Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
Journal of the South African Veterinary Association
versão On-line ISSN 2224-9435
versão impressa ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.93 no.2 Pretoria 2022
http://dx.doi.org/10.36303/JSAVA.238
CASE REPORT
Successful treatment of a South African cat with effusive feline infectious peritonitis with remdesivir
M Bohm
King Edward Veterinary Referral Hospital, South Africa
ABSTRACT
Historically, feline infectious peritonitis (FIP) has been considered almost invariably fatal. The recent COVID-19 pandemic has fuelled research in coronavirus pathophysiology and treatment. An unintended consequence is that we now have an effective treatment accessible for FIP. This paper reports on the successful resolution of immunohistochemistry-confirmed effusive FIP in an adolescent cat in South Africa following monotherapy with remdesivir at 4.9-5.6 mg/kg daily for 80 days.
Keywords: feline infectious peritonitis, FIP, remdesivir, effusive, cats, coronavirus, GS-441524, ascites, treatment, feline
Introduction
Feline infectious peritonitis (FIP) can be difficult to confirm ante mortem, as was the case here. Treatment with a wide variety of drugs has been attempted (Addie et al. 2009) but proved disappointing and most affected cats died or were euthanased. In the last five years, there have been reports of effective treatments but none have been accessible for clinical cases (Dickinson et al. 2020; Izes et al. 2020; Kim et al. 2016; Murphy et al. 2018; Pedersen et al. 2018; Pedersen et al. 2019). The COVID-19 pandemic resulted in the accelerated development and release of a number of drugs, including remdesivir, a prodrug of GS-441524. At the time this patient presented there were no published studies on the use of this molecule to treat cats with FIP though there were encouraging reports on the efficacy of GS-441524 (Dickinson et al. 2020; Murphy et al. 2018; Pedersen et al. 2019). Remdesivir does not cure all cats with FIP (Dr Richard Malik, personal communication), but for the first time, vets can offer clients a specific treatment with a reasonable chance of success.
Patient presentation
An eight-month-old neutered female domestic shorthaired cat was referred for investigation of ascites. The cat was born to a feral queen, was adopted into a house with one other cat at four weeks of age, but had continued contact with feral cats. She received routine vaccinations, deworming and ectoparasite control and was neutered at six months of age.
She was presented to the primary vet one month post ovariohysterectomy with lethargy and muscle loss, voluminous diarrhoea, an increased appetite and isolated instances of vomiting. Haematology documented a haematocrit of 0.36 and 0.38 l/l (reference interval [ref] 0.24-0.45 l/l), a neutrophilia (21 x 109/l, ref 2.5-12.5 x109/l) and a monocytosis (2.62 x 109/l, ref 0-0.8109/l) with mild hypoalbuminaemia (22 g/l, ref 25-40 g/l) and hyperglobulinaemia (53 g/l ref 33-39 g/l). Liver enzymes were within reference intervals. Benchtop tests for both retroviruses were negative. Thoracic radiographs were unremarkable. Abdominal effusion had a specific gravity of 1.042, a total protein of 60 g/l. The total nucleated cell count (TNCC) was 410 cells/µl with 66% non-degenerate neutrophils, 17% lymphoid cells, 16% macrophages and 1% eosinophils on cytology. Based on these parameters, the fluid was classified as a cell-poor exudate, consistent with effusive FIP. Immunohistochemistry on a pellet of cells harvested from 3 ml of abdominal effusion had failed to detect feline coronavirus.
At referral one month after initial presentation, rectal temperature was 40.1 °C, body condition score 2/5 and there was marked muscle wasting as well as obvious abdominal distension. Despite this, the cat remained lively and interactive and continued to eat well. The remainder of the clinical examination, including ocular examination, was unremarkable. Her packed cell volume was 32%. Blood smear revealed a mature neutrophilia. There was no striking monocytosis and platelet numbers were subjectively normal. There was mild anisocytosis and polychromasia. Serum protein electrophoresis (SPE) revealed an alpha 1 globulin spike. Beta globulins and gamma globulins were within feline reference ranges (Figure 1).
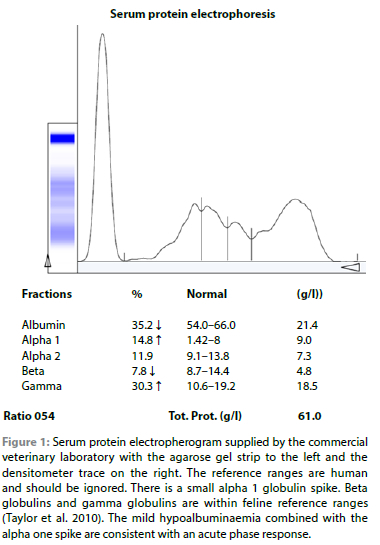
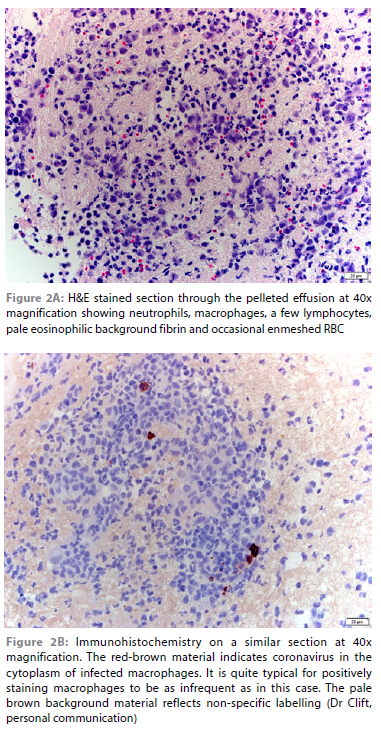
Abdominal ultrasound revealed no significant organ changes. There was a large volume of anechoic fluid; 500 ml of this was removed to improve patient comfort and ultrasound assessment. The effusion was transparent yellow with a total protein of 70 g/l and resulted in an obvious string when a drop was placed between two fingers drawn apart. The TNCC was not repeated. 80 ml of this fluid was submitted for repeat immunohistochemistry after discussion with the laboratory (Dr Sarah Clift, Onderstepoort Veterinary Academic Hospital's Pathology service). Several aliquots were spun down to ensure that there was a substantial pellet on which to perform the immunohistochemistry for feline coronavirus. This was duly detected (Figure 2).
Management and outcome
The cat was treated with remdesivir (Ciplamed Cipremi, 100 mg diluted in 19 ml saline, 2.8 ml per dose) for 80 consecutive days. She received no other treatment during this time. The first three doses of 5.6 mg/kg were given intravenously, the remainder subcutaneously. Each reconstituted vial of lyophilised powder was used over a week (14 mg daily) as studies had shown the molecule to be stable for a week at room temperature or when refrigerated (Nick Bova, BOVA Specials UK Ltd, personal communication). Her dose was not increased when she gained weight as her owner could only afford one vial of remdesivir per week. This meant that at the start of her treatment, she received 5.6 mg/kg per day, but by the end of her treatment, she received 4.9 mg/kg per dose, so was slightly underdosed. Within one week, she was visibly better in herself with reduced ascites. Diarrhoea initially worsened but then resolved as the ascites resolved. One month into treatment, her activity levels had increased, her appetite had decreased from ravenous to normal and she appeared like a normal cat to her owner. No pain on injection and no palpable injection site reactions were noted. Her coat improved, and she regained muscle. At re-examination three months after the start of treatment, she weighed 2.86 kg, had a body condition score of 3/5, normal muscling, and a clinical examination detected no abnormalities. At the time of writing, she remained asymptomatic and had been off treatment for seven months.
Discussion
A prevalent and longstanding theory was that FIP is caused when feline enteric coronavirus, an alphacoronavirus, mutates and in so doing attains the ability to enter the body and infect macrophages (Addie 2012). It was thought that clinical FIP develops when the host mounts an antibody-mediated rather than a predominantly cell-mediated immune response as the virus has adapted to use antibodies to enter and then persist in macrophages, but recent reviews suggest this may be an oversimplification (Addie 2012). Reviews of FIP as recent as 2014 reported that there was no effective cure (Addie 2012; Addie et al. 2009; Pedersen 2014). Palliative care revolved around immunosuppression with prednisolone or other corticosteroids but the disease was considered almost uniformly fatal (Addie et al. 2009) with effusive cases generally progressing more rapidly than cats with granulomatous disease and survival times of days to a few weeks (Addie 2012; Pedersen 2014). Occasional small studies would appear showing promising results with immunomodulators, but these were usually not duplicated in larger trials (Addie et al. 2009; Pedersen 2014). Studies on therapeutic efficacy have been hampered by the difficulty of using cats as a study subject as well as the difficulty in confirming the diagnosis of FIP beyond doubt - with histopathology, immunohistochemistry and/or qRT- PCR often only being available in a small proportion of cases (Izes et al. 2020; Pedersen 2009).
In 2018, treatment of 20 cats naturally infected with FIP with GC376, a 3C-like protease inhibitor, showed promise in as much as the molecule was able to reverse clinical signs. Unfortunately, signs recurred in many cases when treatment was withdrawn after two weeks (Pedersen et al. 2018). In the same year, studies with the experimental nucleoside analogue GS-441524 confirmed that it could be safely used in cats, and 10 cats experimentally infected with FIP were symptom-free after two weeks' treatment at 2-5 mg/kg oid subcutaneously. Two cats relapsed at four and six weeks but recovered after a further two weeks' treatment (Murphy et al. 2018). The following year treatment with 2-4 mg/kg GS-441524 for at least 12 weeks resulted in a lasting clinical cure in 18/31 cats with naturally acquired FIP, while a further seven recovered after one or more relapses were followed with further treatment (Pedersen et al. 2019). All relapses occurred within three months of completing treatment. Most of the cats that died had CNS involvement, but in a subsequent study, three of four cats with naturally acquired FIP with neurological signs recovered when treated with GS-441524 at 5-10 mg/kg (Dickinson et al. 2020). The main side-effects were pain at injection sites and injection site reactions (Pedersen et al. 2019). As GS-441524 is not commercially available, a burgeoning black market developed (Hughes et al. 2021; Izes et al. 2020), causing concerns about purity and efficacy of GS-441524 produced by third parties and ethics of its use.
The current COVID-19 pandemic caused by the related beta-coronavirus SARS-CoV-2 has led to an explosion of knowledge about coronavirdiae in general and a massive drive in developing new treatments. Remedsivir is a prodrug of GS-441524 that received emergency use authorisation by the US FDA for the treatment of COVID-19 in 2020 as an intravenous infusion. It was developed by Gilead, the same company developing GS-441524. Remdesivir generics are now produced by several companies under license from Gilead. Remdesivir was imported into South Africa to assist in the management of the second COVID-19 wave. This also occurred in Australia, where GS-441524 had been in use to treat cats with FIP. Access to GS-441524 was becoming difficult, which resulted in its replacement with remdesivir in some clinical cases with encouraging responses (Hughes et al 2021). Again, the main side-effect was pain at subcutaneous injections sites which could be mitigated against by using thin needles, replacing the needle used for aspiration of the drug with a new needle prior to injection, allowing the drug to warm up to room temperature prior to injection, applying EMLA cream or pre-treating with gabapentin and buprenorphine. In occasional cases where none of these interventions made subcutaneous treatment tolerable to the patient, a peripheral venous catheter was placed and re-sited every four to five days to allow intravenous treatment. Other reported side-effects included mild lethargy lasting a few hours immediately after IV injection, a transient worsening of pleural effusions, and temporary neurological signs including seizures (Hughes et al. 2021).
Recently published studies showed remdesivir's effect in COVID-19 cases to be much more modest than initially anticipated, and its high cost limited its use (Hsu 2020; Liao et al. 2021; Yan & Muller 2021). This means that while the drug is currently accessible, this may be a temporary situation in South Africa. Thankfully there appears to be emerging interest in GS-441524 as a treatment for COVID (Yan & Muller 2020), in part as oral bioavailability appears acceptable at least in the dog (Yan et al. 2021). In addition, BOVA Specials UK Ltd, a compounding pharmacy, launched remdesivir for veterinary use in the UK in August 2021. This means that an effective FIP treatment may eventually be accessible as an unintended benefit of the COVID-19 pandemic, and in the immediate future remdesivir appears to be an option.
Diagnosis of FIP cannot be made based on a single test (Felten & Hartmann 2019; Tasker 2018). In this case, signalment, history, biochemistry and effusion characteristics were consistent with effusive FIP. SPE did not support the diagnosis. Neither did the initial immunohistochemistry on 2-3 ml of effusion. Others have observed a discordance between SPE results and the final diagnosis (Giori et al. 2011). Based on the characteristics of the effusion, the main differential for this patient's clinical signs was lymphocytic cholang(iohepat)itis (Addie 2012; Tasker 2018). Her normal liver enzymes at initial presentation argued strongly against this being the underlying cause.
In this case, immunohistochemistry on a pellet of cells collected from 80 ml abdominal effusion revealed the presence of feline coronavirus. It appears likely that detection was made possible by submitting a much larger volume of abdominal effusion which then allowed the laboratory to harvest a much higher number of cells, increasing the odds of finding cells containing coronavirus, which are typically sparse (Sarah Clift, personal communication). In this case, the final proof was the prompt and complete response to remdesivir as sole therapy.
Acknowledgements
The author would like to thank Aurora Lambrecht of Project Fight FIP for disseminating information on remdesivir, Dr Sarah Clift for her advice on immunohistochemistry and for photographing the sections from this case, and Dr Richard Malik for providing insight and guidance into its storage and most appropriate use in cats with FIP when none was yet published.
Ethical approval
This paper does not involve human patients. The owner of this cat is aware that the case report has been written and provided verbal consent for its publication. This report details the management of a clinical case that was part of my routine clinical caseload. Management was not altered in any way by writing this case report.
ORCID
M Bohm: https://orcid.org/0000-0002-2416-3520
References
Addie, D., Belák, S., Boucraut-Baralon, C., et al., 2009, Feline infectious peritonitis. ABCD guidelines on prevention and management, J Feline Med Surg 11(7), 594-604. https://doi.org/10.1016/j.jfms.2009.05.008. [ Links ]
Addie, D.D., 2012, Feline Coronavirus Infections, in C.E. Greene (ed), Infectious Diseases of the Dog and Cat, 4th edn, Elsevier, United States of America, pp. 92-108. [ Links ]
Dickinson, P.J., Bannasch, M., Thomasy, S.M., et al., 2020, Antiviral treatment using the adenosine nucleoside analogue GS-441524 in cats with clinically diagnosed neurological feline infectious peritonitis, J Vet Intern Med 34(4), 1587-1593. https://doi.org/10.1111/jvim.15780 [ Links ]
Felten, S. & Hartmann, K., 2019, Diagnosis of feline infectious peritonitis: A review of the current literature, Viruses 11(11), 1068. https://doi.org/10.3390/v11111068 [ Links ]
Giori, L., Giordano, A., Giudice, C., et al., 2011, Performances of different diagnostic tests for feline infectious peritonitis in challenging clinical cases, J Small Anim Pract 52(3), 152-157. https://doi.org/10.1111/j.1748-5827.2011.01042.x. [ Links ]
Hsu, J., 2020, Covid-19: What now for remdesivir? BMJ 371, m4457. https://doi.org/10.1136/bmj.m4457. [ Links ]
Hughes, D., Howard, G., Malik, R., 2021, Treatment of FIP in cats with remdesivir, The Veterinarian, 19-27. [ Links ]
Izes, A.M., Yu, J., Norris, J.M., et al., 2020, Current status on treatment options for feline infectious peritonitis and SARS-CoV-2 positive cats, Vet Q 40(1), 322-330. https://doi.org/10.1080/01652176.2020.1845917. [ Links ]
Kim, Y., Liu, H., Galasiti Kankanamalage, A.C., et al., 2016, Reversal of the progression of fatal coronavirus infection in cats by a broad-spectrum coronavirus protease inhibitor, PLoS Pathog 12(3), e1005531. https://doi.org/10.1371/journal.ppat.1005531. [ Links ]
Liao, S.H., Hung, C.C., Chen, C.N., et al., 2021, Assessing efficacy of antiviral therapy for COVID-19 patients: A case study on remdesivir with bayesian synthesis design and multistate analysis, J Formos Med Assoc 120, Suppl 1, S77-S85. https://doi.org/10.1016/j.jfma.2021.04.026. [ Links ]
Murphy, B.G., Perron, M., Murakami, E., et al., 2018, The nucleoside analog GS-441524 strongly inhibits feline infectious peritonitis (FIP) virus in tissue culture and experimental cat infection studies, Vet Microbiol 219, 226-233. https://doi.org/10.1016/j.vetmic.2018.04.026. [ Links ]
Pedersen, N.C., 2009, A review of feline infectious peritonitis virus infection: 1963-2008, J Feline Med Surg 1 1(4), 225-258. https://doi.org/10.1016/J.jfms.2008.09.008. [ Links ]
Pedersen, N.C., 2014, An update on feline infectious peritonitis: diagnostics and therapeutics, Vet J 201(2), 133-141. https://doi.org/10.1016/j.tvjl.2014.04.016. [ Links ]
Pedersen, N.C., Kim, Y., Liu, H., et al., 2018, Efficacy of a 3C-like protease inhibitor in treating various forms of acquired feline infectious peritonitis, J Feline Med Surg 20(4), 378-392. https://doi.org/10.1177/1098612X17729626. [ Links ]
Pedersen, N.C., Perron, M., Bannasch, M., et al., 2019, Efficacy and safety of the nucleoside analog GS-441524 for treatment of cats with naturally occurring feline infectious peritonitis, J Feline Med Surg 21(4), 271-281. https://doi.org/10.1177/1098612X19825701. [ Links ]
Tasker, S., 2018, Diagnosis of feline infectious peritonitis: Update on evidence supporting available tests, J Feline Med Surg 20(3) 228-243. https://doi.org/10.1177/1098612X18758592. [ Links ]
Taylor, S.S., Tappin, S.W., Dodkin, S.J., et al., 2010, Serum protein electrophoresis in 155 cats, J Feline Med Surg 12(8), 643-653. https://doi.org/10.1016/j.jfms.2010.03.018. [ Links ]
Yan, V.C. & Muller, F.L., 2020, Advantages of the parent nucleoside GS-441524 over remdesivir for Covid-19 treatment, ACS Med Chem Lett 11(7), 1361-1366. https://doi.org/10.1021/acsmedchemlett.0c00316. [ Links ]
Yan, V.C. & Muller, F.L., 2021, Remdesivir for COVID-19: Why not dose higher? Antimicrob Agents Chemother 65(4), e02713-20. https://doi.org/10.1128/AAC.02713-20. [ Links ]
Yan, V.C., Pham, C.D., Yan, M.J., et al., 2021, Pharmacokinetics of orally administered GS-441524 in dogs, bioRxiv. https://doi.org/10.1101/2021.02.04.429674. [ Links ]
 Correspondence:
Correspondence:
M Bohm
Email: marlies@wol.co.za














