Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
SA Orthopaedic Journal
versão On-line ISSN 2309-8309
versão impressa ISSN 1681-150X
SA orthop. j. vol.12 no.1 Centurion Jan. 2013
SPINE
Hyperextension at the cervicodorsal junction in osteogenesis imperfecta - a case report
AD BhattaI; S GovenderII
IMD, H Dip Ortho (SA). Registrar, Department of Orthopaedic Surgery, Nelson Mandela School of Medicine, University of KwaZulu-Natal, South Africa
IIMD, FRCS. Professor, Department of Orthopaedic Surgery Spine Unit, Department of Orthopaedic Surgery, King George V Hospital, Nelson Mandela School of Medicine, University of KwaZulu-Natal, South Africa
ABSTRACT
Hyperextension at the cervico-dorsal (CD) junction is rare in osteogenesis imperfecta (OI) with no cases being reported in the literature. We report a three-year-old child with OI (Sillence type III2) who presented with: hyperextension at CD junction; low bone mass; thoracolumbar kyphosis; bilateral anterolateral bowing of femora; and failure to thrive.
Key words: hyperextension, cervico-dorsal junction, osteogenesis imperfecta
Introduction
Osteogenesis imperfecta (OI) results from mutations in genes encoding for type I collagen. Collagen is the major structural protein in bone, ligaments, tendons, skin, sclera and dentin.1
Type I collagen is also an integral component of several extraskeletal tissues leading to dentinogenesis imperfecta, blue sclerae, hearing impairment and hyperlaxity of the skin and ligaments.7
Mutant expression produces non-functional collagen (severe OI) or insufficient quantities of collagen (mild OI).1
The pathogenesis of spinal deformities in OI is still unknown, but is thought to be caused primarily by a combination of vertebral micro fractures due to the fragility of the bones and injury to the vertebral growth plate. Ligamentous laxity, limb-length discrepancy, pelvic obliquity, and abnormalities of the discs are secondary factors.4-6
In severe forms of OI, progression of multiple compression fractures of the spine and vertebral height shortening may be responsible for a global sagittal trunk imbalance.3,4,9,17
Studies have indicated that the incidence increases with age. This may be due to the upright posture and the effect of axial loading on the weak spinal column.4,5,8,9 It has been found4,6,8,10 that the intervertebral discs are stronger than the bone of the vertebral bodies and that compression fractures give the vertebrae a codfish appearance.
We describe a case of hyperextension of the CD junction.
The case report
A three-year-old child was referred with OI with hyperextension at the CD junction.
Perinatal history revealed normal vaginal delivery but delayed developmental milestones. The child was not able to lift her head until the age of three months. The child had sustained multiple long bone fractures following trivial trauma- which included bilateral femora and right humerus. The child was on regular follow-up with endocrinologists- and was treated with zoledronic acid.
On general examination the child had macrocephaly- blue sclera and poor dentition as well as kyphotic deformity of the cervical and thoracic spine. She was not able to stand or crawl- but could sit with support.
The child was spastic (Ashworth 2)15 with altered sensory level from T2 and associated bowel and bladder involvement.
Further examination revealed bilateral anterolateral bowing of femora with associated limb length discrepancy on the left lower limb.
CT scan revealed CD junction hyperextension with elongation of the pedicles and attenuation of the posterior elements (Figure 1). There was no evidence of fractures.
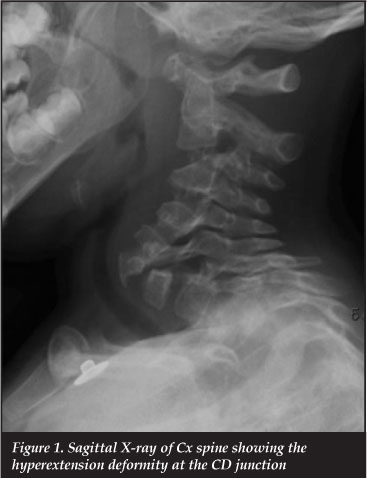
Further images showed scalloping and wedging at the posterior vertebral body at the level of C7 with associated hyperextension.
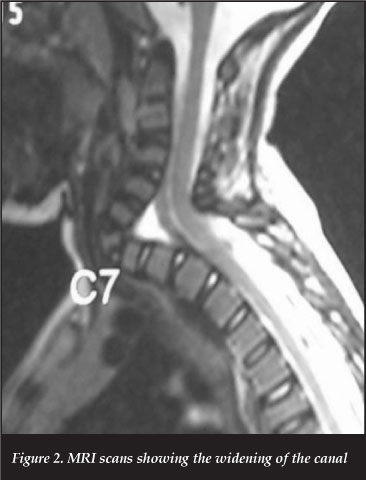
MRI scans showed widening of the canal at the same level (Figure 2).
Discussion
Although OI is a well-known skeletal disease, there have been only a few reports of spondylolisthesis in the lumbar spine.7 Familiarity with the normal developmental anatomy and radiographic features, along with knowledge of the common manifestations of hereditary and systemic diseases, are prerequisite to understanding the disorders that affect the paediatric cervical spine.13
The transition of the 'C'-shaped vertebral column to an 'S' shape occurs as a child starts to sit and stand with development of cervical and lumbar lordosis respectively. The C-spine essentially is free to rotate about the CD junction due to the relative immobility of the trunk during head movement. The C-spine thus acts as a cantilever beam with the 'fixed end' at the CD junction, the location of the highest stresses.
The spine depends upon a balance of forces during growth and development. Pathologies affecting the bony or soft tissue component around the spine could lead to deformity in different planes.
OI disrupts the musculoskeletal matrix resulting in change in the morphology of vertebral bodies which may result in a spinal deformity.
In our patient the hyperextension deformity might be associated with a C7 vertebral micro fracture with posterior wedging and compensatory forward bending moment in order to withstand the weight of the macro-cephalic skull.
Furthermore delayed milestones with the inability to lift the head might have led to persistent kyphosis of the cervical spine in order to maintain the mechanical axis with the thoracic spine.
As shown by Basu et al14 elongation of pedicles with resultant rapid progression of spondylolisthesis leads to persistent hyperlordosis of the spine above.
Pathologies around the CD junction give rise to a kyphotic deformity and are associated with neurological deficits because of the mechanical effects and smaller size of the canal and tenuous blood supply to the lower cervical cord.11
Ronald et al reported three patients with OI showing angular hyperlordosis caused by elongation of the lumbar pedicles and consecutive spondylolisthesis.7 Our patient had elongation of the pedicles and crowding of posterior elements with associated spondylolisthesis and resultant CD junction hyperextension.
During childhood with increased mechanical loads, pedicle elongation and hyperlordosis shows a rapid progression.3 This fits the hypothesis that osteopaenia causes increased micro damage in OI bones, resulting in increased bone remodelling and, with raised mechanical strains, progressive deformations.
Basu et al14 reported successful treatment of patients suffering from spondylolisthesis in OI due to lumbar pedicle elongation with interbody fusion without instrument; whereas Ronald Ivo et al7 treated three patients with spondylolisthesis due to lumbar pedicle elongation with laminectomy and posterolateral fusion.
Our patient was treated with cervical traction for a period of six weeks followed by posterior instrumentation.
Surgery was performed in two stages. Initially the child was kept on cervical traction for a period of six weeks. Pin site care was done on a regular basis and no infection was encountered during the period.
Due to the soft and fragile bony architecture which was encountered during surgery, posterior decompression and instrumentation with pedicle screws and rods was performed (Figure 3).
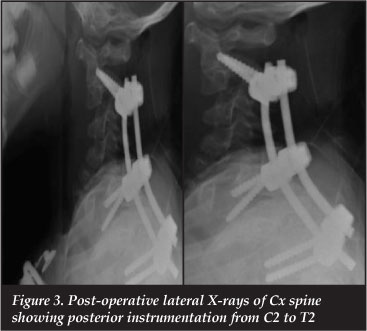
She has now been followed up for 12 months after her surgery. At the latest follow-up her neurology has improved to Frankel D grade.16
Conclusion
Although there are case reports on the management of severe kyphotic deformities of the thoracolumbar spine in OI4,6-8-12 there are no reports addressing the pathology around the CD junction with hyperextension deformity. We have presented a unique case of OI with hyperextension deformity at the CD junction.
The content of this article is the sole work of the authors. No benefits in any form have been or will be received from a commercial party related directly or indirectly to the subject of this article.
References
1. Cole WG. Advances in osteogenesis imperfecta. Clin Orthop 2002;401:6-16. [ Links ]
2. Sillence DO, Rimoin DL, Danks DM. Clinical variability in osteogenesis imperfecta-variable expressivity or genetic heterogeneity. Birth Defects 1979;15:113-29. [ Links ]
3. Abelin K, Vialle R, et al .The sagittal balance of the spine in children and adolescents with osteogenesis imperfecta. Eur Spine J 2008; 17:1697-704. [ Links ]
4. Benson DR, Donaldson DH, Millar EA. The spine in osteogenesis imperfecta. Bone and Joint Sur 1978;60-A:925-29. [ Links ]
5. Norimatsu H, Mayuzumi T,Takahashi H. The development of the spinal deformities in osteogenesis imperfecta. Clin Orthop 1982;162:20-25. [ Links ]
6. Renshaw TS, Cook RS, Albright JA. Scoliosis in osteogenesis imperfecta. Clin Orthop 1979;145:163-67. [ Links ]
7. Ivo R, Fuerderer S, Eysel P. Spondylolisthesis caused by extreme pedicle elongation in osteogenesis imperfecta. Eur Spine J 2007;16:1636-40. [ Links ]
8. Bathgate B, Moseley CF. Scoliosis in osteogenesis imperfecta. Spine State Art Rev 1990;14:121-30. [ Links ]
9. Hanscom DA, Bloom BA. The spine in osteogenesis imperfecta. Orthop Clin North Am 1988;19:449-58. [ Links ]
10. Benson DR, Newman DC. The spine and surgical treatment in osteo-genesis imperfecta. Clin Orthop 1981;159:147-53. [ Links ]
11. Govender S, Parbhoo AH, Kumar KPS. Tuberculosis of cervicodorsal junction. Journal of Pediatric Orthopaedics 2001;21:285-87. [ Links ]
12. Bradford DS. Osteogenesis imperfecta. In: Moe's textbook of scoliosis and other spinal deformities, 3rd ed.1995;555-59. Philadelphia: WB Saunders. [ Links ]
13. Ghanem I, Hage S, Rachkidi R, et al. Pediatric cervical spine instability. J Child Orthop 2008;2:71-84. [ Links ]
14. Basu PS, Noordeen MHH, Elsebaie H. Spondylolisthesis in osteogen-esis imperfecta due to pedicle elongation. Spine 2001;26:506-509. [ Links ]
15. Bohannon RW, Smith MB. Interrater reliability of a modified Ashworth Scale of Muscle Spasticity. Phys Ther 1987;67:206-207. [ Links ]
16. Frankel HL, Hancock DO, Hyslop G, et al. The value of postural reduction in the initial management of closed injuries of the spine with paraplegia and tetraplegia. Paraplegia 1969;7:179-92. [ Links ]
17. Oppenheim WL. The spine in osteogenesis imperfecta: a review of treatment. Connect Tissue Res 1995;31:S59-S63. [ Links ]
 Reprint requests:
Reprint requests:
Dr AD Bhatta
Department of Orthopaedics
Nelson Mandela School of Medicine
Private Bag 7 Congella 4013
Email: aavas_bhatta@hotmail.com
Tel: +27 31 260 4393 Fax: +27 31 260 4518














