Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
Journal of Energy in Southern Africa
versión On-line ISSN 2413-3051
versión impresa ISSN 1021-447X
J. energy South. Afr. vol.26 no.2 Cape Town may. 2015
Energy improvement in induction furnace using foaming slag with variation of carbon injection
Theodore Mwata Kipepe; Xiaowei Pan
Department of Extraction Metallurgy, University of Johannesburg, Johannesburg, South Africa
ABSTRACT
Energy efficiency is nowadays one of the biggest concerns worldwide. It has become one of the critical matters to any country and industry particularly in South Africa. Because of the complexities and challenges found in industries and especially in foundry, many strategies have been established to measure and regulate more efficiently the energy. Many studies have been conducted in steelmaking process and electrical arc furnace using foaming slag to reduce energy consumption. As such, in this case study, the experiment will be conducted in a small foundry metal casting laboratory using an Induction Furnace (IF), which will reduce energy usage by using the foaming slag and as well pointing out the importance and the impact of carbon rate change on the foaming creation. After experiments all results show that the melting time can be reduced from 42min to 35 min, which is 20% time saving, the electrical energy consumption also has been reduced from 13.93kWh to 10kWh which is 39.3% saving. On the other hand, the foaming slag optimum height was reached at 1 cm. Therefore, a simple modelling has been designed and calculations have been made on heat transfer at the surface opening of the Induction Furnace and temperature loss has been reduced from 878°C to 870°C, with heat transfer loss reduced from 27.99kW to 26.38kW which was 6.10% of energy saving.
Keywords: energy efficiency, foaming slag, induction furnace, foundry metal casting
1. Introduction
For many years, trying to be energy efficient has become one of the first priorities all over the world. The energy efficiency goal is to reduce the amount of energy required to provide products and services (Wikipedia, 2014). The efficiency benefits can also be gained through environmental improvements.
More than ever, companies are facing increased competitive pressures to produce high quality products at equal or lower cost. With rising energy prices, companies are put under pressure to bring energy costs in line with standards of best practice. The efficiency is applied to all sectors so that they may be able to control and to use less energy by having a good result at the end of the process.
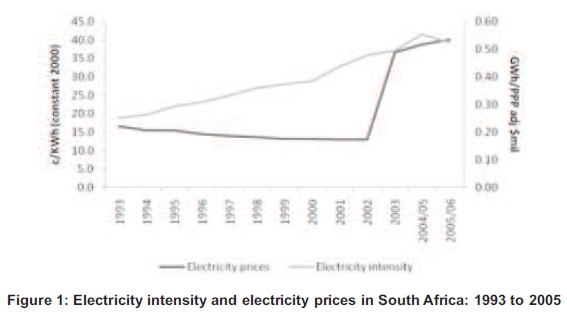
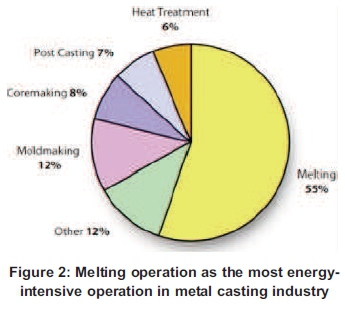
South Africa, for many years, is now facing very big challenges regarding energy consumption usage due to lower price of electricity which was a lack of motivation to save energy and has a negative effect of high energy usage (Rasmeni, 2014) (Figure 1). South African Industrial sectors (agriculture, mining, manufacturing and construction) are some of the biggest consumers of energy and use almost 45 to 49% of energy in South Africa (Davidson, 2006; Matlala, 2004). In the industrial sector, iron & steel use 29% of energy in the industrial sub-sector (Davidson, 2006). Therefore, in the iron & steel sector, foundries are the biggest consumers using different types of furnaces with the Induction Furnace known as the most efficient. Energy efficiency of any foundry largely rides on the efficiency of the melting process. Melting is the most energy-intensive operation in the metal casting industry with 55% of the total energy in the foundries (Incorporated, 2005) (Figure 2). The induction heating process has been considered as high productivity, repeatable quality, and green heating technology compared to other fuel-fired furnaces (Dang, 2012).
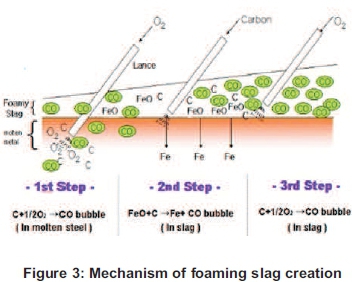
The aim of this paper is to increase the efficiency of the Induction Furnace (IF) by using a foaming slag layer as insulation material to reduce the heat loss at the opening of the Induction Furnace (Figure 3). The importance of this paper is to show the value of the carbon rate change in the creation of the foaming slag and the impact that it plays during this process on the energy efficiency procedure. The use of the foaming slag will decrease the heat loss, which will increase the efficiency of the furnace. As such, a Heat Transfer calculation will be conducted at the opening of the Induction Furnace to measure the heat loss during the process and to show the relation between the foaming slag and the heat transfer at the furnace opening.
2. Experimental methods
2.1 Materials, apparatus and procedure
For this investigation, the materials used were slag particles, oxygen gas, pure carbon particles and cast-iron scraps (Figure 4). In this paper, the experimental processes were done using a small laboratory Induction Furnace of 5 kg, an electrical power station (Figure 5), some alumina lances used to inject gas, a pyrometer to measure the temperature and a thermometer.

Figure 5 shows the schematic diagram of the experimental procedure, which consists of 4 main steps.
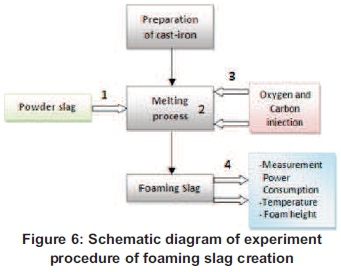
Before any procedure, cast-iron scraps (5 kg) and the carbon particles in different weight of 21.27, 30, 35, 40, 45, and 55g were prepared. First step, the slag particles were poured into the crucible, followed by the cast-iron scrap reduced into small pieces and thereafter the laboratory's induction furnace was switched on.
At the temperature of 900°C when the cast-iron scraps are melting, the slag particles also melted and due to the difference of densities of the two melted elements, the slag has started floating and, at the same time, 100 ml of oxygen gas is injected for 30 seconds into the molten. Thereafter, the carbon is now injected at 21.27g for the first experiment and the procedure went on and on until the last 55g of carbon was injected in the last experiment. The oxygen gas was injected for 30 seconds to ensure the reaction as shown in Figure 3 above. From the starting point until the measurement of the temperature on top of the foaming slag, the melting time and the power consumed were measured. The measurement of the height of the foaming slag was made by inserting an alumina rod into the molten and traces of the foaming slag left on the rod was allowing the reading of the height of the foam (Figure 7).
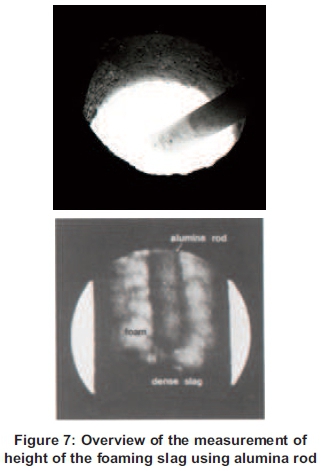
The melting process will be done using a preheated furnace for every experimental procedure due to the results obtained from the basic experimental procedure shown in Table 1.
3. Results and discussion
The results and discussion will give the influences of the carbon rate changes and its effect on parameters mentioned in Table 2.
3.1 Influences of foaming slag height
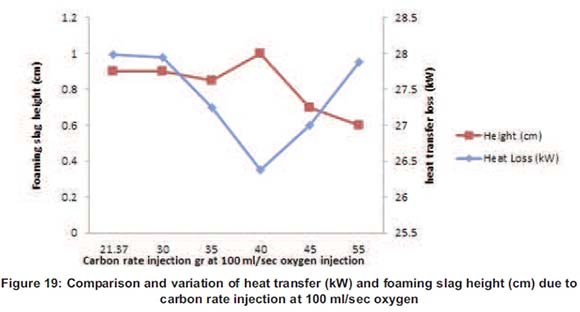
The carbon particles used in the melting process for the foaming slag creation have been poured into the furnace at different rates from 21.27 to 55g. Figure 8 shows the variation of the height according to the carbon poured into the furnace; it also shows the height going up from 0.8 mm to 1cm for 40g of carbon poured. At 45g and 55g, the height started to decrease due to carbon particles which were now in excess and started destroying the foaming slag that was already made.
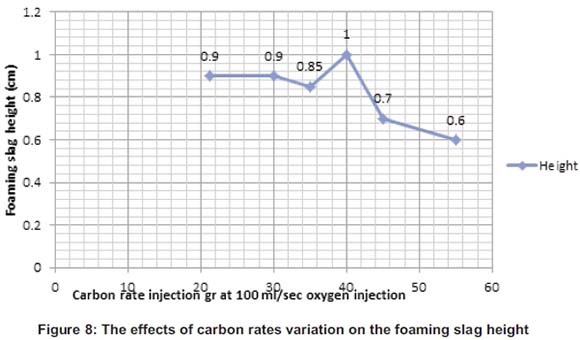
At the optimum, the temperature at the surface was 878°C, the melting time was 42 minutes and the optimum foaming slag height was 1cm, the electrical power consumption used was 13.93kWh.
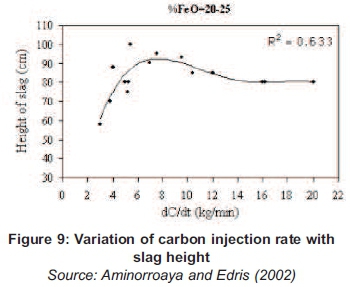
At 40g of carbon poured in the molten, the reaction has reached the optimum and above this amount of carbon, the particles started destroying the bubbles because of the surface tension. A study conducted by Aminorroaya and Edris (2002) on the effect of foamy slag in electrical arc furnaces on electric energy consumption shows that the variation of carbon rate more than 9kg/min has no effect on slag foaming and increasing in electric energy consumption by increasing in melting time (Aminorroaya and Edris, 2002).
3.2 Effect of melting time and temperature of the molten
The melting time and the temperature of the molten during this operation were also influenced by the carbon that was poured into the molten during the procedure. Figures 10 and 11 show that increasing the carbon in the molten until a certain point was contributing to the oxidation which was exothermic and increasing the heat means decreasing the melting time of the cast-iron in the crucible.
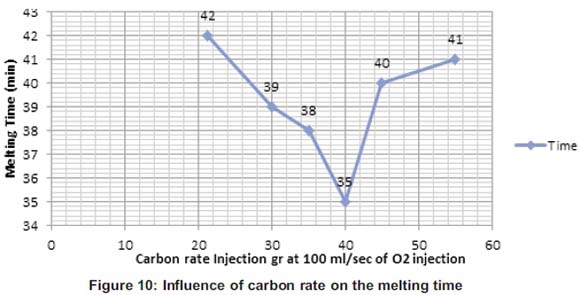
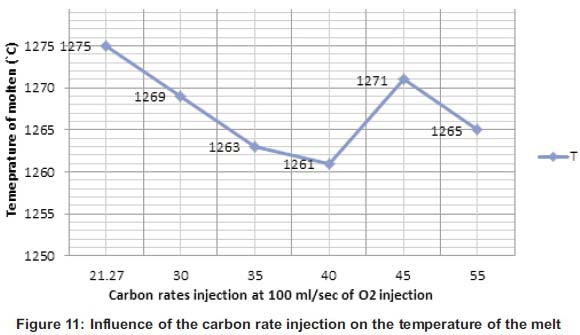
Figure 10 shows that at 21.27g the melting was 42 min and slightly increases for 1min at 43 min. But at 35g and 40g, the melting time starts decreasing and the minima time was at 40g of carbon poured into the molten. From 45g until 55g, the melting time starts increasing again. During the melting process at 35g and 40g the oxygen gas injected at 100ml/sec has reacted until the major percentage of carbon injected was consumed due the reaction. But at 45 and 55g, the oxygen gas at 100ml/sec wasn't enough to oxidize all the carbon and that increases the time due the extra material added in the molten. But Figure 11 shows the relation between the temperature and the carbon rate injected in the molten from 21.27 to 40g, temperature was increasing due to the exothermic reaction which brought extra heat in the molten at 100ml/sec. But from 45 to 55g, heat was brought up and the rate of the oxygen could react with all the carbon particles poured in the molten. Ozturk and Fruehan have shown the impact of temperature on the foaming height. Foaming height on Figure 8 decreases with increasing the temperature (Fruehan, 1995). This shows the co-relation between the carbon and the temperature during the melting process.
3.3 Influence of electrical energy consumption
During the reading of electrical power, the carbon rate injection has shown some variation during the process. In fact, some other parameters have played an important role in the reduction of electrical energy consumption such as the basicity of the slag (Aminorroaya and Edris, 2002). Figure 12 shows the variation of the electrical power consumption. From 21.27g to 40g, the electrical energy went down and slightly started increasing from 40 to 55g.
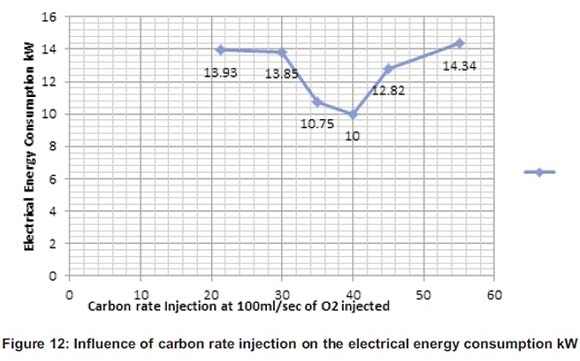
Therefore, from 21.27g to 40g the room temperature was 23°C and from 40 to 55g room temperature was 9° less than normal. Room temperature also played a part on the heating process of the material, the time and eventually on the energy needed to heat the cast-iron. Figure 13 shows that the electrical energy consumption varies with the foaming slag height. If the foaming slag height increases, the electrical energy consumption decreases (Aminorroaya and Edris, 2002).
Aminorroaya and Edris (2002) have also studied and found the influence of the carbon rates on the electrical energy consumption and during their experience, electrical energy decreased with increasing carbon injection rate up to 9kg/min.
3.4 Influence of surface temperature of the foaming slag
The temperature determination on the surface of the foaming slag has all to do with the thickness of the foaming slag and the contribution of the carbon particles. The foam creation is one of the major elements during the process.
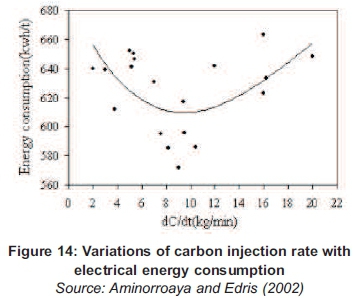
Figure 15 shows that from 21.27g to 40g the temperature decreases due to the carbon rate, which influences the foaming slag height, and increases the thickness of the foaming slag. Increasing the thickness means increasing the height which decreases the heat loss on top of the furnace (Figure 16). At 45g and 55g, the temperature starts increasing due to the fact that the foaming slag started to be destroyed due to the excess of carbon particles in the molten. The carbon particles reacted with the bubble and start destroying them and as such decreasing the foam height (Y. Zhang, 1995). The surface temperature of the foaming slag has also everything to do with the foaming height thickness.
3.5 Heat transfer calculation
The heat transfer in the induction furnace is located and comes from the metal to be heated. The thermal transfer is done to the wall and to the top or opening of the furnace. An Induction furnace is a made of a crucible in graphite, some coils cooled by water circulation (Mauricio V. Ferreira da Luz, 2010). Those coils are protected by an insulation material or refractory.
For a better understanding of the furnace, modelling should be done to see which part of the furnace might be influencing the calculation. The choice of the domain is very important because it shows and gives the kind of heat applied for. The aim is to find some domains which will influence and show which types are used: conduction, convection and radiation. Our focus will be on the thermal problems. Figure 17 gives us a sketch of the furnace and the possible heat and domain which needs to be measured.
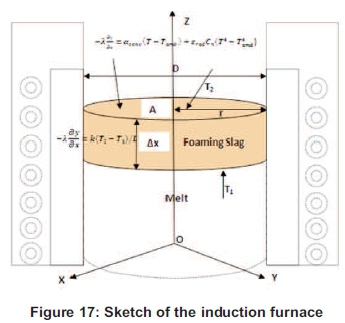
The calculus domain in our case will only be focused on the thermal problem, the inductor region will be considered as one single core and the calculation will be done according to that.
The boundaries of the calculus domain are in fact the surface through that thermal transfer from the furnace parts to the surroundings.
At the surface of the furnace the domain will be given by and the heat transfer is a combination of convection and radiation:
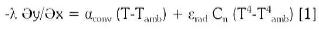
Where, αconv is the convection coefficient. εrad = 0.83 is the total emissivity and Cn = 5.67.10-8 W/m2K4 is Stephan constant, Τ is the surface temperature, Τamb is the temperature of surrounding environment. The coefficients αconv and εrad could depend on temperature and εrad could depend in addition on surface quality.
If the heat transfer calculation in the inductor must be done, the convection heat transfer should be considered because of the exchange between the inductor and the cooling water that circulate through the coils and it given by:
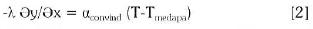
Where αconv ind-apa is the transmissivity between the inductor and cooling water whose medium temperature is Tmed apa(Pasca, 2004). In this case the temperature of the molten, the surface temperature on top of the foaming slag has been considered in Table 2: At the Interface melt and foaming slag the heat transfer is made by conduction:
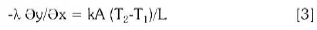
Where k is the thermal conductivity, A is the surface of cylinder; L is the length of the layer.
During the calculation the ∆X changes according to the height of the foaming slag which influences the heat transfer operation.
The total heat transfer loss during the process on top of the furnace will be influence by the conduction, convection and radiation:
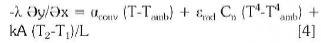
The influence of surface of the cylinder A in this case will be influenced by the height of the foaming slag and represented by L:
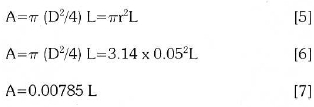
The measurement of the heat transfer will depend on the variation of the thickness of the foam. As such, the results will show the variation of the heat according to the foaming height. The calculation has been made according to the equation 4:
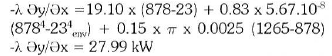
All the results are shown on Table 2 which is the table of variations. The heat transfer calculation for the basic experiments without any kind of foam or insulation has been considered as such the calculation was given by the formula in equation 10. The conduction was considered as zero because the temperature at the interface was the same solid (molten) and the gas.
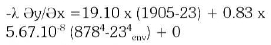
 = 136.5 kW for cold furnace and;
= 136.5 kW for cold furnace and;
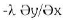 = 156.8 kW for preheated furnace.
= 156.8 kW for preheated furnace.
During the carbon injection into the molten, the reactions below are exothermic because they increased the temperature into the furnace (N. Sahay, June 2014} (Pandey, 2003).

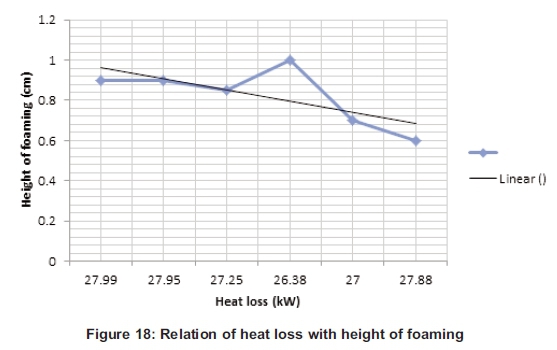
Figure 18 gives the variation of the heat loss compare to the height of the foaming; the figure shows that 1cm was the optimum height reached during the process. From 0.9cm the foaming height stays a little bit constant. When the height increases the heat loss decreases due to the thickness of the foam which reduces the heat loss.
4. Conclusion and summary
The importance of energy efficiency has been demonstrated during the experimental procedure. This paper illustrated the importance of the foaming slag during the melting process and the utility of the foaming slag during the heat loss. The results of the experiments and observations revealed that:
1. At 40g, the use of the foaming slag in the induction furnace has given a melting time saving of 5 minutes which is almost 17.15% of time.
2. The electric energy consumption has been saved from 13.93kW to lOkW at 39.3% of electricity energy. At 40g of carbon injected, the foaming slag obtained its optimum at the height of 1 cm.
3. The foaming slag insulation has helped reduce the temperature radiation from 878°C to 870°C. The thickness of the foam creates a layer which could reduce the heat loss.
4. The heat transfer calculation during the parameters change shows a variation of the heat loss due to the height (Figure 18). The graph line goes down with the heat loss decreasing and that means if the height decreases the heat loss increases. The heat transfer loss during the calculation gives a variation of temperature from 27.99kW to 26.38kW, which give a saving of 6.10%.
5. Comparing the basic experimental heat loss (136.53kW) with the heat loss during the foaming slag (26.38kW) creation almost shows the importance of using foaming slag during the process because in this case, using foaming slag reduces the heat loss to 110,15kW which is almost 19.4% of the energy loss.
6. Usage of foaming slag in the induction furnace in this experiment shows that the foaming slag is theoretically and practically efficient but coming to the heat transfer a tremendous heat loss occurred due to the thickness of the foaming slag, which was very tiny and instead of reducing more heat radiation its reduces less.
7. The improvement of the induction furnace using carbon injection parameters in this research can be implemented to the actual furnaces and not only to the Lab situation.
Acknowledgments
The authors gratefully acknowledged the support of the University of Johannesburg's Foundry Laboratory of Doornfontein Campus for the realization of this project. Our thanks go to the Laboratory technician assistant, Mr. Gilbert. for his availability, time and assistance.
References
Agency, I. E. (2012). Worldwide engagement for sustainable energy strategies. Germany: International Energy Agency. [ Links ]
Aminorroaya, S and Edris, H. (2002). the effect of foamy slag in the electrical arc furnaces on the electric energy consumption. 7th European Electric Steelmaking Conference (pp. 2.447- 2.456). Venice: 7th European Electric Steelmaking Conference. [ Links ]
Dang, H.-S. R-R (2012). Optimization of the In-line induction Heating Process for Hot Forging in Terms of Saving Operating Energy. International Journal of precision engineering and manufacturing , 13 (1085), 1085-1093. [ Links ]
Fruehan, B. O. (1995). Effect of Temperarute on slag foaming. Metallurgical and Materials Transactions B, 26B, 1086- 1088. [ Links ]
Fruehan, B. O. (1995). Effect of temperature on slag foaming. Metallurgicall and Materials Transactions B, 26B, 1086. [ Links ]Fruehan, K. I. (1989). Study on the foaming of CaO-SiO2-FeO slags: Rart 1. Faoming parameters and experimental results. Metallurgical Transaction B, 20B, 509- 511. [ Links ]
Incorporated, B. (2005). Advanced Melting Technologies: energy savings concepts and opportunities for the Metal Casting Industry. Columbia, MD: U.S Department of Energy Energy Efficiency and renewable Energy. [ Links ]
Matlala, T. (2004). Demand Side Management Implementation in South Africa.: Eskom. [ Links ]
Mauricio V Ferreira da Luz, A. B. (2010). Modeling of a Crucible Induction Furnace Taking Into Account the Inter-laminar losses. Brazil: IEEE. [ Links ]
Mohamadi, A. E. (2011). A Collective approach towards green fourndries In South Africa-moving towards green foudnries in SA. Johannesburg: National Foundry Technology Network. [ Links ]
Sahay, A. B. (2014). Dealing with open fire in an underground coal mine vy ventilation control techniques. Journal of the Southern African Institute of Mining and Metallurgy , 114 (6), 447. [ Links ]
Ogunlade Davidson, H. W. (April 2006). Energy policies for sustainable development in South Africa. Cape-Town: Energy Research Centre, UCT. [ Links ]
Randey, B. C. (2003). Laboratory study of channel gasification with stream-air blast in sub-bituminous coal from Rindra Raniganj coalfield. Minetech , 24 (6), 37-49. [ Links ]
Rasca, S. (2004). Rarticularities on numerical modeling of crucible induction furnace. Bucarest. [ Links ]
Wikipedia. (2014). Energy Efficiency. Wikipedia. [ Links ]
Y. Zhang, R. F (1995). Effect of Carbonaceous Rarticles on Slag Foaming. Metallurgical and Materials Transactions B , 26B, 813. [ Links ]
Z.Z. Rasmeni, D. X. (2014). Analyse of energy efficiency and consumption in south african steel foundries. World Foundry Congress. Bilbao: World Foundry Congress. [ Links ]
Received 10 October 2014
Revised 18 May 2015














