Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SA Orthopaedic Journal
On-line version ISSN 2309-8309
Print version ISSN 1681-150X
SA orthop. j. vol.9 n.4 Centurion Jan. 2010
CLINICAL ARTICLE
Occipito-cervical fusion: review of surgical indications, techniques and clinical outcomes
Simon BickI; Robert DunnII
IMBChB(UCT), Registrar
IIMBChB(UCT), MMed(Orth)UCT, FCS(SA)Orth, Associate Professor Spine Surgery Unit, Division of Orthopaedic Surgery, University of Cape Town
ABSTRACT
STUDY DESIGN: A retrospective review of patients undergoing single-surgeon occipito-cervical fusion.
OBJECTIVE: The aim of this study is to evaluate the surgical indications, techniques and clinical outcomes of occipito-cervical fusion, including C2 fixation methods and complications.
MATERIALS AND METHOD: Thirty-four consecutive patients (16 males, 18 females) who underwent occipito-cervical fusion were reviewed. The indications for fusion were instability due to inflammatory diseases (13), trauma (9), congenital abnormalities (9), infections (2) and tumours (1). Nine patients (all but 1 paediatric) underwent fusion with bone grafting and halo immobilisation. Twenty-five patients underwent posterior instrumented fusion. Halo removal was performed after 6 weeks and soft collars were worn for 6 weeks in the instrumented group. Surgical techniques and clinical outcomes (stability, fusion, complications) were reviewed.
RESULTS: Clinical and radiological fusion was attained in all patients available for follow-up, with an average of 2.7 months in the uninstrumented group and 5.2 months in the instrumented group. All fusions resulted in resolution of preoperative pain and an improvement in pre-operative neurology. Two patients demised in the acute postoperative period as a result of the underlying pathology. Eighteen patients required simultaneous decompressions. No instrumentation failures occurred. Superficial wound sepsis occurred in 4 patients, one subsequently requiring instrumentation removal.
CONCLUSION: Occipito-cervical fusion is a safe and reliable procedure, predictably providing stability and improvement in preoperative pain and neurology. Multiple cervical fixation options are available according to surgeon preference and anatomical variants.
Introduction
Occipito-cervical fusion (OCF) is required when the junction is rendered unstable by a variety of pathological conditions including congenital abnormalities, inflammatory diseases, trauma, tumours and infections.
Over the years there has been an evolution of surgical technique as implants have developed to accommodate the challenges of the occipito-cervical junction. These range from onlay bone graft techniques with halo jacket immobilisation to sophisticated instrumentation techniques. Early on, tenuous wire fixation methods were utilised. Prof Brookes Heywood published on the use of the T-plate (usually used in distal radius fractures) as a fixation option.1 His concept was not dissimilar to the modular occipital plates used today.
The unique anatomy and function of the region, the perceived high risk of vascular and neurological complications, and the anatomical variations make OCF a challenging procedure. Currently, the accepted method for OCF is rigid posterior internal fixation utilising segmental modular instrumentation.2-9
We undertook a retrospective review of our patients undergoing OCF assessing surgical indications, technique, clinical outcomes and complications.
Methods and materials
Thirty-four consecutive patients with occipito-cervical disorders undergoing OCF between December 2002 and February 2010 were identified. All procedures were performed by the senior author (RD) at Red Cross Children's Hospital, Groote Schuur Hospital and Constantiaberg Medi-Clinic.
The study was approved by the Research Ethics Committee of the Health Sciences Faculty, University of Cape Town, reference 188/2010.
There were two groups in this cohort, viz. a paediatric (< 16 years) and adult group. There were 12 patients in the paediatric group which included 5 females and 7 males. The average age was 8.6 years (1-16 ± 5.0 years). The adult group included 22 patients, 15 females and 7 males, with an average age of 52.9 years (25-79 ± 15.4).
The presenting complaints at the time of surgery were non-traumatic pain or instability, myelopathy and traumatic instability as shown in Table I. The paediatric myelopathy was due to Down's syndrome, Morquio's syndrome, congenital kyphosis and Conradi-Hunerman syndrome. In the adult group, the myelopathy was largely due to rheumatoid arthritis (5) and 1 tuberculosis. The myelopathies were generally mild and the patients were ambulant pre-operatively. Other paediatric indications included traumatic atlanto-occipital dissociation and chronic granulomatous osteitis.
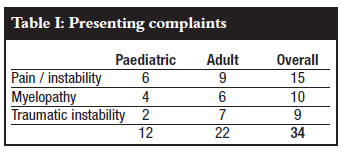
Rheumatoid arthritis (13) predominated as a cause in the adult group, followed by trauma (7), tumour (1) and tuberculosis (1).
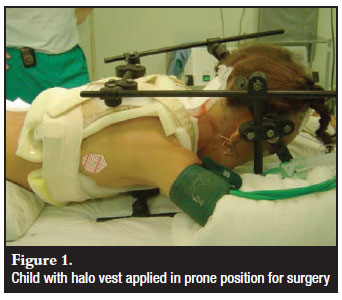
Nine patients (8 paediatric, 1 adult) underwent uninstrumented fusions with halo immobilisation (Figure 1). Twenty-five patients (21 adult, 4 paediatric) underwent instrumented fusions. The PCR / Summit system (DePuy®) was used in 11 patients the Axon (Synthes®) in 14 patients.
Eighteen patients required simultaneous spine decompression due to myelopathy or stenosis. The majority were a posterior C1 arch resection (14), sub-axial laminectomy (3) and 1 necessitating a trans-oral odontoidectomy.
Pre-operative X-rays including dynamic views, CT and MRIs were reviewed to determine stability, the extent of soft-tissue abnormalities (tumour and pannus), bony pathology and their influence on planned fixation types and level.
Patients underwent general anaesthetic induction while supine. The halo group then had the halo applied and halovest assembled. Any misalignment was reduced if possible and confirmed on lateral image. The patients were re-positioned prone on the anterior halo struts and ring. The surgical procedure was performed through the posterior halostruts, and bone graft was harvested from the posterior iliac crest after ensuring the vest allowed adequate access.
In the instrumented group, a Mayfield clamp was applied before re-positioning on a Relton-Hall frame. Fluoroscopy was used to confirm the desired neutral cervical position, the reduction of anatomical malalignment, and the placement of instrumentation. Posterior iliac crest bone graft was utilised in 27 patients. Allograft was used in seven paediatric and trauma patients.
The occipito-cervical area was exposed via a posterior midline approach with sub-periosteal exposure of the skull from the external occipital protuberance (EOP) to the required cervical level.
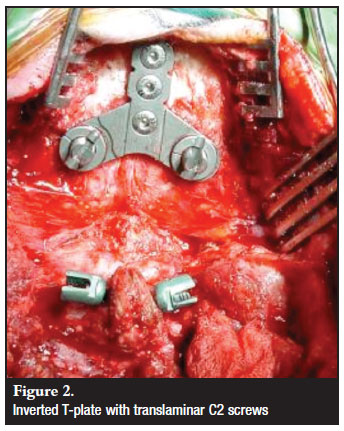
Occipital fixation was achieved with a T-plate fixed in the midline with 4.5 mm bicortical screws (Figure 2). The EOP was burred on the caudal side to facilitate plate placement, both flat against the skull and as cephalad as possible to allow fixation in the thickest bone. Careful drilling and tapping with the use of depth restriction guides was done. After an initial observation of subcutaneous plate-rod articulation prominence, subsequent plates were inverted. Skull plates were used in all patients except in a one-year-old patient where two paramedian plate/rods were applied due to anatomical constraints. Bicortical fixation was used.
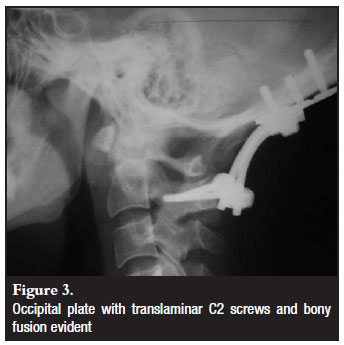
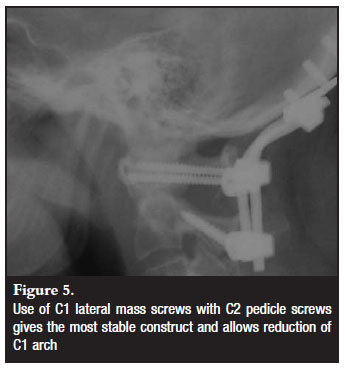
Different cervical fixation methods were employed as determined by the indication for fixation and anatomical variants (Figures 3-5). These included C1/2 transarticular (3), C2 pedicle (14) or translaminar screws (7) and sub-axial lateral mass screws. The default C2 screw was the pedicle screw with the translaminar screw as a bail-out if the pedicle was not possible due to anatomical or technical limitations. All but one construct bypassed C1. In this patient C1 lateral mass screws were utilised. Seven patients were fixed to below C2 level. The fixation option was decided upon intraoperatively according to screw hold and surgeon satisfaction.

The average surgical time in the uninstrumented group was 83 minutes (40-195) and 137 minutes (85-275) in the instrumented group. The average blood loss was 142 ml (50-300) in the uninstrumented group and 513 ml (100-3 300). The 3 300 ml blood loss was due to the vertebral artery injury.
Halo jackets were worn for a minimum of 6 weeks, and removed as soon as possible thereafter as the children required a general anaesthetic for removal. In the instrumented group, a soft cervical collar was worn for 6 weeks postoperatively. Follow-up visits were arranged for 6 weeks, 3 months, 6 months, 1 year and annually thereafter, with radiographic follow-up at each visit.
Fusion was assessed on lateral radiographs as cross-trabeculation of bone mass, absence of peri-screw lucency and absence of instrumentation failure.
Results
Twenty-eight patients were available to follow-up with an average follow-up of 9.9 months (6–48). Four patients failed to return and two demised peri-operatively.
Successful clinical and radiological fusion was obtained in all 28 patients available for follow-up. In the uninstrumented group, all 8 patients fused at an average time of 2.7 months (1.5–4 months). In the instrumented group, 20 patients fused at an average time of 5.2 months (3–12 months).
All patients with pre-operative radicular pain had resolution of their symptoms. Those with myelopathy had improvement to normal or near normal except for 2 infants in the paediatric group who remained myelopathic at follow-up.
The two patients who demised in the acute period postoperatively included an adult who had suffered traumatic atlanto-occipital dissociation and quadriparesis. She succumbed to respiratory complications in ICU. The second patient was a child with Trisomy 21. She required the transoral decompression and suffered a gastric stress ulcer with perforation in ICU post-surgery.
There were no instrumentation failures or revisions required. Two patients had minimal occipital plate lift-off (1–2 mm) on the postoperative films. This did not progress and both went on to successful fusion. One patient had an intra-operative cerebrospinal fluid leak from the occipital drill which stopped on screw insertion. There were no subsequent problems.
Four patients suffered from superficial postoperative wound infections. Two required oral antibiotics and dressings. The other two required washouts in the early postoperative period and settled on oral antibiotics. One subsequently required instrumentation removal at 2 years postop due to recurrence of infection. Once removed, the infection settled and she continued to have pre-operative symptom resolution. Of these two patients requiring wound washouts, one was a rheumatoid arthritis patient using methotrexate at the time of surgery and the other the Down's child. One patient, with the atlanto-occipital dissociation had an intra-operative unilateral vertebral artery violation during C1 screw placement. This settled with local measures. One patient suffered from a postoperative deep vein thrombosis.
Discussion
OCF has progressed a long way since 1927 when Foerster first described the technique using a fibular graft in a patient who had sustained a dens fracture.10 Prior to this, such pathology was viewed as inoperable and a terminal event. In 1928, Juvara and Dimitriu used tibial grafts, and in 1935, Khan and Yglesias reported the first case using iliac crest grafting to stabilise an atlanto-axial dislocation.11-12
For years onlay bone grafting was used with postoperative immobilisation in either a halo immobiliser or SOMI brace. Good results and fusion rates (up to 89%) have been shown using this method, but with prolonged external immobilisation and often initial skeletal traction.13-16
Wire fixation was used to secure the bone graft and assist in stability while awaiting bony fusion.17-23 Brattstrom and Granholm added methylmethacrylate to the fusion mass to increase stability, obviating halo immobilisation postoperatively.24 This technique was adopted by several surgeons, but has largely been abandoned due to high complication rates.21,25-28 Zygmunt published a long-term result on 163 patients with rheumatoid arthritis and OCF with this technique, 24 requiring reoperation, and 16 suffering from wound infection.29 A study by Grob et al showed a 27% non-union rate, unacceptably high.27
Development of wire-based systems continued, with Hartshill-Ransford loops, Luque rods, rectangular Luque- Hartshill systems and hook-claw systems used with wire fixation with good results.30-41 Malleable 5 mm rods (upside-down 'U'-shape), occipital titanium loops and threaded Steinman pins secured to the occiput with wire loops, and the cervical spinous processes or laminae have also shown good results.42-45
However C2 sublaminar wiring or spine-graft block wiring has been associated with cervical redislocation and neurological deterioration after tightening.22,28,43 Wire and cable systems also have the tendency to abrade through bone, affecting stability.46
Concerns over fusion rates and complications with earlier techniques47-53 led Heywood to try plate and screw fixation, with additional wire fixation reserved for long segment fusions only.1 Limited by the technology of the time, he used a distal radius T-shaped plate from the standard 'small fragment trauma set' with routine bone grafting and halo immobilisation. In addition, he studied cadavers and identified that the occiput was thickest in the midline. Twelve out of 14 patients went on to satisfactory fusion, with one failure in a rheumatoid arthritis patient.
Several studies on rigid plate fixation followed.27,54-57 The main successes with this procedure were improved fusion rates and the end of the absolute need for prolonged postoperative immobilisation. This can partly be attributed to the C1-2 transarticular screw, developed by Magerl, and repeatedly shown to be superior to wire-based fixation systems.55-57 Gluf et al, in a review of 353 C1-2 transarticular screw insertions for atlantoaxial instability, noted vertebral artery injury in six screws (1.7%), five malpositioned screws, yet an overall fusion rate of 98%.58
Modular screw-and-rod systems were developed on the success of rigid plate fixation.59-61 Abumi published a series using cervical pedicle screws in a screw-rod construct, with fusion obtained in 24 of 26 patients, significant malalignment correction, and no screw insertion complications.62 Vale described a rigid posterior OCF system, improving the occipital fixation points towards the thicker skull midline under the external occipital protuberance.63
Onlay bone grafting and halo immobilisation has been shown to be an effective procedure for OCF in children and adolescents with upper cervical instability.64-66 This is due to rapid fusion rates, but many adults find prolonged halo restriction unacceptable.
As 50 per cent of the total range of motion of the neck occurs through occiput-C2 level, stable fixation remains key to successful fusion in OCF procedures.67,68 Complete screw systems have been proven biomechanically over wire fixation methods.69-70 C1 wire fixation to the construct significantly increased the construct's stability. Pedicle screws have an advantage over transarticular screws in that the laminae are not required for fixation, hence can be used after posterior decompression.62
Occipital fixation has evolved from traditional wire fixation to screw fixation.27,69 Central screw positioning, just below the superior nuchal line, has been shown to be the thickest and safest region.1,71-73 Bicortical screws have 50% greater pullout strength than unicortical screws or occipital wires, with pullout strength directly related to the skull bone thickness, and no significant difference between cortical and cancellous screw types.74-75 Lateral and midline occipital bicortical screw fixation, using modern rodscrew systems, show similar biomechanical properties.76 Inside-out techniques for occipital screw fixation show biomechanical but not clinical superiority over traditional outside-in methods.71,77-79
More recently C1 lateral mass-C2 pedicle screw combinations have been found to be superior to the C1-2 transarticular method.80-81 Translaminar screws have been reported to be a safe alternative.82 They reduce risk to the vertebral arteries and are recommended for patients with high riding vertebral arteries or small pedicles.
Conclusions
OCF, whether rigid instrumented fusion or onlay bone grating and halo immobilisation, remains a successful, safe and reliable procedure for the stabilisation of the OC junction for a variety of indications. It achieves stability and a marked improvement in pre-operative pain and neurology.
The on-lay fusion with halo immobilisation remains a good option in the paediatric group who experience rapid fusion and tolerate the halo well. In adults the advantage of rigid fixation obviating prolonged immobilisation makes it a valuable option. Modular instrumentation allows individualisation of fixation techniques to minimise risk and maximise stability, based on patient-specific anatomy. C2 screws provide excellent cervical purchase, but the surgeon should be capable of multiple techniques as this option is not possible in all individuals.
References
1. Heywood AWB, Learmonth ID, Thomas M. Internal fixation for occipitocervical fusion. J Bone Joint Surg [Br] 1988; 70: 708-11. [ Links ]
2. Paquis P, Breuil V, Lonjon M, et al. Occipitocervical fixation using hooks and screws for upper cervical instability. Neurosurgery 1999; 44: 324-31. [ Links ]
3. Grob D, Dvorak J, Panjabi M, et al. Posterior occipitocervical fusion. A preliminary report of a new technique. Spine 1991; 16(Suppl 3): S17-S24. [ Links ]
4. Sasso RC, Jeanneret B, Fischer K, et al. Occipitocervical fusion with posterior plate and screw instrumentation: A longterm follow-up study. Spine 1994; 19: 2364-68. [ Links ]
5. Lieberman IH, Webb JK. Occipito-cervical fusion using posterior titanium plates. Eur Spine 1998; 7(4): 308-12. [ Links ]
6. Jeanneret B. Posterior rod system of the cervical spine: a new implant allowing optimal screw insertion. Eur Spine J 1996; 5: 350-56. [ Links ]
7. Abumi K, Takada T, Shono Y, et al. Posterior occipitocervical reconstruction using cervical pedicle screws and plate-rod systems. Spine 1999; 24: 1425-34. [ Links ]
8. Vale FL, Oliver M, Cahill DW. Rigid occipitocervical fusion. J Neurosurg (Spine 2) 1999; 91: 144-50. [ Links ]
9. Nockels RP, Shaffrey CI, Kanter AS, Azeem S, York JE. Occipitocervical fusion with rigid internal fixation: long-term follow-up data in 69 patients. J Neurosurg Spine 2007; 7: 117-23. [ Links ]
10. Foerster O. Die Leitungsbahnen des Schmerzgefühls und die chirurgische Behandlung der Schmerzzustände. Berlin: Urban & Schwarzenburg, 1927. [ Links ]
11. Juvara E, Dimitriu V. Luxation atloido-axoidienne avec phenomes compressifs, Operation secondaire. Lyon Chirurgical 1928; 25: 668. [ Links ]
12. Kahn EA, Yglesias L. Progressive atlanto-axial dislocation. Journal of the American Medical Association 1935; 105: 348. [ Links ]
13. Perry J, Nickel VL. Total cervical-spine fusion for neck paralysis. JBJS [Am] 1959; 41-A: 37. [ Links ]
14. Thompson H. The 'halo' traction apparatus. J Bone Joint Surg 1962; 44-B: 655-61. [ Links ]
15. Newman P, Sweetnam R. Occipito-cervical fusion: an operative technique and its indications. J Bone Joint Surg [Br] 1969; 51-B, 423-31. [ Links ]
16. Elia M, Mazzara JT, Fielding JW. Onlay technique for occipitocervical fusion. Clin Orthop 1992; 280: 170-74. [ Links ]
17. Hamblen DL. Occipito-cervical fusion: Indications, technique and results. J Bone Joint Surg Br 1967; 49: 33-45. [ Links ]
18. Grantham SA, Dick HM, Thompson RC, Stinchfield FE. Occipitocervical arthrodesis: Indications, technique and results. Clinical Orthop 1969; 65: 118-29. [ Links ]
19. Wertheim SB, Bohlman HH. Occipitocervical fusion: Indications, technique and long-term results in thirteen patients. J Bone Joint Surg [Am] 1987; 69: 833-36. [ Links ]
20. McAfee PC, Cassidy JR, Davis RF, North RB, Ducker TB. Fusion of the occiput to the upper cervical spine. A review of 37 cases. Spine 1991; 16: S490-4. [ Links ]
21. Zygmunt S, Saveland H, Brattstrom H, et al. Reduction of rheumatoid periodontoid pannus following posterior occipitocervical fusion visualised by magnetic resonance imaging. Br J Neurosurg 1988; 2: 315-20. [ Links ]
22. Jain VK, Takayasu M, Singh S, et al. Occipital-axis posterior wiring and fusion for atlantoaxial dislocation associated with occipitalization of the atlas. Technical note. J Neurosurg 1993; 79: 142-44. [ Links ]
23. Jain VK, Mittal P, Banerji D, et al. Posterior occipitoaxial fusion for atlantoaxial dislocation associated with occipitalised atlas. J Neurosurg 1996; 84: 559-64. [ Links ]
24. Brattstrom H, Granholm L. Atlanto-axial fusion in rheumatoid arthritis: a new method of fixation with wire and bone cement. Acta Orthop Scand 1976; 47: 619-28. [ Links ]
25. Clark CR, Keggi KJ, Panjabi MM. Methylmethacrylate stabilization of the cervical spine. J Bone Joint Surg 1984; 66A: 40-66. [ Links ]
26. Stambough JL, Balderston RA, Grey S. Technique for occipito-cervical fusion in osteopenic patients. J Spinal Disord 1990; Dec. 3(4): 404-47 . [ Links ]
27. Grob D, Dvorak J, Panjabi MM, et al. The role of plate and screw fixation in occipitocervical fusion in rheumatoid arthritis. Spine 1994; 19: 2545-51. [ Links ]
28. McAfee PC, Bohlman HH, Ducker T, et al. Failure of stabilization of the spine with methylmethacrylate: A retrospective analysis of twenty-four cases. J Bone Joint Surg 1986; 68A: 1145-57. [ Links ]
29. Zygmunt SC, Christensson D, Saveland H, Rydholm U, Alund M. Occipitocervical fixation in rheumatoid arthritis: An analysis of surgical risk factors in 163 patients. Acta Neurochir Wien 1995; 135: 24-31. [ Links ]
30. Malcolm GP, Ransford AO, Crockard HA. Treatment of nonrheumatoid occipitocervical instability. Internal fixation with the Hartshill-Ransford loop. J Bone Joint Surg Br 1994; 76: 357-66. [ Links ]
31. Ransford AO, Crockard HA, Pozo JL, et al. Craniocervical instability treated by contoured loop fixation. J Bone Joint Surg Br 1986; 68: 173-77. [ Links ]
32. Luque ER. The anatomic basis and development of segmental spinal instrumentation. Spine 1982; 7: 270-75. [ Links ]
33. Nakano N, Nakano T. Upper cervical fusion with SSI. Preliminary report. Thirteenth Annual Meeting of the Cervical Spine Research Society, Cambridge, Massachusetts, Dec. 6, 1985. [ Links ]
34. Itoh T, Tsuji H, Katoh Y, Yonezawa T, Kitagawa H. Segmental spinal instrumentation for rheumatoid diseases. Spine 1988; 13: 1234-38. [ Links ]
35. Vanden Berghe A, Ackerman C, Veys E, et al. Occipito-cervical fusion in rheumatoid arthritis. Acta Orthop Belg 1991; 57(Suppl 1): 94-98. [ Links ]
36.. Ellis PM, Findlay JM. Craniocervical fusion with contoured Luque rod and autogeneic bone graft. Can J Surg 1994; 37: 50-54. [ Links ]
37. MacKenzie AI, Uttley D, Marsh HT, et al. Craniocervical stabilization using Luque/Hartshill rectangles. Neurosurgery 1990; 26: 32-36 [ Links ]
38. Sakou T, Kawaida H, Morizono Y, et al. Occipitoatlantoaxial fusion utilizing a rectangular rod. Clin Orthop 1989; 239: 136-44. [ Links ]
39. Zimmermann M, Wolff R, Raabe A, Stolke D, Seifert V. Palliative occipito-cervical stabilization in patients with malignant tumours of the occipito-cervical junction and the upper cervical spine. Acta Neurochir Wien 2002; 144: 783-90. [ Links ]
40.. Faure A, Monteiro R, Hamel O, et al. Inverted-hook occipital clamp system in occipitocervical fixation. Technical note. J Neurosurg (Spine 1) 2002; 97: 135-.41 [ Links ]
41. Paquis P, Breuil V, Lonjon M, et al. Occipitocervical fixation using hooks and screws for upper cervical instability. Neurosurgery 1999; 44: 324-31. [ Links ]
42. Fehlings MG, Errico T, Cooper P, et al. Occipitocervical fusion with a five-millimeter malleable rod and segmental fixation. Neurosurgery 1993; 32: 198-208. [ Links ]
43. Sonntag VKH, Dickman CA. Craniocervical stabilization. Clin Neurosurg 1993; 40: 243-72. [ Links ]
44. Jackson RJ, Gokaslan ZL. Occipitocervicothoracic fixation for spinal instability in patients with neoplastic processes. J Neurosurg 1999; 91(Suppl): 81-89. [ Links ]
45. Singh SK, Rickards L, Apfelbaum RI, et al. Occipitocervical reconstruction with the Ohio Medical Instruments Loop: results of a multicenter evaluation in 30 cases. J Neurosurg (Spine 3) 2003; 98: 239-46. [ Links ]
46. Dickman CA, Papadopoulos SM, Crawford NR, et al. Comparative mechanical properties of spinal cable and wire fixation systems. Spine 1997; 22: 596-604. [ Links ]
47. Roy-Camille R, Saillant G, Mazel C. Treatment of cervical spine injuries by posterior osteosynthesis with plates and screws. In: Kehr P, Weidner A, eds. Cervical Spine 1987; Vol 1. New York: Springer-Verlag. [ Links ]
48. Roy-Camille R, Saillant G, Mazel C. Internal fixation of the unstable cervical spine by posterior osteosynthesis with plates and screws. In: Sherk H, ed. The Cervical Spine. 2nd ed. 1989: 390-403. Philadelphia: JB Lippincott . [ Links ]
49. Roy-Camille R, Gagna G, Lazennec JY. L'Arthrodese Occipito-Cervicale. In Roy-Camille R (ed). Cinqiemes Journees d'Orthopedie de la Pitie 1986. Rachis Cervical Superieur. Paris, Masson 49-51. [ Links ]
50. Smith MD, Anderson P, Grady MS. Occipitocervical arthrodesis using contoured plate fixation. Spine 1993; 18: 1984-90. [ Links ]
51. Grob D, Dvorak J, Panjabi M, et al. Posterior occipitocervical fusion. A preliminary report of a new technique. Spine 1991; 16(Suppl 3): S17-S24. [ Links ]
52. Sasso RC, Jeanneret B, Fischer K, et al. Occipitocervical fusion with posterior plate and screw instrumentation: A longterm follow-up study. Spine 1994; 19: 2364-68. [ Links ]
53. Lieberman IH, Webb JK. Occipito-cervical fusion using posterior titanium plates. Eur Spine 1998; 7(4): 308-12. [ Links ]
54. Magerl F, Seemann P. Stable posterior fusion of the atlas and axis by transarticular screw fixation. In: Kehr P, Weidner A, editors. Cervical Spine. Strassbourg; Springer-Verlag; 1986: 322-27. [ Links ]
55. Magerl F, Grob D, Seemann D. Stable dorsal fusion of the cervical spine (C2-TH1) using hook plates. In: Kehr P, Weidner A, editors. Cervical Spine I: Strasbourg 1985. New York: Springer-Verlag, 1987, pp 217-21. [ Links ]
56. Dickman CA, Sonntag VKH. Posterior C1-C2 transarticular screw fixation for atlantoaxial arthrodesis. Neurosurgery 1998; 43: 275-81. [ Links ]
57. Farey ID, Nadkarni S, Smith N. Modified Gallie technique versus transarticular screw fixation in C1-C2 fusion. Clin Orthop 1999; 359: 126-35. [ Links ]
58. Gluf WM, Schmidt MH, Apfelbaum RI. Atlantoaxial transarticular screw fixation: a review of surgical indications, fusion rate, complications, and lessons learned in 191 adult patients. J Neurosurg Spine 2005; 2: 155-63. [ Links ]
59. Jeanneret B. Posterior rod system of the cervical spine: a new implant allowing optimal screw insertion. Eur Spine J 1996; 5: 350-56. [ Links ]
60. Heidecke V, Rainov NG, Burkett W. Occipitocervical fusion with the cervical Cotrel-Dubousset rod system. Acta Neurochir 1998; 140: 969-76. [ Links ]
61. Korovessis P, Katonis P, Aligizakis A, Christoforakis J, Baikousis A, Papazisis Z, Petsinis G. Posterior compact Cotrel-Dubousset instrumentation for occipitocervical, cervical and cervicothoracic fusion. Eur Spine J 2001; 10: 385-94. [ Links ]
62. Abumi K, Takada T, Shono Y, et al. Posterior occipitocervical reconstruction using cervical pedicle screws and plate-rod systems. Spine 1999; 24: 1425-34. [ Links ]
63. Vale FL, Oliver M, Cahill DW. Rigid occipitocervical fusion. J Neurosurg (Spine 2) 1999; 91: 144-50. [ Links ]
64. Roy L, Gibson D. Cervical spine fusions in children. Clin Orthop Rel Res 1970; 73: 146-51. [ Links ]
65. Koop SE, Winter PRB, Lonstein JE. The surgical treatment of instability of the upper part of the cervical spine in children and adolescents. J Bone Joint Surg Am 1984; 66: 403-11. [ Links ]
66. Letts M, Slutsky D. Occipitocervical arthrodesis in children. J Bone Joint Surg Am 1990; 72: 1166-70. [ Links ]
67. White AA III, Panjabi MM. The clinical biomechanics of the occipitoatlantoaxial complex. Orthop Clin North Am 1978; 9: 867-78. [ Links ]
68. White III AA, Panjabi MM. Clinical biomechanics of the spine. Second edition. Philadelphia: JB Lippincott, 1990: 95-97. [ Links ]
69. Hurlbert RJ, Crawford NR, Choi WG, et al. A biomechanical evaluation of occipitocervical instrumentation: screw compared with wire fixation. J Neurosurg (Spine 1) 1999; 90:84-90. [ Links ]
70. Oda I, Abumi K, Sell LC, et al. Biomechanical evaluation of five different occipito-atlanto-axial fixation techniques. Spine 1999; 24: 2377-82. [ Links ]
71. Lee SC, Chen JF, Lee ST. Complications of fixation to the occipit - anatomical and design implications. British Journal of Neurosurgery 2004; 18(6): 590-97. [ Links ]
72. Ebraheim NA, Lu J, Biyani A, et al. An anatomic study of the thickness of the occipital bone. Implications for occipitocervical instrumentation. Spine 1996; 21: 1725-30. [ Links ]
73. Zipnick RI, Merola AA, Gorup J, et al. Occipital morphology: An anatomic guide to internal fixation. Spine 1996; 21: 1719-24. [ Links ]
74. Haher TR, Yeung AW, Caruso SA, et al. Occipital screw pullout strength: A biomechanical investigation of occipital morphology. Spine 1997; 24: 5-9. [ Links ]
75. Roberts DA, Doherty BJ, Heggeness MH. Quantitative anatomy of the occiput and the biomechanics of occipital screw fixation. Spine 1998; 23: 1100-7. [ Links ]
76. Anderson PA, Oza AL, Puschak TJ, Sasso R. Biomechanics of occipitocervical fixation. Spine 2006; 31: 755-61. [ Links ]
77. Pait TG, Al-Mefty O, Boop FA, et al. Inside-outside technique for posterior occipitocervical spine instrumentation and stabilization: preliminary results. J Neurosurg (Spine 1) 1999; 90: 1-7. [ Links ]
78. Sandhu FA, Pait TG, Benzel E, Henderson FC. Occipitocervical fusion for rheumatoid arthritis using the inside-outside stabilization technique. Spine 2003; 28: 414-19. [ Links ]
79. Caglar YS, Pait TG, Hogue W, Bozkurt M, Ozgen S. Biomechanical comparison of inside-outside screws, cables, and regular screws, using a sawbones model. Neurosurg Rev 2005; 28: 52-58. [ Links ]
80. Harms J, Melcher RP. Posterior C1-C2 fusion with polyaxial screw and rod fixation. Spine 2001; 26: 2467-71. [ Links ]
81. Finn MA, Fassett DR, Mccall TD, Clark R, Dailey AT, Brodke DS. The cervical end of an occipitocervical fusion: a biomechanical evaluation of 3 constructs. J Neurosurg Spine 2008; 9: 296-300. [ Links ]
82. Reddy C, Aditya VI, Channon S, Lim T-H, Torner J, Hitchon P. In vitro biomechanical comparison of transpedicular versus translaminar C-2 screw fixation in C2-3 instrumentation. J Neurosurg Spine 2007; 7: 414-18. [ Links ]
 Reprint requests:
Reprint requests:
Prof RN Dunn
info@spinesurgery.co.za
No benefits were received by any of the authors from a commercial party related directly or indirectly to the subject of this article.














