Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SA Orthopaedic Journal
On-line version ISSN 2309-8309
Print version ISSN 1681-150X
SA orthop. j. vol.10 n.4 Centurion Jan. 2011
CASE REPORT AND REVIEW OF THE LITERATURE
Spinal schistosomiasis in the era of tuberculosis
DE NonyaneI; MT MaribaII; J du ToitIII; S JenaIV; NM MbelleV
IBSc, MBChB(Medunsa) Registrar, Department of Orthopaedics, University of Limpopo, Dr George Mukhari Hospital, Pretoria
IIMBChB, MMed(Ortho),FCS(Ortho)(SA) Professor, Department of Orthopaedics, University of Limpopo, Dr George Mukhari Hospital, Pretoria
IIIMBChB(UP) Registrar, Department of Radiology, University of Limpopo, Dr George Mukhari Hospital, Pretoria
IVMBChB, MMed(Forensics)(Medunsa) Registrar, Department of Anatomical Pathology, University of Limpopo, National Health Laboratory Services, South Africa
VNM MSc, FCS(Path) Senior lecturer, Department of Microbiology, University of Limpopo, National Health Laboratory Services, South Africa
ABSTRACT
Spinal schistosomiasis although rare is a welldescribed entity. We report an unusual manifestation of ectopic spinal schistosomiasis involving T6/T7 with spinal cord compression in a patient on treatment for a suspected TB of the spine. The patient had T6/T7 vertebral destruction with complete paraplegia. This patient was treated at a rural hospital for spinal tuberculosis without neurological improvement and was diagnosed with spinal schistosomiasis by both histopathological means as well as serology. In areas endemic for schistosomiasis and tuberculosis, in patients with paraparesis irrespective of the level of the spinal lesion, schistosomiasis should be looked for as a differential diagnosis, especially in patients with poor response to an antiTB treatment regimen.
Key words: Schistosomiasis, tuberculosis, spine lesion, exposure, early treatment
INTRODUCTION
Africa is by far the most affected by schistosomiasis with 85% of the estimated 193 million global cases occurring on the continent.1 The disease is endemic in several provinces in South Africa. Although the most common presentation is urinary tract schistosomiasis, a diagnosis is easily made in the laboratory (microscopy and serology). Spinal schistosomiasis is well described in the literature and probably underdiagnosed2 as in South Africa paraparesis is commonly caused by tuberculosis far more frequently than schistosomiasis.
We therefore report a rare case of spinal schistosomiasis in which a patient presents with radiological features typical of spinal tuberculosis with poor response to antituberculous therapy. We also review the current literature on spinal schistosomiasis.
Case presentation
A 61yearold male presented at a local hospital (Limpopo province) with progressive loss of motor and sensory function in the lower limbs and loss of urinary and bowel sphincter control. An initial clinical diagnosis of tuberculosis of the spine was made but the patient did not respond to six months of antituberculous therapy. The patient was referred to us as a tertiary hospital due to failure of neurological recovery.
Physical examination revealed an elderly male in good general condition without visceromegaly. The patient was conscious and cooperative. Further assessment showed Frankel A neurological status trunk and lower limbs, with upper motor neuron lesion signs, motor and sensory level at T6. There was no urinary or bowel sphincter control.
Laboratory investigations were done. Haematological investigations were unremarkable except for a mildly raised erythrocyte sedimentation rate (ESR) of 16 mm/hr. There was no evidence of eosinophilia. The HIV ELISA was found to be nonreactive. Radiological examination confirmed T6/T7 vertebral body destruction with disc space narrowing and kyphotic deformity (Figure 1). Magnetic resonance imaging showed destruction of the vertebral bodies with paravertebral soft tissue mass and spinal cord compression anteriorly (Figure 2).
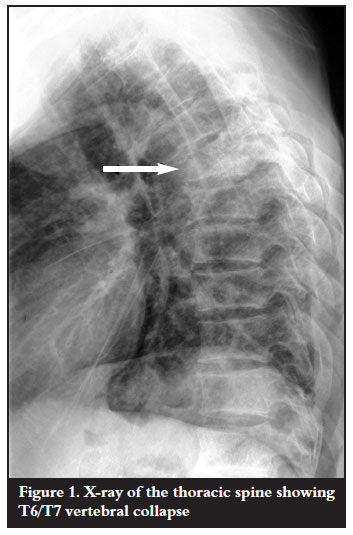
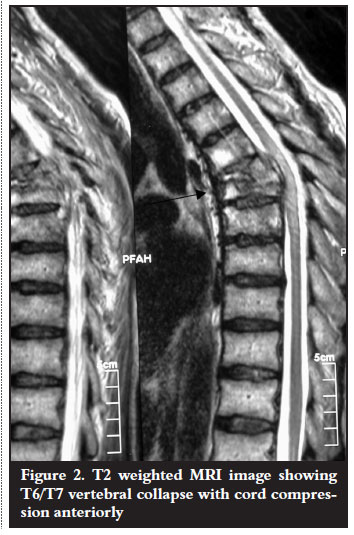
Antituberculous treatment was continued on the basis of the clinical findings. Surgical intervention was indicated as the neurological condition remained unchanged for six weeks. Anterior cord decompression and stabilisation were performed through a rightsided thoracotomy. Intraoperative findings were highly suggestive of spinal tuberculosis (cold abscess and granulation tissue). Biopsy materials were sent for microscopy culture and sensitivity as well as histology. These failed to identify Mycobacterium tubercu! losis on the ZiehlNielson stain and there was no growth on culture. Histopathological results revealed areas of a noncaseating granulomatous inflammation with multiple foreign body giant cells as well as numerous Schistosoma haematobium ova with lateral spines (Figures 3 and 4). Serological results using indirect haemagglutination assay (IHA) showed a titre of 1:128 and an immunofluorescence assay (IFA) result that was positive for both IgG and IgM. No bilharzia ova were identified in the urine.
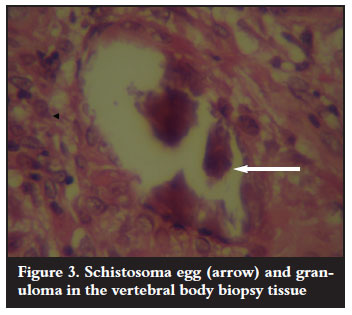
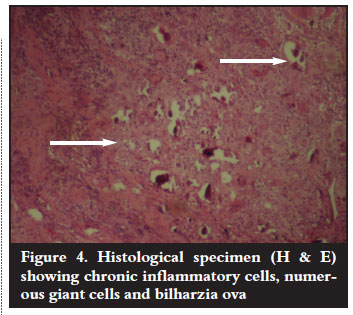
In addition to antituberculous treatment, praziquantel 40 mg/kg and prednisone 20 mg bid daily for two days were added. The patient continued with his spinal rehabilitation programme but with no neurological improvement. The IHA titre repeated after two months was 1:32. The IFA was positive for IgG but negative for IgM antibodies, indicating good response to antibilharzia medication.
Discussion
Schistosomiasis is endemic to Africa, South America and most parts of Asia. Schistosoma mansoni, Schistosoma japonicum and Schistosoma haematobium are the species significant to humans and the most widely distributed. The typical manifestations are urogenital, intestinal and hepatorenal. The involvement of other organs especially the central nervous system is uncommon.3 It is thought that the gravid worms or ova travel via anastomoses from the portal circulation to the internal vertebral plexus of Batson which is valveless. Retrograde spread via the vertebral vein to the spinal cord is postulated. S. mansoni and S. haematobium with their lateral and terminal spines are thought to enable the attachment of these ova in the spinal cord rather than the brain as occurring with S. japonicum. Adult worms and ova have been well demonstrated in vertebral vessels. The presence of ova is the cause of the pathological change of granuloma formation.4 The extent of the granulomatous lesion depends on the degree of infestation and the immune response of the host. Initially the inflammatory response is reversible; however, with chronic untreated disease, the pathology is associated with collagen deposition and fibrosis with a poor prognosis.5
Spinal schistosomiasis is a rare but it is a well described entity in South Africa.6 It is probably underdiagnosed as reported in the literature.7 Through our literature search, no similar case of vertebral schistosomiasis has been described. The clinical and radiological manifestations in our case overlapped that of tuberculosis so significantly that an original diagnosis of tuberculosis spondylitis was accepted. A midthoracic location as in our case is commonly seen in tuberculosis. Features of vertebral collapse and gibbus formation with intervertebral disc destruction are well described in tuberculosis as seen in our case. Moreover, granuloma formation is a feature of both schistosomiasis and tuberculosis. Paraspinal soft tissue abscesses with epidural extension to displace the cord are also typical findings of tuberculosis.8
Conclusion
As the clinical and radiological manifestations of spinal schistosomiasis may be similar to that of tuberculosis of the spine, clinicians should have a high index of suspicion in patients who present with paraparesis from areas endemic to schistosomiasis so as to improve clinical outcomes. The threshold for investigations should be lowered to exclude a treatable cause of paraparesis. A diagnostic biopsy is therefore essential before presumptive treatment.9 Our patient may have had a better outcome if diagnosed and treated for schistosomiasis earlier.
References
1. World Health Organization Public Health input of Schistosomiasis Bulletin WHO 1993;71:62357. [ Links ]
2. Joubert J, Fripp PJ, Hay IT, Davel GH, Van Graan ESJ. Schistosomiasis of the spinal cord underdiagnosed in South Africa? SAMJ 1990;77:29799. [ Links ]
3. Saleem S, Belal AI, ElGhandour NM. Spinal cord schistosomiasis: MR imaging appearance with surgical and pathologic correlation. Am Journ Neurol 2005;26:164654. [ Links ]
4. Budzilovich GN, Most H, Feigin I. Pathogenesis and latency of spinal cord schistosomiasis. Arch Pathol 1964;77:38388. [ Links ]
5. Saxe N, Gorden W. Schistosomiasis of the spinal cord and skin. S Afr Med J 1975;49:57. [ Links ]
6. Haribhai HC, Bhigjee AI, Bill PLA, Pammenter MD, Modi G, Hoffman M, Kelbe C, Becker P. Spinal cord schistosomiasis a clinical, laboratory and radiological study with a note on therapeutic aspects. Brain 1991;114(2):70926. [ Links ]
7. Ruberti RF, Saio M. Schistosomiasis of the spinal cord. Afr Journ Neuro Sc 2002;13(2):14. [ Links ]
8. Obisesan AA, Lagundoye SB, Lawson EA. Radiological features of tuberculosis of the spine in Ibadan, Nigeria. Afr J Med Sci 1977 Jun;6(2):5567. [ Links ]
9. Dunn RN, Zondagh I. Spinal tuberculosis: Diagnostic biopsy in mandatory. SAMJ 2008 May;98(5):36062. [ Links ]
 Reprint requests:
Reprint requests:
Dr DE Nonyane
PO Box 224 Medunsa 0204
Tel: 012 521 4049
Fax: 012 521 4029
Email: lsrauss@ul.ac.za
The content of this article is the sole work of the authors. No benefits in any form have been received or will be received from a commercial party related directly or indirectly to the subject of this article. Informed consent was obtained from the patient.














