Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SA Orthopaedic Journal
On-line version ISSN 2309-8309
Print version ISSN 1681-150X
SA orthop. j. vol.12 n.2 Centurion Jan. 2013
SPINE
Neuromuscular scoliosis - surgical management and outcomes
A PudduI; RN DunnII
IMBChB H Dip Ortho, FC(Ortho) BSc(Hons) AO Spine Fellow: Division of Orthopaedic Surgery, University of Cape Town
IIMBChB(UCT)MMed(Orth)UCT, FCS(SA)Orth Consultant Spine and Orthopaedic Surgeon, Full Professor University of Cape Town Head: Division of Orthopaedic Surgery Head: Orthopaedic Spinal Services, Groote Schuur Hospital
ABSTRACT
Neuromuscular scoliosis affects a heterogeneous group of patients with myopathic, upper and lower motor neuron diseases. Spinal surgery is often required to optimise respiratory, sitting and ambulatory function.
OBJECTIVES: Review of management and outcomes of surgically treated neuromuscular scoliosis.
STUDY DESIGN: Retrospective review of prospectively maintained data, including demographics, intra-operative variables, pre- and post-operative imaging, complications, outcomes and a telephonic follow-up questionnaire.
RESULTS: Ninety-eight patients (45 male and 53 female) were included in the study. The average operating time was 230 (100-525 ± 60.9) minutes and an average of 15.4 (8-19 ± 2.9) levels were fused. Pedicle screw only constructs corrected the primary curve by 63% initially and 56% correction at last follow-up. Hybrid constructs had an immediate correction of 69% and 47% at last follow-up. Although pedicle screw constructs lost less correction when compared to hybrid constructs, this was not a statistically significant difference. Pelvic obliquity was corrected from 14.02 (0-80 ± 15.54) to 4.06 (0-35 ± 7.69) degrees. The majority of the telephonic responses were positive.
CONCLUSION: : Corrective spinal surgery in the neuromuscular patient is demanding with a high rate of complications but outcomes are good, with radiographic correction maintained in the long term and high level of patient and parent satisfaction.
LEVEL OF EVIDENCE: III
Key words: neuromuscular scoliosis, surgery, management, outcomes
Introduction
Neuromuscular scoliosis (NMS) develops in a heterogeneous group of patients with multiple and sometimes unknown underlying causes. The clinical picture is diverse, ranging from high-functioning patients with mild curves to severely debilitated patients with gross scoliosis. Patients may present with long C-shaped curves, short-segment focal scoliosis with compensatory curves, or kyphotic deformities.-
Spinal deformity in NMS may develop early, progress rapidly with growth and often deteriorate after skeletal maturity as opposed to the more common idiopathic type.
Children undergoing surgery for NMS have longer hospital stay, higher total cost, more procedures and more complications than children with idiopathic scoliosis.1 Pelvic obliquity is a cause of great morbidity causing sitting/ambulatory difficulty, skin ulceration and hip dislocation.
NMS is classified into neuropathic and myopathic groups according to its underlying cause. The neuropathic types are subdivided into upper motor neuron lesions (UMNL) and lower motor neuron lesions (LMNL). Common UMNL causes are cerebral palsy (CP), spinal muscular atrophy and Friederich's ataxia whereas myelomeningocoele and polio are LMNL causes. Duchenne muscular dystrophy and Marfan's syndrome are examples of the myopathic type.
Management of NMS patients requires a multi-disciplinary approach with active involvement of parents, caregivers, physiotherapists, occupational therapists, paediatricians and intensive care physicians. Patients with moderate to severe curves are managed non-operatively if they are too ill to undergo major corrective spinal surgery. Thoracolumbar orthoses are poorly tolerated and restrict the already impaired respiratory function.2 Custom-moulded wheelchairs are of some benefit to these ill patients.
Surgery is indicated to correct sitting imbalance due to severe deformity as well lift the chest off the abdominal contents as the patient's trunk collapses. Surgery in this instance may salvage the deteriorating respiratory system and can be life-saving. Discomfort from impingement of the rib cage on the iliac spine causes great morbidity and is relieved by deformity-correcting surgery. Correcting these patients' coronal balance may improve upper limb function by reducing the need for arm support to prevent slouching/falling over.
Surgical fixation techniques have advanced from previous distracting techniques with Harrington rods and sub-laminar wire fixation of Luque, which had poor rotational control. Modern segmental, pedicle screw-based instrumentation techniques have allowed spinal surgeons to not only halt deformity progression, but dramatically correct it in sagittal, coronal and axial planes.
Materials and methods
Following University of Cape Town Institutional Research Council approval (no.228/2012), a retrospective review of prospectively collected data was undertaken.
The senior author's (RND) database was interrogated for neuromuscular scoliosis patients undergoing spinal deformity fusion surgery. All underwent their surgery by the senior author between October 2001 and December 2011. All the procedures were performed at Groote Schuur Hospital, Red Cross War Memorial Children's Hospital, UCT Private Academic Hospital or Constantiaberg Medi-Clinic.
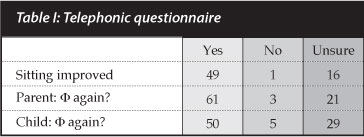
Demographics, intra-operative variables, pre- and postoperative imaging, complications and outcomes were analysed. Patients and caregivers/parents were contacted telephonically and asked three questions:
1. Would the patient opt to have the surgery again now that they know what it involved?
2. Would the parent/caregiver opt to have the surgery again if given the same opportunity?
3. Has seating been improved by surgery?
Data was compiled with the aid of a Microsoft Access database and analysed using a Microsoft Excel spread sheet. The Student t-test was employed to calculate p-val-ues with 1-tail significance levels.
Results
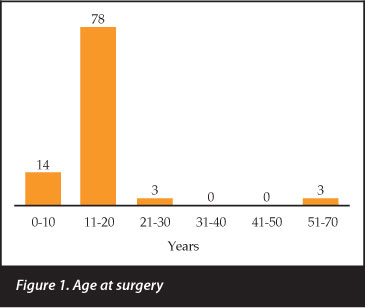
Ninety-eight patients were identified of which 45 were male and 53 female. The median age at surgery was 13.5 (6.2-70.4 ± 9.0) years and the average age at surgery was 15.0 (6.2-70.4 ± 9.0) years. Fourteen patients were younger than 10 years at surgery, 78 were between the ages of 11 and 20 years and three between 21 and 30 years of age (Figure 1). The three oldest patients had Parkinson's disease (59 and 70 years old) and poliomyelitis (56 years old). Their scoliosis was deemed to be due to the underlying disease and not degeneration.
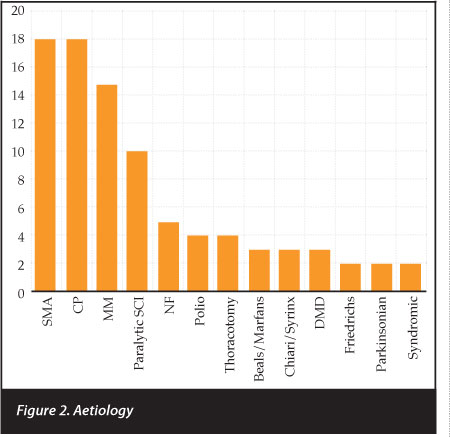
Spinomuscular atrophy and cerebral palsy were the most common underlying diseases (n=18) followed by myelomeningocoele (n=15) and paralytic spinal cord injury (n=10) (Figure 2). The average operating time was 230 (100-525 ± 60.9) minutes with an average of 15.4 (8-19 ± 2.9) levels fused per patient. Seventy-two patients had posterior only surgery. Fifteen had anterior only surgery. The anterior only surgery was all for focal thoracolumbar scolioses. They were attributed to neurofibromatosis (n=2), cerebral palsy (n=3) and myelomeningocoele (n=7). Fifteen patients had anterior and posterior surgery. This was done on the same day in two patients, and staged in 13 patients.
When an anterior operation was performed for anterior only surgery or for combined anterior/posterior surgery, an average of 6 (2-8 ± 1.2) levels were released. The blood loss for this anterior procedure was 200 (50-850 ± 229.3) ml and duration 130 (60-180 ± 37.9) minutes.
For all the procedures combined, the average total blood loss per operation was 1 812.9 (250-8500 ± 1255) ml, which is 119.5 (25-850 ± 101.0) ml per fused level. There were 18 patients with blood loss greater than 2 500 ml. Patients who were fused to the pelvis had significantly (p=0.006) greater blood loss than those not fused to the pelvis: 2170.1 (600-8500 ± 1389.1) ml vs 1361.8 (250-3600 ± 890.5) ml.
A cell saver was used in 35 cases and 1 800 (500-8021 ± 1396.7) ml blood collected per patient. Of the collected blood volume, 650 (200-3498 ± 588.7) ml or 36% was re-infused. Blood loss when using the cell saver was 2 100 (900-8500 ± 1399.5) ml and without was 1 000 (250-000 ± 984.9) ml (p=0.00003).
Posterior spinal fusion was performed on 86 patients. Of these, 38 were with pedicle screw only constructs and 48 with hybrid constructs. A hybrid construct was defined as any construct using transverse process hooks (n=24), sub-laminar wires (n=22) or both hooks and wires (n=2) in conjunction with pedicle screws.
Metal density is defined as the ratio of fixation points utilised to those available. In pedicle screw only constructs the metal density was 0.44 (0-0.75 ± 0.14) whereas in hybrid constructs (hooks and sublaminar wires) the density was 0.34 (0.06-0.75 ± 0.13).
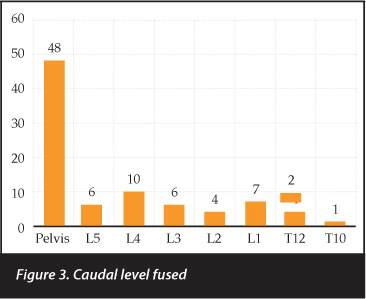
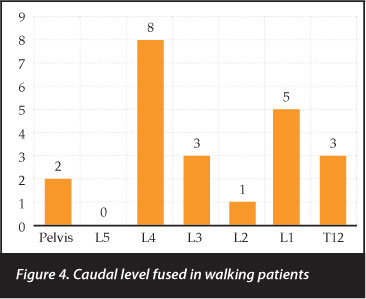
The most caudal level fused was the pelvis in 48 patients followed by L4 (n=10), L1 (n=7), L5 and L3 (n=6 each) (Figure 3). The pelvis was instrumented in only two walking patients; L4 was the most common inferior level in ambulating patients (n=8) (Figure 4).
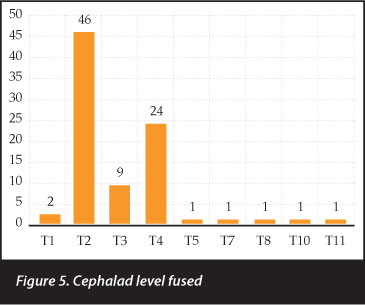
Forty-six patients were fused to T2 superiorly, followed by 24 to T4 and nine to T3 (Figure 5).
Six patients died in the post-operative period at an average of 34.2 (20-51 ± 11.92) months post-operatively. All deaths were unrelated to the surgery and due to the underlying neuromuscular condition. Fifty-two patients had a follow-up of greater than two years and 22 are still in the two-year post-operative period. Eighteen were lost to follow-up after their initial post-op visit. This was usually due to patients from distant areas finding it difficult to travel.
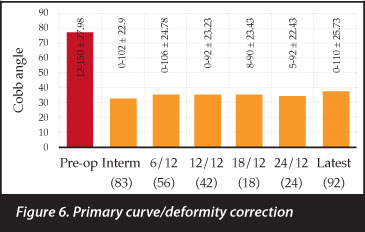
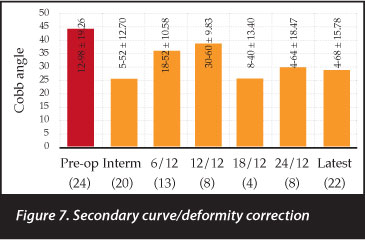
The primary curve was corrected from 77.2 to 32.5 degrees immediately post-operative - a correction of 66% or fraction of 0.66 (0.1-1 ± 0.24). This correction was maintained for the follow-up period (Figure 6) and statistically significant throughout. At last follow-up (27.97 (1-107 ± 23.83) months) the primary curve magnitude was 37.59 (0-110 ± 25.73) degrees. The secondary curves were initially corrected from 44.2 to 25.5 degrees - a correction of 0.62 (-1-1 ± 0.42). This correction was however not maintained in the follow-up period. At 25.52 (1-72 ± 17.78) months the secondary curves measured 29.05 (4-68 ± 15.78) degrees. Only the immediate post-operative secondary curve correction was statistically significant (Figure 7).
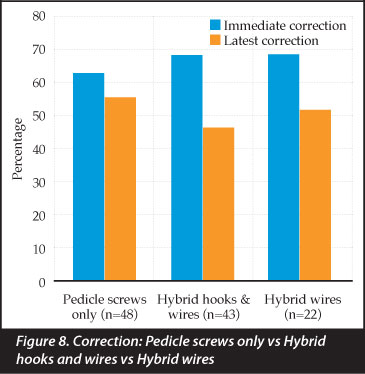
Pedicle screw only constructs corrected the primary curve by 63% which was maintained at 56% at latest follow-up. Hybrid constructs (sublaminar wires and/ or hooks) had an immediate correction of 69% and 47% at last follow-up. Hybrid constructs with only sublaminar wires corrected by 69% initially, declining to 52% at follow up (Figure 8). Although pedicle screw only constructs lost less correction (11.1%) when compared to hybrid constructs (31.1%), this was not a statistically significant difference.
Pelvic obliquity was corrected from 14.02 (0-80 ± 15.54) to 4.06 (0-35 ± 7.69) degrees. This is a correction of 71% (0-56 ± 0.51) (Figure 9). A correction of 72% was achieved when the pelvis was incorporated in the fusion mass (n=48). When the pelvis was not incorporated in the fusion mass, the pelvic obliquity correction was 67% (n=38).
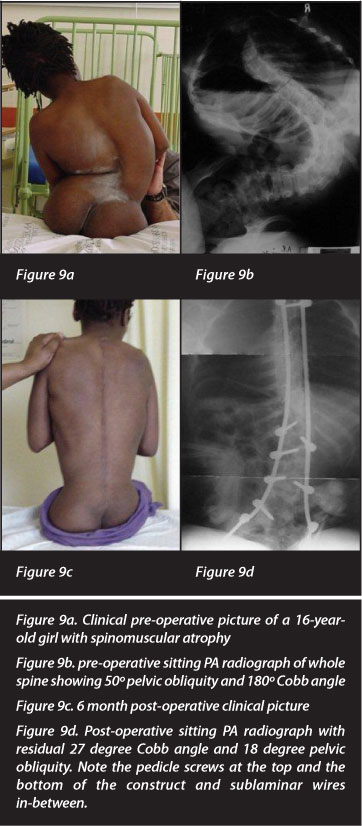
Eighty-six families were telephonically contactable. Some patients/parents simply would not commit to answering positively or negatively - they were grouped under 'unsure' (n=66). The majority of the responses were positive (160 vs 9) and both parent/caregiver and child would opt to have the surgery again if given the opportunity (Table I). Patients who were ambulating (n=20) were excluded from the sitting follow-up question. A patient with post-operative neurological deterioration was the only patient who thought sitting balance was not improved post-operatively.
Complications
Neurological
There were four patients with neurological complications. A 13-year-old boy with hydrocephalus underwent a T2-L2 posterior spinal fusion. Despite doing well initially, he developed myelopathy 6 weeks post-operatively. MRI confirmed T1-2 subluxation and cord compression. Revision surgery was performed, extending the instrumentation to T1 and reducing the subluxation. Despite subsequent wound infection requiring repeated irriga-tion/debridement, he recovered to a normal neurological status over a few weeks.
A 13-year-old boy underwent an uneventful T2-pelvis posterior spinal fusion for cerebral palsy-induced scolio-sis. Although he was wheelchair bound pre-operatively, he had some left leg control and sensation. During the immediate post-operative period he developed cardio-res-piratory instability which was confirmed to be due to ascending spinal oedema. He required inotropic support and mechanical ventilation. At day 15 post surgery he appeared as a C4 complete spinal cord injury, but slowly regained his upper limb neurological function. Unfortunately he remained a T8 complete paraplegic and never regained his lower limb motor function or sensation.
A 15-year-old girl with polio and a severe kyphoscoliosis underwent a T2-L4 posterior spinal fusion. Despite a normal intra-operative wake up test, she developed complete paraplegia a few hours after the procedure. She was taken back to theatre immediately, the instrumentation removed and the spine allowed to return to its pre-operative position. She regained motor function by the morning and returned to normal over the next few months.
A 7-year-old girl with a severe dystrophic kyphoscoliosis from neurofibromatosis with pre-operative myelopathy underwent a T2-T10 posterior spinal fusion and circumferential vertebral column resection to decompress the cord. Her myelopathy worsened post-operatively but slowly recovered in a protracted eight-month hospital stay.
Four patients had unintentional durotomies. These were typically the meningomyelocoele patients with thin skin adherent to the expanded thecal sac. There were no neurological consequences.
Massive blood loss
One patient had a massive blood loss of 8 021 ml. This was partly explained by persistent hypothermia. Via the cell saver 3 498 ml was re-infused, which prevented any further consequences.
Respiratory
Pulmonary oedema developed in a patient on post-operative day 10. This was due to hypoalbuminaemia and negative intrathoracic pressure from the expanded thoracic cage. She was ventilated via tracheostomy for three days and recovered uneventfully.
Mechanical
A 59-year-old female with Parkinson's disease developed a positive sagittal balance despite extensive anterior release (T10-L4) and L5/S1 transforaminal interbody fusion at the initial procedure. She required revision surgery with a pedicle subtraction osteotomy of L3 seven months after index surgery and is now doing very well. Five patients had asymptomatic rod breakages or pedicle screw loosening. These were left alone. One patient had early pull-out of his superior screws. He was managed with a thoracolumbar sacral orthoses.
Infection
There were two early post-op wound infections. These were managed with repeated washouts, vacuum dressings and secondary closure under the cover of appropriate antibiotics for six weeks.
A 6-year-old arthrogrypotic patient presented 10 months post-operatively with late onset sepsis and skin breakdown over the prominent screw heads. This resolved with removal of instrumentation, debridement and six weeks of antibiotics.
Discussion
Scoliosis develops in the majority of neuromuscular patients. The mainstay of management of the scoliosis is surgical arthrodesis of the spine in a functional position." Bracing causes further deterioration of the already compromised respiratory system in the established severely scoliotic patient8 and is only recommended for a select few NMS patients.9 Corrective spinal surgery in patients with a severe physical handicap results in deterioration of function in the initial six months, but is then followed by return to pre-operative function in the next six months.10 Surgery has been shown to improve quality of life in patients with scoliosis related to CP and muscular dystrophy.11
Timing of surgery is a contentious issue with two opposing factors to be taken into account: skeletal growth (thoracic height) and pulmonary development on the one hand and deformity progression with pulmonary restriction and compromise on the other. In normal children, the pulmonary function growth spurt lags behind the height growth spurt on average 0.6 to 0.9 years. This is independent of age and gender.12 Children reach 66.7% of adult T1-T12 height at age 5 and 81.5% at pre-adolescence.13 The first eight years of life have been termed the golden period for thoracic growth and pulmonary development. Considering all these factors, the exact age at surgery is often a subjective decision influenced by many factors including age, gender, underlying neuro-muscular condition and neurological function or loss thereof.
Pre-operative pulmonary function testing is a reliable indicator of post-operative pulmonary complications and prolonged (>3 days) post-operative mechanical ventilation.14-16 However, pulmonary function testing in young children, even with normal mental capabilities, is often not reliable.17 Neither is poor pre-operative pulmonary function (FVC < 30%) or nocturnal non-invasive ventilation a contra-indication to surgery in the NMS patient.18-20 Because of this, we do not routinely perform pulmonary function tests as a pre-operative assessment on our NMS patients but judge surgical suitability by subjective pre-operative respiratory symptoms, respiratory effort and exertion tolerance.
The transthoracic anterior approach allows for release of rigid, severe, short segment curves, but carries greater postoperative respiratory morbidity.21 A combined anterior and posterior approach is required when the anterior approach alone will not allow sufficient surgical access. Many NMS patients preclude chest violation due to underlying respiratory compromise, and the posterior only approach is the mainstay. It is however, not without complications. Modi et al reported on haemo/pneumothorax, pleural effusion, pulmonary oedema, complete spinal cord injury, deep wound infection and death in a series of 50 patients. Unusual complications included coccygodynia.22
Blood loss in NMS surgery is the focus of many recent articles. Most agree that NMS is an independent risk factor for increased blood loss during spinal surgery.23,24 Tranexamic acid has been shown to reduce the blood loss in posterior scoliosis surgery by up to 41%25,26 but high dosage is required to be effective (100 mg/kg before incision and 10 mg/kg/hour during surgery).25 The cell saver is a vital instrument in the armamentarium of the spinal surgeon to decrease or negate allogenic blood transfusion. Consumables are however expensive and this cost needs to be weighed up against the cost of allogenic blood and the associated risk of infection transmission and adverse reactions. In this series of NMS patients, blood loss when using the cell saver was 2 100 ml and 1 000 ml without (p = 0.00003). This difference indicates that the pre-operative blood loss risk assessment was accurate and the cell saver used appropriately. Of course absolute blood loss is not easily interpreted as many of these children have small blood volumes. This may lead to a situation where a proportionally large blood loss is still not large enough to allow cell saver collection as the minimum collection for spinning down at our institution is 800 ml.
An interesting finding of this series was that patients with multiple level sublaminar wires (and pedicle screws at the bottom and/or top of the construct) maintained their correction better than constructs supplemented with hooks. A median of 2.17 (1-6 ± 1.09) hooks were used in 24 patients. These were at the superior end of the construct (transverse process hooks) when pedicle screws could not be inserted confidently. There was, however, not a statistical difference between the loss of correction of pedicle screw only constructs and hybrid constructs using sublaminar wires (Figure 8). The cost of pedicle screws is of concern, as is the risk of neurological injury when passing sublaminar wires.
Pre- and postoperative primary curve Cobb angles in this cohort are comparable to similar series in international liter-ature,27-29 and good correction was achieved throughout the follow-up period (Figure 6). Secondary or compensatory curves were not always included in the fusion mass and subsequently tended to deteriorate (Figure 7). Pelvic obliquity is a cause of great morbidity, and correction thereof often results in improved sitting function, pressure distribution and personal hygiene. It also frees the upper limbs from supporting the trunk while sitting, to allow for more regular activities like feeding and playing. A similar pelvic obliquity correction was achieved with and without incorporation of the pelvis into the fusion mass (71% compared to 67%). The comparable correction is an indication that the inferior level of the fusion construct was well chosen with good pelvic obliquity correction in both groups.
The use of spinal cord monitoring has become the standard practice of care in spinal deformity correction. It has, however, a limited role in NMS patients. Somatosensory-evoked potential (SSEP) monitoring with multiple recording sites has been shown to be effective in early detection of intra-operative spinal cord injury in NMS surgery30,31 but it requires a trained neurophysiologist to be present during surgery. We currently use surgeon-controlled, trans-cranial, motor-evoked potential neuromonitoring for all idiopathic corrections, but have found it ineffective in many NMS cases.
Understandably, the patients themselves were less enthusiastic about the surgical procedure than the parents or caregivers. Most of the parents, who responded negatively to the questionnaire, cited the surgery simply being too much for the child and parent to warrant the outcome. Some thought the rigid spine and child made transferring and caring more difficult.
Conclusion
Although corrective spinal surgery in the neuromuscular patient is demanding and has a high complication rate, few complications have long-term sequelae. Primary curve deformity correction is obtained in the long term. Sitting and walking balance is improved by correcting the pelvic inclination and there is a high satisfaction rate from parents and patients. Corrective spinal surgery in the neu-romuscular patient can halt and correct deformity and prolong life by maintaining or even improving pulmonary function.
University of Cape Town Institutional Research Council approval no. 228/2012
No benefits of any form have been derived by the author from any commercial party related directly or indirectly to the subject of this article.
References
1. Murphy NA, Firth S, Jorgensen TBS, Young PC. Spinal surgery in children with idiopathic and neuromuscular scoliosis. What's the difference? J Ped Ortho 2006;26:216-20. [ Links ]
2. Noble-Jamieson CM, Heckmatt JZ, Dubowitz V, Silverman M. Effects of posture and spinal bracing on respiratory function in neuromuscular disease. Arch Dis Child 1986;61:178-81. [ Links ]
3. Bell D, Moseley C, Koreska J. Unit rod segmental spinal instrumentation in the management of patients with progressive neuromuscular spinal deformity. Spine 1988;14:1301-307. [ Links ]
4. Boachie-Adjei O, Lonstein JE, Winter RB, et al. Management of neuromuscular spinal deformities with Luque segmental instrumentation. J Bone Joint Surg [Am] 1989;71:548-62. [ Links ]
5. Broom MJ, Banta JV, Renshaw TS. Spinal fusion augmented by Luque-rod segmental instrumentation for neuromuscular scoliosis. J Bone Joint Surg [Am] 1989;71:32-44. [ Links ]
6. Herndon WA, Sullivan A, Yngve DA, et al. Segmental spinal instrumentation withsublaminar wires. J Bone Joint Surg [Am] 1987;69:851-59. [ Links ]
7. Whitaker C, Burton DC, Asher M. Treatment of selected neuromuscular patients with posterior instrumentation and arthrodesis ending with lumbar pedicle screw anchorage.Spine 2000;25:2312-18. [ Links ]
8. Olafsson Y, Saraste H, Al-DabbaghZ. Brace treatment in neuromuscular spine deformity. J Ped Ortho 1999;19:376-79. [ Links ]
9. Askin GN, Hallett R, Hare N, Webb J. The outcome of scoliosis surgery in the severely handicapped child: an objective and subjective assessment. Spine 1997;22:44-50. [ Links ]
10. Mercado E, Alman B, Wright J. Does spinal fusion improve quality of life in neuromuscular scoliosis? Spine 2007;19:S120-S125. [ Links ]
11. Wang X, Dockery DW, Wypiy D et al. Pulmonary function growth velocity in children 6 to 18 years of age. Am J Respir Crit Care Med 1993;6:1502-508. [ Links ]
12. Dimeglio A, Canavese F. The growing spine: how spinal deformities influence normal spine and thoracic cage growth. Eur Spine J 2012;21:64-70. [ Links ]
13. Yuan N, Fraire JA, Margetis MM, Skaggs DL, Tolo VT, Keens TG. The effect of scoliosis surgery on lung function in the immediate post-operative period. Spine 2004;19:2182-85. [ Links ]
14. Ten Cate FEA, Van Royen BJ, Van Heerde M, Roerdink D, Plotz FB. The incidence and risk factors of prolonged mechanical ventilation in neuromuscular scoliosis surgery.J Ped Orth 2008;17:203-206. [ Links ]
15. Kang G, Suh S, Lee I. Pre-operative predictors of post-operative complications in neuromuscular scoliosis surgery. J Orthop Sci 2011;16:139-47. [ Links ]
16. Crenesse D, Berlioz M, Bourrier T, Albertini M. Spirometry in children aged 3-5 years: Reliability of forced expiratory maneuvers. Ped Pul 2001;32:56-61. [ Links ]
17. Modi HN, Suh SW, Hong JY, Park YH, Yang JH. Surgical correction of paralytic neuromuscular scoliosis with poor pulmonary functions. J Sp Dis & Tec 2011;24:325-33. [ Links ]
18. Inder G, Eagle M, Metha JS, Gibson, MJ, Bushby K, Bullock R. Correction of neuromuscular scoliosis in patients with preexisting respiratory failure. Spine 2006;31:2478-83. [ Links ]
19. Chong HS, Moon ES, Park JO, et al. Value of preoperative pulmonary function test in flaccid neuromuscular scoliosis surgery. Spine 2011;36:E1392- E1394. [ Links ]
20. Zhang JG, Wang W, Qui GX, et al. The role of preoperative pulmonary function tests in the surgical treatment of scoliosis. Spine 2005;30:218-21. [ Links ]
21. Modi HN, Suh SW, Yang JH, et al. Surgical complications in neuromuscular scoliosis operated with posterior-only approach using pedicle screw fixation. Scoliosis 2009;4:11 [ Links ]
22. Meert KL, Kannan S, Mooney JF. Predictors of red cell transfusion in children and adolescents undergoing spinal fusion surgery.Spine 2002;27:2137-42. [ Links ]
23. Ender A, Murray DJ, Forbes RB. Blood loss during posterior spinal fusion in patients with neuromuscular disease: is there an increased risk? Ped Anesth 2003;13:818-22. [ Links ]
24. Sethna NF, Zurakowski D, Brustowicz RM, et al. Tranexamic acid reduces intraoperative blood loss in pediatric patients undergoing scoliosis surgery. Anesthesiology 2005;102:727-32. [ Links ]
25. Grant JA, Howard J, Luntley J, et al. Perioperative blood transfusion requirements in pediatric scoliosis surgery: the efficacy of tranexamic acid. J Ped Orth 2009;29:300-304. [ Links ]
26. Benson ER, Thomson JD, Smith BG, Banta JV. Results and morbidity in a consecutive series of patients undergoing spinal fusion for neuromuscular scoliosis.Spine1998;23:2308-17. [ Links ]
27. Thacker M, Hui JHP, Wong HK, Chatterjee A, Lee EH. Spinal fusion and instrumentation for paediatric neuromuscular scoliosis: a retrospective review. J Ortho Surg 2002;10:144-51. [ Links ]
28. Oto M, Holmes L, Rogers K, Yilmaz G, Yorgova P, Shah SA. Outcomes of posterior titanium spinal instrumentation in neuromuscular scoliosis patients. Joint Dis & Rel Surg 2012;23:30-34. [ Links ]
29. Modi HN, Hong JY, Mehta SS et al. Surgical correction and fusion using posterior only pedicle screw construct for neuropathic scoliosis in patients with cerebral palsy: a three year follow-up study. Spine 2009;4:1167-75. [ Links ]
30. Owen JH, Sponseller PD, Szymanski J, Hurdle M. Efficacy of multimodality spinal cord monitoring during surgery for neuromuscular scoliosis. Spine 1995;20(13):1480-88. [ Links ]
31. Noordeen MHH, Lee J, Gibbons CER, Taylor BA, Bentley G. Spinal cord monitoring in operations for neuromuscular scoliosis. J Bone Joint Surg (Br) 1997;79-B :53-57. [ Links ]
 Reprint requests:
Reprint requests:
Reprint requests:
Dr A Puddu
Department of Orthopaedics
Greys Hospital Private Bag
9001 Pietermaritzburg 3200
Phone: (033) 897-3000
Email: alberto.puddu@kznhealth.gov.za
CRITERIA FOR AUTHORSHIP AND CO-AUTHORSHIP OF ARTICLES
The following are internationally acknowledged criteria for authors/co-authors.
With the increase in faculty and in research projects, there is a potential for increased confusion and conflict regarding appropriate authorship credit on manuscripts and presentations. The following are some relatively standardised criteria that can be helpful. These may be overstrict when considering clinical studies in which surgeons often do the "hands on work" that create the study but may not perform major analysis and writing functions. However, all authors should read and contribute editing comments prior to submission.
Relman criteria for authorship
In particular, to qualify as an author a person should fulfil at least three of the following five requirements:
1. Conception of idea and design of experiment
2. Actual execution of experiment; hands on lab work
3. Analysis and interpretation of data
4. Actual writing of manuscript
5. Be able to present to a learned gathering a lecture on the work; interpret it, defend it and take responsibility for it.
These are just guidelines. On the other hand it is probably far worse to leave someone off the list who feels they may have contributed than to include someone who did a bit less.
We should all be as inclusive as possible, offer our interested colleagues the opportunity to provide input, analysis and editing of our works to support each other and improve our papers.














