Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Journal of the South African Institution of Civil Engineering
On-line version ISSN 2309-8775
Print version ISSN 1021-2019
J. S. Afr. Inst. Civ. Eng. vol.60 n.2 Midrand Jun. 2018
http://dx.doi.org/10.17159/2309-8775/2018/v60n2a4
TECHNICAL PAPER
Can detrimental carbonation of cement or lime-stabilised road base layers, and the occurrence of biscuit layers as a result of carbonation, be controlled by proper construction techniques only?
C J Semmelink; N J J Jooste; J E Raubenheimer
ABSTRACT
According to international standards, the majority of paved roads in South Africa are considered as low-volume roads. For this reason, the use of stabilised base layers under thin surfacings is still extensively used in the rehabilitation of these roads and the construction of new road pavements. In this spectrum of roads, problems have occurred with regard to the cementation of such layers. In most cases, these issues arise at the very top of these base layers, and the most common explanation has usually been described as "detrimental carbonation" or similar wording. It is especially the early problems within the construction period and soon thereafter that are under scrutiny in this paper.
This paper presents an update on the experience with chemically stabilised base layers in South Africa, and shows how the so-called "detrimental carbonation" is not a situation that can be eliminated during construction. The paper further argues the reason why the problem is material-related, and that a water-driven reaction probably causes it. Other opinions are that carbonation is the only cause and that carbonation is unaffected by moisture. A further opinion is that standard prescribed compaction techniques, curing and the application of prescribed bituminous types of prime can also be the cause of so-called "detrimental carbonation". The conclusion is that the problem is moisture or "water-driven", as water vapour is required for the carbonation reaction to take place.
Keywords: carbonation, cement, relative humidity, moisture, biscuit layer
INTRODUCTION
Ever since the onset of the construction of cement-stabilised base layers, problems have occurred with regard to the cementation of such layers, especially at the top of these base layers under thin surfacings. The most common explanation is usually described as "detrimental carbonation" or similar wording. While a cemented base option may be considered on low-volume roads, this type of design has inherent risks associated with it, both from a design and construction perspective. Crushing calculations assuming a lightly cemented C4 base and using a design tyre pressure of 750 kPa suggest that crushing should be expected in less than a year. Even a C3 base layer may be a risk, since the TRH4 Catalogue assumes a design tyre pressure of 520 kPa, which is no longer applicable to South African roads (GAUTRANS 2004). A soft layer at the top of a chemically stabilised base layer may well also be the result of a number of factors, or a combination of factors. These other related factors that may also contribute to the formation of a soft layer at the top of a cemented layer may include:
-
Stabilisation design, especially with regard to satisfying the initial consumption of stabiliser
-
The addition of too much water during the compaction process
-
Premature spraying of water as part of the curing process, which may have the same effect as above
-
Wet-dry cycles caused by the specified curing process
-
Type of prime and application rate.
The focus of this paper is carbonation during the construction period and soon after opening during the contract's retention period. General knowledge about the problem, and some of the other documents mentioned in this paper, are of the opinion that carbonation referred to as "detrimental carbonation" can be avoided through the construction and curing processes. This paper shows why the authors consider the problem to be moisture or "water-driven", as water vapour is required for the carbonation reaction to take place.
PERSPECTIVES FROM LITERATURE
The main arguments in some other published papers on the subject of carbonation are the following:
-
Goodbrake et al (1979) determined that the percentage of calcium silicate reacting with carbon dioxide reduces significantly below a relative humidity of 50% and is relatively constant (about 75%) at a relative humidity in excess of 50%.
-
Roberts (1981) stated that "... carbona-tion is greatest at ordinary temperatures in the relative humidity range of 50 to 75 percent." (Page 15)
-
Paige-Greene et al (1990) stated: "The influence of relative humidity on the rate of carbonation, although extremely important as a source of moisture necessary for the reaction to occur, is not clearly defined."
-
Ballim and Basson (in Fulton 2002 page 150) stated that no carbonation takes place when the pores are completely dry or when they are fully saturated, and that the rate of carbonation also increases with increasing ambient temperature.
-
Botha et al (2005) presented a paper at the TREMTI Conference in Paris in 2005 indicating that many of the problems in South Africa that were attributed to carbonation, were caused by "water-driven reactions" and were thus material-related and not construction-related.
-
Wikipedia, the free online encyclopaedia, defines humidity as a term for water vapour in the air. Relative humidity measures the current absolute humidity relative to the maximum humidity and is expressed as a percentage (Wikipedia n.d.A.).
-
Wikipedia, the free online encyclopaedia, also states the following about the effect of carbonation on phenolphtha-lein: "The acid-base indication abilities of phenolphthalein also make it useful for testing for signs of carbonation reactions in concrete. Concrete has naturally high pH due to the calcium hydroxide formed when Portland cement reacts with water. The pH of the ionic water solution present in the pores of fresh concrete may be over 14. Normal carbonation of concrete occurs as the cement hydration products in concrete react with carbon dioxide in the atmosphere, and can reduce the pH to 8½ to 9, although that reaction usually is restricted to a thin layer at the surface. When a 1% phenolphthalein solution is applied to normal concrete, it will turn bright pink. If the concrete has undergone carbonation, no colour change will be observed." (Wikipedia n.d.B.)
-
In an abstract in a TREMTI 2010 paper, Paige-Greene (2010) states the following: "During the early 1980s, a number of problems related to the loss of stabilisation and disintegration of stabilised layers in roads (lime and cement) were reported in South Africa. This led to many comprehensive investigations, and it was shown without any doubt that the problems were related to carbonation of the stabilised materials. A paper was presented by Botha et al (2005) at the TREMTI Conference in Paris in 2005 indicating that many of the problems in South Africa that were attributed to carbonation, were actually caused by 'water-driven reactions' and were thus material-related and not construction-related. This paper assesses the fundamental principles of each of the processes and draws conclusions as to their likelihood and the increasing occurrence of stabilisation problems. It is concluded that, although there is indubitable proven field and laboratory evidence for carbonation of stabilised layers, there is no solid scientific evidence for the occurrence of the 'water-driven reactions' in soil stabilisation for roads."
The paper also states that the car-bonation reaction depends on the solubility and diffusion of the components. "The diffusion is controlled by the concentration differences and is an inward diffusion of CO2 gas and carbonate ions (Lagerblad 2005). The gas diffusion is much faster than ion diffusion. Thus the rate of reaction is controlled by the humidity in the material, i.e. how much liquid fills the connected pore system. In dry material, the CO2 can penetrate well, but there is insufficient water for the reaction to take place. ln the saturated condition, only the carbonate ions move, and carbonation is slow. Typically, the reaction is most likely and rapid at humidity levels of 40% to 70% (Lo & Lee 2002; Ballim & Basson 2001; Gjerp & Oppsal 1998)."
ARGUMENTS WHY CARBONATION IS WATER-DRIVEN
Assume for the moment that "poor curing techniques cause detrimental carbonation" of the stabilised base surface during and after the construction thereof.
As everyone involved in road construction should know, all cement-stabilised layers are compacted at optimum moisture content (OMC). However, priming of the base is done at roughly 50% of OMC (i.e. the moisture content where the inter-particle suction forces of remaining water in the layer peak in order to ensure proper adhesion of the road-prime to the cement-stabilised layer). From the earlier statements it is clear that there are only two points in cement-treated materials' moisture regimes (including concrete) where no carbonation takes place, namely 0% (i.e. totally dry) or 100% saturated so that CO2 cannot be taken up in the pores of the material.
From the above-mentioned construction facts it is clear that the cement-stabilised layer is never totally dry or 100% saturated at any time during its life cycle. If one were to try and prevent carbonation from taking place by saturating the layer, the layer would start pumping under traffic, because inter-particle stresses are reduced by the pore water pressure in the saturated voids. So this is not a workable solution. If hypothetically the layer could be dried back rapidly to 0% moisture content, the layer would probably ravel because of the lack of inter-particle suction forces (which is clearly seen on any very dry gravel road), as the added amount of cement is insufficient to ensure chemical bonds between all particles. Furthermore, it is impossible to compact a totally dry material effectively. It is also clear that water vapour is needed for the carbonation reaction to take place (i.e. water-driven reaction).
Because the cement-stabilised layers are primed at about 50% of the OMC, a substantial amount of the inter-particle voids will not be filled with water at the time of priming. The moisture regime of these voids is situated right in the middle of the so-called active carbonation range of 40% to 60% of humidity. As the layer surface heats up during the day, the temperature of the layer also increases, causing the humidity in the inter-particle voids to increase. As the surface is sealed by the prime and surfacing layers, very little of this moisture in suspension as moisture vapour is lost to the atmosphere. Prior to priming, the surface layer's moisture content also fluctuates between totally wet and totally dry each time the stabilised layer is sprayed with water for curing as specified. This causes a number of carbonation cycles in the stabilised layer each day, as no contractor can keep the layer in a totally saturated or totally dry condition all the time.
However, the carbonation reaction does not stop after the curing period, but continues as long as there is free lime available in the material, and the moisture content of the stabilised layer is in the active car-bonation range of 40% to 60% humidity. As the TREMTI 2005 paper showed (Botha et al 2005), the loose surface material was broomed off before priming and surfacing of the cement-stabilised layer (i.e. normal practice). Later pictures (see photographs of the project in the Appendix on pages 38-39) of the same layer surface show that a loose (dry) powdery layer had formed on top of the surface of the cement-stabilised layer just below the prime and the surfacing. This dry layer is very unusual, as the pavement layers under a surfaced road are normally moist below the road surfacing. What is more, the dark pink colour of the sprayed phenolphthalein solution on the material shows that there is still abundant free lime (i.e. cement) available in the material, which rules out detrimental carbonation.
Despite the presence of free lime from the cement, the surface ravelled under the surfacing because there was no inter-particle suction force between the particles, due to the dry material. However, because it is usually moist (damp) below the surfacing, one can reason in this case as well that the dryness of the material was not caused by evaporation of moisture through the prime and bituminous road surfacing, but was possibly caused by exceedingly high intra-particle suction forces of certain minerals (e.g. illite-smectite) in the material matrix that sucked the inter-particle water into the mineral particles themselves to form part of the chemically bound water, which can only be driven off at exceedingly high temperatures in the region of 600°C. Thus normal oven-drying at 100°C will not show the presence of the chemically bound water at all. This is why the authors are convinced that this damage is caused by a water-driven reaction, despite the fact that no proven chemical reaction or proven physical explanation for this dry powdery layer could be given in this case.
Other evidence from literature
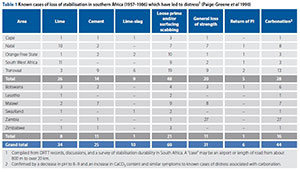
Paige-Greene et al (1990)
Paige-Greene et al (1990) stated: "Distress or failure assessed to be primarily due to loss of stabilisation has occurred in at least 100 cases in southern Africa over the last 30 years." [See Table 1 in Appendix.] "This represents an average rate of about three per year. The most common form of distress is surface disintegration of the primed base during construction and scabbing of the seal in service due to inadequate bond with the base. In many cases a loose layer of disintegrated base course material was noticed between the surfacing and the base. A less frequent occurrence has been a partial or even complete loss of cementation, and a large decrease in strength, leading to rutting, cracking and shearing or pumping. In a few cases an increase in the plasticity index has been found. Lime, cement and lime-slag were used as stabilisers, in amounts ranging between 2% and 5% by mass. Carbonation has been confirmed, or is strongly indicated, to have been a factor in about half of the cases. In the remainder of cases the available information does not permit an assessment of whether or not carbonation was involved." (Page 2)
In the same document (Paige-Greene et al 1990) they also state: "The influence of relative humidity on the rate of carbon-ation, although extremely important as a source of moisture is necessary for the reaction to occur, is not clearly defined. Addis (1986) includes a figure showing that carbonation shrinkage is a maximum at a relative humidity of about 50 per cent and decreases to zero at 100 per cent. Goodbrake et al (1979) have determined that the percentage of calcium silicate that reacted with carbon dioxide declines steeply below a relative humidity of 50 per cent and is more or less constant (about 75 per cent) at a relative humidity in excess of 50 per cent. Roberts (1981) states that carbonation is greatest at ordinary temperatures in the relative humidity range of 50 to 75 per cent." (Page 15)
The following techniques can be used to ascertain whether carbonation has occurred (Paige-Greene et al 1990 pp 18-20):
1. Phenolphthalein (Netterberg 1984 in Paige-Greene et al 1990)
pH < 8.4 pH pH < 11
Colourless ......Pink .............Red
2. Dilute HCI (5N)
Ca(OH)2 , CSH, CAH: no effervescence
CaCO3: effervescence
3. pH (paste or low water: soil ratio)
Ca(OH)2 , OPC: > 12.4
CSH, CAH : = 11.0 to 12.4
CaCO3: ± 8.3
As the reaction products are only stable at a pH of 11 to 12.5, further work is necessary to find a reliable field method of measuring such pH levels. Of all the indicators tried, only phenolphthalein appears to be reliable in all cases. However, this can only indicate when the pH is in excess of about 10 (and less than about 8.4).
De Wet and Taute (1985)
De Wet and Taute (1985) stated: "The best humidity for the test has not yet been ascertained. Carbonation should be fastest at 40-60%. However, this is an air-dry condition which will take time to achieve in the pores of the specimen (which are about 75% saturated with water at OMC) . If vacuum soaking is carried out, the specimen should be placed in the vacuum chamber, a vacuum of 80 kPa applied slowly over a period of 10 minutes, the vacuum tap closed, and water slowly allowed to enter the chamber over a period of 10 minutes to completely cover the specimen and to release the vacuum. Allow to soak for 10 minutes and remove specimens."
The carbonation reaction is therefore shown here to be water-driven.
Lowe and Von Solms (1986)
Lowe and Von Solms (1986) concluded:
-
"In South West Africa [Namibia today] lime stabilisation of calcrete has generally been successful, but on occasion unsuccessful. It seems that the quality of the end product depends on the composition of the calcrete and the reaction of the lime with the material." (Point 4.1)
-
"The presence or not of free lime tested with phenolphthalein, and of CaCO3 as tested with HCl, does not necessarily coincide with the strength of the layer, neither do soft layers within the stabilised layer necessarily contain no free lime. Some base courses having free lime were soft, whereas others without free lime were hard." (Point 4.2)
-
"It seems that on lightly trafficked roads in South West Africa natural calcretes well outside the normal specified plasticity and grading limits may perform equally well, if not better, than the stabilised calcretes." (Point 4.3)
-
"Calcretes which have a low strength should not be corrected by stabilisation with lime. Cement stabilisation may be the answer." (Point 4.4)
Ballantine and Rossouw (1989)
Ballantine and Rossouw (1989) stated: "It is important to note that destructive carbonation is at a minimum when the pH is high. The records show that in most cases where destructive carbonation has occurred the pH was generally below 12. There is strong evidence that carbonation is not always destructive. Tests on stabilised layers, more than ten years old, have shown severe carbonation, yet no loss in strength was evident."
Weinert (1980)
-
Weinert (1980) stated (page 50): "The degree of carbonation depends on the availability of suitable ions and the retention of usually soluble carbonates in weathering rock or the soil. Therefore, although water is required for the formation of carbonates, it must not be present in such quantities that the carbonates are dissolved and removed. This is probably the reason why the formation of certain carbonates, especially calcrete, is most pronounced where the N is between 5 and 10."
-
Weinert (1980) further stated (page 96): "Although the theoretical background of this initial consumption of lime is not fully understood, it appears that it is related to the compositional instability of certain minerals. Such conditions occur especially in minerals which are decomposing into clay, and are apparently intensified when the process tends to produce montmorillonite rather than kaolinite. The conditions also occur in montmorillonitic shales, particularly when the latter contain an appreciable quantity of opaline silica (SiO2.nH2O)."
CONCLUDING REMARKS
-
From the evidence presented it is clear that carbonation cannot take place without a certain amount of water vapour present in the material (i.e. water-driven reaction).
-
Furthermore, it is clear that cement-stabilised layers are never totally dry or saturated during their working life, and therefore the moisture in the layer has a direct bearing on the carbonation of a cement-stabilised layer.
-
Therefore it is misleading to conclude that destructive carbonation is only construction-related, as it is impossible to ensure on site that the stabilised material is either totally dry or totally saturated, be it during construction or the working life of the pavement.
-
In most cases of dispute, one cannot assume that, because the material seems suitable and the laboratory design is workable, the road builder is responsible when the required results are not achieved. This is not a balanced approach, as it is also laboratory-design-based (i.e. using accelerated material testing techniques not reflecting actual construction constraints experienced on site), and not only construction-based. From the evidence of the original report by Paige-Greene et al (1990) it is clear that the authors of that report found that detrimental carbonation had only been confirmed, or was strongly indicated to have been a factor, in about 50% of the cases investigated. Twenty years later in the 2010 TREMTI paper by the same author (Paige-Greene 2010) on the same subject, the conclusion was made that this loose powdery layer underneath the surfacing and prime is caused by carbonation, which implies poor curing techniques, thus making it a construction-related problem only.
-
The authors of this paper are of the opinion that the statement in the 2010 TREMTI paper by Paige-Green (2010) is incorrect, as the dark pink colour of the sprayed phenolphthalein solution on the so-called carbonated material found in the survey done in the investigation as set out in the Appendix, contradicts this statement. One would further expect the whole surface of the "poorly cured" stabilised layer evaluated (see Appendix) to be affected by the detrimental "car-bonation", which is not the case either. In actual fact, this particular road shown in the Appendix has been trafficked for more than ten years without any major surface failures. Keep in mind that all loose surface material had been removed from the stabilised road surface before priming and surfacing the road. From the evidence presented it should be clear that in at least 50% of the cases this loose powdery layer is probably caused by an as yet undefined water-driven reaction which is beyond the control of the road builder and therefore not construction-related. Lowe and Von Solms (1986) came to a similar conclusion.
-
Furthermore, material-related construction problems will only be identified in the laboratory if the laboratory test procedures simulate site construction conditions.
-
"Not everything in a TRH or other document is necessarily valid indefinitely: it only represents the state-of-the-practice or else the best knowledge available at that time. As an interim measure the normal TRH 14 (NITRR 1985b) and TRH 13 (NITRR 1986a) requirements should be applied to the residual values." (Paige-Greene et al (1990) (p 34).
-
It is evident that more research is required to fully understand the cause of this phenomenon.
SUMMARY OF EVIDENCE
1. Water or water vapour is required for the carbonation reaction to take place (i.e. a water-driven reaction).
2. There are only two points in the moisture regime of a cement-stabilised layer where carbonation does not take place, namely totally dry or totally saturated.
3. The structural layers of a road below the prime layer are never totally dry or totally wet.
4. Phenolphthalein colours the cement-stabilised material pink when the material has not carbonated and remains colourless when carbonated, and therefore shows that the road investigated in 2001 had not carbonated (see Appendix).
5. Carbonation was identified to have played a part in the detrimental condition of the cement-stabilised layers in only 50% of sites investigated by Paige-Greene et al (1990). The other 50% was therefore probably caused by another as yet unidentified water-driven reaction.
6. The evidence clearly shows that both detrimental reactions are water-driven, which is beyond the control of road builders and therefore not a construction-related issue only.
7. The safest curing process is therefore to do no curing on site with a water cart, but rather to leave it as is.
REFERENCES
Ballantine, R W & Rossouw, A J 1989. Stabilisation of Soils. Johannesburg: PPC Lime. [ Links ]
Botha, P B, Semmelink, C J & Raubenheimer, J 2005. The real cause of detrimental carbonation in chemically stabilised layers and possible solutions. Paper presented at the Treatment and Recycling of Materials for Transport Infrastructure (TREMTI) Conference, October, Paris. [ Links ]
De Wet, L F & Taute, A 1985. Durability of stabilised materials. Proceedings, Annual Transportation Convention, Pretoria. FB, Paper 1. [ Links ]
Fulton 2002. Fulton's Concrete Technology. Johannesburg: Concrete Institute of South Africa. [ Links ]
GAUTRANS 2004. Stabilisation Manual. Manual 2/04. Johannesburg: Gauteng Department of Public Transport. [ Links ]
Goodbrake, C J, Young, J F & Berger, R L 1979. Reaction of beta-dicalcium silicate and tricalcium silicate with carbon dioxide and water vapor. Journal of the American Ceramic Society, 62(3-4): 168-171. [ Links ]
Lowe, E H & Von Solms, C L 1986. Experience and practice with modified basecourses in South West Africa. Paper presented at the meeting at which TRH 13 [Cementitious Stabilizers in Road Construction] (1986) was introduced to practice. [ Links ]
Paige-Greene, P 2010. A reassessment of problems affecting stabilised layers in roads in South Africa. Paper presented at the Treatment and Recycling of Materials for Transport Infrastructure (TREMTI) Conference, May, Madrid. [ Links ]
Paige-Greene P, Netterberg F & Sampson, L R 1990. The carbonation of chemically stabilised road construction materials: Guide to its avoidance. South African Roads Board Project Report, PR 89/146/1. Pretoria: Department of Transport. [ Links ]
Roberts, M H 1981. Carbonation of Concrete with Dense Natural Aggregate. Building Research Establishment (BRE) Information Paper 6/81. Watford, UK: BRE. [ Links ]
Weinert, H H 1980. The Natural Road Construction Materials of Southern Africa. Cape Town: Academica. [ Links ]
Wikipedia n.d.A. Definition of water vapour. Available at: https://en.wikipedia.org/wiki/Humidity. [ Links ]
Wikipedia n.d.B. Phenolphthalein. Available at: https://en.wikipedia.org/wiki/Phenolphthalein. [ Links ]
 Correspondence:
Correspondence:
Dr Christiaan Semmelink
67 Innes Road, Lynnwood Glen, Pretoria 0081, South Africa
T: +27 12 361 2738
E: conprosol@telkomsa.net
Nicolaas (klasie) Jooste
POBox 173, Newlands0049, South Africa
T: +27 82 654 7222
E: klasiejooste@gmail.com
Jacobus (koos) Raubenheimer
PO Box 13454, Noordstad 9302 South Africa
T: +27 51 250 4251
E: koos.r@raubex.com

DR CHRISTIAAN SEMMELINK Pr Eng, born in 1942, received his tertiary education at the University of Pretoria, including a PhD in Civil Engineering (1992) and an MBA (1979). He was employed by the NITRR at the CSIR for 29 years, where he did research on statistical quality assurance, single and double-surfacing seal designs, the alkali-aggregate reaction problem in concrete and the K-mould research on determining the design properties of treated and untreated road building materials. Together with the late Mr Piet Botha they established that detrimental carbonation was water-driven.

NICOLAAS (KLASIE) JOOSTE Pr Eng retired five years ago as a partner in a consulting civil engineering practice at the age of 65. He holds the degrees BSc (Eng) and BSc (Eng) (Hons), both from the University of Pretoria. His fields of knowledge are roads related, with more than 45 years of experience working predominantly for Provincial and National Roads Authorities.

JACOBUS (KOOS) RAUBENHEIMER Pr Eng retired in 2017 as chairman of the Board of the Raubex Group at the age of 74. He holds a BSc Eng (Civil) degree from the University of Pretoria. He worked for road authorities and a roads contractor for most of his life, and since 1974 for the Raubex Group, all mainly as a road and bridge builder.
Photographs taken during an investigation by Semmelink and Botha in 2001






Photographs taken in March 2012 of the same road