative survey of molluscs in the Mooi River (North West Province) densities of up to 4 483 specimens/m2 were recorded for P. langleyanum (De Kock and Van Eeden, 1969) and Kuiper (1983) mentions a figure of 10 000 or even more per square metre for some species of Pisidium in central European lakes.
A high prevalence of the helminth parasite, Bunodera luciopercae for which the perch is a compatible definite host, has been reported for a population of P. amnicum in a river in eastern Finland (Rantanen et al., 1998). It was also established that the yellow perch was the definite host for B. lucioperca in Canada (Cannon, 1972). To our knowledge the ability of P. langleyanum to act as intermediate host for digenean helminth parasites has not yet been investigated. In view of the reports mentioned above it is recommended that the possible role of P. langleyanum as intermediate host of trematode parasites should be investigated. As reported from elsewhere in the world, some of these parasites could have a negative impact on local fish populations and on the aquaculture industry.
The conclusion of Vaughn and Hakenkamp (2001) that native burrowing bivalves are declining at a catastrophic rate in North America supports the earlier report by Bogan (1993) that freshwater bivalves are threatened and declining globally. Little is known about the conservation status of bivalves in South Africa. The numbers of specimens and diversity of bivalve species, as well as the number of positive sites reported from the Kruger National Park in 1967 (Oberholzer and Van Eeden, 1967) have declined remarkably as revealed by our subsequent surveys mentioned earlier. In a personal communication by JA Day to CC Appleton (Appleton, 2002) it was also mentioned that the bivalve Unio caffer (Krauss, 1848) have become rare in the south-western Cape. Our personal observations led to the conclusion that this species has also become rare in the Mooi River basin, North-West Province where it was relatively common in specific habitats in the 1960s (De Kock and Van Eeden, 1969). With regard to the Pisidium species, it was already mentioned in the introduction that Herbert (1998) is of opinion that P. harrisoni may meet the criteria for Red listing. The absence of P. pirothi from sites in the Kruger National Park from where it was previously collected, was also mentioned earlier.
The relatively widespread distribution and ability of P. langleyanum to exploit a wide variety of water-bodies also make it an ideal candidate for monitoring heavy metal pollution in our country. The shells of preserved specimens of bivalves in the NFSC could also be analysed as a possible indicator of environmental history as is done elsewhere in the world (Carell et al., 1987).
The records of molluscs in the NFSC date back to 1954 and such a long data set could form an important and excellent baseline for future projects dealing with conservation and climatic change. Efforts should therefore be made to update the geographical distribution of the freshwater molluscs in South Africa, an aspect that is long overdue and has been grossly neglected since the early 1980s.
Acknowledgements
The authors wish to thank the following persons for their assistance in processing the data: Professors HS Steyn, head of the Statistical Consulting Service and DA de Waal of the Centre for Business Mathematics and Informatics of the North-West University, Potchefstroom Campus. We are also indebted to the North-West University for financial support and infrastructure.
References
APPLETON CC (2002) Mollusca. In: De Moor IJ and Day JA (eds.) Guides to the Freshwater Invertebrates of Southern Africa. Volume 6, Chapter 3: Arachnida & Mollusca, Araneae, Water Mites & Mollusca. WRC Report No 182/02. Water Research Commission, Pretoria, South Africa. pp 42-125. [ Links ]
APPLETON CC, CURTIS BA, ALONSO LE and KIPPING J (2003) Freshwater invertebrates of the Okavango Delta, Botswana. In: A rapid Biological Assessment of the Aquatic Ecosystems of the Okavango Delta: High Water Survey. RAP Bull. Biol. Ass. 27 58-68; 123-136. [ Links ]
BOGAN AE (1993) Freshwater bivalve extinctions (Mollusca: Unionidae): A search for causes. Am. Zool. 33 599-609. [ Links ]
BREIMAN L, FRIEDMAN JH, OLSHEN RA and STONE CJ (1984) Classification and Regression Trees. Chapman and Hall. [ Links ]
BROWN DS (1978) Freshwater molluscs. In: Werger MJA (ed.) Biogeography and Ecology of Southern Africa. Junk, The Haque. pp 1153-1180. [ Links ]
CANNON LRG (1972) Studies on the ecology of the papillose allocreadiid trematodes of the yellow perch in Algonquin Park, Ontario. Can. J. Zool. 50 1231-1239. [ Links ]
CARELL B, FORBERG S, GRUNDELIUS E, HENRIKSON L, JOHNELS A, LINDH U, MUTVEL H, OLSSON M, SVÄRDSTRÖM and WESTERMARK T (1987) Can mussel shells reveal environmental history. Ambio 16 2-10. [ Links ]
COHEN J (1977) Power Analysis for the Behaviour Sciences. (revised edn.) Academic Press, Orlando, USA. [ Links ]
CONNOLLY M (1939) A monographic survey of the South African non-marine Mollusca. Ann. S. Afr. Mus. 33 1-660. [ Links ]
DE KOCK KN, JOUBERT PH and PRETORIUS SJ (1989) Geographical distribution and habitat preferences of the invader freshwater snail species Lymnaea columella (Mollusca: Gastropoda) in South Africa. Onderstepoort J. Vet. Res. 56 271-275. [ Links ]
DE KOCK KN and VAN EEDEN (1969) Die verspreiding en habitatseleksie van die Mollusca in die Mooirivier, Transvaal. Wetenskaplike Bydraes van die PU vir CHO, Reeks B: Natuurwetenskappe 8 1-119. [ Links ]
DE KOCK KN and WOLMARANS CT (1998) A re-evaluation of the occurrence of freshwater molluscs in the Kruger National Park. Koedoe 41 1-8. [ Links ]
DE KOCK KN and WOLMARANS CT (2005a) Distribution and habitats of the Bulinus africanus species group, snail intermediate hosts of Schistosoma haematobium and S. mattheei in South Africa. Water SA 31 117-126. http://www.wrc.org.za/downloads/watersa/2005/Jan-05/1775.pdf [ Links ]
DE KOCK KN and WOLMARANS CT (2005b) Distribution and habitats of Bulinus depressus and possible role as intermediate host of economically important helminth parasites in South Africa. Water SA 31 491-496. http://www.wrc.org.za/downloads/watersa/2005/Oct-05/1771.pdf [ Links ]
DE KOCK KN and WOLMARANS CT (2005c) Distribution, habitats and role as intermediate host of the freshwater snail, Bulinus forskalii, in South Africa. Onderstepoort J. Vet. Res. 72 165-174. [ Links ]
DE KOCK KN and WOLMARANS CT (2007a) Distribution and habitats of Corbicula fluminalis africana (Mollusca: Bivalvia) in South Africa. Water SA 33 709-715. http://www.wrc.org.za/downloads/watersa/2007/Oct%2007/2069.pdf [ Links ]
DE KOCK KN and WOLMARANS CT (2007b) Verspreiding en habitats van Ceratophallus natalensis (Mollusca: Planorbidae) in Suid-Afrika. SA Tydskr. Natuurwet. Teg. 26 109-119. [ Links ]
DE KOCK KN, WOLMARANS CT, BORNMAN M and MAREE DC (2002) Verspreiding en habitats van Bulinus tropicus, tussengasheerslak van die peervormige bot, Calicophoron microbothrium, in Suid-Afrika. SA Tydskr. Natuurwet. Teg. 21 114-120. [ Links ]
DE KOCK KN, WOLMARANS CT and DU PREEZ LH (2002) Freshwater mollusc diversity in the Kruger National Park: a comparison between a period of prolonged drought and a period of exceptionally high rainfall. Koedoe 45 1-11. [ Links ]
DE KOCK KN, WOLMARANS CT, STRAUSS HD and KILLIAN M (2001) Verspreiding en habitats van Lymnaea natalensis, tussengasheerslak van die lewerbot Fasciola gigantica, in Suid-Afrika. SA Tydskr. Natuurwet. Teg. 20 49-53. [ Links ]
HEARD WH (1962) Distribution of Sphaeriidae (Pelecypoda) in Michigan, USA. Malacologia 1 139-161. [ Links ]
HEARD WH (1965) Comparative life histories of North American pill clams (Sphaeriidae: Pisidium).1,2 Malacologia 2 381-411. [ Links ]
HERBERT DG (1998) Molluscan conservation in South Africa: diversity, issues and priorities. J. Conchol. Spec. Publ. 2 61-76. [ Links ]
HOLOPAINEN IJ (1979) Population dynamics and production of Pisidium species (Bivalvia, Spheariidae) in the oligotrophic and mesohumic lake Pääjärvi, southern Finland. Arch. Hydrobiol./Suppl. 54 466-508. [ Links ]
JAMES MR (1987) Ecology of the freshwater mussel Hydridella mensiesi (Gray) in a small oligotrophic lake. Arch. Hydrobiol. 108 337-348. [ Links ]
KORNIUSHIN AV (2000) Review of the family Spheariidae (Mollusca: Bivalvia) of Australia, with the description of four new species. Rec. Austr. Mus. 52 41-102. [ Links ]
KUIPER JGJ (1964) Contribution to the knowledge of the South African species of the genus Pisidium (Lamellibranchiata). Ann. S. Afr. Mus. 48 77-95. [ Links ]
KUIPER JGJ (1983) The Sphaeriidae of Australia. Basteria 47 3-52. [ Links ]
MANDAHL-BARTH G (1988) Studies on African Freshwater Bivalves. Danish Bilharziasis Laboratory, Charlottenlund, Denmark. [ Links ]
MOUTHON J and DAUFRESNE M (2006) Effects of the 2003 heatwave and climatic warming on mollusc communities of the Saône: a large lowland river and of its two main tributaries (France). Global Change Biol. 12 441-449. [ Links ]
OBERHOLZER G and VAN EEDEN JA (1967) The freshwater molluscs of the Kruger National Park. Koedoe 10 1-42. [ Links ]
PILSBRY HA and BEQUART J (1927) The aquatic mollusks of the Belgian Congo, with a geographical and ecological account of Congo malacology. Bull. Am. Mus. Nat. Hist. 53 69-602. [ Links ]
POTTS WJE (1999) Decision Tree Modeling Course Notes. SAS Institute Inc.Cary, USA. [ Links ]
POURANG N (1996) Heavy metal concentrations in superficial sediments and benthic macroinvertebrates from Anzali wetland, Iran. Hydrobiol. 331 53-61. [ Links ]
RANTANEN JT, VALTONEN ET and HOLOPAINEN IJ (1998) Digenean parasites of the bivalve mollusc Pisidium amnicum in a small river in eastern Finland. Dis. Aquat. Org. 33 201-208. [ Links ]
VAUGHN CC and HAKENKAMP CC (2001) The functional role of burrowing bivalves in freshwater ecosystems. Freshwater Biol. 46 1431-1446. [ Links ]
VINCENT B, VAILLANCOURT G and LAFONTAINE N (1981) Cycle of development, growth, and production of Pisidium amnicum (Mollusca: Bivalvia) in the St. Lawrence River (Quebec). Can. J. Zool. 59 2350-2359. [ Links ]
WOLMARANS CT and DE KOCK KN (2006) The current status of freshwater molluscs in the Kruger National Park. Koedoe 49 39-44. [ Links ]
* To whom all correspondence should be addressed.
+2718 299-2380; fax: +2718 299-2370;
e-mail: kenne.dekock@nwu.ac.za














