Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
SA Orthopaedic Journal
versão On-line ISSN 2309-8309
versão impressa ISSN 1681-150X
SA orthop. j. vol.11 no.1 Centurion Jan. 2012
CLINICAL ARTICLE
Clinical utility of tissue polymerase chain reaction in the diagnosis of spinal tuberculosis
T Monni MBChB(Pret)I; A Visser MBChB(Pret), DTM & H(Wits), PG(Dip)TM(UFS), MMed(Clin Path)(Pret), FC Path(SA)(Clin Path)II; HF Visser MBChB(Pret), MMed(Orth Surg)(Pret)III; SN Motsitsi MBChBIV
ISenior Registrar, Department of Orthopaedic Surgery, University of Pretoria
IIConsultant, Division of Clinical Pathology, University of Pretoria/National Health Laboratory Services
IIIOrthopaedic Surgeon, Life Eugene Marais Hospital
IVHead of Department of Orthopaedic Surgery, Kalafong Hospital, University of Pretoria
ABSTRACT
BACKGROUND: An estimated 14 million people worldwide have active tuberculosis (TB). About 3% of these patients will have osteoarticular involvement and approximately 25% to 60% will have an infectious focus in the spine. Early diagnosis is essential as prompt treatment is associated with improved outcome and reduced mortality. This is particularly true within a high HIV-1 seroprevalence setting.
MATERIALS AND METHODS: All patients admitted to Kalafong District Hospital from January 2008 to December 2010 with a clinico-radiological diagnosis of spinal TB were included in this study. In all cases Ziehl-Nielsson (ZN) microscopy, TB culture, TB polymerase chain reaction (PCR), and histology with ZN stains were collected, and the turnaround times for these assays recorded. HIV testing was performed on patients who gave consent for the procedure.
RESULTS: In total, 29 patients were included in this study. Seventeen patients consented to HIV testing of which 11 were confirmed to be positive. It was determined that sensitivity for culture and PCR were comparable at 77% and 72% respectively. Furthermore, when looking at the subgroup of HIV-1 positive patients specifically, both assays performed better, with sensitivities of 88% and 82% respectively. The TAT for assays was highly variable, with PCR and histology having comparable times.
CONCLUSIONS: PCR testing for spinal TB shows promising results especially within the HIV-1-positive population. Although this type of testing theoretically offers a shorter turnaround time, results were available in similar time frames as for histology. Therefore, on-site testing should be offered in hospitals with high case loads of TB, and combination testing should be used rather than opting for a single testing modality.
Key words: Mycobacterium tuberculosis, molecular diagnosis, skeletal TB
Introduction
Infection with Mycobacterium tuberculosis remains a major health problem worldwide.1,2 The global incidence, as estimated by the World Health Organization (WHO), is said to have increased by 0.4% per annum.3 South Africa has also seen a significant rise in incidence, with rates increasing from 190 cases per 100 000 in 1980 to 339 per 100 000 in 2001.3 This is largely driven by the HIV-1 pandemic4 but other causes of immunosuppression, including malnutrition, IV drug abuse, alcoholism, cirrhosis, diabetes mellitus, pharmacological suppression,5-6 ageing7-13 and transplants14 may also precipitate activation of latent TB.
The WHO estimates that of the 14 million patients worldwide with active TB, approximately 3% will have skeletal infection.15 Of these, approximately 25% to 60% will have the infectious focus in the spine.16 Spinal TB produces an indolent and slow-growing infection17 and is characteristically paucibacillary.18 For this reason, diagnosis by demonstration of the micro-organisms is often problematic.19
Despite the diagnostic pitfalls, early accurate identification of the organism and determination of antibiotic sensitivity is essential, as early appropriate treatment is associated with improved outcome and reduced mortality.20 This is particularly true within a high HIV-1 sero-prevalence setting.20 Solid-media culture-based testing can require up to 8 weeks for identification, which is reduced to approximately 3 weeks with the use of liquid culture assays.19 The promise of rapid diagnosis exists with the wide implementation of molecular platforms like polymerase chain reaction assays (PCR). Turnaround times are being reported to be as short as 24 hours.21
In our clinical setting, patients are investigated utilising various modalities, including culture, histology, TB PCR and microscopy. Each of these modalities has varying sensitivities and specificities. The aim of this study was to evaluate the clinical utility of each of these modalities, both in terms of diagnostic accuracy as well as turnaround times.
Materials and methods
Patient population
All patients admitted to the Kalafong District Hospital from January 2008 to December 2010 with a diagnosis of spinal TB, were included in this study. For the purpose of this study, a diagnosis of spinal TB was based on a combination of suggestive clinical features, in conjunction with typical radiological findings associated with spinal TB. HIV-1 serology results were included where available. All patients were evaluated regarding the site of infection. This was described as both the number of vertebrae affected in each patient, as well as the level of infection.
Laboratory parameters
HIV-1 status was determined using the HIV Combi Assay (Roche Diagnostics, Mannheim, Germany). All positive results were confirmed using the HIV Ag/Ab Combo Assay (Abbott, Santa Clara, USA). Direct microscopy for M. tuberculosis was performed with a Ziehl-Nielsson stain (ZN).22 Quantification of acid-fast bacilli was performed using parameters specified by the Centers for Disease Control and Prevention (CDC).22 Mycobacterial cultures were performed the MGIT system (BD Diagnostic Systems, Sparks, MD) and subsequent sensitivities were determined using the agar proportion method by BACTEC MGIT 960 (BD Diagnostic Systems, Sparks MD). Histological examination of biopsy samples obtained from the affected spinal structures was performed using both haematoxylin and eosin (H&E) stains as well as ZN stain. The H&E stained samples were examined for features of myco-bacterial infection including granulomata and Langerhans cells. The ZN stain was examined for acid-fast bacilli. Biopsy samples were submitted for molecular testing using the GeneXpert Diagnostic System (Cepheid, Sunnydale, CA). This platform utilises real-time PCR technology and has been validated for direct use on diagnostic samples.23,24
Of note, the local laboratory, situated at the study site, performed microscopy and cultures. The biopsy samples were sent to the nearest academic centre for evaluation and the PCR was performed by a local private laboratory, as a referred test.
Results were obtained retrospectively for all assays (microscopy, culture, histology and PCR testing) from the laboratory database. The turnaround times were documented in terms of days from submission of the sample to availability of verified results.
Statistics
In order to determine the sensitivity and specificity of each of the assays, laboratory-based positivity was defined as either a positive finding on culture or histology compatible with tuberculous infection with acid-fast bacilli noted on ZN stain of the biopsy. ZN positivity in isolation was not considered as a positive result as various environmental mycobacteria may lead to false positivity. PCR positivity was not included as this was the assay used for comparison of diagnostic utility.
Results
Patient population
In total, 29 patients were included in this study. The median age was 46 years (28 to 70 years). In total, 11 of the patients were confirmed to be HIV-1-positive, six were HIV-1-negative and, in 12 cases, the HIV-1 status was not determined. Site of infection affected predominantly the lower thoracic spine, lumbar and sacral spine (Figure 1).

Only 11 of the patients had infection in only one vertebra, with the remainder having two vertebrae affected.
Of the patients included, 18 had confirmed spinal TB by either histology, ZN stain or culture positivity. Patients considered to be truly negative for spinal TB were diagnosed with a range of other conditions. Malignant disease was the most common, accounting for more than half of these patients. Two patients were diagnosed with multiple myeloma, one patient with thyroid carcinoma, one with B-cell lymphoma and one with adenocarcinoma. In three patients, no histological diagnosis could be made and in one case, chronic osteomyelitis was diagnosed.
Diagnostic accuracy (Table I)
As expected, ZN staining performed very poorly as a standalone diagnostic test. Although a positive result was highly specific in this setting, the negative predictive value was only 45%. The sensitivity for both culture and PCR were in excess of 70%, but this was found to be even higher in the HIV-1 positive population group (82% and 88% respectively). Culture had a higher method efficiency and negative predictive value. As per study definition, no false positive results were noted for platforms other than PCR testing, rendering the specificity at 100%. Of note, the single case of a positive PCR with negative culture, histology and ZN was not investigated further, and is only considered false positive based on the case definition defined in this study. This finding is further supported by the fact that Mycobacterium tuberculosis was not isolated in any of the cases where an alternative diagnosis was obtained.
Clinical utility
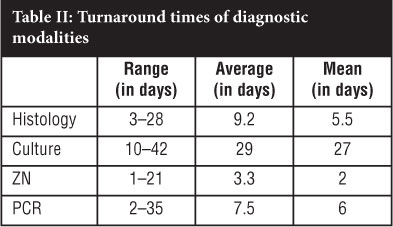
Culture results were obtained between 10 days and 6 weeks from submission to the laboratory (mean of 27 days). All samples with positive cultures were further investigated for possible resistance, and all strains identified were fully susceptible to rifampicin and INH. Although ZN stains generally yield a rapid result, sensitivity and specificity are so poor that they cannot be utilised in this clinical context.
These results reflect the markedly large range in turnaround times for all the assays. This is understandable in culture, as a negative result can only be reported after a six-week incubation period. However, for the other assays, the reason for this variability is unknown. Contrary to what was expected, the mean and average turnaround times for histology and PCR were comparable at 5.5 to 6 and 7.5 to 9 days respectively (Table II).
Discussion
The confirmation of infection with M. tuberculosis remains a diagnostic dilemma despite advances in radiological and laboratory testing. In fact, radiological findings are often so very similar for TB and various malignancies,25-28 that some authors advocate the use of microbiological or histological confirmation in all cases.29
Confirmation with culture can take as long as eight weeks with solid media culture, and alternative testing platforms are therefore sorely needed.7,10,30-36 Recently, various forms of serological testing including the interferon-gamma assay37 have been suggested for diagnosis. This has very limited utility in a high prevalence setting, as most individuals will show some degree of reactivity, irrespective of disease activity.17 Molecular testing seems to be more promising and is performed by amplifying and detecting nucleic acids specific to the micro-organism in question. These assays are often capable of delivering results within 24 hours.38 They also promise superior sensitivities and specificities, depending on number and actual sites targeted for amplification,16,39,40 as well as clinical sites sampled41-43 and the HIV-1 status of the patient.44 Use of molecular methods has the added advantage of improved laboratory safety, as live, infectious organisms are not amplified by culture.
Histology is still considered to be the gold standard for diagnosis by some authors, as the diagnosis is often made within the context of a local tissue reaction in conjunction with acid-fast bacilli.18 Histology requires good sampling techniques, and poor quality biopsies often have poor diagnostic utility. In this setting, molecular testing on tissue samples may render superior results, as very little genetic material is needed to be amplified and detected.45
Suffice it to say that the diagnostic test of choice should not only provide accurate results, but should also do so in a timely manner, to ensure the early initiation of appropriate therapy.46 Both histology and tissue PCR requires invasive sampling. Therefore, PCR testing will only be preferred if it offers a quicker turnaround time. In this study, histology and PCR turnaround times were very similar. Davies and co-workers suggested that provided the case load is sufficient, testing should be offered on site, as this had a big impact on acquiring timely results.19 Furthermore, utilising various diagnostic assays in a complementary fashion, rather than considering any one assay as a gold standard, may further improve diagnostic yield.45
In conclusion, PCR testing for spinal TB shows promising results especially within the HIV-1 positive population. Although this type of testing theoretically offers a shorter turnaround time, results were available in similar time frames as for histology. Therefore, on-site testing should be offered in hospitals with high case loads of TB, and combination testing should be used rather than opting for a single testing modality.
No benefits of any form have been received or will be received from a commercial party related directly or indirectly to the subject of this article.
References
1. Dye C, Scheele S, Dolin P, Pathania V, Raviglione M. Consensus Statement: Global Burden of Tuberculosis. Estimated incidence, prevalence, and mortality by country. JAMA 1999;282(7):677-86. [ Links ]
2. Raviglione M, Snider D, Kochi A. Global epidemiology of tuberculosis. morbidity and mortality of a worldwide epidemic. JAMA 1995;273(3):220-26. [ Links ]
3. WHO annual report on global TB control - summary. Wkly Epidemiol Rec 2003;78:122-28. [ Links ]
4. Chretien J, Papillon F. Tuberculosis and mycobacterioses in the AIDS era. Rev Prat 1990;40(8):709-14. [ Links ]
5. Dye C, Scheele S, Dolin P, Pathania V, Raviglione M. Global burden of tuberculosis: estimated incidence, prevalence, and mortality by country. JAMA 1999;282:677-86. [ Links ]
6. Leibert E, Schluger N, Rom W. Spinal tuberculosis in patients with humna immunideficiency virus infection: Clinical presentation therapy and outcome. Tubercle and Lung Disease 1996;77(4):329-34. [ Links ]
7. Baker C, Cartwright C, Williams D, Nelson S, Peterson P. Early detection of central nervous system M. tuberculosis with the genprobe neucleic acid amplification assay: utility in an inner city hospital. Clin Infect Dis 2002;35:339-42. [ Links ]
8. RIndi L, Lari N, Bonanni D, Garzelli C. Detection of Mycobacterium tuberculosis genotypic groups by a duplex realtime PCR targeting the katG and gyr A genes. J Microbiol Methods 2004;59:283-87. [ Links ]
9. Ruiz M, Torres M, Llanos A, Arroyo A, Palomares J, Aznar J; Direct detection of rifampin- and isoniazid-resistant Mycobacterium tuberculosis in auramine-rhodamine positive sputum specimens by real-time PCR. J Clin Microbiol 2004;42:1585-89. [ Links ]
10. Takahashi T, Nakayama T, Tamura M, Ogawa K, Tsuda H, Morita A. Nested polymerase chain reaction for assessing the clinical course of tuberculous meningitis. Neurology 2005;64:1789-93. [ Links ]
11. Takahashi T, Tamura M, Takahashi S, Matsumoto K, Sawada S, Yokoyama E, Nakayama T, Mizutani T, Takasu T, Nagase H. Quantitative nested real-time PCR Assay for assessing the clinical course of tuberculosis meningitis. Journal of the Neurological Sciences 2007;255:69-76. [ Links ]
12. Thwaites G, Nguyen D, Nguyen H, Hoang T, Do T, Nguyen T. Dexamethasone for the treatment of tuberculous meningitis in adolescents and adults. N Engl J Med 2004;351:1741-51. [ Links ]
13. Wada T, Maeda S, Tamaru A, Imai S, Hase A, Kobayashi K. Dual-probe assay for rapid detection of drug-resistant MycobacteriuM tuberculosis by real-time PCR. J Clin Microbiol 2004;42:5277-85. [ Links ]
14. Ip M, Yuen K, Woo P. Risk factors for pulmonary tuberculosis in bone marrow transplant receipients. Am J Respir Crit Care Med 1998;158:1173-77. [ Links ]
15. Tuberculosis [http://www.who.int/mediacentre/factsheets/fs104/en/] [ Links ]
16. Abou-Raya S, Abou-Raya A. Spinal tuberculosis: overlooked? J Int Med 2006;260:160-63. [ Links ]
17. Jain A. Tuberculosis of the spine. JBJS 2010;92(7):905-13. [ Links ]
18. Tuli S. General principles of osteoarticular tuberculosis. Clin Orthop Rel Res 2002;398:11-19. [ Links ]
19. Davies A, Newport L, Billigton O, Gillespie S. Length of time to laboratory diagnosis of MycobacteriuM tuberculosis infection: comparison of in-house methods with reference laboratory results. J Infect 1999;39:205-208. [ Links ]
20. Hargreaves N, Kadzakumanja O, Whitty C, Salaniponi F, Harries A, Squire S. 'Smear-negative' pulmonary tuberculosis in a DOTS programmes: poor outcomes in an area of high HIV seroprevalence. Int J Tuber Lung Dis 2001;5(9):847-54. [ Links ]
21. Negi S, Gupta S, Khare S, Lal S. Comparison of various microbiological tests including polymerase chain reaction for the diagnosis of osteoarticular tuberculosis. Ind J Med Microbiol 2005;23:245-48. [ Links ]
22. Winn W, Allen S, Janda W, Koneman E, Procop G, Schreckenberger P, Woods G. Koneman's color atlas and textbook of diagnostic microbiology, 6th edn. Philedelphia: Lippencott Williams & Wilkins; 2006. [ Links ]
23. Boehme C, Nicol M, Nabeta P, Michael J, Gotuzzo E, Tahirli R, Gler M, Blakemore R, WOrodria W, Gray C et al. Feasibility, diagnostic accuracy, and effectiveness of decentralised use of the Xpert MTB/RIF test for diagnosis of tuberculosis and multidrug resistance: a multicentre implementation study. Lancet 2011. [ Links ]
24. Helb D, Jones M, Story E, Boehme C, Wallance E, Ho K, Kop J, Owens M, Rodgers R, Banada P et al. Rapid detection of MycobacteriuM tuberculosis and Rifampicin resistance by use of on-demand, near-patient technology. J Clin Microbiol 2010;48:229-37. [ Links ]
25. An H, Vaccaro H, Dolinskas C. Differentiation between spinal tumors and infection iwht magnetic resonance imaging. Spine 1991;16(Suppl 8):334-38. [ Links ]
26. Arizono T, Oga M, Shiota E. Differentiation of vertebral osteomyelitis and tuberculous spondylitis by magnetic resonance imaging. Int Orthop 1995;19:319-21. [ Links ]
27. Hoffman F, Crosier J, Cremin B. Imaging in children with spinal tuberculosis: a comparison of radiography, computer tomography and magnetic resonance imaging. J Bone Joint Surg Br 1993;75:233-39. [ Links ]
28. Lolge S, Maheswari M, Shah J. Isolated solitary body tuberculosis: study of seven cases. Clin Radiol 2003;58:545-82. [ Links ]
29. Jutte P, Altena Rv, PRas E, Thijn C. Causes of misdiagnosis and mistreatment of spinal tuberculosis with radiotherapy in nonendemic areas: a pitfall in diagnosis and treatment. Spine 2005;30(11):300-304. [ Links ]
30. Kox L, Kuijper S, Kolk A. Early diagnosis of tuberculosis meningitis by polymerase chain reaction. Neurology 1995;45:2228-32. [ Links ]
31. Lee B, Tan J, Wong S, Tan C, Yap H, Low P. DNA amplification by the polymerase chain reaction for the rapid diagnosis of tuberculosis meningitis. Comparison of protocols involving three mycobacterial DNA sequences, IS6110, 65kDA antigen and MPB64. J Neurol Sci 1994;123:173-79. [ Links ]
32. Liu P, Shi Z, Lau Y, Hu B. Rapid diagnosis of tuberculous meningitis by a simplified nested amplification protocol. Neurology 1994;44:1161-64. [ Links ]
33. Nakajima H, Ashida K, Yamasaki H, Shinoda K, Ohsawa N. Intracranial tuberculoma with spontaneous recovery. Rincho Skinkeigaku 1995;35:521-25. [ Links ]
34. Scarpellini P, Racca S, Cinque P, Delfanti F, Gianotti N, Terreni M. Nested polymerase chain reaction for diagnosis and monitoring treatment response in AIDS patients with tuberculous meningitis. AIDS 1995;9:895-900. [ Links ]
35. Shankar P, Manjunath N, Mohan K, Prasad K, Behari M. Rapid diagnosis of tuberculous meningitis by polymerase chain reaction Lancet 1991;337:5-7. [ Links ]
36. Takahashi T, Nakayama T. Novel technique of quantitative nested real-time polymerase chain reaction assay for MycobacteriuM tuberculosis DNA. J Clin Microbiol 2006;44:1029-39. [ Links ]
37. Lai C, Tan C, Liu W, Lin S, Huang Y, Liao C, Hsueh P. Diagnostic performance of an enzyme-linked immunospot assay for interferon-gamma in skeletal tuberculosis. Eur J Clin Microbiol Infect Dis 2011;30(6):767-71. [ Links ]
38. Pandey V, Shawla K, Acharya K, Rao S, Rao S. The role of polymerase chain reaction in the management of osteoarticular tuberculosis. International Orthopaedics 2009;33(3):801-805. [ Links ]
39. Cheng V, Yam W, Hung I, Woo P, Lau S, Tang B, Yuen K. Clinical evaluation of the polymerase chain reaction for the rapid diagnosis of tuberculosis. J Clin Pathol 2004;57:281-85. [ Links ]
40. Kang E, Choi J, Seo B, Oh Y, Lee C, Shim J. Utility of polymerase chain reaction for detecting MycobacteriuM tuberculosis in specimens from percutaneous transthoracic needle aspiration. Radiology 2002;225:205-209. [ Links ]
41. Afonze D, Mir A, Kirmani A, Rehman S, Eachkoti R, Siddiqi M. Improved diagnosis of central nervous systeM tuberculosis by MPB64-target PCR. Braziian Journal of Microbiology 2008;39:209-13. [ Links ]
42. Condos R, McClune A, Rom W, Schluger N. Peripheral-blood-based PCR assay to identify patients with active pulmonary tuberculosis. Lancet 1996;347:1082-1085. [ Links ]
43. Kolk A, Schuitema A, Kuijper S, Leeuwen Jv, Hermans P, Embden Jv. Detection of MycobacteriuM tuberculosis in clinical samples by using polymerase chain reaction and a nonradioactive detection system. J Clin Microbiol 1992;30:2567-75. [ Links ]
44. Ribeiro M, Barouni A, Augusto C, Augusto M, Lopes M, Salas C. PCR identification of MycobacteriuM tuberculosis complex in a clinical sample from a patient with symptoms of tuberculous spondylosdiscitis. Brazilian Journal of Medical and Biological Research 2007;40:1-4. [ Links ]
45. Dijk Vv, McCleod A, Botha P, Shipley J, Kapnoudhis M, Beukes C. The diagnosis of skeletal tuberculosis of polymerase chain reaction. Cent Afr J Med 2000;46(6):144-49. [ Links ]
46. Getahun H, Harrington M, O'Brien R, Nunn P. Diagnosis of smear-negative pulmonary tuberculosis in people with HIV infection or AIDS in resource-constrained settings: informing urgent policy changes. Lancet 2007;369:2042-49. [ Links ]
 Reprint requests:
Reprint requests:
Dr T Monni
Email: tonimonni@hotmail.com
Cell: +2783 412 6716














