Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SA Orthopaedic Journal
On-line version ISSN 2309-8309
Print version ISSN 1681-150X
SA orthop. j. vol.11 n.4 Centurion Jan. 2012
CLINICAL ARTICLE
A randomised controlled trial of steroid injection in the management of plantar fasciitis
M AbdihakinI; K WafulaII; S HasanIII; J MacLeodIV
IMBChB, MMed. Department of Surgery and Orthopaedics, Aga Khan University Hospital, Nairobi, Kenya
IIMBChB, MMed. Department of Surgery and Orthopaedics, Aga Khan University Hospital, Nairobi, Kenya
IIIBSc, MBChB, MMed, FACS. Department of Surgery and Orthopaedics, Aga Khan University Hospital, Nairobi, Kenya
IVMD, MSc, FRCS(C), FCS(ECSA), FACS. Department of Surgery and Orthopaedics, Aga Khan University Hospital, Nairobi, Kenya
ABSTRACT BACKGROUND: The management of plantar fasciitis varies among clinicians because no single treatment has strong scientific evidence to support its use to improve outcome. Steroid injections are more commonly used in our practice with varied and unpredictable response. This study set out to compare the effects of steroid injections with conservative regimen (analgesics, stretch exercises and insoles) versus conservative management alone for the treatment of plantar fasciitis.
METHODS: A double blind randomised controlled trial. Eighty-eight patients with plantar fasciitis were randomised to two treatment arms, 47 to the steroid injection arm and 41 to the control arm. Both arms received standard conservative management. Visual analogue scale (VAS) and Foot Function Index (FFI) scores of the patients were recorded at study entry, at one month and at two months.
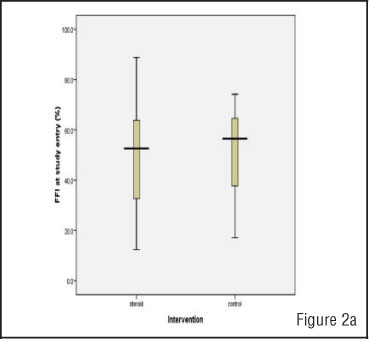
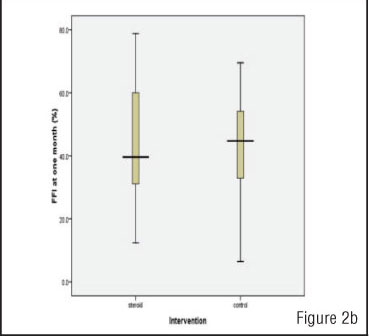
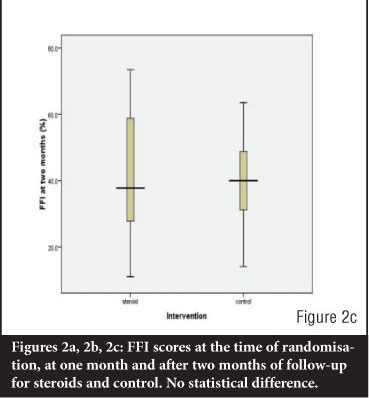
RESULTS: The mean age of the overall study cohort was 42.9 years (SD 9.1). Forty-eight per cent were male. There was a significant reduction in mean pain scores, after one month and after two months in both the steroid and the control arms (p<0.005). The mean FFI at study entry was 50.5 (SD 18.2). At one month and two months follow-up, the mean FFI scores were 43.4 (SD 16.2) and 41.4 (SD 15.3), respectively (P<0.001). There was no statistically significant difference in pain scores between patients who received steroid injections 7.36 (SD 1.6) and those in the control arm 7.22 (SD 1.7) at one month (P = 0.73) (CI-0.64 to 0.9). There was also no statistically significant difference in pain scores between the two groups of patients: steroid injections 6.77 (SD 1.8) and the controls 6.54 (SD 1.7) at two months (P = 0.59) (CI-0.61 to 1.06). FFI scores at one month were 43.25 (SD 17.63) for the steroid group and 43.79 (SD 14.73) for the control group (P = 0.88) (CI-7.78 to 6.6). At two months, the scores were 42.26 (SD 17.19) for the steroid arm and 40.48 (SD 13.30) for the control arm (P = 0.615) (CI-5.21 to 8.75).
CONCLUSIONS: Patients with plantar fasciitis improve significantly with conservative management. At one and two months, compared to a control group of conservative management alone, injection of steroids does not make a difference in improving pain and functionality.
Key words: heel spur, plantar fasciitis, steroids, injection, conservative management
Introduction
Plantar fasciitis is the most common cause of foot pain and accounts for up to 15% of all foot symptoms requiring professional care among adults.1 The incidence peaks between the ages of 40 and 60 years but younger in people who are avid runners.1 However, the condition can occur in both active and sedentary adults of all ages.2 Despite the ease of clinical diagnosis, both the aetiology and treatment of plantar fasciitis remain controversial and poorly understood.3
Histological findings of plantar fasciitis include myxoid degeneration, micro-tears in the fascia, collagen necrosis, and angiofibroblastic hyperplasia4 consistent with a degenerative process without inflammation. Therefore, steroid injections which are predominantly anti-inflammatory in action are questioned in their usefulness as a treatment option for plantar fasciitis. Further, most patients with plantar fasciitis eventually improve over time irrespective of the modality of treatment provided.5
There is no current standard of care and the existing management protocols are mainly from expert opinion-derived practice guidelines. The step-ladder approach is the most widely accepted approach. The use of steroid injections remains controversial as it has no clear improved outcome over other conservative treatments and importantly, significant possible underappreciated side effects.
Unfortunately no studies have been done in East Africa to determine the response to steroid injections despite the widespread use of steroid injections in the region to treat the condition. There is no published data on outcomes in the treatment of plantar fasciitis in Kenya. Eight out of nine orthopaedic surgeons surveyed at a single institution routinely used steroid injections in the management (unpublished data). Therefore, this study aims to study whether steroid injections should be used in the treatment of plantar fasciitis and whether they improve outcome over conservative management without steroid injections.
Methodology
Study design
This study was a double blind randomised controlled study with an intervention arm: methylprednisolone acetate 40 mg plus conservative treatment and a control arm without steroid injection and conservative treatment. The patients in the control arm received saline and lidocaine instead of the steroid. Conservative management included: Diclofenac 50 mg three times daily or 75 mg twice daily; stretching exercises with patients being taught stretch exercises; orthotics - prefabricated insoles and heel splints obtained from the Physiotherapy Department; shoe recommendations - avoiding flat shoes and walking barefoot. All patients above the age of 18 years with a clinical diagnosis of plantar fasciitis were included in the study cohort. Exclusion criteria for the study included: patients with rheumatologic disease; pregnant patients; patients who had received steroid injections within the last three months for any reason; patients on steroids for any reason, patients with other conditions causing foot pain that was either due to trauma, arthritis or neurologic conditions; and patients unable to give consent.
Demographic patient data were also collected including the body mass index and occupation which was then classified as active or sedentary.
Ethical approval for the study was granted by the institutional review board.
Patients were recruited at the Accident and Emergency unit, affiliated outreach clinics or at the orthopaedic clinics, and started on conservative management. Questionnaires were supplied to doctors in these units who assessed the visual analogue scale and notified the principal investigator for possible accrual of patients into the study.
Upon first contact, the baseline level of pain was recorded. Visual analogue scores were taken on a 10 cm VAS in which 0 represents no pain and 10 represents the worst pain imaginable in each subject's experience. The FFI Questionnaire was used to determine the disability caused by the patient's plantar fasciitis and to determine the degree of improvement with treatment. The FFI was developed to measure the impact of foot pathology on function in terms of pain, disability and activity restriction. The FFI is a self-administered index consisting of 23 items divided into three sub-scales. Both total and sub-scale scores are produced. It is a time efficient self-reported measure that patients can complete in 3 minutes or less.
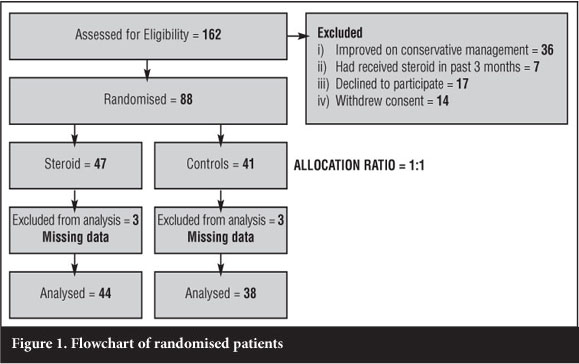
Patients were reviewed within three weeks of contact at the Orthopaedic outpatient clinics by experienced orthopaedic surgeons, and if no improvement was noted as defined by a failure to demonstrate a drop of more than 50% in their VAS score, patients were then enrolled into the study and randomised to one of two arms (Figure 1):
- One arm with steroid injections (1 ml methylprednisolone acetate 40 mg/ml, plus lidocaine 1 ml of 1%, injected at medial aspect of heel-pad at point of maximal tenderness) and conservative management.
- The second arm (control arm) with 1 ml saline plus lidocaine injection (1 ml of 1%) and conservative management (analgesics, stretch exercises, orthotics, shoe recommendations).
Consent was obtained at the stage of randomisation. All injections were given by the consultant orthopaedic surgeons or rotating senior residents. The steroids and saline/lidocaine were packaged in the same manner by the Pharmacy Department. The patient was given a prescription without the exact intervention indicated which could only be understood by the pharmacist. Patients were assigned the allocated intervention as per a randomly computer-generated randomisation table which was kept in the pharmacy and not accessed by the physician. Thus, the patient and the physician were blinded to the intervention.
Both groups of patients continued conservative management throughout the study period. Patients were asked to score their heel pain on a VAS at randomisation and later, at one month and at two months. Patients also filled the FFI Questionnaire at the time of recruitment to the study, at one month and at two months.
Sample size determination
The sample size was calculated based on previous studies with an expected 30% difference in improvement in pain scores between the intervention arm (steroid injection) and control arm (saline injection). A p-value of less than 0.05 was considered significant with a power of 80%. The calculated sample size was 41.9 in each treatment arm. Therefore, a sum total of 84 patients was required to detect this difference.
Data analysis
Data was entered into SPSS version 16.0 for Windows on completion of the study. All analyses were done using all randomised patients with complete data. Demographic characteristics of the two arms were summarised using descriptive statistics. Changes from baseline to one and two months for outcomes were measured using continuous and categorical scales (VAS, FFI) and compared between the two study arms. The tests for statistical significance between groups were performed using the Student's t test for quantitative data (the difference between means) and Chi-square test for categorical data (the difference between proportions) with a p<0.05 and confidence intervals of 95% being considered statistically significant.
Results
Patient demographics
There were 119 eligible patients of whom 88 consented to be included in the study. Seventeen declined participation at the outset and 14 opted for conservative management alone. A total of 82 patients (six had missing values) were analysed for the primary outcomes: 44 steroid arm, 38 control arm.
The mean age of the patients of the overall study cohort was 42.9 years (SD 9.1) with 42 males and 46 females. Eighty-seven per cent of the study patients were active individuals in occupations that required prolonged walking. The mean Body Mass Index (BMI) was 31.7 with 54% of the patients having a BMI above 30. Eleven study participants had flatfoot (12.5%). The characteristics of both groups were similar at study entry (Table I).
Pain score results over time
At study entry and randomisation, 40% of patients recorded their pain as being 10 out of 10 while 80% of patients recorded their pain as being 8 and above. There was a significant drop in mean pain scores at enrolment, after one month and after two months for both the steroid and control arms. The mean VAS score was 8.6 (SD 1.4) at study entry; 7.2 (SD 1.7) at one month; and 6.6 (1.7) at two months (P<0.001). The mean morning pain was 7.5 (SD 2.2) at enrolment; 6.2 (SD 1.9) at one month; and 5.6 (SD 1.8) at two months (P<0.001). Twelve per cent of patients had VAS scores of 10 out 10 at one month post-treatment and 53% of the patients had VAS scores less than 8 at one month.
At study entry, 60.4% of patients had VAS scores of more than eight regarding their morning pain. There was a significant improvement in morning pain among the participants after one month and two months with 25.0% and 15.5% of the patients, respectively, recording their morning pain at VAS scores more than 8. Up to 34% of patients had VAS scores less than 5 at one month and 45% had VAS scores less than 5 at two months of treatment.
Pain score comparison between study groups
At one month and two months post-treatment, there was no significant difference in the degree of worst pain and morning pain experienced between the two groups, as shown in Table II.
Foot function index scores
The mean FFI score at the time of randomisation was 50.5 (SD 18.2). At one month and two months follow-up, the mean FFI scores were 43.4 (SD 16.2), and 41.4 (15.3) respectively. There was a significant reduction in mean FFI scores in both groups (P<0.001) at one month and at two months. There was no significant difference in worst pain experienced, morning pain and FFI Scores between the two groups (Steroid and Control) at one month and two months as shown in Table II.
There was no significant difference in FFI scores between the two groups at study entry at one month and at two months (Figures 2a, 2b, 2c).
Discussion
Our trial demonstrated that in patients with plantar fasciitis who were managed with a conservative treatment regimen, the addition of steroid injections to this treatment regimen did not improve patient outcome when compared with controls. There was no clinical or statistically significant change in visual analogue scores recorded by patients in terms of worst pain experienced and morning pain at one month and at two months of follow up between the group that received a steroid injection and the control group. However, both study arms did show a significant improvement in symptoms from baseline to one and two months post-treatment. Furthermore, we demonstrated that the FFI scores improved in both groups but again there was no significant difference between the two groups. These findings suggest that the use of steroid injections in the management of this condition did not add any benefit.
The demographics of the patients in our study are consistent with the findings in other studies on plantar fasciitis with the mean age of the patients being 42.9 years.4-7 Most patients reported an active lifestyle with occupations involving prolonged walking or standing. This corresponds with findings in other studies.8-10 The mean BMI of our study population was 31.7 consistent with the predominance of the disease among the overweight and obese populations. BMI is not only an important risk factor but also an important predictor of the extent of functional loss reported by patients and it appears from other studies that interventions designed for obese patients with plantar fasciitis should address obesity.1
In comparison to the majority of the other published trials, the pain scores at entry into our study were much higher with a mean of 8.6.11-12 Our study enrolled patients who failed initial conservative management while other studies enrolled all plantar fasciitis patients, and therefore had an overall lower mean score.
In the study by Crawford and colleagues, it was shown that injection of 1 ml of prednisolone acetate (25 mg) with 1 ml of local anaesthetic resulted in significantly greater improvement in pain at one month than did injection of local anaesthetic alone; the mean (± SD) changes in the pain score, measured on a 10 cm VAS, were 2.0 ± 2.9 and 0.06 ± 3.0, respectively.11 At three and six months, there were no differences between the groups in pain measures, but a high rate of loss to follow-up precluded the drawing of conclusions. Moreover, it was not clear what other treatments the patients in the two different arms received. Therefore there could be a non-random distribution of other conservative treatment options that confounded their results.
The mean reduction in pain scores in all the patients in the study regardless of the treatment may be related to the effect of conservative therapy alone. This has been shown in multiple randomised controlled trials.13-15 Others have reported results similar to this study. In the Cochrane review on interventions for treating plantar heel pain, steroid injections were recognised as a popular method of treating the condition but only seemed to be useful in the short term (one month) and only to a small degree.16
Baseline FFIs were equal for both groups in our study. There was clearly a significant difference in both groups at one and two months but the difference between the steroid and control groups did not achieve statistical significance. The difference between the groups was also not relevant clinically.
This study had several limitations. A total of six patients (7%) who were randomised were not analysed due to the missing values noted during analysis. This group of patients however, had the same demographic characteristics as the analysed group and any effect on the primary outcome was minimal. Nevertheless, they were considered as protocol violations. The follow-up period of only two months may be considered a short follow-up period. However, previous studies have not shown a significant change in pain scores beyond the first month, thus we also were unlikely to find a different result if we had a longer follow-up.
The FFI and the VAS may have several limitations in our set up. These include varied foot shape subjects and difficulty in assessing the differences in the sex, age, education and cultural backgrounds of the studied patients. Lastly, it is also difficult to assess the compliance of patients with several conservative treatment modalities prescribed other than the interventions given. However, there was a general improvement in both groups thus suggesting that the compliance across the two arms was relatively consistent.
Conclusion
One and two months after steroid injection for the treatment of plantar fasciitis, we detected no difference with respect to pain or function when compared with a control arm of conservative treatment alone. Therefore, in conclusion, steroid injection as applied in our randomised double-blind controlled trial showed no improvement in patient outcome over the use of a conservative treatment regimen alone in treating plantar fasciitis in the first two months after initiation of treatment. The wide-spread use of steroid injections to treat the symptoms of plantar fasciitis is not supported by our study. A smaller difference in improvement may be found in future studies that are powered to detect less than 30% improvement between groups.
Conflict of interest
The authors did not receive grants or outside funding in support of their research or preparation of this manuscript. They did not receive payments or other benefits or a commitment or agreement to provide such benefits from a commercial entity. No commercial entity paid or directed, or agreed to pay or direct, any benefits to any research fund, foundation, educational institution, or other charitable or non-profit organisation with which the authors are affiliated or associated.
References
1. Riddle DL, Pulisic M, Sparrow K. Impact of demographic and impairmentrelated variables on disability associated with plantar fasciitis. Foot Ankle Int. 2004 May;25(5):311-17. [ Links ]
2. Cole C, Seto C, Gazewood J. Plantar fasciitis: evidence-based review of diagnosis and therapy. Am Fam Physician. 2005 Dec 1;72(11):2237-42. [ Links ]
3. Neufeld SK, Cerrato R. Plantar fasciitis: evaluation and treatment. J Am Acad Orthop Surg. 2008 Jun;16(6):338-46. [ Links ]
4. Lemont H, Ammirati KM, Usen N. Plantar fasciitis: a degenerative process (fasciosis) without inflammation. J Am Podiatr Med Assoc. 2003 May-Jun; 93(3):234-37. [ Links ]
5. Buchbinder R. Clinical practice. Plantar fasciitis. N Engl J Med. 2004 May 20;350(21):2159-66. [ Links ]
6. Wolgin M, Cook C, Graham C, Mauldin D. Conservative treatment of plantar heel pain: long-term follow-up. Foot Ankle Int. 1994 Mar;15(3):97-102. [ Links ]
7. Rompe JD, Furia J, Weil L, Maffulli N. Shock wave therapy for chronic plantar fasciopathy. Br Med Bull. 2007;81-82:183-208. [ Links ]
8. Riddle DL, Schappert SM. Volume of ambulatory care visits and patterns of care for patients diagnosed with plantar fasciitis: a national study of medical doctors. Foot Ankle Int. 2004 May;25(5):303-10. [ Links ]
9. Riddle DL, Pulisic M, Pidcoe P, Johnson RE. Risk factors for plantar fasciitis: a matched case-control study. J Bone Joint Surg Am. 2003 May;85-A(5):872- 77. [ Links ]
10. Sadat-Ali M. Plantar fasciitis/calcaneal spur among security forces personnel. Mil Med. 1998 Jan;163(1):56-57. [ Links ]
11. Crawford F, Atkins D, Young P, Edwards J. Steroid injection for heel pain: evidence of short-term effectiveness. A randomized controlled trial. Rheumatology (Oxford). 1999 Oct;38(10):974-77. [ Links ]
12. Porter MD, Shadbolt B. Intralesional corticosteroid injection versus extracorporeal shock wave therapy for plantar fasciopathy. Clin J Sport Med. 2005 May; 15(3):119-24. [ Links ]
13. Martin RL, Irrgang JJ, Conti SF. Outcome study of subjects with insertional plantar fasciitis. Foot Ankle Int. 1998 Dec;19(12):803-11. [ Links ]
14. Pribut SM. Current approaches to the management of plantar heel pain syndrome, including the role of injectable corticosteroids. J Am Podiatr Med Assoc. 2007 Jan-Feb;97(1):68-74. [ Links ]
15. Lynch DM, Goforth WP, Martin JE, Odom RD, Preece CK, Kotter MW Conservative treatment of plantar fasciitis. A prospective study. J Am Podiatr Med Assoc. 1998 Aug;88(8):375-80. [ Links ]
16. Crawford F, Thomson C. Interventions for treating plantar heel pain. Cochrane Database Syst Rev. 2003(3):CD000416. [ Links ]
 Reprint requests:
Reprint requests:
Dr Mohamed Abdihakin
Nairobi, Kenya
PO Box 30270-00100
Tel: +254 203662125
Email: abdi.mohammed@aku.edu