Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
SA Orthopaedic Journal
versión On-line ISSN 2309-8309
versión impresa ISSN 1681-150X
SA orthop. j. vol.9 no.3 Centurion ene. 2010
CLINICAL ARTICLE
Anterior shoulder instability with bony deficiencies and the author's preferred method of treatment
Joe F de Beer
MBChB, MMed(Orth). Cape Shoulder Institute, Panorama, South Africa
ABSTRACT
Anterior shoulder instability in the athlete is a common problem. It is imperative that in order to prevent unnecessary time away from sport that the correct decisions and interventions are taken at the appropriate times. The last decade has seen a change in our perceptions of the surgical options for this type of patient. We now have a better understanding of the pathology, the surgical options and their outcomes and the natural history both of the condition and surgery.
Introduction
In 1999 arthroscopic soft tissue reconstruction of the shoulder was at the forefront for the pioneers of shoulder surgery. The debate was between open and arthroscopic soft tissue reconstruction of the shoulder. Bony augmentation of the shoulder was little talked about.
Initial enthusiasm for arthroscopic techniques was dampened by poor results with certain arthroscopic techniques such as trans-glenoid suturing and bio-absorbable tacks. As techniques for arthroscopic stabilisation developed, results now rival those of the traditional open procedures.1 However, despite advanced techniques concerns have been raised with regard to high recurrence rates in the bone-deficient shoulder in the contact athlete.2
Shoulder injuries are a common occurrence during elite sport. The incidence is highest during contact sports such as elite level rugby. In English Professional Rugby Union shoulder injuries result in an average of 241 player days lost per club per season. Not unsurprisingly most injuries occur during matches, with the commonest mechanism of injury being the tackle. The injury that causes the greatest absence and which has the highest recurrence rate is shoulder instability.3
Diagnosis and evaluation
When instability does occur it is variable in its presentation. In its most straightforward form, the frank dislocation, the diagnosis is obvious to both player and clinician. It can however be more subtle in its presentation with feelings of apprehension or weakness in the tackle. It may present simply as a painful shoulder.
All episodes of conclusive and suspected shoulder instability require the same detailed evaluation. It is critical when treating patients with high sporting ambitions that the correct diagnosis and treatment is instigated from the first episode. For a patient with a promising career a wrong diagnosis or treatment path can spell the end of a career. A single dislocation with a significant bony lesion may lead to failed conservative management and redislocation in the next season can potentially end a career.
The last 10 years have brought about a resurgence of interest in bony deficiency of the shoulder with instability. Bony deficiency of the glenoid was thought to be uncommon. This was probably because we didn't look. It is rarely enough to have a single AP X-ray view of the shoulder. In the paper by Edwards et al4 the osseous glenoid lesion was only visible on the AP view in 73 of the 139 cases. Even at arthroscopy the diagnosis may be unclear - you won't see it if you only view from the posterior portal.
The frequency of glenoid bone deficiency was highlighted by three publications in 2003. Griffith et al5 showed variable flattening of the anterior glenoid in 42 of 46 shoulders that had previously dislocated. Sugaya et al6 examined 100 shoulders with recurrent instability using three-dimensional CT scanning and showed abnormal morphology in 90%. Fifty glenoids demonstrated an osseous fragment and 40 had flattening of the anterior glenoid. Edwards et al4 demonstrated osseous lesions of the glenoid in 139 of 160 (87%) shoulders with a diagnosis of instability. These findings bring into question the frequency of pure Bankart lesions where the labrum is avulsed without any bone. It seems that the Sharpey's fibres avulse at least a fragment of bone in most young individuals during a traumatic anterior dislocation. This fact led us to question the actual existence of ALPSA (anterior labro-periosteal sleeve avulsion) lesions:7 the frequency of bony avulsion may imply that we are looking into a non-union of a bony lesion rather than a periosteal sleeve avulsion.
Radiographic evaluation of the unstable shoulder starts with adequate plane X-ray views. Special attention must be given to the sclerotic glenoid line. Loss of this line is highly specific for glenoid bone loss8 although sensitivity is moderate. The Bernegeau view is of particular help in evaluating the anterior column of the glenoid. This view is highly radiographer-dependent and taken with repeated accuracy only when there is the luxury of an image intensifier to guide the beam. In order to ensure that the correct diagnosis is made, in all cases, a 3D-CT or a CT scan with an en-face sagittal reconstruction of the glenoid is needed.6,9,10 Recently different radiological methods to measure the size of a bony defect have been described and validated.5,6,11-17 To measure the size of a defect, one must know the original size of the glenoid. There are two ways to obtain this information. The first is by comparing the CT of the contralateral shoulder, which can only be done if the contralateral shoulder has no history of instability or other disorders.5,15,16 The second is by placing a best-fitting circle on the inferior part of the glenoid (Figure 1), which will reconstruct the shape of the inferior glenoid.6,11,14 Finally the size of the defect can be expressed in different ways: as a percentage of the surface of the best-fitting circle on the inferior glenoid (Figure 1); as a percentage of original width; as a percentage of the length of the glenoid. The same information can be obtained from MRI scanning.13

Glenoid bone loss can also be measured arthroscopically with reference to the glenoid bare spot.18 Viewing from the antero-superior portal is required to demonstrate the bone loss adequately. In fact, the 'inverted pear' appearance of the glenoid, when the inferior part of the glenoid appears narrower than the superior part and the usual pear-like appearance is now 'upside down', is an arthroscopic appearance of the glenoid when viewed from the antero-superior portal.19 To assess glenoid bone loss while viewing from antero-superiorly a calibrated probe is inserted from the posterior portal. The distances from the posterior and anterior glenoid margins to the bare spot are measured. The percentage glenoid width deficiency can then be calculated as described by Lo et al20 as the difference between the two measurements divided by twice the distance from the posterior margin to the bare spot.
This measurement has been found to be accurate, especially for anterior located defects,21,22 which is the case in the majority of patients with glenoid bone loss.10,23 It is common practice for shoulder surgeons to view the glenoid from the posterior portal only, which may make it difficult to appreciate the degree of anterior glenoid bone loss (Figure 2).

It is, however, one thing to recognise bony deficiency of the glenoid but another to determine its significance. We defined 'significance' for the purpose of our paper in 2000 to be glenoids of the 'inverted pear' configuration.19 This configuration means that there has been 25% bone loss of the inferior pole of the glenoid.20 The 25% cut-off has since been widely quoted. The effect of sizeable glenoid defects on shoulder instability has been confirmed in cadaveric studies. Itoi et al24 performed sequential glenoid osteotomies to determine their effect on humeral head translation. They determined that glenoid deficiencies of a width greater the 21% of the glenoid length may be best served by restoration of the glenoid arc for reasons both of stability and range of movement. The same was found by Yamamoto et al, who performed a biomechanical cadaver study on the anterior located defects.25 The benefits of reconstructing these defects with bone graft has been shown by Montgomery et al.26 What experience has taught us is that there is an inverse relationship between the amount of bone loss tolerated and the demands placed on the shoulder, i.e. the higher the demand, the less bone loss is tolerated. We therefore deem significant even small bony deficiencies in the elite athlete.
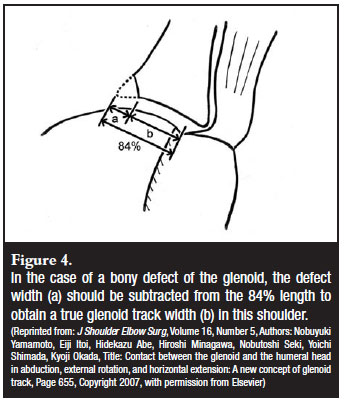
Bony deficiency on the humeral side, namely an engaging Hill-Sach's lesion, also needs to be taken into account. The size of the Hill-Sach's lesion will be evident from the CT scan but its true significance is best determined at arthroscopy. One also has to realise that the glenoid bone loss from the edge of the glenoid and the Hill-Sach's lesion result in a functional-mechanical phenomenon referred to as an 'engaging Hill-Sach's lesion'. The combined effect of the bone loss on the humeral head and the defect of the anterior glenoid results in the 'engagement' or 'hooking' of the defect on the anterior edge of the glenoid. Evaluation by air arthroscopy has the best chance of showing the engagement of the lesion before the increased soft tissue tension from the arthroscopy fluid prevents it (Figure 3). The concept of the engaging Hill-Sachs was further developed by Yamamoto et al who performed a cadaveric study.27 They showed a consistent pattern of the glenohumeral contact area when the arm is abducted and externally rotated. This contact area shifts from inferomedial (arm adducted) to superolateral (arm abducted and externally rotated). They called this zone of shifting contact the glenoid track. They confirmed this pattern in three patients with 3D-CT scans. The width of the glenoid track from the attachment of the cuff was 84% of the glenoid width. If the medial margin of a Hill-Sach's lesion lies outside the glenoid track, this will cause an engaging Hill-Sach's. On the other hand, if there is a bony defect of the glenoid, this will narrow the glenoid track, which can cause the medial margin of a relatively small Hill-Sach's lesion to be outside the glenoid track (Figure 4). This model therefore combines the effect of a glenoid defect and the Hill-Sach's lesion. This will hopefully lead to a further understanding and improvement of the treatment of gleno-humeral instability.

Balg and Boileau28 have provided a very useful index to help us in the decision-making process for shoulder instability surgery. The instability severity index score takes into account six significant pre-operative factors: age under 20 years (2 points), competitive sports (2 points), contact or forced overhead activity (1 point), anterior or inferior hyper-laxity (1 point) and on the AP X-ray a visible Hill-Sach's lesion in external rotation (2 points) and loss of normal inferior glenoid contour (2 points). A score of 3 or less associated with a recurrence rate of 5% with arthroscopic stabilisation, 6 or less a 10% recurrence rate, and greater than 6 the recurrence rate escalates to 70%. They have suggested that a patient with a score of more than 6 would be better served by open surgery, i.e. a Latarjet procedure.
Treatment
An important factor in the treatment pathway is to determine when intervention should be undertaken. This not only means whether surgery is indicated but when surgery should be undertaken.
Surgery should certainly be considered for the first-time dislocator. Depending on the age of the patient, the stage of their career and the pathology identified, it is often best to proceed expeditiously to definitive treatment, although one may choose to allow a player to play out all or part of a season following an instability episode if the situation dictates.
Operative treatment
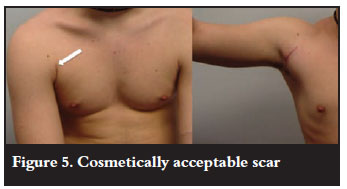
The most important factor when determining the operation of choice is its success rate. Most papers report the percentage risk of further dislocations. Although this is a gross indicator of success, particularly in this group of patients, return to their previous level of sport is most important. This requires the return to normality of other parameters such as the patient's perceived joint stability, muscle strength and range of motion. Many scores have been developed which better account for these additional factors. It is also important to take into account recovery times and the complication rate. Most patients will choose the operation with the highest success rate irrespective of whether it is an open or arthroscopic procedure. In an age where the majority of our surgery is arthroscopic it can be the case that we shy away from unfamiliar surgery preferring an arthroscopic over an open procedure. Is there really a role for the 'trial' of arthroscopic Bankart repair, especially in this group of patients? The scar from open surgery, including a Latarjet procedure, can still be cosmetically very acceptable (Figure 5).
Since the publications of the early part of this decade more attention has been paid to the results in patients with significant deficiencies of the glenoid. It may be possible with careful attention to technique that acceptable results can be achieved with arthroscopic techniques. It certainly appears important to separate the patients with a sizeable avulsion fragment from those with more attritional bone loss. Mologne et al29 reported a series of patients in whom arthroscopic suture anchor repair was performed in patients with a glenoid of the inverted pear type. Bone loss was between 20 and 30%. They had 2 (9.5%) recurrences in the 21 patients followed for 34 months. There were however no recurrences in the group in whom a bony fragment was present and incorporated into the repair. Sugaya et al30 in their 2005 paper also reported excellent results at a similar follow-up using arthroscopic repair incorporating osseous Bankart lesions in patients with average glenoid bone loss of 25%. Open surgery may also be able to yield good results without bony reconstruction. Pagnani31 reported no recurrences in his group of patients with large glenoid defects who had undergone open capsular repair. The numbers in this series are however small and their case is yet to be proven.
It has been our practice to perform bony reconstruction of the glenoid using a modification of the Latarjet32 procedure. It is of course possible to reconstruct the glenoid in multiple ways: Eden-Hybinnette procedure, J-shaped bone grafts and various other autograft and allograft techniques.
While various operations have been described to specifically address humeral head bony deficiency, such as Remplissage and humeral head allografts, we have found that this form of bony deficiency can be adequately addressed by augmenting the articular arc of the glenoid and thus preventing engagement.
The Congruent-Arc Latarjet33 is a modification of the technique described by Latarjet in 1954.32 The advantage of the Latarjet procedure over free grafting techniques is that the graft remains pedicled and therefore is at least in part vascularised. The advantage of this specific modification of the Latarjet procedure is that by rotating the graft through 90 degrees the concavity of the inferior surface of the coracoid matches that of the glenoid negating the need for contouring of the graft (Figure 6). Rotation of the coracoid in this manner has been shown to optimise gleno-humeral contact forces when compared to the traditional Latarjet technique.34 The optimal position for the graft is flush with the glenoid surface. Grafts placed medially result in increased pressures with high edge-loading. Bone grafts placed in a proud position not only increase the peak contact forces antero-inferiorly but also increase the postero-superior glenoid pressure indicating a shift posteriorly.

There are several key steps that we believe are critical to the rapid recovery and success of this modified Latarjet operation. Most important is probably the soft tissue management. The operation is performed through a split in subscapularis at the junction of the superior third and inferior two-thirds. The capsule is elevated from the glenoid neck, and the edge is repaired to the glenoid, therefore effectively lengthening it and lateralising. The other fundamental factor in the operation's success is achieving bony union of the coracoid onto the glenoid neck. The rate of bony union is maximised by the decortication of the surface to be applied to the edge of the glenoid by osteotomising the pectoralis minor insertion. By making this osteotomy slightly oblique the surface area is increased. The opposing surfaces of the coracoid graft and surface on the glenoid neck must match perfectly, as incongruity of those surfaces contributes to non-union of the graft. Partially threaded compression screws ensure graft compression which aids in bony union.
Postoperative rehabilitation
Owing to the lack of damage to subscapularis and the lengthening of the capsule there is no soft tissue repair to protect postoperatively and the rehabilitation can be accelerated. Return to activity and ultimately sport is only limited by the rate of bony union of the coracoid block. At present research is being undertaken into CT techniques to determine the exact time of bony union of the coracoid graft to the edge of the glenoid - once that is demonstrated it would be safe to allow the player back to his full activities.
Our present approach is based on experience and not any specific 'measurements': range of motion with the 'hand in vision', thus avoiding abduction and external rotation, is encouraged from day one. The sling is used only for comfort and usually discarded in the first couple of weeks. Early rehabilitation and gym work, not placing undue strain on the bone graft, is started when comfort allows. Non-contact ball work is allowed from 8 weeks with return to full contact after three months.
With the resurgence of interest in this type of surgery this type of operation is now being performed arthroscopically.35 The arthroscopic variant has not been performed for long enough for an opinion to be expressed on the success rate yet.
Operative complications
Complications from this operation are rare if undertaken by experienced surgeons. Attention to detail is the key. Acute complications such as musculo-cutaneous nerve injury are thus preventable. It is our practice to identify the nerve before coracoid osteotomy (Figure 7). The identity of the nerve can be checked by intra-operative nerve stimulation. This allows for safe soft tissue release and coracoid mobilisation after the coracoid is osteotomised. Even the most experienced surgeons can struggle to identify the nerve after the coracoid has been osteotomised.

Non-union or strong fibrous union of the coracoid is probably not uncommon but rarely problematic. Screw breakage or coracoid displacement has been rarely seen and usually relates to over-aggressive rehabilitation and return to contact sport before adequate healing of the graft. Recurrent instability, which in this demanding category of patients has previously been reported to be high with an arthroscopic soft tissue repair, has been decreased with this procedure to 5%.2 The most important factor in preventing long-term complications from this procedure is accurate coracoid placement. The graft should be placed at the level of or just slightly medial to the surface of the glenoid. It should never be placed more lateral than the surface as this results in a high rate of arthritis.36 In order to get this most critical step right the most important factor is exposure. The release of the subscapularis from the underlying capsule is critical to allow for an adequate capsulotomy and insertion of the Fukuda or other humeral head retractor, A lever retractor placed on the glenoid neck further enhances exposure. Once the soft tissue is cleared from the glenoid neck, usually with the bony Bankart remnant, the bed must be made flat.
It is useful at this point to look at the CT scans to determine the plane of the bony loss. It is best to have this surface perpendicular to the plane of the glenoid. The slightly oblique osteotomy of the medial surface of the coracoid will then bring the concave undersurface of the coracoid into the correct plane. Strict attention to these details will avoid poor coracoid placement. Malposition rates for the coracoid are in some series alarmingly high with 35 of 58 (58%) being malpositioned on plain radiographs in the series report by Allain et al.36
Conclusion
The keys to the treatment of anterior shoulder instability in this category of patient are therefore to make the right diagnosis and to perform the correct operation at the optimum time. Although the decision-making process is still an art we are now better armed to make the correct choices for our patients.
References
1. Hobby J, Griffin D, Dunbar M, Boileau P. Is arthroscopic surgery for stabilization of chronic shoulder instability as effective as open surgery? A systematic review and meta-analysis of 62 studies including 3044 arthroscopic operations. J Bone Joint Surg Br 2007 Sep;89(9):1188-96. [ Links ]
2. SS, De Beer JF, Barth JR, Cresswell T, Roberts C, Richards DP. Results of modified Latarjet reconstruction in patients with antero inferior instability and significant bone loss. Arthroscopy. 2007 Oct;23(10):1033-41. [ Links ]
3. Headey J, Brooks JH, Kemp SP. The epidemiology of shoulder injuries in English professional rugby union. Am J Sports Med. 2007 Sep;35(9):1537-43. [ Links ]
4. Edwards TB, Boulahia A, Walch G. Radiographic analysis of bone defects in chronic anterior shoulder instability. Arthroscopy. 2003 Sep;19(7):732-9. [ Links ]
5. Griffith JF, Antonio GE, Tong CW, Ming CK. Anterior shoulder dislocation: quantification of glenoid bone loss with CT. AJR Am J Roentgenol. 2003 May;180(5):1423-30. [ Links ]
6. Sugaya H, Moriishi J, Dohi M, Kon Y, Tsuchiya A. Glenoid rim morphology in recurrent anterior glenohumeral instability. J Bone Joint Surg Am. 2003 May;85-A(5):878-84. [ Links ]
7. Neviaser TJ. The anterior labroligamentous periosteal sleeve avulsion lesion: a cause of anterior instability of the shoulder. Arthroscopy 1993;9(1):17-21. [ Links ]
8. Jankauskas L, Rudiger HA, Pfirrmann CW, Jost B, Gerber C. Loss of the sclerotic line of the glenoid on anteroposterior radiographs of the shoulder: A diagnostic sign for an osseous defect of the anterior glenoid rim. J Shoulder Elbow Surg 2009 Jun 23. [ Links ]
9. Stevens KJ, Preston BJ, Wallace WA, Kerslake RW. CT imaging and three-dimensional reconstructions of shoulders with anterior glenohumeral instability. Clin Anat 1999;12(5):326-36. [ Links ]
10. Griffith JF, Antonio GE, Yung PS, Wong EM, Yu AB, Ahuja AT, et al. Prevalence, pattern, and spectrum of glenoid bone loss in anterior shoulder dislocation: CT analysis of 218 patients. AJR Am J Roentgenol 2008 May;190(5):1247-54. [ Links ]
11. Baudi P, Righi P, Bolognesi D, Rivetta S, Rossi Urtoler E, Guicciardi N, et al. How to identify and calculate glenoid bone deficit. Chir Organi Mov 2005 Apr-Jun;90(2):145-52. [ Links ]
12. Huysmans PE, Haen PS, Kidd M, Dhert WJ, Willems JW. The shape of the inferior part of the glenoid: a cadaveric study. J Shoulder Elbow Surg 2006 Nov-Dec;15(6):759-63. [ Links ]
13. Huijsmans PE, Haen PS, Kidd M, Dhert WJ, van der Hulst VP, Willems WJ. Quantification of a glenoid defect with three-dimensional computed tomography and magnetic resonance imaging: a cadaveric study. J Shoulder Elbow Surg 2007 Nov-Dec;16(6):803-9. [ Links ]
14. Barchilon VS, Kotz E, Barchilon Ben-Av M, Glazer E, Nyska M. A simple method for quantitative evaluation of the missing area of the anterior glenoid in anterior instability of the glenohumeral joint. Skeletal Radiol 2008 Aug;37(8):731-6. [ Links ]
15. Chuang TY, Adams CR, Burkhart SS. Use of preoperative three-dimensional computed tomography to quantify glenoid bone loss in shoulder instability. Arthroscopy 2008 Apr;24(4):376-82. [ Links ]
16. Diederichs G, Seim H, Meyer H, Issever AS, Link TM, Schroder RJ, et al. CT-based patient-specific modeling of glenoid rim defects: a feasibility study. AJR Am J Roentgenol 2008 Nov;191(5):1406-11. [ Links ]
17. Magarelli N, Milano G, Sergio P, Santagada DA, Fabbriciani C, Bonomo L. Intra-observer and interobserver reliability of the 'Pico' computed tomography method for quantification of glenoid bone defect in anterior shoulder instability. Skeletal Radiol 2009 Nov;38(11):1071-5. [ Links ]
18. Burkhart SS, De Beer JF, Tehrany AM, Parten PM. Quantifying glenoid bone loss arthroscopically in shoulder instability. Arthroscopy 2002 May-Jun;18(5):488-91. [ Links ]
19. Burkhart SS, De Beer JF. Traumatic glenohumeral bone defects and their relationship to failure of arthroscopic Bankart repairs: significance of the inverted-pear glenoid and the humeral engaging Hill-Sachs lesion. Arthroscopy 2000 Oct;16(7):677-94. [ Links ]
20. Lo IK, Parten PM, Burkhart SS. The inverted pear glenoid: an indicator of significant glenoid bone loss. Arthroscopy 2004 Feb;20(2):169-74. [ Links ]
21. Griffith JF, Yung PS, Antonio GE, Tsang PH, Ahuja AT, Chan KM. CT compared with arthroscopy in quantifying glenoid bone loss. AJR Am J Roentgenol 2007 Dec;189(6):1490-3. [ Links ]
22. Provencher MT, Detterline AJ, Ghodadra N, Romeo AA, Bach BR, Jr., Cole BJ, et al. Measurement of glenoid bone loss: a comparison of measurement error between 45 degrees and 0 degrees bone loss models and with different posterior arthroscopy portal locations. Am J Sports Med 2008 Jun;36(6):1132-8. [ Links ]
23. Saito H, Itoi E, Sugaya H, Minagawa H, Yamamoto N, Tuoheti Y. Location of the glenoid defect in shoulders with recurrent anterior dislocation. Am J Sports Med 2005 Jun;33(6):889-93. [ Links ]
24. Itoi E, Lee SB, Berglund LJ, Berge LL, An KN. The effect of a glenoid defect on anteroinferior stability of the shoulder after Bankart repair: a cadaveric study. J Bone Joint Surg Am 2000 Jan;82(1):35-46. [ Links ]
25. Yamamoto N, Itoi E, Abe H, Kikuchi K, Seki N, Minagawa H, et al. Effect of an anterior glenoid defect on anterior shoulder stability: a cadaveric study. Am J Sports Med 2009 May;37(5):949-54. [ Links ]
26. Montgomery WH, Jr., Wahl M, Hettrich C, Itoi E, Lippitt SB, Matsen FA, 3rd. Anteroinferior bone-grafting can restore stability in osseous glenoid defects. J Bone Joint Surg Am 2005 Sep;87(9):1972-7. [ Links ]
27. Yamamoto N, Itoi E, Abe H, Minagawa H, Seki N, Shimada Y, et al. Contact between the glenoid and the humeral head in abduction, external rotation, and horizontal extension: a new concept of glenoid track. J Shoulder Elbow Surg 2007 Sep-Oct;16(5):649-56. [ Links ]
28. Balg F, Boileau P. The instability severity index score. A simple pre-operative score to select patients for arthroscopic or open shoulder stabilisation. J Bone Joint Surg Br 2007 Nov;89(11):1470-7. [ Links ]
29. Mologne TS, Provencher MT, Menzel KA, Vachon TA, Dewing CB. Arthroscopic stabilization in patients with an inverted pear glenoid: results in patients with bone loss of the anterior glenoid. Am J Sports Med 2007 Aug; 35(8):1276-83. [ Links ]
30. Sugaya H, Moriishi J, Kanisawa I, Tsuchiya A. Arthroscopic osseous Bankart repair for chronic recurrent traumatic anterior glenohumeral instability. J Bone Joint Surg Am 2005 Aug;87(8):1752-60. [ Links ]
31. Pagnani MJ. Open capsular repair without bone block for recurrent anterior shoulder instability in patients with and without bony defects of the glenoid and/or humeral head. Am J Sports Med 2008 Sep;36(9):1805-12. [ Links ]
32. Latarjet M. [Treatment of recurrent dislocation of the shoulder.]. Lyon Chir 1954 Nov-Dec;49(8):994-7. [ Links ]
33. De Beer JF BS, Roberts CP, van Rooyen K, Cresswell T, du Toit DF. The Congruent-Arc Latarjet. Techniques in Shoulder and Elbow Surgery 2009;10(2):62-7. [ Links ]
34. Ghodadra N GA, Shewman E, Goldstein J, Verma NN, Bach BR, Romeo AA, Provencher MT. Normalization of glenohumeral articular contact pressures after either Latarjet or iliac crest bone grafting procedure: Impact of graft type, position, and coracoid orientation. JBJS - Am Volume In Press. [ Links ]
35. Lafosse L, Lejeune E, Bouchard A, Kakuda C, Gobezie R, Kochhar T. The arthroscopic Latarjet procedure for the treatment of anterior shoulder instability. Arthroscopy 2007 Nov;23(11):1242 e1-5. [ Links ]
36. Allain J, Goutallier D, Glorion C. Long-term results of the Latarjet procedure for the treatment of anterior instability of the shoulder. J Bone Joint Surg Am 1998 Jun;80(6):841-52. [ Links ]
 Reprint requests:
Reprint requests:
Dr JF de Beer
Bloomberg House Bloulelie Crescent
Cape Town 7506
South Africa
No benefits of any form have been received from a commercial party related directly or indirectly to the subject of this article.














