Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SA Orthopaedic Journal
On-line version ISSN 2309-8309
Print version ISSN 1681-150X
SA orthop. j. vol.9 n.3 Centurion Jan. 2010
CLINICAL ARTICLE
Management of open tibia fracture following fresh water contamination
CH SnyckersI; A VisserII; E HoosienIII; T MonniIV; NA IsmailV
IMBChB(Pret) Dip(PEC)SA MMed(Ort)(Pret) FC(Ort)SA. Consultant: Department Orthopaedic Surgery, University of Pretoria
IIMBChB(Pret) DTM&H PG (Dip) TM. Senior Registrar: Department Clinical Pathology, University of Pretoria; National Health Laboratory Services TAD
IIIMBChB(Pret). Registrar: Department Medical Microbiology, University of Pretoria; National Health Laboratory Services TAD
IVMBChB(Pret). Senior Registrar: Department Orthopaedic Surgery, University of Pretoria
VMBChB(Medunsa) FC Path (Micro) DTM&H(Wits) PDIC(Stell). Consultant: Department Medical Microbiology, University of Pretoria; National Health Laboratory Services TAD
Introduction
The treatment of contaminated open fractures of the tibia remains problematic. Research has shown that the combination of early wound debridement, fracture stabilisation, soft tissue cover and empiric antibiotic use can reduce the incidence of infective complications.1 Empiric choice of antimicrobial(s) should target the most likely pathogens at the site of infection. Staphylococci and Streptococci are the most common aetiological agents2-3 for wound sepsis in the orthopaedic setting, and are typically covered by the first generation cephalosporins.
Wound infections secondary to exposure to water sources poses a unique challenge in that a different spectrum of microorganisms are expected and varies according to the type of water source. A recent article published by Ribeiro et al3 focusing on the management of burns patients, noted that water exposure due to irrigation of the wounds as part of first aid management resulted in Gram-negative colonisation and infection. Thus, different empiric antimicrobials were suggested according to the water sources used for irrigation. Local epidemiology and resistance patterns need to be taken into account when deciding on the initial empiric antibiotic regimen.
Case report
A 26-year-old male was involved in a motor vehicle accident, in which the car hit a fire hydrant. The patient was found unconscious and lying in a pool of water. He was intubated at the accident scene to secure his airway. The subsequent clinical and radiological examinations after stabilisation in casualties, revealed a traumatic subarachnoid haematoma (SAH) and a Gustilo-Anderson grade IIIB open tibia and fibula fracture (Figure 1). The limb was neurovascularly intact. The neurosurgeon and orthopaedic surgeon were consulted to manage this patient further.

The management of the open fracture commenced in the resuscitation bay with a thorough rinsing of the wound with sterile water, followed by sterile dressings and splintage with an above-knee Plaster of Paris backslab. He received his first dose of antibiotics (cephazolin 2g stat IV) along with tetanus prophylaxis and analgesia. The time span from injury to initial irrigation was approximately 6 hours owing to delayed transport to the emergency unit.
Once stabilised, the patient underwent a craniotomy to evacuate the SAH. An intracranial pressure monitor was inserted. The open fracture was surgically debrided by extending the wounds for better exposure and removal of all foreign material and devitalised tissue. The bone ends were delivered through the wound and thoroughly cleaned. The wound was rinsed out with copious amounts of a chlorhexidine water solution. A monolateral external fixator was used to hold the reduction (Figure 2). Several tissue samples were taken during the procedure and submitted for microbiological analysis. These initial samples rendered negative cultures. The defect was managed with a gentamicin/Intrasite/Opsite pouch. Postoperatively the supportive care continued in the trauma ICU. Triple intravenous antibiotic therapy (cephazolin, gentamycin and metronidazole) was commenced empirically, owing to the degree of contamination.

The patient returned to theatre 48 hours post initial debridement for a second wound inspection. The plastic surgeon was consulted to assist with soft tissue coverage of the exposed bone. Although there was some local induration and erythema around the wound, no frank pus was noted. These changes were in keeping with the local trauma. The wound bed was deemed acceptable for a reconstructive procedure and a local soleus flap was performed. This was covered by a split skin graft. Multiple intraoperative tissue samples and swabs were again collected for microbiological analysis, as per protocol. The procalcitonin (PCT) levels were monitored post-operatively and this showed an initial reduction (Figure 3).
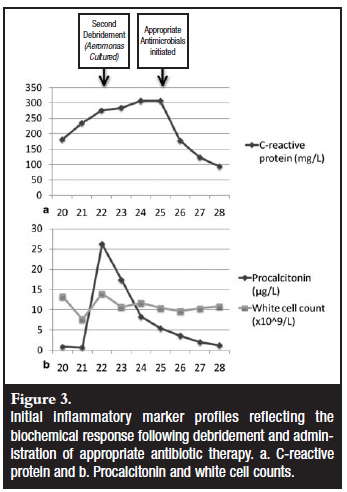
The subsequent wound inspection performed in the Intensive Care Unit revealed a flap survival of only 80%, with tissue breakdown on the inferior suture line. The local wound bed was indurated and clinically infected. Aeromonas hydrophylia was isolated in pure culture on the four tissue samples and two pus swabs that were submitted. It was sensitive to cefepime, the carbapenems, ciprofloxacin as well as the aminoglycosides (gentamycin and amikacin). The antimicrobial treatment was changed to cefepime and the wound was treated locally with vacuum-assisted closure (VAC dressing).
Despite the wound showing improvement with a notable downward trend in the inflammatory markers (Figure 3) after the correct antibiotic was started, a 6x3 cm soft tissue defect remained at the flap's inferior edge. Furthermore, a 2x2 cm area of tibia stripped of periosteum was exposed. As the local flap had already shown early failure, the next reconstructive option was a free flap. The plastic surgeon was concerned that both the available tissue locally, and the patient's general condition, could lead to flap failure.
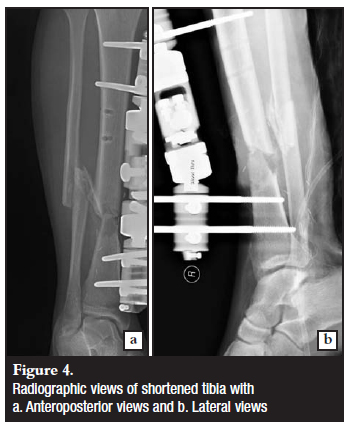
It was decided to manage this problem with an acute shortening of the tibia to achieve soft tissue coverage and optimal dead space management. The soleus flap was mobilised to allow for the tibia shortening of 4 cm (Figures 4 and 5). The soft tissue defect could now be successfully closed with repositioning of the original flap and an additional split skin graft.
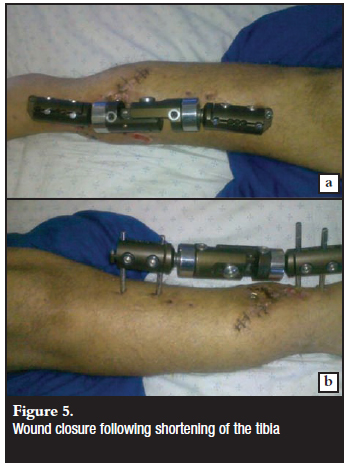
The wounds have subsequently healed well with no further breakdown of the flap.
Discussion
The clinical case highlights the challenges faced when dealing with open tibia fractures. In this case there are two main management principles to note: first, the empiric choice of antimicrobials where water exposure has occurred and secondly, the use of acute limb shortening in the management of a fracture.
Management of water-contaminated wounds
Aeromonas hydrophilia is a Gram-negative bacillus found in fresh water masses. It was first described as a cause of gastroenteritis. Subsequently, it has been associated with infection in burns patients and secondary infection following treatment with therapeutic leech therapy.4
Fresh water
Fresh-water contamination is typically associated with Aeromonas species (spp.), Pseudomonas spp. Plesiomonas spp. and Ewardsiella tarda2,3 infections. Corynebacteria and Bacillus spp. are Gram-positive organisms also found in fresh water collections, and Gram-positive antimicrobial cover is therefore important as part of initial empiric treatment.
Brackish water sources are typically found to have higher salt content and host more salt-tolerant bacteria like Agrobacterium sanguineum.5 If water masses are subject to the effects of humans' or animals' waste disposal, coliforms are more common. Ribeiro et al suggest the use of fluoroquinolones, trimethoprim-sulfamethoxazole or third or fourth generation cephalosporins and carbapenems reserved to settings where polymicrobial infections are suspected3 (Figure 6). It is the opinion of the authors that carbapenem use should be restricted to settings of microbial resistance. Within this clinical setting, pseudomonal cover is imperative, and a first-line empiric choice of a fourth generation cephalosporin or a fluoroquinolone is likely to be the best choice. This distinction may be difficult to make, as polymicrobial infections can be expected in most of these cases. Therefore, the findings on the initial Gram stain may guide decisions on antimicrobial choice.
Soil-contaminated water
Soil-contaminated water carries the added risk of containing various fungi, especially from the zygomycetes group, as well as Bacillus cereus.3,6 Spore-forming bacteria such as Bacillus cereus can be associated with rapidly progressive necrotising infection with myonecrosis, clinically mimicking clostridial infection.7 All patients with contaminated wounds should receive tetanus prophylaxis. In soil-contaminated wounds, this is of particular importance.3 Empiric antimicrobial choice advocated by Ribeiro et al3 included combination therapy of vancomycin, clindamycin, imipenem or a fluoroquinolone with an aminoglycoside (Figure 6).
The choice of vancomycin is purely to cover Bacillus spp. and the authors feel that a stat dose is indicated in all soil-contaminated wounds, which should be continued if Bacillus spp. is subsequently cultured. Antifungal cover in the form of Amphotericin B, together with aggressive debridement is suggested if the presence of fungal elements is confirmed with an intra-operative frozen section.
Salt (sea) water
Pathogens associated with seawater exposure include Vibrio vulnificus, V. cholera, Shewanella algae, S putrefaciens and other Shewanella spp.3,8-9 Infection with V. vulnificus is notoriously invasive and carries a higher mortality rate than infection by the other organisms.10 In this setting, use of combination therapy containing doxycycline with either a fluoroquinolone, ceftazidime or cefotaxime is suggested3 (Figure 6). Mycobacterium marinum and Erysipelothrix rhusiopathiae are two uncommon organisms also seen following seawater exposure. However, they are associated with chronic infections and would require a different treatment regimen directed at the infecting organism.
Acute shortening of open long bone fractures as a means to ensure soft tissue coverage
Tibia fractures remain the most common long bone fracture and up to 24% of these fractures are open.1 These open fractures are often the result of high-energy transfer leading to severe soft tissue and bone injury and contamination.
The host and limb factors should be determined at an early stage. This will help with early goal directed therapy to optimise the biology in the zone of injury. Hence, by minimising the overt inflammatory and anti-inflammatory responses locally, one can limit the systemic responses that may be immunosuppressive.
Appropriate initial management can profoundly affect the overall outcome of the injury by minimising the risk of infection.11
The Gustilo-Anderson classification is widely used with regard to wound classification in the pre-operative and intra-operative setting.12 It has assisted us in communicating the severity of the injury to colleagues and gives a prediction of the outcome. Patzakis documented that infection rates vary from 0-2% in GA type I injuries, to 2-10% in type II injuries and 10-50% in type III injuries.13 Gustilo stated the single most important factor to managing open fractures is the surgical debridement.12 This view was challenged by studies showing the efficacy of early antibiotic use.14 In the first randomised placebo-controlled trial there was a significant reduction in infection with antibiotic use. The timing of its initiation was also important. The rate of infection was significantly higher in those patients who received antibiotics more than 3 hours post injury compared to those who received it within 3 hours.13 Subsequent studies determined the efficacy of a first generation cephalosporin in the management of open tibias.15-16 Although the evidence is insufficient, these studies also suggest possible additional Gram-negative antimicrobial cover in severely contaminated wounds. In keeping with international literature, our policy is to institute empiric first generation cephalosporin treatment as soon as possible for all Gustilo-Anderson I-IIIA fractures (low energy) and to consider triple antibiotic cover in heavily contaminated wounds, farmyard injuries and Gustilo-Anderson IIIA (high energy), IIIB and IIIC fractures.17
As our patients often present late, we still feel an urgent systematic debridement combined with empiric antibiotic use remain the gold standard in the treatment of open fractures. Irrigation is used as a supplement in removing foreign debris and decreasing the bacterial load. Although there are some animal models in the literature that point to the efficacy of irrigation, there is no clear evidence in the literature as to the type or amount needed.18 Furthermore, the use of high pressure irrigation has been questioned recently.19 For these reasons our treatment protocol for heavily contaminated open tibia fractures is to use copious amounts of low pressure lavage, both from a tap-with-hose pre-operatively as well as a bulb syringe intra-operatively. Although controversial, we advocate the initial surgical debridement to be within 6 hours of the injury. This is the recommendation given by most guidelines.14
Definitive treatment of open fractures remains a challenge. Fracture contamination as well as the disruption of the biological soft tissue envelope creates a perfect environment for superficial and deep infection and other related complications. The ultimate aim of treatment is to restore the limb's soft tissue and bone integrity while reducing the complication rate. Prompt decontamination of the wounds, fracture stabilisation and the restoration of the mechanical axis should be the goals of the primary debridement. Despite a recent study showing the effectiveness of immediate intramedullary nailing and meticulous soft tissue management, these facilities are not always available or practical.20 The role of immediate intramedullary nailing in the late presenting patient has not been extensively studied. For this reason we still advocate the use of a monolateral external fixator (where possible) as part of the initial debridement as this approach delivers acceptable results. In a recent metaanalysis the union rate of external fixation was 94%.21 Conversion to internal fixation can be done during the wound inspection procedure.22 Although this practice has recently been challenged, we still find this a useful technique when dealing with heavily contaminated wounds.
Restoration of the soft tissue envelope with vascularised tissue should be done as a matter of urgency.11 The literature is uncertain as to the ideal timing of the soft tissue reconstruction. The evidence strongly suggests that a delay increases infection rates. These defects should be closed within 5-7 days but ideally before 72 hours.23,24 The improved circulation of the wound bed has many benefits which include improved tissue oxygenation, an increase in antibiotic delivery to the area and thus a decrease in infection. Furthermore, restoration of the soft tissue acts as an effective barrier to potential nosocomial contamination.
Whether soft tissue reconstruction is achieved through the use of local rotational flaps, fasciocutaneous flaps or free tissue transfer, depends on the local wound and systemic factors. The location, size and volume of the defect often remain the determining factors. Local rotational flaps are typically used in the proximal and middle thirds of the tibia with gastrocnemius used for the former and soleus for the latter.11 The distal third of the tibia is normally successfully covered by free tissue flaps.
The breakdown of a flap can be devastating. An important point for consideration was made by Pollak et al25 in the LEAP (Lower Extremity Assessment Project) study. Rotational flaps may be compromised as they often reside within the zone of injury. It was found that these flaps had a higher wound complication rate compared to free flaps (44% vs 22%). One must always take into account the extent of the local tissue trauma. This can often only be gauged effectively at the wound inspection procedure.
Infection can also lead to flap failure. Poor empiric antibiotic choice can negatively affect the outcome and result in local sepsis. Once the flap has failed, reconstruction options become difficult. The wound bed is often not suitable for a free flap. Subsequent delay during wound bed preparation with vacuum-assisted coverage or repeated debridement can lead to local bone sequestration, osteitis or osteomyelitis.26
In this case, our solution to this limb salvage problem was to shorten the tibia in order to gain some soft tissue 'length' to close the defect. This was done by repositioning the monoplanar external fixator. The idea of converting this to a ring fixation external construct at a later stage for limb lengthening was kept in mind. A further operation would possibly be required to obtain union of the docking site once the soft tissue and bone have been appropriately lengthened.27
This easy and effective method of wound closure is often overlooked in the literature.17,28,29 In a recent review of open tibia fractures no reference was made to acute shortening.1,11 The literature accepts that up to 4 cm of the tibial diaphysis can be shortened acutely. The vascularity of the limb should, however, be well-supervised intra- and postoperatively.28 This applies to both the arterial supply and venous drainage of the limb. Limb shortening can cause kinking of these vessels, which inevitably results in obstruction. The often troublesome soft tissue defects can be effectively reduced in size to allow for closure. This method is effective if no plastic surgery is available.
Conclusion
This case illustrates that the treatment of open tibia fractures remains a challenge. Blind adherence to protocols can have adverse effects. Attention should be given to the mechanism as well as the environment of an injury.
Water contamination poses a unique challenge as the bacterial contamination varies and choosing of empiric antimicrobials can be difficult. For fresh water the use of a fourth-generation cephalosporin with or without an aminoglycoside should be used whereas salt-water contamination necessitates the use of a third-generation cephalosporin and doxycycline.
Soft tissue defects and flap breakdown can be troublesome to manage. Acute shortening can be an effective way to bridge soft tissue defects in certain situations. Limb vascularity should be diligently followed up.
References
1. Melvin J, Dombroski D, TOrbert J, Kovach S, Esterhai J, Mehta S. Open tibial shaft fractures: I. Evaluation and initial wound management. Journal of the American Academy of Orthopaedic Surgeons 2010;18(1):10-9. [ Links ]
2. Noonburg G. Management of extremity trauma and related infections occuring in the aquatic environment. J Am Acad Orthop Surg 2005;13:243-53. [ Links ]
3. Ribeiro N, Heath C, Kierath J, Rea S, Duncan-Smith M, Wood F. Burn wounds infected by contaminated water: Case reports, review of the literature and recommendation for treatment. Burns 2010;36:9-22. [ Links ]
4. Janda J. Recent advances in the study of the taxonomy, pathogenicity, and infectious syndromes associated with genus Aeromonas. Clin Microbiol Rev 1991;4(4):397-410. [ Links ]
5. Auerbach P,Yajiko D,Nassos P,Kizer K,Morris J,Hadley W. Bacteriology of the freshwater environment: implications for clinical therapy. Ann Emerg Med 1987;16:1016-22. [ Links ]
6. Andresen D, Donaldson A, Choo L, Knox A, Klaassen M, Ursic C. Multifocal cutaneous mucormycosis complicating polymicrobial wound infections in a tsunami survivor from Sri Lanka. Lancet 2005;365:876-8. [ Links ]
7. Drobniewski F. Bacillus cereus and related species. Clin Microbiol Rev 1993;6:324-38. [ Links ]
8. Heath C, Garrow S, Golledge C. Vibrio cholera: a fatal cause of sepsis in Northern Australia. Med J Aust 2001;174:480-1. [ Links ]
9. Steinberg J, Del-Rio C. Gram-negative and Gram-variable bacilli. In: Livingstone EC, editor. Principles and practice of infectious diseases. New York: Elsevier; 2005. p. 2751-61. [ Links ]
10. Bross M, Soch K, Morales R, Mitchell R. Vibrio vulnificus infection: diagnosis and treatment. Am Fam Physician 2007;76:539-44. [ Links ]
11. Melvin J, Dombroski D, Torbert J, Kovach S, Esterhai J, Mehta S. Open tibial shaft fractures: II. Definitive management and limb salvage. Journal of the American Academy of Orthopaedic Surgeons 2010;18(2):108-17. [ Links ]
12. Gustilo R, Anderson J. Prevention of infection in the treatment of one thousand-and-twenty-five open fractures of long bones: Retrospective and prospective analysis. J Bone Joint Surg Am 1976;58:453-8. [ Links ]
13. Patzakis M, Wilkins J. Factors influencing infection rate in open fracture wounds. Clin Orthop Relat Res 1989;243:36-40. [ Links ]
14. A report by the British Orthopaedic Association/British Association of Plastic Surgeons Working Party on the management of open tibial fractures. Br J Plast Surg 1997 September 1997;50:570-83. [ Links ]
15. Hauser C, jr CA, Eachempati S. Council of the Surgical Infection Society: Surgical Infection Society Guideline: Prophylactic antibiotic use in open fractures. An evidence-based guideline. Surg Infect (Larchmt) 2006;7:379-405. [ Links ]
16. Patzakis M, Jr JH, Ivler D. The role of antibiotics in the management of open fractures. J Bone Joint Surg Am 1974;56:532-41. [ Links ]
17. El-Rosasy M. Acute shortening and re-lenghthening in the management of bone and soft-tissue loss incomplicated fractures of the tibia. J Bone Joint Surg Br 2007;89(B):80-8. [ Links ]
18. Anglen J. Wound irrigation in musculoskeletal injury. J Am Acad Orthop Surg 2001;9:219-26. [ Links ]
19. Hassinger S, Harding G, Wongworawat M. High-pressure pulsatile lavage propagates bacteria into soft tissue. Clin Orthop Relat Res 2005;439:27-31. [ Links ]
20. Kakar S, Tornetta P. Open fractures of the tibia treated by immediate intramedullary tibial nail insertion without reaming: A prospective study. J Orthop Trauma 2007;21:153-7. [ Links ]
21. Giannoudis P, Papakostidis C, Roberts C. A review of the management of open fractures of the tibia and femur. J Bone Joint Surg Br 2006;88:281-9. [ Links ]
22. Blachut P, Meek R, O'Brien P. External fixation and delayed intramedullary nailing of open fractures of the tibial shaft: A sequential protocol. J Bone Joint Surg Am 1990;72:729-35. [ Links ]
23. Fisher M, Gustilo R, Varecka T. The timing of flap coverage, bonegrafting, and intramedullary nailing in patients who have a fracture of the tibial shaft with extensive soft-tissue injury. J Bone Joint Surg Am 1991;73:1316-22. [ Links ]
24. Godina M. Early microsurgical reconstitution of complex trauma of the extremities. Plast Recontr Surg 1986;78:285-92. [ Links ]
25. Pollak A, McCarthy M, Burgess A. Short-term wound complications after application of flaps for coverage of traumatic soft-tissue defects about the tibia: The Lower Extremity Assessment Project (LEAP) Study Group. J Bone Joint Surg Am 2000;82:1681-91. [ Links ]
26. Bhattachayya T, Mehta P, Smith M, Pomahac B. Routine use of wound vacuum-assisted closure does not allow coverage delay for open tibia fractures. Plast Recontr Surg 2008;121:1263-6. [ Links ]
27. Mehkail A, Abraham E, Gruber B, Gonzalez M. Bone transport in the management of posttraumatic bone defects in the lower extremity. J Trauma 2004;56:368-78. [ Links ]
28. Lerner A, Fodor L, Soudry M, Peled I, Herer D. Acute shortening: Modular treatment modality for severe combined bone and soft tissue loss of the extremities. J Trauma 2004;57:603-8. [ Links ]
29. Nho S, Helfet D, Rozbruch S. Temporary intentional leg shortening and deformation to facilitate wound closure using the Ilizarov/Taylor spatial frame. J Orthop Trauma 2006;20:419-24. [ Links ]
 Reprint requests:
Reprint requests:
Dr A Visser
Tel: +2782 780 1051
E-mail: adele@up.ac.za














