Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Water SA
On-line version ISSN 1816-7950
Print version ISSN 0378-4738
Water SA vol.34 n.5 Pretoria Oct. 2008
Interplay of factors involving chlorine dose, turbidity flow capacity and pH on microbial quality of drinking water in small water treatment plants
CL ObiI; JO IgumborII, *; MNB MombaIII; A SamieIV
IAcademic and Research Directorate, Walter Sisulu University, Nelson Mandela Drive, Mthatha, Eastern Cape, South Africa
IIDepartment of Public Health, University of Venda, South Africa
IIIDepartment of Water Care, Tshwane University of Technology, Pretoria
IVDepartment of Microbiology, University of Venda, South Africa
ABSTRACT
In an endeavour to ascertain the interplay of factors involving chlorine dose, turbidity and flow capacity on microbial quality of drinking water in small water treatment plants (SWTPs), data from a previous study were analysed.
The findings showed that most of the SWTPs were not producing water of safe microbial quality. Fifty one percent (51%) and seventy three (73%) of the SWTPs were below the stipulated limits for residual chlorine in final water and water at the point of use respectively. Current flow capacity was a major determinant of the microbial water quality indicators but no association was found between the dose of chlorine used for water treatment and the microbial water quality indicators. However, a combination of the amount of chlorine dose used up during treatment, flow capacity and change in turbidity contributed to about 65% of the amount of heterotrophic plate counts removed from raw water. Current flow capacity contributed less than 14% of the variation in chlorine dose used in water treatment at the plants. Turbidity tended to correlate and contribute more to the prediction of total coliform counts while faecal coliform counts were determined by current flow capacity and conductivity. Treatment plants with current flow capacity of over 50 Mℓ/d tended to be more efficient in heterotrophic plate count removal.
In conclusion, this study noted that most of the SWTPs were using a chlorine dosage that was below the amount required by their respective current flow capacity; possible micro-organism resistance to chlorine and the significant effect of the level of turbidity on the microbial quality of treated water.
Keywords: microbial indicators, chlorine dose, current flow capacity, turbidity, water treatment plants
Introduction
Over the past decade, the South African Government has continued to demonstrate a commitment to improve access to safe water supply and also to bridge the urban-rural disparities in access to potable water. This is evidenced by the marked reduction in the number of South Africans without access to potable water supply from over 15 m. (12 m. of whom were living in rural areas) in 1994 to less than 4 m. in 2004 (StatSA, 2004). This effort is geared towards facilitating social development and equity, self-reliance and health promotion under its National Reconstruction and Development Plan (RDP).
Crucial to this process are the small water treatment plants (SWTPs) in both rural and peri-urban areas. The SWTPs are defined as water treatment systems that are installed in areas which are not well serviced, and which do not normally fall within the confines of urban areas. SWTPs include water supplies from boreholes and springs that are chlorinated; small treatment systems for rural communities; treatment plants of small municipalities and establishments such as rural hospitals, schools, clinics and forestry stations. Most of these applications fall within the category of small plants of less than 2.5 Mℓ/d. Plants of up to 25 Mℓ/d may sometimes also fall into this category (DWAF, 1998).
SWTPs strive to provide water of adequate quantity and quality to the communities they serve but achieving this objective has been fraught with many challenges (Momba et al., 2006; Obi et al., 2007). Such challenges are lack of relevant technical capacity for water treatment processes, including chlorine dose calculations, flow-rate determinations and basic repairs of equipment. There are also administrative issues facing SWTPs. These include poor working conditions, frequent chemical stock depletion, lack of maintenance culture and a lack of emergency preparedness (Obi et al., 2007).
In the light of the above challenges, it is essential to establish the interplay between the amount of chlorine dose used-up during the water treatment process, changes in turbidity and current flow capacity of the treatment plants and the quality of treated water. Nevondo and Cloete (1999) and Momba et al. (2004) also expressed similar concerns over the ramifications of the challenges facing the SWTPs on water quality. The inability of SWTPs to reduce the turbidity of drinking water to limits that are safe for human consumption has also been reported by previous studies (Obi et al., 2007; Momba et al., 2006; Momba et al., 2004). Water turbidity level is known to significantly affect the microbial quality of drinking water either by encouraging the growth and survival of micro-organisms or by decreasing the efficiency of chlorine in water treatment (Health Canada, 2006; Snead et al., 1980; Hoff, 1978).
Water quality is commonly measured by indicators such as heterotrophic plate count (HPC), total coliform count (TCC) faecal coliform counts (FCC) and E. coli, based on their presence, concentration, sensitivity to disinfection, relative survival in water when compared to other water-borne pathogens of faecal origin and the efficiency of their detection processes. Each indicator has notable limitations given that they are proxy measures of water quality and to minimise the indicator-specific limitations in water quality monitoring, it is often advisable to use multiple indicators at a time. This is particularly important in view of reports that have suggested that the absence of an indicator organism does not mean the absence of other pathogenic organisms (Zamxaka, 2004). Beyond microbial measurements of water quality, safe water also has physico-chemical qualities such as temperature, pH, salinity, hardness, dissolved oxygen and phosphates. These qualities are readily affected by climatic events and impact on the survival of micro-organisms and efficiency of treatment processes (DWAF, 1996). This study which was predicated on the statistical analyses of data obtained from a previous study (Obi et al., 2007) , was conducted to ascertain the interplay of factors with respect to chlorine dose, turbidity and flow capacity.
Materials and methods
Study site
The study sites were rural and peri-urban establishments in Limpopo Province. They included: Phiphidi, Vondo, Mutale, Dzingahe, Mudaswali, Damni, Dzindi, Mutshedzi, Tshedza, Tshifhire, Tshakhuma regional, Tshakhuma, Makhado, Messina, Shikudu, Mhinga, Malamulele, Nkowankowa, Semerela, Thapane, Nkambako, Letsitele, Tzaneen, George's Valley. In the Mpumalanga Province they included Lydenburg, Hazyview, White River RE, White River Country Estate, Malelane, KaNyamazane regional, KaNyamazane, Nelspruit, Matsulu, Sabie, Machadodorp, Kruger Dam), Hendrina, Waterval, Belfast, Witbank, Presidentsrus, Middelburg, Dullstroom (Obi et al., 2007). Limpopo and Mpumalanga Provinces are situated in the north-eastern regions of South Africa and are among the least resourced provinces.
Plant survey
A survey of technical and administrative issues of various water treatment plants was conducted. The survey started with a short introductory meeting followed by a plant tour and interview with the plant operators and other superintendents, councillors or representatives. The plant tour was done in the presence of the superintendent of the plant or his representative and at least one plant operator. Different units comprising the plants were inspected in detail to monitor their functions on the day of the survey. Information concerning administrative issues such as the training of operators, their salaries, benefits, decision making, ownership of the SWTPs, security and any upgrade of the plant was sought through the use of questionnaires. Measurements were made to determine the size of the baffling chambers, sedimentation tanks and filters. Type of coagulant, filtration types, disinfection types, lime dosage as well as dosing pumps were determined during the plant tour. The operators present at the plant at the time of survey were interviewed individually (Obi et al., 2007).
Sample collection
A total of 55 rural and peri-urban SWTPs and distribution systems from the two provinces were visited and samples were collected from raw water, final water and water at the point of use. These sites were conveniently selected based on their accessibility and willingness of plant management to participate in the study. Water samples from each collection point were collected in 2 sterile 500 mℓ plastic bottles. The physicochemical parameters were measured immediately using the sample from one bottle and the other one was immediately kept in ice in a cooler box and transported back to the laboratory within 24 h of collection for the microbiological analysis. The survey was conducted from 26 August 2004 to 10 March 2005 (Obi et al., 2007).
Physico-chemical analysis
Physico-chemical analyses were conducted on site. Values measured included turbidity, temperature, conductivity and pH of water. The JENWAY pH meter 3150 was used for the measurement of the pH; The CRISON CM35 conductivity meter was used for the measurement of conductivity whilst the HACH Model 2100P portable turbidimeter was used to measure the turbidity of the samples. All the measurements were done in triplicate and their averages were considered (Obi et al., 2007).
Microbiological analysis
Microbiological analyses including data on heterotrophic plate counts, total coliforms, faecal coliforms and enteric bacteria were generated as already reported (Obi et al., 2007).
Data analysis
This study used means, standard deviations and percentages to describe the microbial and physicochemical indicators across the water samples and the general performance of the SWTPs. Pearson correlation of the variables was used to establish associations between the various parameters of water quality. Following this, the means of correlating variables was compared across their attributes, using t-tests, analysis of variance and the LSD post hoc test. The contribution of varied variables to the prediction of another variable was done using a stepwise regression analysis.
Results
The results presented in this section are further to an earlier publication by Obi et al. (2007). The earlier publication provided a basic description of the microbial and physicochemical properties of water samples collected from SWTPs in Limpopo and Mpumalanga Provinces. Using the same data, the results below demonstrate the interplay between the microbial and physicochemical properties of the water samples, chlorine does and current flow capacity at the designated SWTPs.
The currents analysis revealed that a greater proportion of the SWTPs were able to attain the stipulated safety limits for faecal coliform counts when compared to the proportion that met the safety limits for heterotrophic plate counts and total coliform counts respectively (Fig. 1). The 29 out of 53 (54.7%) SWTPs that exceeded the safety limits for heterotrophic plate count (0 cfu/100 mℓ-1) at point of use in the two provinces supply water to the following places: Thabazimbi, Tzaneen, Waterval Boven, White River regional, Letsetele, Dzingahe, Modimole, White River Country Estate, Dullstroom, Nkowankowa, Bela Bela, Nelspruit, Ohrigstadt, Nkambako, Presidentsrus, George's Valley, Middelburg (Vaalbank), Witbank, Sabie, Burgersfort, Seshego, Mutale, Machadodorp, Malelane, Vondo, KaNyamazane old plant, Phiphidi, Malamulele, Middelburg (Kruger Dam). At these sites heterotrophic plate counts at point of use ranged from 2 cfu/100 mℓ-1 in Thabazimbi SWTP catchment area in Limpopo Province to 6 500 cfu/100 mℓ-1 in Middelburg (Kruger Dam) catchment in Mpumalanga Province.
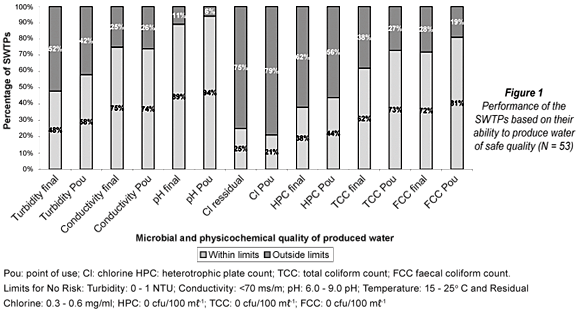
In terms of the physicochemical parameters, 31 out of the 53 SWTPs exceeded the safety limits for turbidity at the point of use (Fig. I). The pattern of water quality measures in Fig. I demonstrates that the proportion of SWTPs that attained safe water limits increased at point of use when compared to the proportions that attained safe water quality limits for the final water. It was also observed that a majority of the plants were unable to meet the stipulated safety limits for residual chlorine. The data further revealed that 51% and 73% of the SWTPs supplied water below the stipulated limits for residual chlorine in final water and water at the point of use respectively while 24% and 6% were above the recommended limits for residual chlorine in final water and water at the point of use respectively.
Effect of location, current flow capacity and type of ownership on treated water quality
Table 1 shows the measures of association between location of the SWTPs, its current flow capacity, type of ownership and the various indicators of the quality of final water and water at the point of use. A further comparison of the means of the variables that were found to be associated with location, by province, showed that SWTPs in Mpumalanga Province had a significantly higher dose of residual chlorine (0.7747 mg/mℓ) in their final water when compared to mean residual chlorine in final water samples from the SWTPs in Limpopo Province (0.4027 mg/mℓ) (p < 0.030). The average residual chlorine in final water samples from the SWTPs in Mpumalanga Province also exceeds the stipulated safety limit of between 0.3 mg/mℓ and 0.6 mg/mℓ. Similarly, the differences in the mean of microbial and physic-cochemical variables such as the association with the categories of current flow capacity and type of ownership are detailed in Tables 2 and 3 respectively.
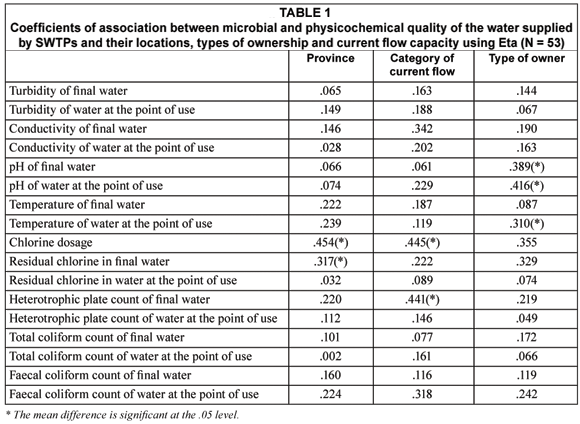
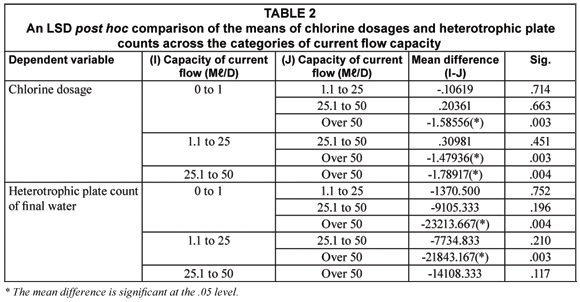
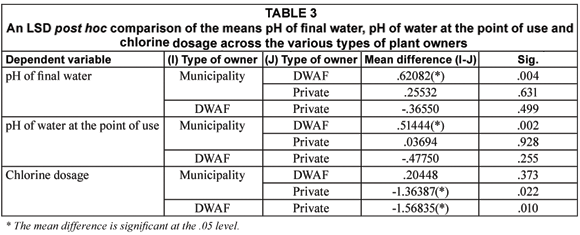
Inter-relations between physic-cochemical and microbial water qualities
Table 4 shows the Pearson's correlation coefficients of the physicochemical properties of the water produced at the various SWTPs. The figures show that the turbidity of the water samples tended to be associated with its total coliform counts. Significant relationships were also noted between the faecal coliform count of water at the point of use and heterotrophic plate count of raw water and heterotrophic plate count of final water respectively (Table 4).
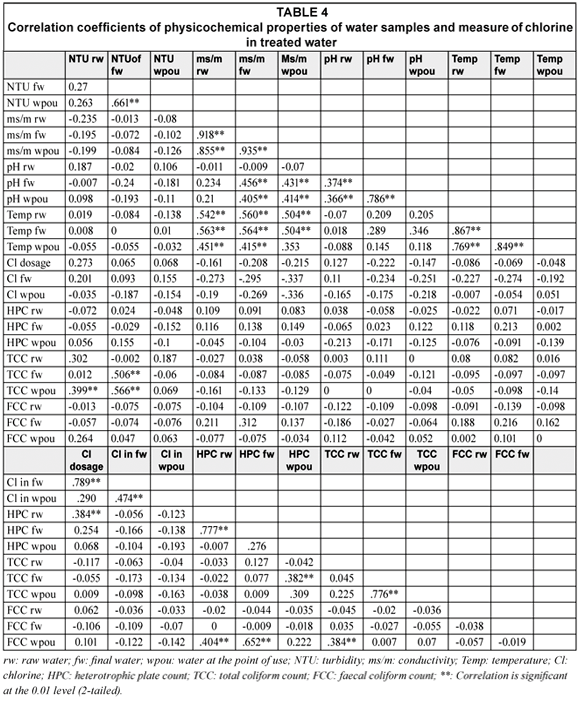
A positive correlation was also noted between chlorine dose and residual chlorine in final water (r = .684; p < .001), chlorine dose and current flow capacity (r = .371; p < .001) and chlorine dose and heterotrophic plate count of raw water (r = .384; p < .001). Similarly residual chorine in final water was more strongly associated with residual chlorine at the point of use (r = .531; p < .001).
Predictors of microbial water quality
Table 5 shows a linear regression of the various measures of the physicochemical indicators, current flow capacity and chlorine dosage on each of the measures of the microbial qualities of final water and water at the point of use. The results show that turbidity and current flow capacity were the common predictors of microbial water quality. Current flow capacity contributed about 32%, and 10% to the heterotrophic plate counts in final water and faecal coliform counts of water at the point of use respectively. The table also shows that turbidity of raw water, final water and water at the point of use contributed close to 60% of the total coliform counts of water at the point of use. Turbidity of raw water also contributed about 10% of the total coliform counts of raw water.
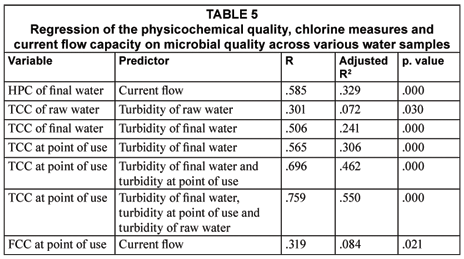
Table 6 shows that turbidity of final water was the main contributor to total coliform counts of final water and water at the point of use. Chlorine dosage did not contribute to any of the predictors in Tables 5 and 6.
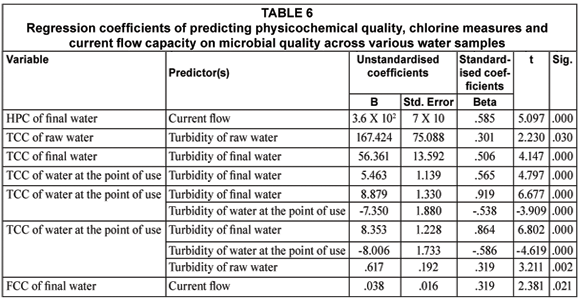
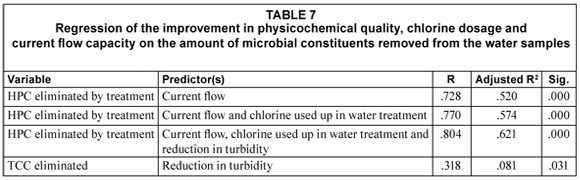
A linear regression of the quantity of chlorine used up during disinfection and improvement in physic-cochemical qualities following treatment was done for each microbial the indicators. Tables 7 and 8 illustrate that current flow capacity was a major predictor of improvement in the heterotrophic plate count of the water samples following treatment. The effect of current flow capacity on the improvement of heterotrophic plate count increased in combination with the amount of chlorine used up during the disinfection of raw water and reduction in water turbidity (Table 7). The amount of particulate matter removed (measured as the turbidity) following treatment, contributed to improvement in total coliform count of the water samples (Table 7).
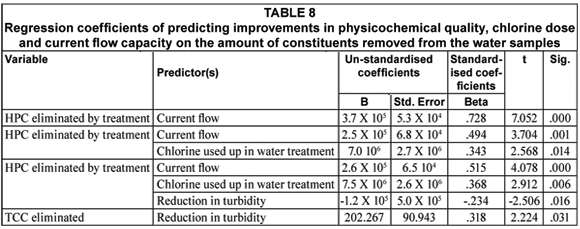
Discussion
This study was carried out to establish the effect of chlorine dosing and current flow capacity on the efficiency of SWTPs. This is a follow-up on the findings of previous studies that indicated the inability of SWTP operators to regulate and maintain both factors (Obi et al., 2007; Momba et al., 2003). This study concludes that current flow capacity of the SWTPs is a major determinant of the efficiency of SWTPs in improving water quality while chlorine dose has either limited or no effect on raw water quality improvement for human consumption.
The observed lack of effect of chlorine may be due to several factors. This pointer could also be an indication of the poor understanding around the mechanisms responsible for coliform bacterial survival in treated water as suggested by LeChevallier et al. (1988). It is, however, known that the efficiency of disinfectants such as chlorine is dependent on its concentration, contact time, turbidity, temperature and pH (LeChevallier and Au, 2004; Ridgway and Olson, 1982). Other factors that could affect bacterial sensitivity to chlorination could include attachment to surfaces, encapsulation, aggregation and low nutrient growth (Reilly and Kippin, 1983; Clark, 1984; LeChevallier et al., 1981).
As an indicator of the accuracy of the concentration of chlorine used in the SWTPs, this study found that the interrelation between chlorine dose and current flow capacity of the plants was less than 14%, but standard regulations demand that chlorine dose should be calculated in relation to the plants' current flow capacity. The study noted that majority of the SWTPs visited had low levels of residual chlorine in final water and water at point of use. In addition, these two measures were strongly related to the dosing of chlorine used in water treatment. In this way, more accurate higher dosages of chlorine would have resulted in increases in residual chlorine levels so as to comply with the stipulated safety limits. Sufficient residual chlorine is meant to maintain water quality from the SWTPs to the point of use, offer protection against contaminants in the distribution process and prevent reactivation and growth of coliforms and bacteria (LeChevallier and Au, 2004). A similar survey of water treatment plants in South Africa has revealed that the SWTPs were running at only about 5% of the actual chlorine requirement (Momba et al., 2004). The strong correlations between the levels of the respective microbial indicators in final water and water at point of use, suggest that the micro-organisms detected at point of use may have come from the treated water and have thrived due to low residual chlorine levels. In addition, studies have questioned the effectiveness of residual chlorine in treated water maintenance (Hudson et al., 1983; Reilly and Kippin, 1983; Wierenga, 1985). Other researchers such as Clark (1984) and Reilly and Kippin (1983) have isolated encapsulated bacteria from chlorinated drinking water, indicating resistance to chlorine. Stewart and Olson (1986) demonstrated that the aggregation of Acinetobacter increased its resistance to chlorine twofold.
Similar studies have reported an increased microbial resistance to disinfection as a result of their attachment to surfaces, particles or other organisms including micro-organisms (LeChevallier et al., 1988; Herson et al., 1987; Ridgway and Olson, 1982; LeChevallier et al., 1981). It is assumed the microbial agents in water are shielded from disinfection by their attachment to particles and surfaces in water. This may explain the significantly high effect of turbidity on the presence of microbial agents in final water and water at point of use. This finding is critical in the light of the fact that the majority of the SWTPs did not meet the required safety limits for turbidity of final water and water at point of use. In the same way, total coliforms and heterotrophic plate counts had significant correlations with turbidity and were also less likely to be removed from raw water following treatment, when compared to the amount of faecal coliform removed. It has been demonstrated that heterotrophic bacteria grown on metal coupons were 2 400 times more resistant to free chlorine than the unattached cells (LeChevallier et al., 1988).
Turbidity levels have also been extensively suggested to increase micro-organism resistance to disinfectants (Health Canada, 2006). This point remains crucial with the poor reduction of water turbidity across the SWTPs that participated in this study. LeChevallier (1981) demonstrated that an increase in turbidity from 1.0 to 10.0 NTU would result in an eightfold reduction in the disinfection efficiency at a fixed chlorine dose.
All the same, the marked removal of the respective microbial indicators from treated water may have resulted from a combination of the other treatment processes such as coagulation, sedimentation, flocculation and filtration. Tanner and Ongerth (1990) have for instance reported that slow sand filtration alone has the capacity to remove about 99.5% of faecal coliform and total coliform in raw water. Coagulation and sedimentation can remove up to 74% of viruses, 87% of bacterial and 94% of algae (Gimbel and Clasen, 1998). Similar to the findings of this study, other studies have reported that flow rate and raw water quality are a major determinant of the efficiency of the treatment processes to remove the microbial constituents from raw water (Tanner and Ongerth, 1990; USEPA, 1991).
In terms of flow capacity, water treatment plants with a flow capacity of over 50 Mℓ/d were more likely to supply water of improved quality. This remains a major concern as SWTPs by definition (DWAF, 1996) do not fall into this category and incontrovertibly may be less efficient.
The microbial indicators of water quality, such as faecal coliform counts, total coliform counts and heterotrophic plate counts are relied upon as predictors of water contamination by other entero-pathogens (Byamukama et al., 2000). Other researchers have assessed whether the presence of either of these indicators could respectively predict the presence of each other (Edberg and Smith, 1989). Such efforts have yielded varying results. Riley and Kippin (1983) found that low heterotrophic counts (0 to 50 CFU/mℓ) could not account for total coliform counts. Whereas Goshko et al. (1983) found that above 30 CFU/mℓ, heterotrophic plate counts had no correlation with total coliform counts. The findings of Goshko et al. (1983) corroborates the findings of this study, as only heterotrophic plate counts of water at point of use (mean = 40 CFU/mℓ) correlated with total coliform counts. While this remains beyond the scope of this study, the correlations between the primary microbial water quality indicators displayed no consistent pattern. Turbidity was, however, a major predictor of the total coliform counts and heterotrophic plate counts. This finding accords with similar studies that have demonstrated strong associations between turbidity, bacterial coliform counts and heterotrophic plate counts (Goshko et al., 1983; Reilly and Kippin, 1983). This association has been used to explain the persistence of bacterial coliforms in chlorinated water with a turbidity values ranging between 3.8 and 84 NTU. The theory behind this is that microbial growth is most extensive on particle surfaces as a result of the nutrients absorbed by these particles, thus promoting more active growth of micro-organisms, compared to their growth in free suspension (Brock, 1966; Stotzky, 1966).
In conclusion, this study marks the first attempt to document the intricate and intercalating dynamics of turbidity, current flow capacity, chlorine dose, pH and ownership of SWTPs on microbial water quality in Limpopo and Mpumalanga Provinces.
Acknowledgements
The authors are grateful to the Water Research Commission (WRC), National Research Foundation (NRF) for providing the funds for this project and the Belgian Technical Cooperation (BTC) for the study fellowship awarded to Mr Igumbor. We would also like to thank the administrators in various municipalities and offices of DWAF and Biowaters, as well as the supervisors, superintendents and operators of the various plants visited.
References
BROCK TD (1966) Principles of Microbial Ecology. Prentice-Hall: Englewood Cliffs, NJ. p. 72. [ Links ]
BYAMUKAMA D, KANSIIME F, MACH RL and FARNLEITNER AH (2000) Determination of Escherichia coli contamination with chromocult coliform agar showed a high level of discrimination efficiency for differing faecal population levels in tropical waters of Kampala, Uganda. Appl. Environ. Microbiol. 66 864-868. [ Links ]
DEPARTMENT OF WATER AFFAIRS AND FORESTRY (DWAF) (1996) South African Water Quality Guidelines (Volume 1), Domestic Uses (2nd edn.). Department of Water Affairs & Forestry, Pretoria, South Africa. [ Links ]
DEPARTMENT OF WATER AFFAIRS AND FORESTRY (DWAF) (1998) Quality of Domestic Water Supplies. Assessment Guide 1 (2nd edn.). Department of Water Affairs & Forestry, Department of Health and Water Research Commission of South Africa. [ Links ]
EDBERG SC and SMITH DB (1989) Absence of association between total heterotrophic and total coliform bacterial from public water supply. Appl. Environ. Microbiol. 55 (2) 380-384. [ Links ]
GIMBEL R and CLASEN J (1998) International report: removal of micro-organisms by clarification and filtration processes. Water Supply 16 203-208. [ Links ]
GOSHKO MA, MINNIGH HA, PIPES W0 and CHRISTIAN RR (1983) Relationship between standard plate counts and other parameters in distribution systems. J. Am. Water Works Assoc. 75 568-571. [ Links ]
HEALTH CANADA (2006) Relationship between Turbidity and other Water Quality Parameters. Available at: http://www.hc-sc.gc.ca/ewh-semt/pubs/water-eau/doc_sup-appui/turbidity/chap_5_e.html. Accessed on 22/07/07. [ Links ]
HERSON DS, McGONIGLE B, PAYER MA and BAKER KH (1987) Attachment as a factor in the protection of Enterobacter cloacae from chlorination. Appl. Environ. Microbiol. 53 1178-1180. [ Links ]
HOFF JC (1978) The Relationship of Turbidity to Disinfection of Potable Water. Evaluation of the Microbiology Standards for Drinking Water. EPA-570/9-78-OOC. US Environmental Protection Agency, Washington, DC. [ Links ]
HUDSON LD, HANKINS JW and BATTAGLIA M (1983) Coliforms in a water distribution system: a remedial approach. J. Am. Water Works Assoc. 75 564-568. [ Links ]
LECHEVALLIER MW and KWOK-KEUNG AU (2004) Water Treatment and Pathogen Control. IWA Publishers, London. [ Links ]
LECHEVALLIER MW, CAWTHON CD and LEE RG (1988) Factors promoting survival of bacteria in chlorinated water supplies. Appl. Environ. Microbiol. 54 (3) 649-654. [ Links ]
LECHEVALLIER MW, EVANS TM and SEIDLER RJ (1981) Effect of turbidity on chlorination efficiency and bacterial persistence in drinking water. Appl. Environ. Microbiol. 42 159-167. [ Links ]
MOMBA NMB, TYAFA Z and MAKALA N (2003) Rural water treatment plants fail to provide water to their consumers: Alice water treatment plant in the Eastern Cape Province of South Africa S. Afr. J. Sci. 100 307-310. [ Links ]
MOMBA NMB, TYAFA Z, MAKALA N, BROUCKAERT BM and OBI CL (2006) Safe drinking water still a dream in rural areas of South Africa. Case study: the Eastern Cape Province. Water SA 32 (5) 715-720. http://www.wrc.org.za/downloads/watersa/2006/WISA%20special%20ed/17.pdf [ Links ]
NEVONDO TS and CLOETE TE (1999) Bacterial and chemical quality of water supply in Dertig Village settlement. Water SA 25 215-220. http://www.wrc.org.za/archives/watersa%20archive/1999/April/apr99_p215.pdf [ Links ]
OBI CL, MOMBA NMB, SAMIE A, IGUMBOR JO, GREEN E and MUSIE E (2007) Microbiological, physicochemical and management parameters impinging on the efficiency of small water treatment plants in Limpopo and Mpumalanga Provinces of South Africa. Water SA 33 (2) 229-237. http://www.wrc.org.za/downloads/watersa/2007/Apr%2007/2022.pdf [ Links ]
OBI CL, PO BESSONG, MNB MOMBA, N POTGIETER, A SARNIE and IGUMBOR EO (2004) Profiles of antibiotic susceptibilities of bacterial isolates and physico-chemical quality of water supply in rural Venda communities, South Africa. Water SA 30 (4) 515-520. http://www.wrc.org.za/archives/watersa%20archive/2004/Oct-04/10.pdf [ Links ]
REILLY JK and KIPPIN JS (1983) Relationship of bacterial counts with turbidity and free chlorine in twodistribution systems. J. Am. Water Works Assoc. 75 309-312. [ Links ]
RIDGWAY HF and OLSON BH (1982) Chlorine resistance patterns of bacteria from two drinking water distribution systems. Appl. Environ. Microbiol. 44 972-987. [ Links ]
SNEAD MC, OLIVIERI VP, KRUSE CW and KAWATA K (1980) Benefits of Maintaining a Chlorine Residual in Water Supply Systems. EPA-600/2-80-010. U.S. Environmental Protection Agency, Washington, DC. [ Links ]
STATISTICS SOUTH AFRICA (StatSA) (2004) General Household Survey 2004. Statistics South Africa: Pretoria. [ Links ]
STOTZKY G (1966) Influence of clay minerals on microorganisms. III. Effect of particle size, cation exchange capacity, and surface area on bacteria. Can. J. Microbiol. 12 1235. [ Links ]
TANNER SA and ONGERTH JE (1990) Evaluating the performance of slow sand filters in Northern Idaho. J. Am. Water Works Assoc. 82 (12) 51-61. [ Links ]
UNITED STATES ENVIRONMENTAL PROTECTION AGENCY (USEPA) (1991) Optimizing Water Treatment Plant Performance Using the Composite Correction Program. Cincinnati, OH, Office of Research and Development, United States Environmental Protection Agency. [ Links ]
WIERENGA JT (1985) Recovery of coliforms in the presence of a free chlorine residual. J. Am. Water Works Assoc. 77 83-88. [ Links ]
ZAMXAKA M, PIRONCHEVA G and MUYIMA NYO (2004) Microbiological and physico-chemical assessment of the quality of domestic water sources in selected rural communities of the Eastern Cape Province, South Africa. Water SA 30 (3) 333-340. http://www.wrc.org.za/archives/watersa%20archive/2004/July-04/6.pdf [ Links ]
* To whom all correspondence should be addressed.
+2721 447 0822 Ext. 130; fax: +2721 448 6157;
e-mail: judeigumbor@yahoo.co.uk; jude.igumbor@arkonline.org












