Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
SA Orthopaedic Journal
versión On-line ISSN 2309-8309
versión impresa ISSN 1681-150X
SA orthop. j. vol.11 no.3 Centurion ene. 2012
CLINICAL ARTICLE
Fibromatosis: Where are we now?
Leonore du PlessisI; Theo LB le RouxII; Pauline GreylingIII
IMBChB(UP) Orthopaedics Registrar. Department of Orthopaedics, 1 Military Hospital. University of Pretoria, Faculty of Health Sciences, School of Medicine, South Africa
IIMBChB(Pret), MMed(Orth)(Pret), FCS(Orth)SA Head. Department of Orthopaedics, 1 Military Hospital; and Consultant in the Dept. of Orthopaedic Surgery, Steve Biko Academic Hospital. University of Pretoria, Faculty of Health Sciences, School of Medicine, South Africa
IIIMBChB(UP) Orthopaedics Registrar. Department of Orthopaedics, Kalafong Hospital. University of Pretoria, Faculty of Health Sciences, School of Medicine, South Africa
ABSTRACT
PURPOSE OF THE STUDY: Fibromatosis is a benign but locally aggressive tumour. A high rate of recurrence was noted in a number of patients treated by the senior author at a tumour and sepsis unit despite the fact that a wide surgical excision had been performed. The question was raised whether there are any alternate treatment modalities with a higher success rate available currently. A retrospective study and review of the literature was performed in order to ascertain whether new treatment modalities which can prevent recurrence more successfully have been developed recently.
MATERIALS AND METHODS: A retrospective study was performed. The files of all patients who presented at an orthopaedic practice with confirmed fibromatosis on histological examination in the past 19 years were reviewed. The following was looked at: age of the patient at first presentation; gender; tumour site; surgery performed; histological results; first line of treatment and recurrence rate. Patients were also contacted telephonically in order to ascertain whether any recurrence managed by another orthopaedic surgeon had been attended to.
RESULTS: We evaluated 17 patients of which eight were males and nine females. The mean age was 25.87 years (range 2-52years). All of the primary sites were extra-abdominal. Median follow up was 3.9 years (0-9) with a mean recurrence rate of 2.3 times. All the patients were treated with a wide marginal surgical excision without adjuvant therapy.
CONCLUSION: Fibromatosis has a high recurrence rate using current surgical treatment modalities. Complete surgical excision does not lead to a good outcome. The literature review revealed that a wide variety of treatment modalities, both surgical and non-surgical, are available. Non-surgical treatment modalities include: hormones; non-steroidal anti-inflammatory drugs; chemotherapy; radiotherapy. Wide surgical excision remains the mainstay of treatment but a multidisciplinary approach is necessary in order to optimise the efficacy of this treatment.
LEVEL OF EVIDENCE: Level III
Key words: Fibromatosis, retrospective study, histology, treatment modality, desmoid tumour
Introduction
Fibromatosis is a benign but locally aggressive tumour with a very high rate of recurrence. It has an infiltrative growth pattern which makes complete excision difficult. The National Comprehensive Cancer Network subdivides soft tissue sarcomas into four subdivisions: soft tissue sarcoma of extremity/trunk; retroperitoneal or intraabdominal soft tissue sarcoma; gastrointestinal stromal tumours and desmoid tumours.1-3 We will be focusing on the extra-abdominal division of desmoid tumours.
A high rate of recurrence was noted in a number of patients treated at the Pretoria tumour and sepsis unit despite the fact that a wide surgical excision had been performed. The question was raised whether there are more current alternate treatment modalities with a higher success rate. We looked at previously published articles regarding treatment modalities over the past few years and specifically did a review on the new treatment modalities published during the past 5 years.
Materials and methods
A retrospective study was performed. We evaluated all the patients who presented at the Pretoria tumour and sepsis unit with confirmed fibromatosis on histological examination during the past 19 years. The following was studied: age of the patient at first presentation; gender; tumour site; surgery performed; histological results; first line of treatment and recurrence rate. Patients were furthermore contacted telephonically in order to ascertain whether any recurrence managed by another orthopaedic surgeon had occurred.
Results (Table I)
Seventeen patients were evaluated. Desmoid tumour had to be confirmed histologically in order to be included in this study. One patient had Dupuytren's disease on histology and was excluded from this study. Seven patients were males and nine females (ratio: 0:0.8). The mean age was 25.87 years (range 2- 52years). All of the primary sites were extra abdominal, the highest percentage being on the extremities 62.5% (10/16). Median follow-up was 3.9 years (0-9) with a mean recurrence rate of 2.3 times. All the patients were treated with a wide surgical excision margin. No adjuvant therapy was given. Seven of the patients had complete surgical removal which was confirmed histologically; in all multiple recurrences occurred. The histopathologist was unable to determine the surgical margin in six of the specimens. Only three patients had no recurrence. No genetic screening was done on any of these patients.
Aetiology and histopathology
Fibromatosis might be associated with genetic predisposing conditions (familial adenomatous polyposis; Gardner syndrome); hormonal influences (increased oestrogen levels, oral contraceptives, peripartum) and surgical trauma (especially at abdominal incision sites). Though it does not have the same histopathological features as a soft tissue sarcoma, clinical features do correlate with soft tissue sarcomas. Fibromatosis has a severe infiltrative pattern, with a high rate of recurrence and functional morbidity due to aggressive surgery.4
It is important to be familiar with the pathological features of this disease in order to treat it appropriately. We know that it is a benign proliferation of fibroblastic cells associated with the WNT (group of proteins)/APC (adenomatous polyposis coli)/p-catenin pathway which drives the process of formation of these tumours.5-8 The WNT/p-catenin pathway plays a role in transcription in the nucleus and cell adhesions. The APC (which is a tumour suppressor gene) controls the level of the β-catenin (which is an oncogene) by means of phosphorylation and this is indirectly controlled by the WNT pathway.5,6 β-catenin accumulation was originally demonstrated in desmoid tumours in FAP (familial adenomatous polyposis) patients who had a deficient APC complex. CTNNB1 (gene encoding β-catenin) was seen in a large population of sporadic desmoid tumours and leads to activation of the WNT pathway, thus increasing the β-catenin.5 The increased β-catenin level activates the T-cell factor. This causes transcription of the COX-2 (cyclooxyge-nase-2) leading to activation of the PDGFRA (platelet-derived growth factor receptor α) and PDGFRB (platelet-derived growth factor receptor β).9
Wu et al10 did a study on mice and found a correlation between fibromatosis and mesenchymal progenitor cells supporting tumour genesis via the β-catenin pathway. Matono et al11 evaluated 74 samples and came to the conclusion that there is overproduction of the VEGF (vascular endothelial growth factor) in desmoid tumours and especially in the recurrent group. They could not however find any correlation between the 'β-catenin mutation and VEGF mRNA' expression.
The exact reason for resolution of the tumour at the 'wait-and-see' principle is unknown and needs further investigation which could help with treatment and even prevention of this disease.
The histologist determined the positive or negative surgical margin which we are referring to in this article. It describes the margin of the tumour in the specimen which was resected and evaluated. 'Positive' means tumour at the border of the specimen excised (possible incomplete excision) and 'negative' means a clear, tumour-free surgical margin.
Treatment modalities
Conservative treatment
Barbier et al12 evaluated 26 cases of extra-abdominal fibromatosis of which 11 had no previous surgery and 15 recurred after surgery. They found that 24 of the cases stabilised at a mean of 14 months; only two regressed and showed evolution at 23 months. They came to the conclusion that a 'wait-and-see' principle can be optimal especially in those with a high risk of severe functional or cosmetic deformities after surgery. A few other smaller studies were also discussed in this article and showed similar results. They suggested that surgery makes the tumour more aggressive. The patient populations in these studies were however small.
Stoeckle et al13 evaluated 106 patients over a period of 123 months and came to the conclusion that the tumours stabilised after 3 years and some even regressed thereafter. They suggested a 'wait-and-see' approach and medical management in all tumours except those located primarily in the lower trunk wall/girdle. In the case of these tumours response was more favourable when surgery was performed.
Pignatti et al4 evaluated 83 cases and suggested that conservative management can be considered for non-progressive recurrent lesions. A retrospective study done in France by Bonvalot et al14 on 112 patients found a similar event-free survival: 65% versus 68%. The gender, age, tumour size and treatment period were not statistically significant but there was a correlation between the tumour site and quality of surgery with extremity tumours and positive surgical margins having a poorer outcome. A study done by Salas et al15 also found that age, tumour size, tumour site and surgical margins had a significant impact on progression-free survival, and suggested dividing desmoid tumours into prognostic subgroups to decide on management.
The above studies suggest that the least invasive method be used as first-line treatment; this however excludes patients with severe pain; a massive tumour or severe loss of function. Each patient still needs to be evaluated individually.
Radiotherapy options and the influence of the surgical margin
Gluck et al16 did a retrospective study and reviewed 95 patients treated at their institute over 24 years with surgery, radiotherapy or both. They found equivalent local control rates in all the groups; no difference between the outcomes of positive or negative surgical margins. They noted a higher recurrence rate (36.8% versus 16.8%) in head and neck tumours versus trunk, extremities, pelvis and retroperitoneal tumours. It is however important to note that the head-and-neck group was very small in relation to the other group (19 patients versus 54 patients).
They suggested adjuvant radiotherapy for patients with recurrence after surgery (possibly in positive surgical margins); site of origin - head and neck; and a poor functional outcome after surgery.
Melis et al11 published a review on how important a negative surgical margin is and came to a conclusion that you should not resect desmoid tumours due to the consequence of a poor functional outcome in order to achieve a negative surgical margin. In this review it was concluded that treating recurrent lesions with surgical excision and radiotherapy had better results than surgery alone.
Gronchi et al18 came to the conclusion after reviewing 203 cases retrospectively (35-year follow-up) that microscopic positive disease does not necessarily affect disease-free outcome in patients with primary lesions but definitely at recurrence. They found that 15% of those patients who presented with a primary lesion were cured even though they had a positive margin on histology. Forty of their patients had adjuvant radiation with a disease-free survival rate of 18% versus 12% at 5 years for those who were not treated with adjuvant radiation therapy.
Merchant et al19 evaluated 189 patients of whom 105 complied with their inclusion criteria (with primary lesions) and found that there was no statistically significant difference microscopically between a positive or negative surgical margin with a local recurrence rate of 22% in those with a positive resection margin and 24% in those with a negative resection margin (p=0.51). There was also no difference in the group treated with adjuvant radiation therapy. Seven of the 31 patients (23%) who had radiation developed recurrence versus the 23% (11 of 14 patients) recurrence rate of those who didn't have radiation (p=0.82).
Spear et al20 evaluated 101 patients who had surgery alone, radiation therapy alone, or both, and found that they had better local control in those patients with a negative surgical margin as well as those with a positive margin and radiation therapy.
Ballo et al21,11 came to the conclusion that radiation therapy is effective in unresectable masses alone or in combination with a positive surgical margin. Tumours with negative surgical margins had a 10-year relapse rate of 21%; positive surgical margins had a relapse rate of 54%; and radiation therapy a relapse rate of 24% with a 15% expected control in those patients who were treated with radiotherapy over the long term.
Nuyttens et al23 did a Medline search in 2000 and compared 22 articles. They came to the conclusion that radiotherapy alone or surgery plus radiotherapy had a better outcome than surgery alone.
There are numerous studies stating that adjuvant radiotherapy has a better outcome in selected cases11,20-22,24-29 but also many others stated no difference in the outcome 3,4,18,19,23,24,30,31
Definitive radiotherapy must be considered in a patient where surgery will be severely debilitating.21-23 The benefits of radiation therapy versus the complications of radiation therapy such as wound breakdown, infection, secondary malignancies, lymphedema and pathological fractures should however be weighed up carefully when planning treatment.32
Neoadjuvant radiation therapy has not been proven to have a better outcome.21 Studies are however limited.
Intra-operative electron radiotherapy (IOERT) has been studied by Roeder et al.33 They evaluated 30 individualised patients in whom surgical intervention alone would have had severe complications. They had a 3-year local control rate of 82% and concluded that IOERT might be feasible in a multimodal approach.
Radiofrequency ablation has been suggested as an alternative treatment option for desmoid tumours but studies are limited.34,35
Percutaneous cryoablation in small desmoid tumours have been suggested by Kujak et al.36 They found that this treatment modality appeared to have adequate local control of the tumour as well as pain relief. This was however a retrospective study of only five patients.
When reviewing the above and many other studies one can come to the conclusion that surgical margin is a controversial measurement of recurrence. There is furthermore no clear indication as to whether radiation therapy should be administered or not. It does however make sense to give radiation therapy first in those patients with a high possibility of morbidity post-surgery. A multidisciplinary team approach is necessary in order to correctly individualise treatment. The surgeon, oncologist, pathologist and patient must be part of the decision-making team.
Systemic therapy
Systemic therapy has been experimented with for years in order to see if there is a less invasive way of managing desmoid tumours than with surgery and radiation therapy. Some of the systemic treatment options are: cytotoxic agents (anthracyclines); molecular targeted agents: imatinib; and antiestrogen hormonal agents (e.g. tamoxifen, toremifene),interferon and NSAIDs.
De Camargo et al37 did a retrospective review of 68 patients seen at their institution who received some form of systemic therapy from 1994-2001.The median follow-up was 63 months. They found the following: partial response (PR): 19%; stable disease (SD): 58%; progressive disease (PD) 23%. It was concluded that systemic therapy is an option for patients with debilitating disease. They also noted that anthracyclines and hormonal therapy had a better response rate than dacarbazine/temozolomide or thyrosine kinase inhibitors. This is however a retrospective study over 13 years, the type of treatment and dosages were surgeon-dependent and could have also been influenced by the severity of the disease.
Tyrosine kinase inhibitor (imatinib)
The exact mechanism of action for imatinib on a desmoid tumour is still being investigated. There have been a few studies evaluating the target of imatinib, and some of the possible target sites were: PDGFRct, PDGFRp, c-KIT, PDGFct, PDGFp and macrophage colony stimulating factor.38-43 We know it is a tyrosine kinase inhibitor. Table II summarises the studies we evaluated in order to see what the outcome of imatinib in desmoid tumours is.
After reviewing the studies in Table II, the following conclusion was reached: there is definitely an indication for imatinib in the treatment of desmoid tumours with stable disease (SD) ranging between 28% and 84% and of progressive disease (PD) of 10%-35%. What was however concerning was the number of patients who defaulted in two of the studies. In the study by Penel et al44 50% of patients defaulted due to: disease progression (9), haematological toxicity (4), refusal of treatment (6), and investigator's decision. Heinrich et al45 had a 92% default rate due unsatisfactory results; this study however included 186 different malignancies with imatinib-sensitive tyrosine kinases or PDGFRa. To date, these are not necessarily the targeting receptor on those tumours.
Anti-oestrogen therapy
Bocale et al49 published a systematic review in 2011 evaluating the efficacy of anti-oestrogen therapy. They evaluated 168 patients. Forty-one articles were reviewed. The anti-oestrogen therapy treatment was started as first-line treatment in 92 patients for a period of 9 months and in 34 patients with recurrent disease following surgery; they were unable to determine if it was started as first-line treatment or recurrence in 42 patients. They found the following:
Outcome of anti-oestrogen therapy in FAP/Gardner's syndrome and desmoid tumour as shown in Table III.
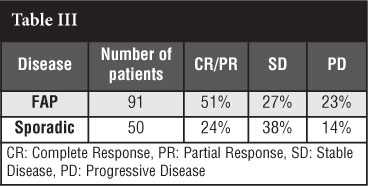
Monotherapy with anti-oestrogen had a significantly higher CR/PR than those treated with anti-oestrogens and NSAIDs.
Tamoxifen and NSAID had a 35% response rate versus 58% in those treated with tamoxifen alone; they however had a higher stabilisation rate. The authors came to the conclusion that this could be biased as more aggressive desmoid tumours might have been treated with combination treatment rather than monotherapy.
Tamoxifen and toremifene are equally effective. Anti-oestrogen treatment is definitely an option for systemic therapy in desmoid tumours. It has fewer side effects than the other systemic drugs and is tolerated better by the patients.
Hong et al50 did a study on mice and came to the conclusion that testosterone regulates the β-catenin level and cell proliferation and that anti-testosterone might be effective in the treatment of aggressive fibromatosis. This however needs to be further investigated with a prospective human trial.
Chemotherapy
The French Sarcoma Group Garbay et al51 did a retrospective study of 62 patients who had chemotherapy. Forty-four patients had combination chemotherapy and 18 monotherapy of which 13 patients had an anthracycline-containing regimen. They found a higher response rate in the anthracycline group versus the nonanthracycline-containing group: 54% versus 12% (p=0.0011). Forty-eight per cent did not need any other treatment intervention after chemotherapy and the most commonly used regimen was methotrexate-vinblastine.
A retrospective study on 39 patients by Constantinidou et al52 found similar results (Table IV).
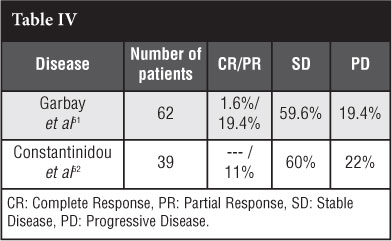
Chemotherapy can have severe side effects such as: mucositis, vomiting, neutropaenia, neurotoxicity, cardiotoxicity (irreversible) and treatment-induced malig-nancy.51,52 Though chemotherapeutic agents are a possibility for the treatment of desmoid tumours, the significant morbidity due to the side effects of this treatment modality have to be considered. The benefit must outweigh the risk.
NSAIDs
COX-2 is involved in the cell proliferation due to β-catenin stabilisation in desmoid tumours.53 Nishida et al54did a prospective and consecutive case-control trial where they treated 22 patients with meloxicam (10 mg daily) for a median period of 20 months. All the specimens showed a strong positive staining with COX-2 on immunohisto-chemistry. Two patients defaulted due to gastritis, pneumonia or diarrhoea. Of the 20 patients who continued with the study, 95% had a good outcome (stable disease or better) (complete response: 1; partial response: 7, progressive disease: 1; not determined: 2; stable disease: 11). This was however a small prospective study and a bigger randomised trial will be ideal in order to evaluate the above results adequately.
Conclusion
After reviewing the literature and evaluating the outcome of our patients the following treatment regimen was decided upon:
The 'wait-and-see' principle is valid in a patient who is asymptomatic, stable and with non-progressive disease. They have to be followed up closely every 3-6 months to monitor the disease progress. Rather intervene if there is any doubt regarding the disease progression.
Surgery has a high recurrence rate, whether a positive or negative surgical margin is achieved. Radiotherapy in a patient with a positive surgical margin would not necessarily have changed the outcome of our patients. You have to take the side effects of radiation into consideration before exposing your patient to this modality as these side effects will also limit the success of your surgical intervention, if needed, later (e.g. wound breakdown).
Radiation therapy can be considered in patients with large tumours causing functional incapacity, neurovascular involvement or where surgery will cause severe morbidity (unresectable tumour).
Systemic therapy can be given in any recurrent tumour, unresectable tumour, progressive disease or where the least-invasive treatment is to be used as first-line treatment.
Radical surgery for an unresectable tumour is rarely needed and radiotherapy or systemic therapy should be considered first.
The most important concept is to individualise each patient and to have a multidisciplinary team approach involving the surgeon, radiation and medical oncologist, pathologist, radiologist, psychologist, occupational therapist, physiotherapist and most importantly the patient's opinion.
References
1. National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology - Soft Tissue Sarcoma. 2011; Available at: http://www.nccn.org, 2012. [ Links ]
2. De Bree E, Keus R, Melissas J, Tsiftsis D, van Coevorden F. Desmoid tumors: need for an individualized approach. Expert Rev AnticancerTher. 2009 Apr;9(4):525-35. [ Links ]
3. Tolan S, Shanks JH, Loh MY, Taylor B, Wylie JP. Fibromatosis: benign by name but not necessarily by nature. Clin Oncol (R Coll Radiol) 2007 Jun;19(5):319-26. [ Links ]
4. Pignatti G, BarbantiBrodano G, Ferrari D, Gherlinzoni F, Bertoni F, Bacchini P, et al. Extraabdominal desmoid tumor: a study of 83 cases. Clinical Orthopaedics & Related Research 2000 June;375:207-13. [ Links ]
5. Lazar AJ, Hajibashi S, Lev D. Desmoid tumor: from surgical extirpation to molecular dissection. Curr Opin Oncol. 2009 Jul;21(4):352-59. [ Links ]
6. Lips DJ, Barker N, Clevers H, Hennipman A. The role of APC and beta-catenin in the aetiology of aggressive fibromatosis (desmoidtumors). Eur.J.Surg.Oncol. 2009 Jan;35(1):3-10. [ Links ]
7. Ferenc T, Wronski JW, Kopczynski J, Kulig A, Sidor M, Stalinska L, et al. Analysis of APC, alpha-, beta-catenins, and N-cadherin protein expression in aggressive fibromatosis (desmoidtumor). Pathol Res Pract. 2009;205(5):311-24. [ Links ]
8. Jilong Y, Jian W, Xiaoyan Z, Xiaoqiu L, Xiongzeng Z. Analysis of APC/beta-catenin genes mutations and Wnt signalling pathway in desmoid-type fibromatosis. Pathology 2007 Jun;39(3):319-25. [ Links ]
9. Signoroni S, Frattini M, Negri T, Pastore E, Tamborini E, Casieri P, et al. Cyclooxygenase-2 and platelet-derived growth factor receptors as potential targets in treating aggressive fibromatosis. Clin Cancer Res. 2007 Sep 1;13(17):5034-40. [ Links ]
10. Wu C, Amini-Nik S, Nadesan P, Stanford WL, Alman BA. Aggressive fibromatosis (desmoidtumor) is derived from mesenchymal progenitor cells. Cancer Res. 2010 Oct 1;70(19):7690-98. [ Links ]
11. Matono H, Tamiya S, Yokoyama R, Saito T, Iwamoto Y, Tsuneyoshi M, et al. Abnormalities of the Wnt/[beta]-catenin signalling pathway induce tumour progression in sporadic desmoid tumours: correlation between [beta]-catenin widespread nuclear expression and VEGF overexpression. Histopathology 2011 September;59(3):368-75. [ Links ]
12. Barbier O, Anract P, Pluot E, Larouserie F, Sailhan F, Babinet A, et al. Primary or recurring extra-abdominal desmoid fibromatosis: assessment of treatment by observation only. Orthopaedics & traumatology, surgery & research 2010 Dec;96(8):884-89. [ Links ]
13. Stoeckle E, Coindre JM, Longy M, Binh MB, Kantor G, Kind M, et al. A critical analysis of treatment strategies in desmoid tumours: a review of a series of 106 cases. Eur J Surg Oncol. 2009 Feb;35(2):129-34. [ Links ]
14. Bonvalot S, Eldweny H, Haddad V, Rimareix F, Missenard G, Oberlin O, et al. Extra-abdominal primary fibromatosis: Aggressive management could be avoided in a subgroup of patients. Eur J Surg Oncol. 2008 Apr;34(4):462-68. [ Links ]
15. Salas S, Dufresne A, Bui B, Blay JY, Terrier P, Ranchere-Vince D, et al. Prognostic factors influencing progression-free survival determined from a series of sporadic desmoidtumors: a wait-and-see policy according to tumor presentation. J Clin Oncol. 2011 Sep 10;29(26):3553-58. [ Links ]
16. Gluck I, Griffith KA, Biermann JS, Feng FY, Lucas DR, Ben-Josef E. Role of radiotherapy in the management of desmoidtumors. Int J Radiat Oncol Biol Phys. 2011 Jul 1;80(3):787-92. [ Links ]
17. Melis M, Zager JS, Sondak VK. Multimodality management of desmoidtumors: how important is a negative surgical margin? J Surg Oncol. 2008 Dec 15;98(8):594-602. [ Links ]
18. Gronchi A, Casali PG, Mariani L, Lo Vullo S, Colecchia M, Lozza L, et al. Quality of surgery and outcome in extra-abdominal aggressive fibromatosis: a series of patients surgically treated at a single institution. J Clin Oncol. 2003 Apr 1;21(7):1390-97. [ Links ]
19. Merchant NB, Lewis JJ, Woodruff JM, Leung DH, Brennan MF. Extremity and trunk desmoid tumors: a multifactorial analysis of outcome. Cancer 1999 Nov 15;86(10):2045-52. [ Links ]
20. Spear MA, Jennings LC, Mankin HJ, Spiro IJ, Springfield DS, Gebhardt MC, et al. Individualizing management of aggressive fibromatoses. Int J Radiat Oncol Biol Phys. 1998 Feb 1;40(3):637-45. [ Links ]
21. Ballo MT, Zagars GK, Pollack A, Pisters PW, Pollack RA. Desmoid tumor: prognostic factors and outcome after surgery, radiation therapy, or combined surgery and radiation therapy. J Clin Oncol. 1999 Jan;17(1):158-67. [ Links ]
22. Ballo MT, Zagars GK, Pollack A. Radiation therapy in the management of desmoidtumors. International Journal of Radiation Oncology*Biology*Physics 1998 12/1;42(5):1007-14. [ Links ]
23. Nuyttens JJ, Rust PF, Thomas CR,Jr, Turrisi AT,3rd. Surgery versus radiation therapy for patients with aggressive fibromatosis or desmoidtumors: A comparative review of 22 articles. Cancer 2000 Apr 1;88(7):1517-23. [ Links ]
24. Jelinek JA, Stelzer KJ, Conrad E, Bruckner J, Kliot M, Koh W, et al. The efficacy of radiotherapy as postoperative treatment for desmoid tumors. Int J Radiat Oncol Biol Phys. 2001 May 1;50(1):121-25. [ Links ]
25. Micke O, Seegenschmiedt MH. German Cooperative Group on Radiotherapy for Benign, Diseases. Radiation therapy for aggressive fibromatosis (desmoidtumors): results of a national Patterns of Care Study. Int J Radiat Oncol Biol Phys. 2005 Mar 1;61(3):882-91. [ Links ]
26. McDonald ES, Yi ES, Wenger DE. Best cases from the AFIP: extraabdominaldesmoid-type fibromatosis. Radiographics 2008 May-Jun;28(3):901-906. [ Links ]
27. Rudiger HA, Ngan SY, Ng M, Powell GJ, Choong PF. Radiation therapy in the treatment of desmoid tumours reduces surgical indications. Eur J Surg Oncol. 2010 Jan;36(1):84-88. [ Links ]
28. Baumert BG, Spahr MO, Von Hochstetter A, Beauvois S, Landmann C, Fridrich K, et al. The impact of radiotherapy in the treatment of desmoid tumours.An international survey of 110 patients. A study of the Rare Cancer Network. Radiat Oncol. 2007;2:12. [ Links ]
29. El-Haddad M, El-Sebaie M, Ahmad R, Khalil E, Shahin M, Pant R, et al. Treatment of aggressive fibromatosis: the experience of a single institution. Clin Oncol (R Coll Radiol) 2009 Dec;21(10):775-80. [ Links ]
30. Lev D, Kotilingam D, Wei C, Ballo MT, Zagars GK, Pisters PW et al. Optimizing treatment of desmoid tumors. J Clin Oncol. 2007 May 1;25(13):1785-91. [ Links ]
31. Kumar V, Khanna S, Khanna AK, Khanna R. Desmoid tumors: experience of 32 cases and review of the literature. Indian J Cancer 2009 Jan-Mar;46(1):34-39. [ Links ]
32. Biermann JS. Desmoid tumors. Curr Treat Options Oncol. 2000 Aug;1(3):262-66. [ Links ]
33. Roeder F, Timke C, Oertel S, Hensley FW, Bischof M, Muenter MW, et al. Intraoperative Electron Radiotherapy for the Management of Aggressive Fibromatosis. International Journal of Radiation Oncology*Biology*Physics 2010 3/15;76(4):1154- 60. [ Links ]
34. Tsz-Kan T, Man-Kwong C, Shu Shang-Jen J, Ying-Lee L, Wai Man-Wah A, Hon-Shing F. Radiofrequency ablation of recurrent fibromatosis. J Vasc Interv Radiol. 2007 Jan;18(1 Pt 1):147-50. [ Links ]
35. Ilaslan H, Schils J, Joyce M, Marks K, Sundaram M. Radiofrequency ablation: another treatment option for local control of desmoidtumors. Skeletal Radiol. 2010 Feb;39(2):169-73. [ Links ]
36. Kujak JL, Liu PT, Johnson GB, Callstrom MR. Early experience with percutaneous cryoablation of extra-abdominal desmoidtumors. Skeletal Radiol. 2010 Feb;39(2):175-82. [ Links ]
37. De Camargo VP, Keohan ML, D'Adamo DR, Antonescu CR, Brennan MF, Singer S, et al. Clinical outcomes of systemic therapy for patients with deep fibromatosis (desmoidtumor). Cancer 2010 May 1;116(9):2258-65. [ Links ]
38. Goncalves A, Monges G, Yang Y, Palmerini F, Dubreuil P, Noguchi T, et al. Response of a KIT-positive extra-abdominal fibromatosis to imatinibmesylate and KIT genetic analysis. J Natl Cancer Inst. 2006 Apr 19;98(8):562-63. [ Links ]
39. Liegl B, Leithner A, Bauernhofer T, Windhager R, Guelly C, Regauer S, et al. Immunohistochemical and mutational analysis of PDGF and PDGFR in desmoidtumours: is there a role for tyrosine kinase inhibitors in c-kit-negative desmoid tumours?. Histopathology 2006 Dec;49(6):576-81. [ Links ]
40. Tamborini E, Negri T, Miselli F, Lagonigro MS, Pricl S, Pilotti S. Re: Response of a KIT-positive extra-abdominal fibromatosis to imatinibmesylate and KIT genetic analysis. J Natl Cancer Inst. 2006 Nov 1;98(21):1583-84. [ Links ]
41. Wcislo G, Szarlej-Wcislo K, Szczylik C. Control of aggressive fibromatosis by treatment with imatinibmesylate. A case report and review of the literature. J Cancer Res Clin Oncol. 2007 Aug;133(8):533-38. [ Links ]
42. Heinrich MC, McArthur GA, Demetri GD, Joensuu H, Bono P, Herrmann R, et al. Clinical and molecular studies of the effect of imatinib on advanced aggressive fibromatosis (desmoidtumor). J Clin Oncol. 2006 Mar 1;24(7):1195-1203. [ Links ]
43. Dufresne A, Bertucci F, Penel N, Le Cesne A, Bui B, Tubiana-Hulin M, et al. Identification of biological factors predictive of response to imatinibmesylate in aggressive fibromatosis. Br J Cancer 2010 Aug 10;103(4):482-85. [ Links ]
44. Penel N, Le Cesne A, Bui BN, Perol D, Brain EG, Ray-Coquard I, et al. Imatinib for progressive and recurrent aggressive fibromatosis (desmoidtumors): an FNCLCC/French Sarcoma Group phase II trial with a long-term follow-up. Ann Oncol. 2011 Feb;22(2):452-57. [ Links ]
45. Heinrich MC, Joensuu H, Demetri GD, Corless CL, Apperley J, Fletcher JA, et al. Phase II, open-label study evaluating the activity of imatinib in treating life-threatening malignancies known to be associated with imatinib-sensitive tyrosine kinases. Clin Cancer Res. 2008 May 1;14(9):2717-25. [ Links ]
46. Mace J, Sybil Biermann J, Sondak V, McGinn C, Hayes C, Thomas D, et al. Response of extraabdominaldesmoidtumors to therapy with imatinibmesylate. Cancer 2002 Dec 1;95(11):2373-79. [ Links ]
47. Chugh R. Wathen JK. Patel SR. Maki RG. Meyers PA. Schuetze SM. Priebat DA. Thomas DG. Jacobson JA. Samuels BL. Benjamin RS. Baker LH. Sarcoma Alliance for Research through Collaboration (SARC). Efficacy of imatinib in aggressive fibromatosis: Results of a phase II multicenter Sarcoma Alliance for Research through Collaboration (SARC) trial. Clin Cancer Res. 2010 Oct 1;16(19):4884-91. [ Links ]
48. Kasper B, Dimitrakopoulou-Strauss A, Strauss LG, Hohenberger P. Positron emission tomography in patients with aggressive fibromatosis/desmoid tumours undergoing therapy with imatinib. Eur J Nucl Med Mol Imaging 2010 Oct;37(10):1876-82. [ Links ]
49. Bocale D, Rotelli MT, Cavallini A, Altomare DF. Anti-oestrogen therapy in the treatment of desmoid tumours: a systematic review. Colorectal Dis. 2011 Dec;13(12):e388-95. [ Links ]
50. Hong H, Nadesan P, Poon R, Alman BA. Testosterone regulates cell proliferation in aggressive fibromatosis (desmoid tumour). Br J Cancer 2011 Apr 26;104(9):1452-58. [ Links ]
51. Garbay D, Le Cesne A, Penel N, Chevreau C, Marec-Berard P, Blay JY, et al. Chemotherapy in patients with desmoidtumors: a study from the French Sarcoma Group (FSG). Ann Oncol. 2012 Jan;23(1):182-86. [ Links ]
52. Constantinidou A, Jones RL, Scurr M, Al-Muderis O, Judson I. Advanced aggressive fibromatosis: Effective palliation with chemotherapy. Acta Oncol. 2011 Apr;50(3):455-61. [ Links ]
53. Poon R, Smits R, Li C, Jagmohan-Changur S, Kong M, Cheon S, et al. Cyclooxygenase-two (COX-2) modulates proliferation in aggressive fibromatosis (desmoidtumor). Oncogene 2001 Jan 25;20(4):451-60. [ Links ]
54. Nishida Y, Tsukushi S, Shido Y, Wasa J, Ishiguro N, Yamada Y. Successful treatment with meloxicam, a cyclooxygenase-2 inhibitor, of patients with extra-abdominal desmoidtumors: a pilot study. J Clin Oncol. 2010 Feb 20;28(6):e107-109. [ Links ]
 Reprint requests:
Reprint requests:
Dr L du Plessis
Email: Leonoreduplessis@gmail.com














