Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
Journal of the South African Veterinary Association
versión On-line ISSN 2224-9435
versión impresa ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.93 no.1 Pretoria 2022
http://dx.doi.org/10.36303/JSAVA.2022.93.1.492
CASE REPORT
Widespread metastasis of a spermatocytic seminoma with concomitant hepatic peliosis in a Southern African hedgehog (Atelerix frontalis)
N O'DellI, II; JH O'DellII, III; L van der WeydenIV
IDepartment of Paraclinical Sciences, Faculty of Veterinary Science, University of Pretoria, South Africa
IICentre for Veterinary Wildlife Research, Faculty of Veterinary Sciences, University of Pretoria, South Africa
IIIDepartment of Production Animal Studies, Faculty of Veterinary Science, University of Pretoria, South Africa
IVWellcome Sanger Institute, Wellcome Genome Campus, United Kingdom
ABSTRACT
A six-year-old intact male Southern African hedgehog (Atelerix frontalis) presented with a history of chronic mild to moderate weight loss, and sub-acute hind limb ataxia that progressed to complete paralysis, at which point the hedgehog was euthanised. At autopsy, a large multinodular pale mass had completely replaced the left testicle and transcoelomically metastasised to the diaphragm and the peri-renal area, from where it then invaded the vertebral column and spinal cord. Multifocal, irregular to round, well-demarcated, blood-filled, proliferative lesions were also present in the hepatic parenchyma. Histological analysis of both the testis and metastatic lesions revealed diffuse sheets of neoplastic cells with moderate pale cytoplasm, large irregular to round nuclei and mostly one prominent magenta nucleolus, consistent with metastatic seminoma. The neoplastic cells were negative for periodic acid-Schiff (PAS) stain and positive for CD117 by immunohistochemistry (IHC). Taken together with the morphology of the neoplastic cells and the advanced age of the animal, this is suggestive of a spermatocytic seminoma. Histological analysis of the liver revealed multifocal lesions consisting of large anastomosing blood-filled spaces bordered by compressed hepatocytes, consistent with hepatic peliosis. This is the first report of a neoplasm in the Southern African hedgehog (Atelerix frontalis), the first report of a metastatic seminoma in a hedgehog, together with diagnosis of spermatocytic subtype, and the first report of a hedgehog with concomitant hepatic peliosis.
Keywords: seminoma, Southern African hedgehog, Atelerix frontalis, spermatocytic, hepatic peliosis
Introduction
Seminomas are derived from the germ cells of the spermato-genic epithelium within the seminiferous tubules and are the only germ cell tumour that occurs frequently in the testicles of domestic animals. They are most prevalent in dogs, however, they have also been reported in other species, including horses (Farjanikish et al. 2016), goats (Cosentino et al. 2019), sheep (Shortridge & Cordes 1969) and rabbits (Banco et al. 2017), and most often occur in older animals (Agnew & MacLachlan 2017). However, seminomas have not been previously reported in a hedgehog. The present report describes a spermatocytic seminoma in a hedgehog with widespread metastasis and concomitant hepatic peliosis.
Case presentation
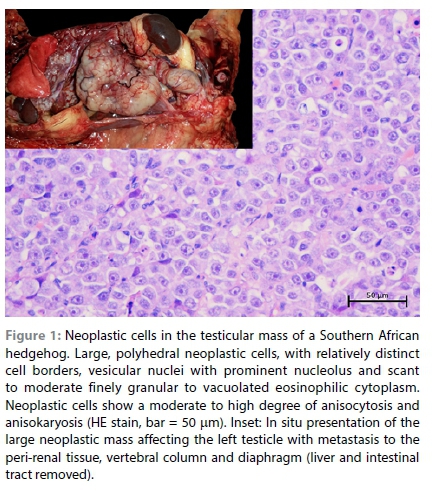
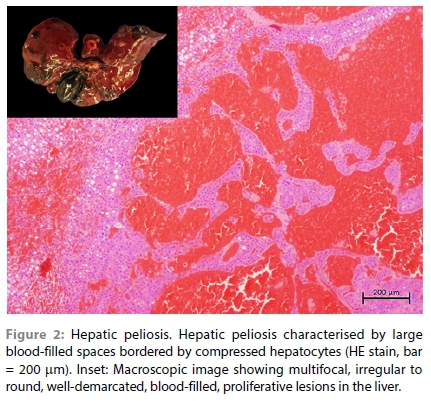
In this report, a captive six-year-old male intact Southern African hedgehog (Atelerix frontalis) presented with a two-week history of chronic mild to moderate weight loss, and subsequently developed hind limb ataxia. The ataxia became progressively worse over a period of two to three days, to the stage of complete paralysis with vocalisation (suggestive of pain), at which point the hedgehog was euthanised and an autopsy performed. Examination of the reproductive tract revealed a large, multinodular, pale, neoplastic mass that completely replaced the left testicle and extended along the spermatic cord to the peri-renal tissue and into the lumbar vertebral column and the associated spinal cord around L2 (Figure 1). Similar multifocal proliferative to plaque-like lesions were present on the peritoneal surface of the diaphragm (Supplementary Figure 1). The liver showed the presence of multifocal, irregular to round, well-demarcated, blood-filled, proliferative lesions in the hepatic parenchyma, with some of the lesions displaying a pale periphery with a dark red sunken centre (Figure 2).
Tissue samples from the liver and the neoplastic lesions in the testis, peri-renal tissue, vertebral column and diaphragm were fixed in 10% buffered formalin, embedded in paraffin wax and sectioned for histopathological evaluation. Sections (4 μιη) were stained with haematoxylin and eosin (HE) or periodic acid-Schiff (PAS). Immunohistochemistry (IHC) was performed on selected sections to evaluate the expression of CD117 (c-KIT) using a polyclonal rabbit anti-human CD117 antibody (Dako, A4502). After dewaxing, sections were treated with 3% hydrogen peroxide in methanol for 15 minutes, then antigen retrieval was heat induced in Tris-EDTA buffer (pH 9) for 21 minutes. The Super Sensitive™ Polymer-HRP IHC Detection System (Biogenex) was used according to the manufacturer's instructions, with Vector NovaRED® Substrate Kit (Peroxidase; Vector Labs) as the chromogen and Mayer's haematoxylin as the counterstain. All sections were examined by light microscopy by a veterinary pathologist.
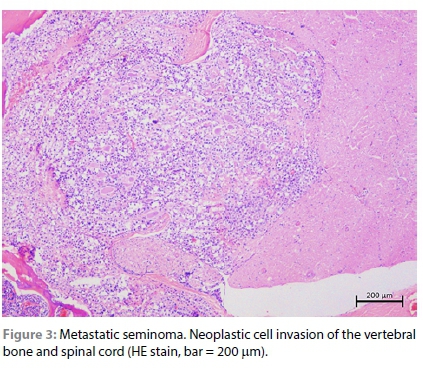
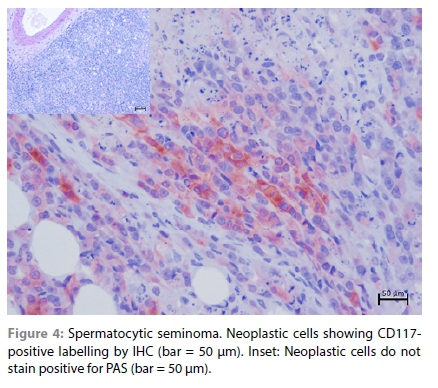
The samples from the lesions in the testis, peri-renal tissue, vertebral column and diaphragm revealed similar changes that consisted of diffuse sheets of neoplastic cells. The neoplastic cells were generally large in size, polyhedral in shape, with relatively distinct cell borders and a moderate to high degree of anisocytosis and anisokaryosis. The neoplastic cell nuclei were vesicular with variable chromatin coarseness containing mostly a large single magenta nucleolus. The scant to moderate cytoplasm was amphophilic to eosinophilic with a finely granular to vacuolated appearance (Figure 1). Degenerate cells were scattered throughout the sheets of neoplastic cells, resulting in the typical "starry-sky" appearance. In the larger mass, the degenerate cells often resulted in larger, fused areas of necrosis characterised by necrotic cellular debris and associated with occasional neutrophils (Supplementary Figure 2a). Only a few scattered areas of lymphocyte infiltration were observed in this case and this was not a common feature (Supplementary Figure 2b). The mitotic rate was high with an average of eight mitoses per high power field and bizarre mitoses were commonly observed (Supplementary Figure 2c). In the section of the diaphragm, sheets of neoplastic cells could be seen invading between the myocytes (Supplementary Figure 2d). In the section of the spinal cord, invasion of the vertebral bone and the spinal cord by neoplastic cells was clearly observed (Figure 3). The diagnosis was consistent with malignant seminoma that had metastasised to the peri-renal tissue, spinal cord and diaphragm. Since this is the first detailed histological analysis of seminoma in a hedgehog, it was unknown whether the division of seminoma into the classical (SE) and spermatocytic (SS) subtypes was feasible. The seminoma in this case was negative for PAS staining (Figure 4) and ~20% of the neoplastic cells revealed moderate to strong membranous and cytoplasmic labelling for CD117 (Figure 4). This staining pattern, together with the morphology of the cells and the advanced age of the animal, suggests it was most likely a SS. The multifocal lesions in the liver of this case consisted of large anastomosing blood-filled spaces bordered by compressed hepatocytes (Figure 2). The surrounding hepatocytes often revealed large lipid vacuoles and in the paler areas, the architecture of the hepatic parenchyma was slightly perturbed resembling nodular hyperplasia (Figure 2). The diagnosis was hepatic peliosis.
Discussion
Primary testicular tumours in domestic animals typically originate from either the germ cells (such as seminomas, teratomas, embryonal carcinomas and yolk sac carcinomas) or the sex cord-stromal elements of the gonad (such as Leydig and Sertoli cell tumours). The most commonly occurring primary testicular tumour in domestic animals is seminoma (Agnew & MacLachlan 2017). In humans, there are two subtypes of seminoma, specifically SE, which arises from gonocytes, is found in younger men and comprises > 95% of the cases, and SS, which arises from primary spermatocytes, tends to occur in older men and has more benign behaviour than SE (Aggarwal & Parwani 2009; Looijenga et al. 2007; Patel et al. 2020). However, although SS is generally noted for its reduced propensity to metastasise relative to SE (Patel et al. 2020), there have been reports, albeit rare, of metastatic SS in humans (Horn et al. 2011; Steiner et al. 2006). SE can be recognised by the PAS stain (due to the presence of glycogen) and IHC for expression of placental alkaline phosphatase (PLAP) (Moch et al. 2016), whereas SS, derived from more differentiated cells, does not express PLAP and is always PAS-negative (Cummings et al. 1994). Both subtypes of seminomas have been observed in canines (with SS being more prevalent than SE [Bush et al. 2011; Grieco et al. 2010; Kim et al. 2010; Looijenga et al. 1994]), and rabbits (Banco et al. 2017), together with the same PAS/PLAP staining patterns as observed in humans. In addition to PAS/PLAP, CD117 (also known as c-KIT) IHC has also been used to distinguish between the subtypes, with SE reported to be PAS/PLAP and CD117-positive, and SS to be PAS/PLAP and c-KIT-negative (Grieco et al. 2010). However, some studies have shown that 40-73% of the cases morphologically diagnosed as SS, showed CD117 positivity to some extent (Bush et al. 2011; Thorvaldsen et al. 2012), and studies of SS in humans have also reported relatively high proportions of the tumours being CD117-positive (Kraggerud et al. 1999). In addition, the CD117 signal in the positive sections of the SS tumours can vary considerably between the samples, with the majority (18/32, 56% cases) showing a staining area of < 25% of the tumour tissue (Thorvaldsen et al. 2012). This CD117 positivity has led to the suggestion that some cases of SS may originate from primordial cells in which expression of the CD117 protein has not been downgraded (Kraggerud et al. 1999; Thorvaldsen et al. 2012).
Peliosis is a rare benign vascular condition characterised by dilatation of hepatic sinusoids and the presence of blood-filled spaces within the liver. The clinical course of hepatic peliosis is variable, ranging from liver rupture and death (Kitchell et al. 2000), to an incidental finding that has been reported to mimic metastatic liver tumours (Maruyama et al. 2020; Tateishi et al. 1998). In humans, hepatic peliosis has been documented in patients with cancer, immunodeficiencies, infections, and after the use of certain medications (such as steroids) (Tsokos & Erbersdobler 2005). Importantly, hepatosplenic peliosis has been observed in patients with seminoma (Engel & Jacobsen 1993; Perez et al. 2012). Hepatic peliosis has been observed in dogs and cats, both secondary to infection with Bartonella species (Álvarez-Fernández et al. 2018) and without infection (in cats) (Buchmann et al. 2010). However, it has never been previously reported in a hedgehog.
Conclusion
This is the first report of a metastatic seminoma in a hedgehog, as well as the first report of a tumour in a South African hedgehog (Atelerix frontalis). This is also the first report of hepatic peliosis in a hedgehog, and as such should be considered a differential diagnosis in hedgehogs showing the presence of multiple or diffuse lesions in the liver. In addition, this report is the first to demonstrate the effectiveness of PAS staining and CD117 IHC in seminoma subtyping in hedgehogs, as is used in humans, dogs and rabbits. It is hoped that further investigations of hedgehogs, in particular Southern African hedgehogs, in wildlife reserves, zoological facilities or as pets, will add to a better knowledge and understanding of the tumour types and concomitant pathologies developed by this species.
Acknowledgements
The authors would like to thank the Histopathology laboratory in the Department of Paraclinical Sciences at the Faculty of Veterinary Science, University of Pretoria for processing of the tissue samples, and performing the staining and immunohis-tochemistry.
Conflict of interest
The authors declare they have no financial or personal relationships that may have inappropriately influenced them in writing this article.
Funding Source
LvdW was supported by the Wellcome Trust grant 108413/A/15/D. NO'D and JHO'D did not receive any specific grants from any funding agencies in the public, commercial, or not-for-profit sectors for this research.
Ethical approval
The owner of the hedgehog gave written consent for the case to be written up. The Research Ethics Committee of the Faculty of Veterinary Science at the University of Pretoria approved this case report (REC 092-20).
ORCID
Ν O'Dell ® https://orcid.org/0000-0003-4793-1948
JH O'Dell https://orcid.org/0000-0002-4966-4073
Lvan der Wevden https://orcid.org/0000-0002-0645-1879
References
Aggarwal, A., Parwani, A.V., 2009, Spermatocytic seminoma, Archives of Pathology and Laboratory Medicine 133, 1985-1988. https://doi.org/10.5858/133.12.1985. [ Links ]
Agnew, D.W., MacLachlan, N.J., 2017, Tumors of the genital systems, in DJ. Meuten (ed.), Tumors in Domestic Animals, 5th edn., pp. 689-722, Blackwell Publishing, Iowa. https://doi.org/10.1002/9781119181200.ch16. [ Links ]
Álvarez-Fernández, A., Breitschwerdt, E.B., Solano-Gallego, L., 2018, Bartonella infections in cats and dogs including zoonotic aspects, Parasites & Vectors 11, 624. https://doi.org/10.1186/s13071-018-3152-6. [ Links ]
Banco, B., Ferreira da Silva, J., Cotti Cometti, S., et al., 2017, Immunohistochemical expression of placental alkaline phosphatase in five cases of seminoma in rabbits. Journal of Comparative Pathology 156, 366-370. https://doi.org/10.1016/j.jcpa.2017.01.008. [ Links ]
Buchmann, A.U., Kempf, V.A., Kershaw, O., Gruber, A.D., 2010, Peliosis hepatis in cats is not associated with Bartonella henselae infections, Veterinary Pathology 47, 163-166. https://doi.org/10.1177/0300985809354347. [ Links ]
Bush, J.M., Gardiner, D.W., Palmer, J.S., et al., 2011, Testicular germ cell tumours in dogs are predominantly of spermatocytic seminoma type and are frequently associated with somatic cell tumors, International Journal of Andrology 34, e288-e295. https://doi.org/10.1111/j.1365-2605.2011.01166.x. [ Links ]
Cosentino, I.O., Alvarez Balaro, M.F., Beatriz da Silva Carvalho, A., et al., 2019, Metastatic seminoma in a male alpine goat: clinical and histopathological approach, Acta Scíentíae Veterinariae 47, 405. https://doi.org/10.22456/1679-9216.93422. [ Links ]
Cummings, O.W., Ulbright, T.M., Eble, J.N., Roth, L.M., 1994, Spermatocytic seminoma: an immunohistochemical study, Human Pathology 25, 54-59. https://doi.org/10.1016/0046-8177(94)90171-6. [ Links ]
Engel, P., Jacobsen, G.K., 1993, An unusual case of retroperitoneal seminoma and fatal peliosis of the liver, Histopathology 22, 379-382. https://doi.org/10.1111/j.1365-2559.1993.tb00139.x. [ Links ]
Farjanikish, G., Sayari, M., Raisi, A., Shirian, S., 2016, Diffuse type testicular seminoma in a stallion, Comparative Clinical Pathology 25, 1 133-1 136. https://doi.org/10.1007/s00580-016-2316-z. [ Links ]
Grieco, V., Banco, B., Giudice, C., et al., 2010, Immunohistochemical expression of the KIT protein (CD117) in normal and neoplastic canine testes, Journal of Comparative Pathology 142, 213-217. https://doi.org/10.1016/jjcpa.2009.07.002. [ Links ]
Horn, T., Schulz, S., Maurer, T., et al., 2011, Poor efficacy of BEP polychemotherapy in metastatic spermatocytic seminoma, Medical Oncology 28, S423-S425. https://doi.org/10.1007/s12032-010-9739-1. [ Links ]
Kim, J-H., Yu, C-H., Yhee, J-Y., et al., 2010, Canine classical seminoma: a specific malignant type with human classifications is highly correlated with tumor angiogenesis, BMC Cancer 10, 243. https://doi.org/10.1186/1471-2407-10-243. [ Links ]
Kitchell, B.E., Fan, T.M., Kordick, D., et al., 2000, Peliosis hepatis in a dog infected with Bartonella henselae, Journal of the American Veterinary Medical Association 216,519-523. https://doi.org/10.2460/javma.2000.216.519. [ Links ]
Kraggerud, S.M., Berner, A., Bryne, M., et al., 1999, Spermatocytic seminoma as compared to classical seminoma: an immunohistochemical and DNA flow cytometric study, APMIS 107, 297-302. https://doi.org/10.1111/j.1699-0463.1999.tb01557.x. [ Links ]
Looijenga, L.H., Olie, R.A., Van der Gaag, I., et al., 1994, Seminomas of the canine testis. Counterpart of spermatocytic seminoma of men?, Lab Investigation 71, 490-496. [ Links ]
Looijenga, L.H., Stoop, H., Hersmus, R., et al., 2007, Genomic and expression profiling of human spermatocytic seminomas: pathogenetic implications, International Journal of Andrology 30, 328-335. https://doi.org/10.1111/j.1365-2605.2007.00779.x. [ Links ]
Maruyama, H., Takahashi, K., Ishikawa, N., et al., 2020, A rare case of peliosis hepatis in a patient with chronic renal failure and renal cell carcinoma, Clinical Journalof Gastroenterology 13, 403-407. https://doi.org/10.1007/s12328-019-01068-5. [ Links ]
Moch, H., Cubilla, A.L., Humphrey, P.A., et al., 2016, The 2016 WHO classification of tumours of the urinary system and male genital organs, European Urology 70, 93-105. https://doi.org/10.1016/j.eururo.2016.02.029. [ Links ]
Patel, P.M., Patel, H.D., Koehne, E.L., et al., 2020, Contemporary trends in presentation and management of spermatocytic seminoma, Urology 146, 177-182. https://doi.org/10.1016/j.urology.2020.10.002. [ Links ]
Pérez, P., Pila Peláez, R., Rosales Torres, P., et al., 2012, Hepatosplenic peliosis: A case report and a literature review, Revista Colombiana de Gastroenterologia 27, 43-51. [ Links ]
Shortridge, E.H., Cordes, D.O., 1969, Seminomas in sheep, Journal of Comparative Pathology 79, 229-232. https://doi.org/10.1016/0021-9975(69)90009-7. [ Links ]
Steiner, H., Gozzi, C., Verdorfer, I., et al., 2006, Metastatic spermatocytic seminoma--an extremely rare disease, European Urology 49, 183-186. https://doi.org/10.1016/j.eururo.2005.08.020. [ Links ]
Tateishi, T., Machi, J., Morioka, W.K., 1998, Focal peliosis hepatis resembling metastatic liver tumor, Journal of Ultrasound Medicine 17, 581-584. https://doi.org/10.7863/jum.1998.17.9.581. [ Links ]
Thorvaldsen, T.E., Nødtvedt, A., Grotmol, T., Gunnes, G., 2012, Morphological and immunohistochemical characterisation of seminomas in Norwegian dogs, Acta Veterinaria Scandinavica 54, 52. https://doi.org/10.1186/1751-0147-54-52. [ Links ]
Tsokos, M., Erbersdobler, A., 2005, Pathology of peliosis, Forensic Science International 20, 25-33. https://doi.org/10.1016/j.forsciint.2004.05.010. [ Links ]
 Correspondence:
Correspondence:
nicolize.odell@up.ac.za














