Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
Journal of the South African Veterinary Association
versão On-line ISSN 2224-9435
versão impressa ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.93 no.2 Pretoria 2022
http://dx.doi.org/10.36303/jsava.516
SHORT COMMUNICATIONS
Intra- and inter-operator variability of refractometric total proteins measurement of canine plasma
F VenierI; W JamontI; K McLennanII; C RosaI
INorthwest Veterinary Specialists, United Kingdom
IIDepartment of Biological Sciences, University of Chester, United Kingdom
ABSTRACT
Refractometric total proteins are commonly used in practice as a quick and inexpensive way to measure total protein concentration in bodily fluids. Little information is available about how the operator performing the measurement affects the results.
The aim of our study was to determine the inter- and intra-operator variability of refractometric total proteins measured on canine plasma using a temperature-compensated handheld refractometer.
A pooled sample of canine lithium-heparin plasma was created using leftover samples from dogs presented to our hospital. The sample was then divided into three aliquots. Total proteins of these aliquots were measured by veterinary nurses, interns, residents and specialists working at our hospital.
Statistical analysis revealed excellent inter-operator (ICC 0.99, CI 95% 0.971-1.00) and intra-operator (ICC 0.997, CI 95% 0.9900.999) variability.
Having different operators measuring refractometric total plasma proteins in practice should not affect the results. This suggests different operators can be used when monitoring total plasma proteins of a patient over time and when designing a study that involves this test.
Keywords: dog, plasma, refractometer, total proteins, variability
Introduction
Handheld temperature-compensated refractometers were introduced into the medical field in the 1960s. They allow measurement of the total proteins dissolved in a solution by measuring the angle of refraction between room air and the sample of interest. Because proteins are the main solute of plasma and body fluids other than urine, refractometers are commonly used as a quick and inexpensive way to measure total protein concentration in these bodily fluids in both humans and veterinary patients (George 2001). They are also commonly used for the determination of urine-specific gravity (USG). Studies investigating the effect on the refractometer model and sample storage on measurement of USG are available in the literature (Du Preez et al. 2020; Mosch et al. 2020; Rowe et al. 2021; Tvedten & Noren 2014). Other studies have focused on refractometric measurement of total proteins and investigated how the results are affected by serum bilirubin (Gupta & Stockham 2014) and urea (Legendre et al. 2017). Information about the effect of the operator on refractometric measurements is less defined. Studies have assessed interobserver variability of USG. Du Preez et al. (2020) and Rowe et al. (2021) found good agreement between different operators when assessing USG with a refractometer. A study by Mosch et al. (2020) also found good inter-operator variability and intra-operator variability was acceptable. There are currently no similar studies investigating refractometric measurement of plasma proteins.
The aim of our study was to determine inter- and intra-operator variability of plasma proteins measured using a handheld refractometer. We also investigated the effect of the operator's job role (veterinary nurse, intern and clinicians). We hypothesised that operators that did the measurement more frequently (nurses and interns), would show less inter-operator variability compared to people that used the refractometer less often.
Material and methods
Leftover samples of canine whole blood in lithium-heparin-coated tubes were centrifuged at 2 500 g (Clinispin Horizon 842 Vet) for five minutes. Plasma was then used to create a pooled sample that was then divided in three identical aliquots (A, B and C) on the first day of the data collection and then stored at 4 °C until completion of the study. Data were collected over six days. Canine total proteins are stable in heparinised plasma for three days when stored at +4 °C (Thoresen et al. 1992), but information about the stability of this analyte for longer storage time is not available. Two of the researchers (FV and WJ) evaluated the refractometric total proteins of the three aliquots at the beginning of the data collection, after 24 hours and at the end of the data collection. This was done to assess stability of total proteins during the data collection period. All measurements were performed using a handheld temperature-compensated refractometer (RHCN-200ACT Combined Refractometer).
Qualified veterinary nurses, interns and clinicians working at our hospital were enrolled in the study. Participants were asked to measure refractometric total proteins of samples A, B and C. All participants were provided with the three samples, the refractometer, disposable transfer pipettes (drop size 50 µL), distilled water and a standard form to record their results. Participants were all blinded to the composition of the samples and to the results obtained by other people enrolled in the study and they were asked not to share their results with others. The number of participants was limited to the number of colleagues available during the period of data collection. Calibration of the refractometer was performed by each participant, and it was checked by one of the researchers (WJ) after each measurement. The researcher also calibrated the refractometer again after each participant completed the study.
All statistical analysis was carried out using R 4.1.3, (R Core Team, 2022). Data was tested for normality using the Kolmogorov-Smirnov Test in the "stats" package (R Core Team, 2022). Data on job role was not normally distributed (nurse: D = 1, p < 0.01; intern: D = 1, p < 0.01; resident D = 1, p < 0.01), nor was aliquot (sample A: D = 1, p < 0.01; sample B: D = 1, p < 0.01; sample C: D = 1, p < 0.01) according to the Kolmogorov test. Time was also not normally distributed (0h: D = 1, p < 0.01; 24 h: D = 1, p < 0.01; 144 h: D = 1, p < 0.01). Levene's test for homogeneity of variance using the "car" package (Fox & Weisberg 2019) showed that the data for the operators job role was non-significant (F = 2.271(2,150), p = 0.763; nurse: F =1.54(2,90), p = 0.221; intern: F = 0.231(2,9), p = 0.80; resident: F = 0.473(2,45), p = 0.626), as was the data for aliquot (F = 2.203 (2,150), p = 0.114), sample A: F = 0.503(2,48) p = 0.61; sample B: F = 0.269(2,48), p = 0.765; sample C: F = 0.194(2,48), p = 0.825). To account for any observer effects across the three aliquots, a repeated measures mixed effects model using maximum likelihood to account for the non-normality, but nonsignificant variability of the data (package "nlme", Pinheiro et al. 2022) was used to determine if the job role or aliquot sample had a significant effect on the measurement of total proteins. To assess if any change in the concentration of total proteins occurred in the samples over time, a repeated measures one-way ANOVA was carried out using the "stats" package (R Core Team, 2022) followed by pairwise comparison using a Bonferroni-corrected p-value. Levene's test for time was also non-significant (F = 0.031 (2,15), p = 0.97). Inter- and intra-operator variability was determined by calculating the Intraclass Correlation Coefficient (ICC). Inter-operator variability was assessed using a two-way random effects model for absolute agreement using mean of multiple raters via the "irr" package in R (Gamer et al. 2019). Intra-operator variability was assessed using a two-way mixed effects model for absolute agreement, of the average score. The ICC was interpreted following recommendations by Koo and Li (2016): < 0.50 poor correlation; 0.50-0.75 moderate correlation; 0.75-0.90 good correlation; > 0.90 excellent correlation. Statistical significance was set at p < 0.05.
Results
Fifty-one people were enrolled in the study. These included 31 nurses, four interns and 16 clinicians (residents or specialists). The data collection was completed over six days to allow enrolment of an adequate number of participants. Measurements were performed by 28 participants on day 0, 18 participants on day 1 and by 5 participants on day 6.
Repeated reassessment of the total protein concentration of aliquots A, B and C, showed a statistically significant difference over time (F = 5.466 (2,15), p = 0.02), with differences between 24 h and 144 h being significant (p = 0.024) and differences between 0 h and 24 h (p > 0.05) and 0 h and 144 h (p = 0.058) not being significantly different. The difference between total protein concentration at the start of the study and at its end was 3 g/L for all samples (5% increase).
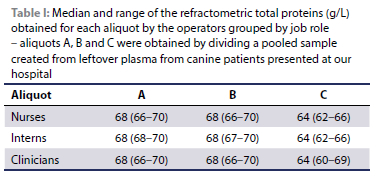
A significant difference was found between the total proteins concentration of sample C compared to sample A and B (p < 0.05), with measurements of sample C being 4% lower. Intraclass correlation coefficient showed excellent correlation between operators when data from nurses, clinicians and interns were considered together (ICC 0.99) with a small confidence interval (CI 95% 0.971-1.00, p < 0.01). The ICC for intra-operator variability was also excellent (ICC = 0.997, p < 0.01), with a small confidence interval (CI 95% 0.990-0.999).
The measurements were not affected by the job role of the operator (p = 0.302) (Table I).
Discussion
In our study, excellent correlation was found between repeated measurements of total proteins on lithium-heparin plasma samples using a handheld temperature-compensated refractometer, performed by the same operator on different aliquots obtained from a pooled sample, suggesting measurements using this technique are repeatable. This information is relevant in clinical practice where total proteins may be monitored over time on the same subject to determine procession of disease or response to treatment.
Our results also show excellent correlation between different operators measuring refractometric total proteins. These results are relevant in clinical practice, where it is unlikely that the same operator will be performing all the measurements. They are also relevant for research because they suggest having different operators measuring refractometric total proteins should not affect the data collected. This is especially relevant for retrospective studies, where information about who performed the measurements may not be available. Total protein measurements were not affected by the job role of the operator, suggesting that experience and familiarity with the technique didn't have a significant impact on the results. Participants to our study included qualified veterinary nurses, interns (qualified veterinarians with at least one year of experience) and clinicians (residents, post-residency clinicians and boarded specialists). This was a convenience sample limited to the staff working in our hospital. Veterinary students, nurse students and laboratory technicians may also be asked to measure refractometric total proteins in veterinary practice. Further studies are needed to determine if our results can also be applied to these professional figures as their familiarity with the test is likely to be different from our study sample.
Analytical, pre-analytical and post-analytical factors can all affect the result of a test. Our study was designed to determine variability of total proteins concentration secondary to the operator performing the test and other sources of variability were not assessed.
Total proteins concentration in sample C was 4% lower than samples A and B. Since all three samples were created from the same pooled sample, this result was unexpected. This could have been due to inadequate mixing of the pooled sample before dividing it into the three aliquots. There is no guideline that defines the allowable total error for refractometric total proteins, however current guidelines define the allowable total error for total proteins to be 10% (Harr et al. 2013). Since the difference between aliquot C and the other aliquots was within this limit, the discrepancy between the readings was deemed acceptable.
The concentration of total proteins in our pooled sample increased by 5% during the data collection. Dehydration of the sample is a possible explanation. The increase was not considered clinically relevant because it was lower than the allowable total error for total proteins (Harr et al. 2013).
Conclusion
Inter- and intra-operator variability of refractometric measurement of total proteins is low. Low inter-operator variability suggests that having different operators measuring total proteins on canine plasma using a refractometer should not affect the results. The job role of the operator did not affect the variability of the results in our study, but our study sample included only qualified veterinarians and nurses. Further studies are needed to determine if our findings can also be applied to other professional figures that may be required to measure total proteins in clinical practice.
Acknowledgements
The authors would like to thank all the colleagues that participated in this study.
Conflict of interest
The author(s) declare that they have no financial or personal relationship(s) that may have inappropriately influenced them in writing this article.
Funding source
No funding was received for this study.
Ethical approval
The author/s declare that this submission is in accordance with the principles laid down by the Responsible Research Publication Position Statements as developed at the 2nd World Conference on Research Integrity in Singapore, 2010. Prior to the commencement of the study, ethical approval was obtained from the following ethical review board, the Ethics Review Panel of the Royal College of Veterinary surgeons (Ref 2020-31-Venier). All participants gave written consent to be enrolled in the study.
ORCID
F Venier https://orcid.org/0000-0003-3782-5746
K McLennan https://orcid.org/0000-0002-8888-540X
C Rosa https://orcid.org/0000-0002-0967-4381
References
Du Preez, K., Boustead, K., Rautenbach, Y., et al., 2020, Comparison of canine urine specific gravity measurements between various refractometers in a clinical setting, Vet Clin Pathol 49(3), 407-416. https://doi.org/10.1111/vcp.12892. [ Links ]
Fox, J. & Weisberg, S., 2019, An {R} Companion to Applied Regression, Third Edition, Thousand Oaks, CA: Sage, Available from: https://socialsciences.mcmaster.ca/jfox/Books/Companion/. [ Links ]
Gamer, M., Lemon, J., Singh, I.F.P., 2019, irr: Various Coefficients of Interrater Reliability and Agreement. R package version 0.84.1, Available from: https://CRAN.R-project.org/package=irr. [ Links ]
George, J.W., 2001, The usefulness and limitations of hand-held refractometers in veterinary laboratory medicine: an historical and technical review, Vet Clin Pathol 30(4), 201-210. https://doi.org/10.1111/j.1939-165X.2001.tb00432.x. [ Links ]
Gupta, A. & Stockham, S.L., 2014, Refractometric total protein concentrations in icteric serum from dogs, J Am Vet Med Assoc 244(1), 63-67. https://doi.org/10.2460/javma.244.1.63. [ Links ]
Harr, K.E., Flatland, B., Nabity, M., et al., 2013, ASVCP guidelines: allowable total error guidelines for biochemistry, Vet Clin Path 42(2), 424-436. https://doi.org/10.1111/vcp.12101. [ Links ]
Koo, T.K. & Li, M.Y., 2016, A guideline of selecting and reporting intraclass correlation coefficients for reliability research, J Chiropr Med 15(2), 155-163. https://doi.org/10.1016/j.jcm.2016.02.012. [ Links ]
Legendre, K.P., Leissinger, M., Le Donne, V., et al., 2017, The effect of urea on refractometric total protein measurement in dogs and cats with azotemia, Vet Clin Pathol 46(1), 138-142. https://doi.org/10.1111/vcp.12464. [ Links ]
Mosch, M., Reese, S., Weber, K., et al., 2020, Influence of preanalytic and analytic variables in canine and feline urine specific gravity measurement by refractometer, J Vet Diagn Invest 32(1), 36-43. https://doi.org/10.1177/1040638719896785. [ Links ]
R Core Team 2022. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria, Available from: https://www.R-project.org/. [ Links ]
Rowe, J.C., Hokamp, J.A., Braatz, J.N., et al., 2021, Interobserver reliability of canine urine specific gravity assessed by analog or digital refractometers, J Vet Diagn Invest 33(3), 611-614. https://doi.org/10.1177/1040638721998295. [ Links ]
Pinheiro. J., Bates, D., DebRoy, S., Sarkar, D., R Core Team 2022. _Nlme: Linear and Nonelinear Mixed Effects Modeles. R package version 3.1-155, Available from: https://CRAN.R-project.org/package=nlme> [ Links ].
Thoresen, S.I., Havre, G.N., Morberg, H., et al., 1992, Effects of storage time on chemistry results from canine whole blood, heparinized whole blood, serum and heparinized plasma, Vet Clin Path 21(3), 88-94. https://doi.org/10.1111/j.1939-165X.1992.tb00591.x. [ Links ]
Tvedten, H.W. & Noren, A., 2014, Comparison of a Schmidt and Haensch refractometer and an Atago PAL-USG Cat refractometer for determination of urine specific gravity in dogs and cats, Vet Clin Pathol 43(1), 63-66. https://doi.org/10.1111/vcp.12110. [ Links ]
 Correspondence:
Correspondence:
F Venier
Email: francesca.venier@nwspecialists.com














