Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SA Orthopaedic Journal
On-line version ISSN 2309-8309
Print version ISSN 1681-150X
SA orthop. j. vol.9 n.3 Centurion Jan. 2010
CLINICAL ARTICLE
Is tetanus toxoid still necessary today?
BE RamasuvhaI; EJ D'AltonII; R GoleleIII
IMBChB(Natal). Registrar, Department of Orthopaedics, Dr George Mukhari Hospital, Pretoria
IIMBChB(Pret), MMed(Orth). Consultant, Dr George Mukhari Hospital, Pretoria
IIIMBChB(Natal), MFGP(SA), MMed(Orth), FCS(Orth)(SA). Professor and Head of Department Orthopaedics Surgery, University of Limpopo (Medunsa Campus)
Introduction
Infection is a common complication of open fractures. Among these infections is tetanus which is caused by Clostridium tetani. Although there is a decrease of tetanus infection worldwide due to immunisation programmes and the use of anti-tetanus toxoid, it is important for health workers to recognise the symptoms and signs of this disease and start treatment as soon as possible because of the high morbidity and mortality rate.
Two cases from our institution are documented; they presented with an open fracture of the tibia and an open fracture of the ankle, and complicated by developing tetanus.
Case 1
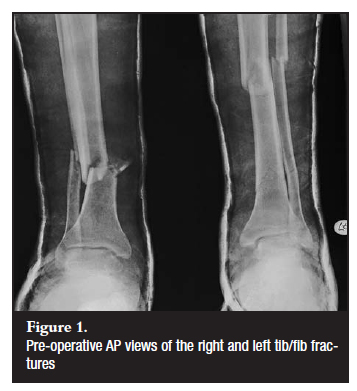
Patient A is a 24-year-old immigrant from Zimbabwe who presented to our emergency room following a pedestrian motor vehicle accident. He sustained a grade I open right tibia/fibula fracture, closed left tibia/fibula fracture and scalp lacerations (Figure 1). The neurovascular status of both limbs was intact and the Glasgow coma scale (GCS) was 15/15. There was no evidence that the patient had been given anti-tetanus toxoid in the emergency room.
On day 1 post admission the GCS dropped to 10/15. Blood pressure was 140/100; the patient also had tachycardia and was tachypnoeic. Blood investigations showed a neutrophilia of 85.3%; other parameters were normal. An emergency CAT scan of the brain was ordered and the results were within normal limits. Later on the same day the patient developed a fever of 38ºC.
Following this the patient developed seizures and was started on phenytoin. Consequently the diagnosis of tetanus was made and the patient was admitted to the Intensive Care Unit (ICU). At this stage the patient had generalised convulsions, opisthotonus, spasms and pyrexia.
In ICU the patient was treated in an isolated, dark room. He was intubated, ventilated and started on penicillin G and metronidazole. Both tib/fib fractures were immobilised in an above-knee Plaster of Paris after minimal debridement in ICU. Tetanus immunoglobulin was also administered together with anticonvulsants.
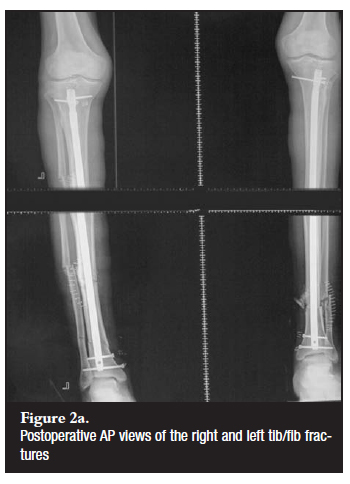
The patient was discharged from ICU on day 18 with a GCS of 5/10. He was on a tracheostomy-mask but had no convulsions or tetanospasms. Feeding was through a nasogastric tube. His condition subsequently improved and the tracheostomy was closed. He underwent both physiotherapy and occupational therapy. Two weeks later the patient was taken to the operating theatre for a delayed intramedullary nailing of both tibias (Figures 2a and b).

The patient has since been discharged and is mobilising without crutches.
Case 2
Patient B is a 40-year-old patient who was admitted with an open fracture/dislocation of the right ankle after falling into a ditch while running. He presented with a wound over the medial malleolus through which the tibia was protruding (Figure 3). Wash-out and a closed reduction were immediately done in the emergency room and the fracture was immobilised in a plaster cast.

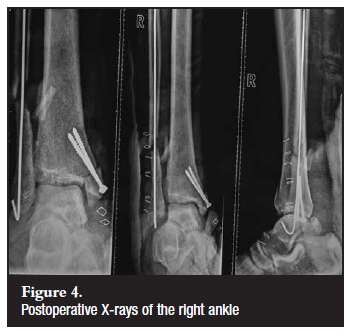
After admission the patient refused surgery and signed a refusal of hospital treatment form. Later that day he changed his mind and was prepared for theatre. In this case there was also no evidence that anti-tetanus toxoid had been administered in the emergency room. The patient was taken to theatre for a wound debridement and fixation of the fractured ankle with a K-wire on the lateral malleolus and two medial malleolar screws. The intra-operative finding was a grade 3b open fracture/dislocation of the ankle with a 10 cm wound over the medial malleolus (Figure 4).
On day 5 postoperatively, the patient developed tonicclonic seizures. There was no previous history of epilepsy. The patient was apyrexial. He had spasms, tenesmus and opisthotonus. The spatula test was positive. Blood investigations showed a neutrophilia of 84.6% with other parameters being normal. The patient was subsequently admitted to the ICU and started on tetanus immunoglobulin, anticonvulsants (phenytoin), metronidazole, penicillin G, as well as supportive measures.
He demised 10 days later in ICU.
Discussion
Tetanus is a devastating disease involving muscle spasm and autonomic instability. It is caused by the exotoxin (tetanospasmin) of Clostridium tetani, which is a Grampositive, anaerobic, spore-forming bacillus. This bacterium needs low oxygen tension for its survival and it is very sensitive to heat. It thrives in a hot, damp climate and soil rich in organic material.1 The spores are resistant to commonly used antiseptics.
The occurrence of tetanus is worldwide, with approximately 800 000 to 1 million deaths a year. Depending on the type of tetanus the mortality rate worldwide is 45%.1 Most cases and deaths occur in sub-Saharan Africa.2 It is more prevalent in males above the age of 40 when childhood immunity has deteriorated. Both our patients were males and they were 24 and 40 years old. The patients were also from densely populated areas and their immunisation status was unknown. Transmission of infection is through contaminated wounds which may be apparent or occult. In 25% of cases the portal of entry is not evident at the time of diagnosis because the spores are able to survive in the body for years and produce disease at a later time when conditions allow.3 Wounds that are more than 6 hours old, stellate, greater than 1 cm in depth, have devitalised tissue, are secondary to missile injury, crush injury, or burn, or those that are heavily contaminated with soil or faeces, are at particularly high risk for colonisation by contracting Clostridium tetani.4 The bacterium produces two toxins, tetanolysin and tetanospasmin. The latter is the one responsible for the disease. Tetanolysin is thought to damage the tissues around the wound in order to facilitate multiplication of the bacteria. Tetanospasmin is a powerful neurotoxin which is transmitted to the central nervous system via neural pathways by retrograde axonal spread.5 It becomes bound to the gangliosides in the central nervous system where it suppresses inhibitory influences on motor and autonomic neurons. It inhibits the release of glycine and gammaamino butyric acid (GABA).1,2,3,6 The inhibition of motor neurons leads to unopposed muscle contractions, spasms and seizures.
The incubation period of tetanus varies between 3 and 21 days with a mean of 7 days.3 The further the site of inoculation from the central nervous system, the longer the incubation period; and the shorter the incubation period the deadlier the disease.1 Patient A had scalp lacerations as well which were most probably the sites of inoculation; thus he had a shorter incubation period, with symptoms developing on the second day of admission.
There are four clinical types of tetanus, namely generalised, which is the most common type (more than 80%); local; cephalic; and neonatal.6 The generalised type is further divided into mild (grade I), moderate (grade II), severe (grade III), and more severe (grade IV) depending on the severity of symptoms, as well as the presence of autonomic disturbances.
Our patients fell within grade II and III which are moderate to severe types because they had respiratory embarrassment that needed ventilation. Local tetanus presents as rigidity of muscles around the site of inoculation and has a low mortality rate of 1%. It can also present as the first signs of generalised tetanus. Cephalic tetanus involves the cranial nerves and usually presents as cranial nerve palsies and muscle spasms, usually affecting the 7th, 6th, 3rd, 4th, and 12th cranial nerves in decreasing order of frequency. Aetiology is most commonly thought to be head injury and otitis media.6 Neonatal tetanus is generalised tetanus in newborns that occurs around the first week of life. It occurs particularly in developing countries where children are often born at home.
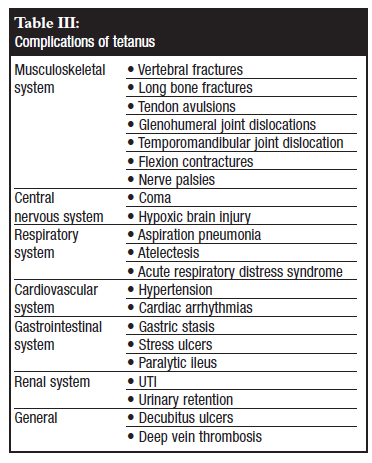
Generalised tetanus usually presents with a descending pattern where the first sign is the locked jaw or trismus due to spasm of the masseter muscle. Risus sardonicus, which is the 'ironical smile of tetanus' can occur due to spasms of the facial muscles, followed by difficulty in swallowing, nuchal rigidity, and opisthotonus (spasm of the extensors of the neck, back, and legs forming a backward curvature). Lastly, the patient can also present with rigidity of abdominal muscles. Severe muscle spasms can result in vertebral fractures, long bone fractures, and tendon avulsions.6
Due to the autonomic nervous system involvement, the patient may present with pyrexia, which occurred with our first patient. Hypertension, tachycardia and sweating may also occur. Spasms may occur frequently and are triggered by external stimuli such as noise and light and continue for a period of 3 to 4 weeks with full recovery taking several months to occur. This is the time it takes for the new neurons to grow. Mortality in the acute phase of the disease is due to diaphragmatic paralysis and laryngeal or glottic spasms. These patients are managed by intubation and mechanical ventilation.
There are no laboratory tests to diagnose tetanus.1-8 Occasionally the white cell count is raised as in both our patients, but this is not diagnostic. Gram stains and cultures are positive in less than a third of the patients. The diagnosis is made clinically. Prior immunisation does not rule out the disease.7,8
A bedside 'spatula test' can aid in making a diagnosis early in the disease. A spatula is inserted into the patient's pharynx. The test is positive if the patient bites the spatula because of reflex masseter spasm. It is negative if the patient gags and tries to expel the spatula.6 While there is no documentation on whether the spatula test was carried out on Patient A, Patient B had a positive spatula test (explained below).
The goals of tetanus control are primarily (i) to eliminate maternal and neonatal tetanus globally; and (ii) to achieve and sustain high coverage of three doses of DTP (diphtheria, tetanus and pertussis) and of appropriate booster doses in order to prevent tetanus in all age groups. A childhood tetanus immunisation schedule of five doses is recommended. The primary series of three doses of DTP3 (DTwP OR DTaP) should be given in infancy (age <1 year), with a booster dose of a tetanus toxoid-containing vaccine ideally at age 4-7 years and another booster in adolescence, e.g. at age 12-15 years. The exact timing of the booster doses should be flexible to take account of the most appropriate health service contacts in different countries. When a high percentage of children, including girls, attend school, school-based immunisation programmes should be used where feasible to deliver the booster doses. Special efforts to reach school non-attendees will be needed.2,9,10 It is recommended that one midlife booster dose be given to adults at the age of 50 to 65 years.11 Adverse effects of tetanus toxoid include local oedema/erythema, regional adenopathy, fever, malaise, headache, anaphylaxis, brachial plexus, and other peripheral neuropathy, and Guillain-Barré syndrome.4,12
A multidisciplinary team approach is mandatory when managing a patient with acute tetanus. Treatment involves neutralising tetanospasmin, removing the source of the toxin, and providing supportive care for muscle spasms, respiration and autonomic instability. Early airway management with intubation and ventilatory support has been shown to decrease the mortality rate significantly. It is therefore important for the patient to be treated in ICU isolated in a quiet and dark room.
Human tetanus immunoglobulin (TIG) is given to neutralise the circulating tetanospasmin. The recommended dose of TIG is 500-6 000 units intramuscularly. TIG helps remove the unbound toxin but cannot remove the toxin that is bound to the nerve endings.1,6,8 Intrathecal administration of TIG is theoretically attractive because one hopes to bind the toxin as it crosses from postsynaptic to presynaptic neuron, but there are not enough studies to prove the efficacy of this. Because TIG does not offer immunity to tetanus, anti-tetanus toxoid must also be given at the contralateral side of the TIG dosing. Giving tetanus toxoid in the emergency department after a wound does more for tetanus immunity for the next injury rather than the current injury.2 Tetanus vaccines are produced by formaldehyde inactivation of tetanus toxin to produce the toxoid.9,13 It is therefore safe to use tetanus toxoid in immunocompromised patients.
Antibiotics are used to prevent the on-going production of toxins by eliminating Clostridium tetani. Metronidazole is the drug of choice because it penetrates the tissues better and the mortality rates are lower than when using penicillin alone, which was the former drug of choice.4,12,14 Penicillin also has GABA-antagonist properties which may potentiate the effects of tetanospasmin. Third-generation cephalosporins, tetracycline and erythromycin are used in patients with sensitivity to penicillin3,15 (Tables I and II).


Wound debridement of necrotic and devitalised tissues must also be performed together with removal of foreign bodies and contaminants, repeated after 48 hours and again if necessary until the wound is clean.
During debridement, 2 cm margins around the wound should be excised; while abscesses should be incised and drained.4 The wounds should be left open and concern for the treatment of infection should outweigh concern over skin closure. In rare instances amputation of the affected limb might also be necessary. Physiological amputation with a tourniquet and dry ice may also be necessary till a definitive amputation is done.15
Benzodiazepines such as diazepam and midazolam are used to suppress spasms and rigidity associated with tetanus. Neuromuscular blocking agents like vecuronium are used with intermittent positive pressure ventilation because the patient usually spends a long time in ICU. Intrathecal baclofen (Lioresal) has been used to wean the patients from a ventilator and also facilitates in stopping diazepam infusion.1 Tracheostomy protects the airway against laryngospasm and should be done electively in severe cases of tetanus when it is estimated that the patient will spend longer than 10 days on ventilatory support.
Recovery involves synthesis of new presynaptic components and their transport to the distal axon. This accounts for the typical 2- to 3-week period before clinical improvement begins. Our patient A was discharged on day 18 post admission to the ICU which falls within the above time frame.
Treatment of autonomic instability is a subject of ongoing research as it has not been universally accepted. For example, unopposed alpha blockade can result in reflex worsening of tachycardia, and unopposed beta blockade can worsen hypertension and has an increased risk of sudden death.6,11
Subsequent morbidity is caused by complications (Table III) that occur during the acute phase of the disease rather than by the disease itself. Full recovery from tetanus is slow and usually takes 2 to 4 months during which time the patient will have to undergo a lot of physiotherapy and occupational therapy.
It is important for the survivors to receive a complete immunisation programme as the disease itself does not offer immunity.
Conclusion
• Anti-tetanus toxoid must be given in all open injuries within 6 hours of being seen in the emergency room. It is also important for the treating ward doctors to double check that anti-tetanus toxoid was indeed given in the emergency room.
• A history of prior immunisation should not dissuade a physician from making a diagnosis of tetanus.
• Adequate debridement must be done on all patients with wounds and if tetanus infection is suspected, wounds must be left open for later closure.
• Always have a high index of suspicion in patients who present with seizures following an open fracture, particularly if there is no history of epilepsy, head injury or history of alcohol abuse.
• For people who come into frequent contact with soil and also those in the agriculture industry, immunisation should be continued into adulthood.
Acknowledgements
The authors would like to acknowledge the assistance of Dr ME Moagi of the Department of Neurology and Dr NC Muleba of the Department of Intensive Care Unit at Dr George Mukhari Hospital.
References
1. Loscalzo IL, Ryan J, Loscalzo J, Sama A, Cadag S. Tetanus: A clinical diagnosis. American Journal of Emergency Medicine 1995;13:488-9. [ Links ]
2. Rhee P, Nunley M.K, Demetriades D, Velmahos G, Doucet J.J. Tetanus and Trauma: A Review and Recommendations. Journal of TRAUMA® Injury, Infection, and Critical Care 2005;58:1082-8. [ Links ]
3. Mallick IH, Winslet MC. A review of the epidemiology, pathogenesis and management of tetanus. International Journal of Surgery 2004;2:109-12. [ Links ]
4. Wakim N, Henderson S.O, Tetanus. Topics In Emergency Medicine 2003;25:256-61. [ Links ]
5. Kefer M. Tetanus. American Journal of Emergency Medicine 1992;10:445-8. [ Links ]
6. Hsu S, Groleau G. Tetanus in the emergency department. Journal of Emergency Medicine 2000;20:357-65. [ Links ]
7. Culbertson TA, Kalliainen LK, Buchele BA. Tetanus and the plastic surgeon. Annals of Plastic Surgery 2004;53:162-5. [ Links ]
8. Livorsi DJ, Eaton M, Glass J. Generalised tetanus despite prior vaccination and a protective level of anti-tetanus antibodies. American Journal of the Medical Sciences 2010;339(2):200-1. [ Links ]
9. Tetanus vaccine - WHO position paper. Weekly Epidemiological Record, No.20, 19 May 2006. [ Links ]
10. Gardner P. Issues related to the decennial tetanus-diphtheria toxoid booster recommendations in adults. Infectious Disease Clinics of North America 2001;15:143-53. [ Links ]
11. Wright DK, Lalloo UG, Nayiager S. Autonomic nervous system dysfunction in severe tetanus: current perspectives. Critical Care Medicine 1989;17:371-5. [ Links ]
12. Ahmadsyah I, Salim A. Treatment of Tetanus: an open study to compare the efficacy of procaine penicillin and Metronidazole. British Med Journal 1985;291:648-50. [ Links ]
13. Cooke MW. Are current UK tetanus prophylaxis procedures for wound management optimal? Emergency Medicine Journal 2009;26:845-8. [ Links ]
14. Sanford JP. Tetanus - forgotten but not gone. New England Journal of Medicine 1995;332:812-3. [ Links ]
15. Buchman MT, Weis EB. Tetanus complicating an open trimalleolar fracture. A case report. Journal of Bone and Joint Surgery (Br) 1984;66:139-41. [ Links ]
 Reprint requests:
Reprint requests:
Dr BE Ramasuvha
PO Box 224 Medunsa 0204
Tel: +27 12 521 4049, Fax: +27 12 521 4029
Email: lstrauss@ul.ac.za
This article was not submitted to an ethical committee for approval. The content of the article is the sole work of the authors. No benefits of any form have been derived from any commercial party related directly or indirectly to the subject of this article.














