Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
SA Orthopaedic Journal
versão On-line ISSN 2309-8309
versão impressa ISSN 1681-150X
SA orthop. j. vol.10 no.1 Centurion Jan. 2011
CLINICAL ARTICLE
Thirty three years of clinical experience with crosslinking of polyethylene in cemented total hip replacement
CJ GrobbelaarI; FA WeberII; TA du PlessisIII
IMBChB, MMed Surg (Orth), MD Orth Professor (extra-ord) University of Pretoria, Department of Orthopaedic Surgery, University of Pretoria
IIMBChB, MMed Surg (Orth), FRCS. Professor University of Witwatersrand, Johannesburg; Private Arthroplasty Practice, Johannesburg
IIIMSc, DSc. Gammatron, Pretoria
ABSTRACT
In response to the realisation, in the early 70s, that polyethylene debris emerged as an important cause of osteolysis and implant looseness, a new hip implant was designed and developed in Pretoria (Figure 1).
Although this was a Charnley lookalike, one of its most important goals was to reduce polyethylene wear and particle disease to the absolute minimum. A programme to gamma-crosslink pre-manufactured polyethylene cups was undertaken under the expert guidance of the third author1. The process was unique in the sense that surface crosslinking was augmented in the presence of acetylene as mediating gas in the crosslinking chamber.
In vitro mechanical tests were promising and were published extensively locally and abroad2. From 1977 to 1983 a cohort of 2200 hip replacements was performed by the first and second authors combined3. This publication acknowledges the Johannesburg cohort, but will focus on the 1775 cases performed by the Pretoria practice, with valuable co-operation and comparisons with the Johannesburg group. From our 1775 operations, we were able to follow up 272 hips at 10 years, and 97 operations for 10-33 years (mean 20.2 years) and this follow-up process is still ongoing.
It was concluded that there was an almost seven to eight times improvement in the polyethylene cup wear, from the international figure of ± 0.1mm per year down to 0.015 mm per year. Not only the longevity, but also the arthroplasty quality was greatly improved. Polyethylene crosslinking has now reached international acceptance in the written as well as the electronic press4.
Introduction
Total replacement of major joints has been successful over the medium term for over 50 years and has greatly improved the lives of millions of older patients around the globe. The important question is: can we consistently repeat these spectacular results in younger patients, and if so, for how long? That would constitute the real test for any prospective implant. Over the years, researchers world-wide have changed so many features of Sir John Charnley's original concept - new metals, ceramic and other bearing materials, cementless designs, new tribological concepts, head size and neck diameter variations. To date there has been no true solution; in fact, quite often the more we strayed from Sir John Charnley's concept, the more we have seemed to falter.
As early as 1976, the first and third authors embarked on a polyethylene improvement programme, subjecting ultra high molecular weight polyethylene (UHMWPE) samples to cobalt-60 gamma rays in an acetylene, gas-saturated environment. Crosslinking was enhanced in the superficial 300 µ of the material, in agreement with earlier work by Hagiwara5. After in vitro testing was completed and extensively published1,3,6,7,8,14, pre-manufactured acetabular cups and knee tibial inserts were crosslinked by this method. Probably the first international publication on crosslinking in orthopaedics appeared by authors 1 and 3 in the August 1978 edition of theBritish Journal for Bone and Joint Surgery2.
A surgical protocol followed in 1977 involving two combined series of 1775 and 430 total hip replacements in Pretoria and Johannesburg respectively, in which all operations were personally performed by the first and second authors. The protocol ended in 1984.
With the arrival of ceramics, the shelved gamma crosslinking protocol was almost forgotten until 1997, when the 14 to 20 year follow-up study revealed an almost unbelievable reduction of wear3. The sustenance of interface resulted in an extremely low incidence of polyethylene granuloma-related implant loosening.
The principle of the Pretoria method of crosslinking
The scientific input was entirely provided by the third author, at the time a radiation chemist at the Atomic Energy Board at Pelindaba. Although Mitsui9 and Hagiwara5 have described the method in principle in 1973, our application of the method in orthopaedics was unique in 1977. Of key importance was the presence of acetylene gas during crosslinking. This was described in detail by Du Plessis1. Although the gamma crosslinking process is complex and needs much more elaboration, the following are the main observations10,11:
• Liberation of hydrogen gas and formation of 'hot' hydrogen atoms CH2-CH2 + gamma = H + CH--CH2-. Crosslinking can now begin. This process is enhanced by the presence of acetylene as mediating gas.
• This is a diffusion-dependent process, accelerating crosslinking in the superficial 300 µ of the premanufactured polyethylene implant. Deeper than 300 µ, the normal attenuated regulated process leaves the material only moderately crosslinked. All other methods, to our knowledge, can only produce a crosslinked homogeneous material, where in fact a significant number of failures due to brittleness have been reported.
• An inhomogeneous material is created with superior wear resistance at the surface, while the desirable viscoelastic properties in the deeper material ensure superior shock-resistant qualities - an analogy with case-hardening in metallurgy. The creation of inhomogeneity is unique to the Pretoria (GammalinkTM) method of crosslinking.
Materials and methods
Case summary
A cohort of 272 total hip arthroplasties was studied. All operations were performed between 1976 and 1983 by the same surgeon. Only patients with a 10 year minimum follow-up were included. There were 97 cases followed over a 10-33 year postoperative period in this retrospective study (Table I). Inclusion and exclusion criteria are given in Table II, and the demographics in Table III. Of particular interest was the relative youth of our patients.



All hips studied utilised a stainless steel monoblock femoral design with a 30 mm head diameter, articulating on a gamma crosslinked UHMWPE acetabular socket. Clinical follow-up was according to Merle d' Aubigne and Postel6 at 6 months post operation at follow-up 1, when interfaces were considered to have stabilised, and again at their most recent consultation at follow-up 2. Wear measurement at follow-up 2 was according to the Digital Magnification Method (DMM), which became available only in 200213 . Earlier measurements were according to a combination of Charnley/Griffiths and Livermore methods15 , used mainly in follow-up 1 examinations. Interface widening and osteolysis were measured on the same digital magnified radiographs.
Results and discussion
Our study involved four important aspects of cemented arthroplasty follow-up. The four variables and degrees of deterioration are given in Table IV.
Wear was the independent variable, with pain, interface changes and osteolysis being the dependent variables.
In keeping with numerous world-wide Meier-Kaplan studies, revision was traditionally considered the final outcome. However, in our study, pain, radiographic deterioration and osteolysis (PIO) were considered extremely important causes of failure and therefore had to be included in the present study.
The Meier-Kaplan study was thus unsuitable, and different computerised statistical models, e.g. Fisher's Exact Test, Frequency Procedure and Chi Square Test were employed.
These methods are described elsewhere in this issue, in 'the associations' and 'failures' articles.
Four groups were clearly identified:
• Group 1: From a total of 97 replacements there were seven wear-related revisions. These seven cases had associated pain, osteolysis and interface widening, and they constituted Group 1.
• Group 2: A further nine cases also had in excess of 1 mm total wear but they had less pain, less interface widening and especially less osteolysis. Since their average total wear per case over 21 years was equal or more than 1 mm, they were considered impending failures and they constituted Group 2.
• Group 3: Thirteen cases had very little wear and almost no associated PIO and they constituted Group 3.
• Group 4: The rest of the series, 68 cases, had neither measurable wear nor other associated signs. Eightyone cases or 83.5% thus had an excellent long-term prognosis with zero wear at 20.12 years.
Wear breakdown of the four individual groups is given in Table V.
The average individual annual wear of the 97 cohort series was 0.015, verified by advanced computerised analysis20. Table V further depicted associations of wear with pain, osteolysis and interface widening.
Follow-up polyethylene wear after 10-33 years (mean 20.12) from a previous analogue study is given in Table VI.
The findings of this analogue study were later verified during computerised analysis.
Table VI also showed a new feature: where <1 mm wear was present, 15 cases presented with improvement (narrowing) of the interface. This was highly significant, and suggested an excellent prognosis and therefore justifies further statistical analysis, which was recently completed and awaits publication.
Examples
Refer to Figures 2-7 for examples.


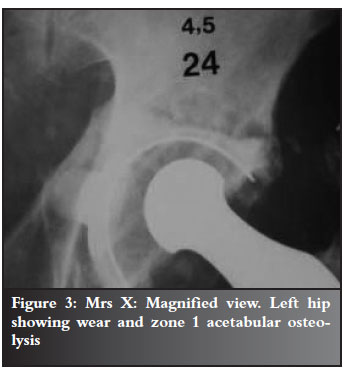


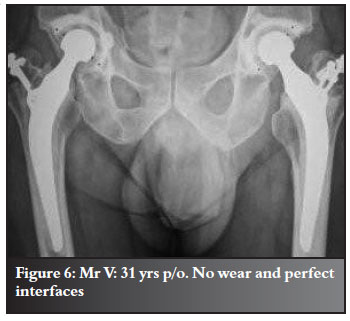
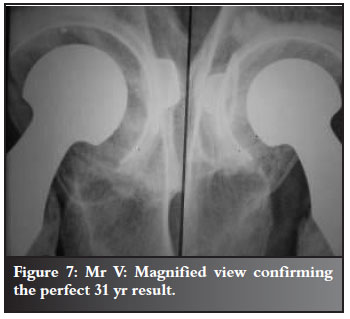
Discussion and review of the literature
Discussion and review of the literature Perhaps the greatest single contribution to total hip replacement of all times was the introduction of polyethylene as bearing material in the late 50s. The polyethylene used in the low-friction arthroplasty of Sir John Charnley rapidly gained a reputation as a wonder biomaterial, and with good reason. Unfortunately, when studying the life expectancy of the early 70s polyethylene on steel hips, they proved to wear at the average rate of 0.1mm/yr4,15. This wear has limited the expected life-span of the implant to 10 years. In his famous article, 'The problem is osteolysis', William Harris16 many years later confirmed the importance of progressive polyethylene particle-induced granulomas, leading to inevitable destruction of the arthroplasty.
Many surgeons around the globe realised the problem and explored various tribological options: ceramics on ceramics (COC), ceramics on polyethylene (COP), metal on metal (MOM), and even ceramics on metal (COM). None of these material combinations proved to offer a perfect solution. Valuable research by Muratoglu26 and numerous publications appeared by Fisher et al17 , McKellop18,19, Saikko23 , Nevelos24 and Claude Rieker25 . Problems were stripe wear, carbon pullout, implant fractures and or loosening, metallic pseudo tumours, metaliron systemic release and unknown longterm prognosis. Catastrophic wear with MOC hips and carbon pullout was reported by Valenti27, while dissemination of metal irons through the entire body caused great concern for Firkins et al28 .
The authors of this study had sufficient confidence in polyethylene, and suspected that wear and generation of polyethylene particles was really the only important weakness which had to be addressed. At that time (the early 70s) there were no reports of the orthopaedic use of crosslinked polyethylene in world literature, although Pilz had already proved in 1954 that polyethylene was the one polymer that was particularly suited to radiation crosslinking. Mitsui9 in 1972, and Hagiwara5 in 1973, demonstrated the role of acetylene and other gases in the augmentation of the radiationinduced crosslinking process. In 1976 the third author published on the mechanical improvement of polyethylene after polyethylene radiation in acetylene medium1, followed by the first article on crosslinking in the JBJS, by the first author2. This was also the first article on this topic in any international journal. This article was recently referred to in 72 publications and many more conference papers.
An answer to the following questions is of great importance to verify the true value of polyethylene crosslinked material:
a. Is gamma crosslinking really a worthwhile improvement? Wear resistance and long-term life expectancy improved by 6.6 to 8 times after crosslinking, according to various literature reports3,4,16,18. The present study showed annual individual wear as low as 0.015 mm, which is 6.66 times better than the 0.1 mm international figure of virgin polyethylene. John Fisher, in his multiple articles quoted an eight-fold reduction in wear with gamma crosslinked polyethylene17,28. Even when the SBA (specific biologic activity) is taken into consideration, the biological improvement is still at least four times better than with virgin polyethylene. Other researchers who quoted wear figures in the order of six to eight times lower with crosslinked polyethylene are McKellop22 , Willert19, Taylor20, Ian Clarke22, FA Weber3, Martell JM33, Dumbleton J29 and Harris WH17. Clearly, in terms of multiple clinical and experimental reports, there can be no doubt about the excellent long-life potential of crosslinked polyethylene in total hip replacement.
b. Negative aspects of crosslinking: There is undoubtedly valid concern; improved wear resistance against wear brings along the negativity of higher elastic modulus and more brittleness. We attempted to address this problem all along. High doses of radiation improved crosslinking, but also cause the formation of more free radicals due to hydrogen liberation - the combination of hardness and oxidation thus leads to brittleness. We have largely overcome this problem by radiation in an acetylene medium, which augments the crosslinking process and captures the free radicals. This allows us to apply much lower radiation doses, e.g. 100-140 KGy instead of, for example, Oonishi's project30 that utilised 1000 KGy! Wear and surface cracking was reported after crosslinking by Letitia Bradford et al31. However, they examined Durasul acetabular liners from 20 patients. The Durasul crosslinking method adopted by Zulzer and propagated by Muratoglu26 differed vastly from the Pretoria method: Zulzer employed a pre-manufactured radiation process, whereby the resin was crosslinked, and then moulded into acetabular cups. The result was a completely different homogeneous material from the inhomogeneous Pretoria acetylene augmented post-manufactured implant. The authors can only vouch for this particular process which was started in our department 30 years ago. None of the seven failures in our series of 97 showed any evidence of surface cracking even though wear was present in all seven cases. Thus, we believed that the method of crosslinking is of the utmost importance.
c. Crosslinked polyethylene under stressful situations: Recently, Kelly32 has demonstrated no increase in wear of crosslinked polyethylene, even with 44 mm head sizes. Wall thickness of liners was only 3.8 mm without detrimental effect. Also recently, Shia and Martell33 reported on younger patients with crosslinked cups and no signs of wear or brittleness after 4 years. This was also confirmed by Muratoglu26. It can thus be said that crosslinking of polyethylene provides us with a mandate to use the head size of our choice - an important advantage in terms of stability.
d. The current status of gamma crosslinked polyethylene: Current usage of crosslinking is still increasing in the USA and Europe. In 2008, 70% of all hip implants performed in the USA already employed a crosslinked polyethylene cup. Kurtz4 went on to state that in 2010 an estimated 175 000 American total hip patients will benefit from crosslinking polyethylene technology in a single year, while 1.5 million patients will have received a crosslinked acetabular cup over the past 12 years. In the USA every major orthopaedic company, except one, has developed their own version of crosslinking, e.g. Crossfire, Longevity, Marathon, XPLE, Durasul, X3, Arcom XL and Acumatch XL. These companies commenced crosslinking only in 1998. Kurtz4 reported purely on American projects, and his study unfortunately excluded earlier 'rest of the world' studies, e.g. the pioneering South African project as far back as 1976, in spite of being published as a full paper in the JBJS in 19782. Kurtz also mentioned the lack of long-term results with crosslinking. This publication hopefully satisfies that deficiency, with a follow-up of 10-33 years, and 1775 original gamma crosslinked hip implant operations.
Summary and conclusion
In this paper we report on a large series of crosslinked hip implants, with the longest postop follow-up that was properly researched, regularly reported on and finally statistically analysed. The small number of patients left after 20.12 years' mean follow-up (3.75%) is not unusual in this type of project. The 10-33 year remaining patients was 37% indicating the large drop out within the first 10 years. No wear at all was registered during the first 10 years.
The very low rate of polyethylene wear not only protects the long-term survival of the arthroplasty, but also improves the general quality of the arthroplasty. Graham Gie, in his recent report on the 33 year Exeter hip followup stated: 'No polyethylene wear = no osteolysis'.
In view of the serious problems emerging regularly with alterative materials and combinations, like MOM, COC and COM, we believe that it will be premature and naïve to reject polyethylene in favour of these relatively untested material combinations.
Polyethylene has come a long way, and by continuously improving the material and implant designs, the future of polyethylene seems assured.
Finally, this study has also produced 16 cases of postarthroplasty improvement of the interface. This is a new and exciting development and typically occurred in cases with no measurable wear, where excellent longevity can be expected. It justifies a statistically validated report which is currently underway.
References
1. Du Plessis TA, Grobbelaar CJ, Marais F. The improvement of polyethylene prostheses through radiation crosslinking. Radiat Phys Chem. 1997; 9: 647-50. [ Links ]
2. Grobbelaar CJ, Du Plessis TA, Marais F. The radiation improvement of polyethylene prostheses. A preliminary study. J Bone Joint Surg Br. 1978; 60: 370-77. [ Links ]
3. Grobbelaar CJ, Weber FA, Spirakis A, Du Plessis TA, Cappaert GGA, Cacik JN. Clinical experience with gamma irradiated crosslinked polyethylene - a 14 to 20 year followup report. SA Bone Joint Surg. 199; 1X3: 140-8. [ Links ]
4. Kurtz S, Medel FJ, Manley M. Wear in lightly crosslinked polyethylenes. Current orthopaedics. 2008; 22: 392-99. [ Links ]
5. Hagiwara M, Tapana T, Shinohara I, Kagiya T. radiation induced crosslinking of polyethylene in the presence of various acetylenic compounds. Polymer Letters. 1973; 11: 613-19. [ Links ]
6. Du Plessis TA. Van kinetika tot kunsgewrigte - die versnelde stralingskruisbinding van hoedigtheid polietileen. SA Tydskrif vir Natuurwettenskap en tegnologie. 1984; 3: 16-19. [ Links ]
7. Grobbelaar CJ. Kunstowwe in geneeskunde. Technicon. 1978; 7: 26-32. [ Links ]
8. Du Plessis TA, Grobbelaar CJ, Marais F. The improvement of polyethylene prostheses through radiation crosslinking. Presented at the international conference on bioengineering: Cape Town, April, 1977. [ Links ]
9. Mitsui H, Hosoi F. Acceleration of a fluorinecontaining monomeracetylene system for the gamma radiation induced crosslinking of polyethylene. Polymer. 1972; J3: 108-13. [ Links ]
10. Chapiro A. radiation chemistry of polymeric systems. Interscience publishers, NewYork. 1962; 407-410. [ Links ]
11. Dole M, Kelly CD, Rose DG. The pile irradiation of polyethylene. J Am Chem Soc. 1954; 76: 4304. [ Links ]
12. Grobbelaar CJ, Du Plessis TA. Straling in die gewrigte. Kernaktief. 1979; 20: 2-4. [ Links ]
13. Grobbelaar CJ. A new method to measure polyethylene cup wear in total hip replacement - a preliminary report. SA Orth J. 2007; 6: 18-24. [ Links ]
14. Griffith MG, Charnley JC. Socket wear in Charnley low friction arthroplasty of the hip. Clinic Orthop. 1978; 137: 37-42. [ Links ]
15. Wroblewski BM, Siney PD, Flemming PA. Charnley low friction torque arthroplasty. J Bone Joint Surg Br. 2009; 91: 447-50. [ Links ]
16. Harris W. The problem is osteolysis. Clin Orthop. 1995; 46: 311-313. [ Links ]
17. Fisher J, Zhongmin J, Tipper J, Stone M, Ingham E. Tribology of alternate joints. Clin Orth and rel res.2006; 453: 25-34. [ Links ]
18. McKellop HA, et al. Friction, lubrication and wear of chrome cobalt prostheses...Proc of 38th Ann Meeting Am Orth Soc Meeting. 1992; 402. [ Links ]
19. Willert H, Bucham GH et al. Histopath changes around metal on metal joints. Osteologie. 2000; 3: 165. [ Links ]
20. Taylor S, Manley T, Sutton K. The role of stripe wear in alumina ceramiconceramic bearings. J of Arthrop. 2007; 22 (7) Suppl 3: 47-51. [ Links ]
21. Gruber FW, Bock A, Tratting S. Cystic lesion of the groin due to metalosis. J of Arthrop. 2007; 22 (6): 923-27. [ Links ]
22. McKellop H, Clarke IC, Markolf K, Amstutz H. Friction and wear properties of polymer, metal and ceramics. J Biomed Mater Res.1981; 15: 619-53. [ Links ]
23. Seikko V. A 12 statio anatomical hip joint simulator. Proc Imech E. 2005; 219; 437-48. [ Links ]
24. Nevelos J, et al. Microseparation of aluminaalumina artificial hip joints. J of Arthop. 200; 15 (6): 783-95. [ Links ]
25. Riecker CB, et al. A tribology of large diameter MOM hips. Orth Clin North Am. 2004; 36: 135-42. [ Links ]
26. Muratoglu OK, et al. Larger diameter femoral heads used in conjunction with highly crosslinked polyethylene. J of Arthropl. 2001; 16 (8): 24-30. [ Links ]
27. Valenti J, et al. Catastrophic wear in metal on ceramic total hip arthroplasty. J of Arthropl. 2007; 22 (6): 920 - 22. [ Links ]
28. Firkins PJ, et al. Dissemination of metal irons. Biomet. 2003; 24: 469-79. [ Links ]
29. Dumbleton JH, Shen C. The friction and wear of very high molecular weight polyethylene. J of Applied Polymer Science.1974; 18: 3493-97. [ Links ]
30. Oonishi H, Clarke IC. Crosslinked polyethylene - longterm results. Presented at alternate bearing surface Conf,Philadelphia. 2000. [ Links ]
31. Bradford Letitia, Baker DA, Graham J. Wear and surface cracking in early retrieved highly crosslinked polyethylene. J Bone Joint Surg. 2004; 86-A (6): 1271 - 82. [ Links ]
32. Kelly NH, et al. High stressed conditions do not increase wear in highly crosslinked polyethylene. Clin Orth rel res. 2010; 468 (2): 418-23. [ Links ]
33. Shia DS, Martell JM, Verner JJ. Clinical performance of a highly crosslinked polyethylene at 2 years. J. of Arthrop 2003; 6 Suppl. 1. [ Links ]
 Reprint requests:
Reprint requests:
CJ Grobbelaar
PO Box 2083
Brooklyn Square
Pretoria 0075
Email:
The content of this article is the sole work of the authors. No benefits of any form have been received from a commercial party related directly or indirectly to the subjectof this article. All subjects included in this study providedtheir written informed consent.














