Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SA Orthopaedic Journal
On-line version ISSN 2309-8309
Print version ISSN 1681-150X
SA orthop. j. vol.10 n.1 Centurion Jan. 2011
CLINICAL ARTICLE
Surgery for myelopathy in spinal deformity
BR GarrettI; RN DunnII
IMBChB(Wits), FCS(SA)Orth Registrar, Department of Orthopaedics
IIMBChB(UCT), FCS(SA)Orth, MMed(Orth) Associate Professor and Head of Spinal Services Groote Schuur Hospital, Department of Orthopaedic Surgery, University of Cape Town, Cape Town, South Africa
ABSTRACT
AIM: To review surgical management and outcome in patients with myelopathy as the presenting complaint due to an underlying spinal deformity.
METHODS: A retrospective review of 15 patients over an eight-year period (2002-2009) was performed. The duration and severity of symptoms were recorded, the deformity assessed by radiological measures and surgical management and neurological outcome was reviewed.
RESULTS: The average age at surgery was 18.2 years with the average duration of recognised deformity being 60.3 months. Myelopathic symptoms were present for an average of 8 months before surgery with only two patients presenting acutely.
Seven patients had congenital spinal deformities, three previous tuberculosis, two neurofibromatosis, one diastrophic dysplasia, one adolescent idiopathic scoliosis and one previously fused myelomeningocoele with pseudarthrosis.
Two patients had severe scoliotic curves, three had moderate kyphosis (45-60º), five had severe kyphosis (>90º), and five complicated multi-planar helical deformities.
The surgical procedures included one posterior only, two anterior only, five staged anterior and posterior procedures, four combined anterior and posterior procedures and three posteriorly-based circumferential vertebral column resection procedures.
Two patients deteriorated neurologically and 13 remained the same or improved: three ASIA B to B/C/D, three C to E, seven D to D/E.
Other complications included two aborted anterior procedures due to impossible access, one revision for incomplete decompression, one revision for graft displacement, one deep wound infection and one wound breakdown.
CONCLUSION: This heterogeneous group of patients represents an extreme surgical challenge. Once neurological compromise develops, recovery or improvement with surgery is possible, but with associated risk of complete paralysis. However, deterioration is inevitable without surgery.
High thoracic deformities are a frequent cause and should be identified and referred early.
In this scenario, we currently recommend surgical management by means of a posteriorly based, circumferential decompression and corrective fusion.
Introduction
Neurological deterioration is the biggest concern to spinal deformity surgeons. In an effort to avoid this, pre-emptive surgery is recommended, and intra-operative risks minimised with meticulous planning, spinal cord monitoring (when available) and careful surgical technique.
Infrequently a patient presents with myelopathy as the complaint and subsequently a spinal deformity is recognised as the underlying cause. This is an extremely challenging group of patients. They have a 'sick spinal cord' and often severe multi-planar rigid deformities. One has to balance the risk of acute neurological deterioration with surgery versus the slow inevitable loss of function with the natural history of the disease. The surgeon has to weigh up waiting until the neurological status is bad enough to justify the risky intervention, or to intervene early for a potentially better outcome, the latter being associated with the possibility of drastic deterioration and complete paralysis.
Deformity of the spinal column has a number of causes: congenital, post-infectious, degenerative or iatrogenic. They may present to the spinal surgeon in a variety of ways, viz. pain, cosmesis concerns or neurological dysfunction. Severe kyphotic or kypho-scoliotic deformities which present primarily with neurological deterioration and myelopathy have a variable aetiological and age distribution, curve magnitude, curve location, and curve flexibility.1 Also the duration and severity of symptoms varies. These factors influence the state of compromise of the spinal cord, and influence the timing and surgical approach and instrumentation required for surgical intervention. The surgery is technically difficult and dangerous, but may prevent progressive neurological deterioration, facilitate recovery and restore sagittal balance and stability to the spine.
Congenital vertebral malformations are classified by Winter et al.2 Type I, the anterior failure of vertebral body formation, is the commonest, and if left untreated can develop severe rigid kyphotic deformity, causing spinal cord compression in 10-18% of cases.2,3 Morphological evaluation is difficult and meticulous pre-operative planning and a detailed knowledge of the anatomy is imperative. An abnormal vascular supply to the spinal cord may jeopardise surgical techniques.
Post-tuberculous infection, the so-called 'late-onset' paraplegia in patients with healed disease, may be produced by localised pressure on the spinal cord by a transverse ridge of bone anterior to the spinal cord or constricting scarring of and around the dura.4 Stretching of the spinal cord over an anterior internal gibbus may lead to interstitial gliosis to produce neurologic complications. Reactivation of quiescent disease at the apex or proximal or distal to the kyphosis also is a possibility.5
Surgical treatment of severe and rigid angular kyphotic deformities, particularly those with neurologic deficit, is particularly risky.
The spinal cord may experience mechanical stresses due to the angular kyphosis itself, or as a result of intra-operative manipulation, which includes: distraction, compression and translation. It may also occur as a result of deformity correction which includes kinking or dural buckling due to spinal shortening.6
A reliable and safe treatment requires thorough neurologic decompression and adequate correction of the deformity without further compromising the spinal cord. Numerous reconstructive techniques exist, using the anterior and posterior approaches or the single posterior approach with osteotomies and vertebral column resection.7,8
We present our experience with this group of patients.
Materials and methods
A retrospective review of patient records and radiographs of 15 consecutive patients treated at the Red Cross Children's Hospital, Groote Schuur Hospital and Constantiaberg Hospital, Cape Town, from 2002 to 2009 (eight years) was performed.
Treatment was on a case-by-case basis, guided by the primary surgeon's evolving experience, the deformity encountered, and the patients' age. All patients who presented with myelopathy caused by a chronic underlying kyphosis or kyphoscoliotic deformity (not acute trauma or active tuberculosis) were included.
Patient demographics, previous diagnosis and treatment received, duration of symptoms prior to referral and surgery, neurological Frankel grading pre-op, post-op, and at follow-up, type of surgical management, details of procedure type, approach, time taken, blood-loss, bone graft used, complications and outcomes were tabulated on a Microsoft Access database and analysed accordingly.
Results
Of the 15 consecutive patients studied, two were female and thirteen were male. The median age at the time of surgery was 18.2 years (4.2-50 ± 11.6). At presentation, the average duration of the awareness of a deformity was 60.3 months (1.4-169 ± 67.6) and the duration of myelopathic symptoms/signs was 8 months (0.2-31 ± 10.8).
Seven patients had congenital deformities. These were typically a focal kyphoscoliosis in the proximal thoracic spine, with an average kyphosis angle of 75º (45-100º). Three patients had a focal kyphosis of 90º due to healed tuberculosis. Two patients had dystrophic curves secondary to neurofibromatosis. One patient had diastrophic dysplasia. This was a young patient with kyphosis of the cervical spine. One patient with adolescent idiopathic scoliosis had an acute spontaneous deterioration and was found to have stenosis at the thoraco-lumbar junction. Another myelomeningocoele patient with a previously fused spinal deformity had a pseudarthrosis of the thoracic spine (Figure 1).
The majority of the deformities occurred in the proximal thoracic spine. The apex of the deformity was at the following levels: one C5, three T3, two T4, two T5, one T6, two T7, two T10, one T11 and one L1.
Nine were kyphoscoliotic deformities (60%), four were kyphotic curves (27%) and two were scoliotic curves (13%).
Neurological impairment was assessed according to the Frankel grading system. Of the eight patients who were preoperatively grade D, seven returned to normal (grade E) but one deteriorated to grade A. Three of the four grade C patients improved to normal but one deteriorated to grade A. The three Frankel B patients either improved (one to C, one to D) or stayed the same (one remained grade B). Improvement occurred in 80% of patients. However, 13% were rendered completely paraplegic (Figure 2).

The choice of surgery represents an evolution over time. Initially anterior only and combined procedures were performed. This progressed to the currently preferred method of vertebral column resection (VCR). Two were anterior only procedures and one posterior only. Nine were combined anterior and posterior procedures (five staged, four simultaneous), and three were posteriorly based circumferential decompressions with fusion (VCR). With this varied group of patients, the surgical time, blood loss, bone graft and instrumentation used was individualised.
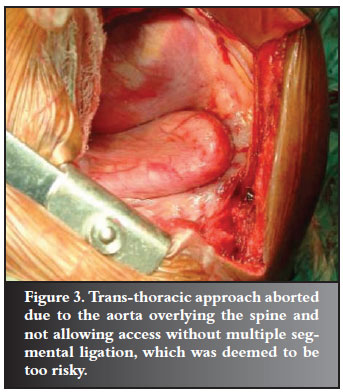
Two procedures were aborted due to inability to access the apex of the curve through the anterior approach. One was due to the severe 'helical' nature of the deformity.
The other was due to an aorta limiting access to the spine. The spinal cord would have been devascularised as multiple segmental artery ligation would have been required to mobilise it (Figure 3).
Two patients required revision surgery. One was for dislodgement of the strut graft. This occurred in a child who was too small for instrumentation. The other revision was for inadequate decompression of the stenosis visualised on postoperative CT imaging. This had been requested after the myelopathic symptoms failed to improve after the first attempt. Two cases of wound infection were managed with debridement and appropriate antibiotics.
Discussion
Lonstein et al9 who reviewed the literature up until 1980 regarding this scenario, found 39% to be due to congenital deformities, 16% due to neurofibromatosis, 11% due to healed tuberculosis, and 14% due to idiopathic scoliosis. Of the available data the deformities noted were: 68% kyphoscoliosis, 17% kyphosis and 15% scoliosis. This matches our cohorts' profile.
Lonstein et al9 then reported on 43 of their own patients. The severity and duration of deformity was also similar to our findings, as was the spectrum of neurological impairment. The average kyphotic curve was 95º and the average duration of symptoms was 15.9 months. Eight patients had minor neurological changes (Frankel D equivalent), 27 were paraparetic (Frankel B and C) and eight were paraplegic (Frankel A). Regarding surgery, anterior decompression fared best in their hands with a 65% (16/25) success rate of neurological improvement.
The majority of patients in the early years of our study received a similar treatment of anterior decompression and strut grafting with minimal correction. The main problem with this approach is surgical access and the inability to correct the deformity. It is often technically difficult to work through the chest in abnormal bony architecture as one is not certain of when the spinal cord will be encountered. If no correction is obtained the area of decompression needs to be more extensive to prevent the cord from kinking at the residual bony corners cranially and caudally.
More recently, decompression by posteriorly based vertebral column resection with correction of angular deformities has become popular in the literature. Several techniques have been described. Kawahara10 describes a 'closing-opening' wedge osteotomy. He inserts the instrumentation, followed by laminectomy and vertebrectomy. The deformity is then corrected, anterior support is placed and the rods are connected posteriorly.
Shimode et al11 performed seven posterior wedge osteotomies for spinal deformity patients (three patients had neurological deficits pre-operatively, all improved postoperatively). Chang et al12 proposes 'in-situ rod bending' after vertebrectomy. The important concept with this technique is for the correction hinge to be in the same plane as the spinal cord so that no stretching of the 'at risk' cord occurs.
Vertebral column resection surgery is technically demanding and requires a steep learning curve in order to perform effectively and efficiently. Surgical times are often long with potential for massive blood loss. The advantage is that the cord is visualised throughout. Controlled correction can be done while observing changes in the thecal sac morphology position and thus assessing that the dangerous tensile forces are not applied. Unfortunately the risk of catastrophic neurological deterioration is ever present, and cannot always be predicted. Spinal cord monitoring is highly recommended during this technique (Figure 4).
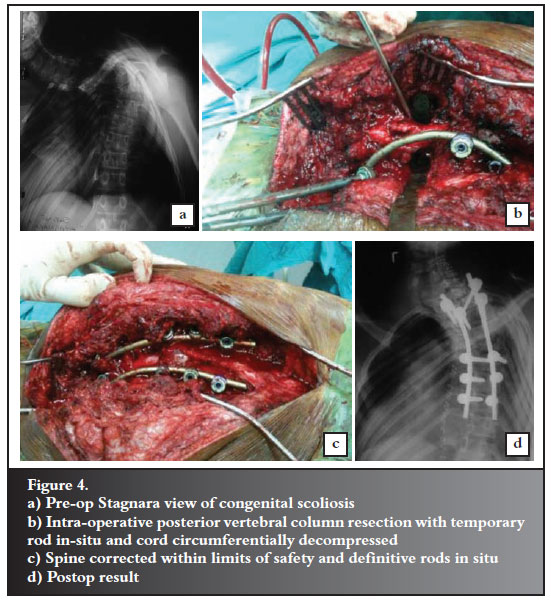
Neurological deterioration is not limited to the intra-operative period, and one of our deteriorations occurred at 48 hours post-op. This was assumed to be due to a vascular event as it was sudden in onset. This highlights the need for continued vigilance in the post-op period. Mean blood pressures should be maintained as physiological levels both intra-op and post-op when patients may be behind on fluids and under the effects of sedation.
Every attempt to be made to avoid patients progressing to such severe deformity as myelopathy is an increasing risk. When present it should be dealt with sooner rather than later.
Conclusion
Once myelopathy is established in the deformity patient, neurological improvement is possible with aggressive surgery (12/15); however acute (and permanent) neurological deterioration remains a risk (2/15).
Therefore early recognition of deformity with high 'myelopathy risk' is mandatory.
We have identified the proximal thoracic kyphotic deformity as being a prevalent cause for this. These patients should be monitored vigilantly and referred early rather than awaiting neurological signs to develop.
Our limited experience suggests that once myelopathy is established, the posterior vertebral column resection (VCR) with shortening and re-alignment is the safest and most effective surgical option for decompression and correction of deformity. However adequate support structures need to be in place including anaesthetic and spinal cord monitoring. A thorough informed consent process is also mandatory.
References
1. Macagno AE, O'Brien MF. Thoracic and thoracolumbar kyphosis in adults. Spine 2006;31:S161-70. [ Links ]
2. Winter RB, Moe JH, Wang JK. Congenital kyphosis: Its natural history and treatment as observed in a study of one hundred and thirty patients. J Bone Joint Surg 1973;55A:223-56. [ Links ]
3. McMaster MJ, Singh H. Natural history of congenital kyphosis and kyphoscoliosis: A study of one hundred and twelve patients.J Bone Joint Surg 1999;81A:1367-83. [ Links ]
4. Jain AK. Treatment of tuberculosis of the spine with neurologic complications. CORR 2002;398:75-84. [ Links ]
5. Jain AK. Tuberculosis of the spine. A review. CORR 2007;460:39-49. [ Links ]
6. Kawaham H, Tomita K. Influence of acute shortening on the spinal cord: an experimental study. Spine 2005;30:6130-20. [ Links ]
7. Suk S, Kim JH, Kim WJ, et al. Posterior vertebral column resection for severe spinal deformities. Spine 2002;27:2374-82. [ Links ]
8. Lenke LG, O'Leary PT, Bridwell KH et al. Posterior vertebral column resection for severe pediatric deformity. Spine 2009;34:2213-21. [ Links ]
9. Lonstein JE, Winter RB, Moe JH et al. Neurologic deficits secondary to spinal deformity. A review of the literature and report of 43 cases. Spine 1980;5:331-55. [ Links ]
10. Kawahara N, Tomita K, Baba H, et al. Closing-opening wedge osteotomy to correct angular kyphotic deformity by a single posterior approach. Spine 2001;26:391-401. [ Links ]
11. Shimode M, Kojima T, Sowa K. Spinal wedge osteotomy by a single posterior approach for correction of severe and rigid kyphosis or kyphoscoliosis. Spine 2002;27:2260-67. [ Links ]
12. Chang K, et al. Correction hinge in the compromised cord for severe and rigid angular kyphosis with neurologic deficits. Spine 2009;34(10):1040-45. [ Links ]
 Reprint requests:
Reprint requests:
Prof RN Dunn
Department of Orthopaedic Surgery. H49 OMB Groote Schuur Hospital
7925 Observatory Cape Town
Tel: (021) 4045387, Fax: 0866 715 294
Email: info@spinesurgery.co.za
Approval for the study was obtained from the Surgical Departmental Research Committee and the Ethics Committee. The content and preparation of this paper is the sole work of the authors. No benefit of any form wasor will be received from a commercial party relateddirectly or indirectly to the subject of this article.














