Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
Journal of the South African Veterinary Association
versión On-line ISSN 2224-9435
versión impresa ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.85 no.1 Pretoria ene. 2014
CASE REPORT
Epineurial repair of an iatrogenic facial nerve neurotmesis after total ear canal ablation and lateral bulla osteotomy in a dog with concurrent cranio-mandibular osteopathy
Ignacio Calvo; Irene Espadas; Gawain Hammond; Kathryn Pratschke
Small Animal Hospital - Division of Small Animal Sciences, School of Veterinary Medicine, University of Glasgow, United Kingdom
ABSTRACT
A 7-year-old male entire West Highland white terrier was referred to the Small Animal Hospital at the University of Glasgow for bilateral, chronic, medically unresponsive otitis media and externa. A history of cranio-mandibular osteopathy was also reported. Bilateral total ear canal ablation and lateral bulla osteotomy was performed with the aid of a pneumatic burr. Extensive bone proliferation was present bilaterally originating from the caudal mandibular ramus and tympanic bulla which incorporated the horizontal canal on each side. The right facial nerve was identified leaving the stylomastoid foramen and running in a cranial direction through a 1.5 cm diameter cuff of bone surrounding the horizontal canal and external acoustic meatus. Despite careful dissection, a facial nerve neurotmesis ensued which required microsurgical epineurial repair. Neurologic examination performed 12 h post-operatively revealed abnormalities consistent with right facial nerve paralysis. At 3 months, the facial nerve function was found to have improved significantly and was assessed to be normal four months after surgery. To the authors' knowledge, this clinical communication described the first reported clinical case where unilateral facial nerve paralysis resulting from iatrogenic facial nerve neurotmesis was successfully treated by microsurgical epineurial repair.
Introduction
Iatrogenic neurotmesis of the facial nerve has been previously suggested as an uncommon complication of total ear canal ablation and lateral bulla osteotomy (TECA-LBO) (Levine & Esrig 2000).
The facial nerve exits the stylomastoid foramen caudal to the external acoustic meatus and courses ventral to the horizontal canal in close proximity to the middle ear, travelling in a rostral direction. Reported clinical signs of facial paralysis are ipsilateral drooping of ear and lip, widened palpebral fissure, drooling, absence of spontaneous and provoked blinking that may result in corneal ulceration, absence of nostril abduction during inspiration, deviation of nostril towards normal side (unless in a chronic case when the nostril deviates to affected side and lips retract farther than normal) and facial spasms. If the affected portion of the facial nerve is located between the medulla and the middle ear, unlike in this case, dysfunction of the parasympathetic supply of the lacrimal gland will lead to neurogenic keratoconjunctivitis sicca, potentially causing recurrent corneal ulceration, mucous ocular discharge, conjunctival hyperemia and chronic keratitis (Dewey & Cerda-Gonzalez 2008; Garosi, Lowrie & Swinbourne 2012).
Supportive treatment with artificial tears to prevent corneal drying has been recommended, particularly in cases where the paralysis is expected to be transient (neurapraxia) or as a palliative treatment in cases where no other therapeutic options are available (e.g. idiopathic facial nerve paralysis) (Jeffery & Granger 2013). Experimental facial nerve neurotmesis and successful epineurial microsurgical repair has been previously reported (Salerno, Bleicher & Stromberg 1990; Sundine et al. 2003).
This clinical communication describes the first reported clinical case where bilateral TECA-LBO has been performed in a dog with concurrent cranio-mandibular osteopathy and the first report where unilateral facial nerve paralysis resulting from iatrogenic facial nerve neurotmesis has been successfully treated with microsurgical epineurial repair.
Case presentation
A 7-year-old entire male West Highland white terrier was referred to the Small Animal Hospital at the University of Glasgow for management of bilateral, chronic, medically unresponsive otitis media and externa. A history of cranio-mandibular osteopathy was also reported. Physical examination revealed a pain-free decreased range of jaw opening and discomfort on bilateral ear canal palpation. Bone proliferation was obvious on palpation of the ear canals.
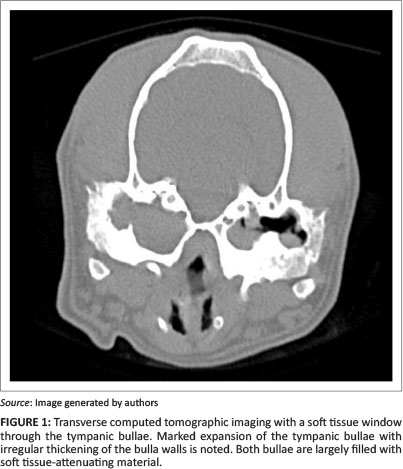
Otoscopy and computed tomography (CT) were performed under sedation and showed narrowed horizontal ear canals, chronic otitis externa and otitis media, severe hyperplasia of the ear canal epithelium resulting in occlusion of the horizontal ear canal, dystrophic mineralisation within the horizontal canal wall and soft tissue-fluid opacity within the tympanic bullae (Figure 1). Bilateral total ear canal ablation with lateral bulla osteotomy was advised and performed as previously described (Mason, Harvey & Orsher 1988); although, as a result of the extensive bone proliferation, a pneumatic burr was used to perform the lateral bulla osteotomy and to free the right facial nerve.
The horizontal canals were ossified and extensively surrounded by proliferative bone originating from the caudal mandibular rami and tympanic bullae (Figure 2). The right facial nerve was identified leaving the stylomastoid foramen and running in a rostral direction for 0.5 cm before becoming encased within a 1.5 cm outer diameter bony tunnel surrounding the horizontal canal and external acoustic meatus. Gentle ventral retraction of the right facial nerve at the entry point of the bony tunnel was performed to aid protection during burring of the bony tunnel. Despite careful attempts to preserve and protect the nerve, facial nerve neurotmesis occurred. The rostral portion of the transacted nerve was released from the bone cuff with a burr and the TECA-LBO completed. On the left side, the facial nerve lay on the surface of the proliferative bone and could be retracted during TECA-LBO.

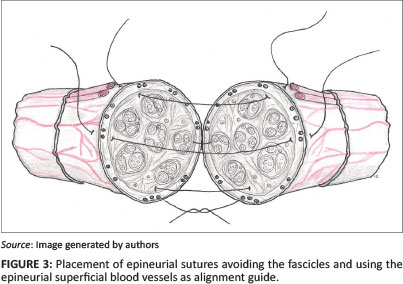
Epineurial repair of the transected nerve was performed as previously described (Matsuyama, Mackay & Midha 2000). Four simple interrupted (8-0 Nylon) sutures (Ethicon, Livingston, Scotland) were placed with the aid of a Zeiss surgical microscope. The nerve repair was accomplished by first placing two lateral sutures through the epineurium, 180 degrees apart. The nerve alignment was ensured by aligning the superficial epineurial longitudinal vasculature (Figure 3). Two more single interrupted sutures were placed at 90 degrees from the previously placed sutures.
Neurologic examination performed 12 h post-operatively revealed the following abnormalities on the right side: drooping of the lip, widened palpebral fissure and absence of spontaneous and provoked blinking. At 3 months after surgery, follow-up neurologic examination revealed normal palpebral fissure widening, blinking ability and menace response had returned. The owner reported that the ability to close the right eye returned 2.5 months after surgery. Mild drooping of the right lip and food retention were still present; however, the owner reported that they were still improving. Based on clinical examination and the owner's opinion, comfort levels and overall demeanour were markedly improved at the 3-month follow-up compared with before surgery. A 4-month post-operative examination showed the lip to be positioned normally and no food retention. Pain-free decreased range of jaw opening was still present.
Discussion
Cranio-mandibular osteopathy has been reported in several breeds, including West Highland white terriers.
It is characterised by an extensive, irregular periosteal proliferative reaction of the mandible. This mandibular periosteal reaction is usually bilateral and can extend to the petrous temporal bone, tympanic bullae and cranium; however, aetiology and pathogenesis of this disease are poorly understood (Thompson et al. 2011).
When performing a TECA-LBO, the exuberant bony periosteal proliferative reaction at the level of the tympanic bullae is not only a reason for concern from the neurologic point of view (i.e. facial nerve involvement) but also from a haemorrhage point of view. Severe haemorrhage during TECA-LBO surgery, normally as a result of damage to the retroauricular vein, is an uncommon but potentially devastating complication that may result in the death of the patient (Smeak 2011). It is important to remember that the retroauricular vein lies just rostral to the external auditory meatus and is most often damaged during detachment of the horizontal ear canal epithelium from the bone or when excessive curettage is performed in this area. Although haemorrhage was not encountered in this case, extreme caution is advisable during burring of the external meatus rostrally.
Facial nerve neurotmesis has been previously reported as a TECA-LBO complication, but in the majority of these reports the facial nerve was deliberately surgically severed. This was commonly performed in an attempt to completely remove a neoplastic process; although, accidental transection during deep dissection of the horizontal ear canal has also been reported in up to 4% - 6% of TECA-LBO procedures (Smeak 2011). In this case, the right facial nerve was identified and protected, but, unfortunately, gentle ventral blunt retraction resulted in neurotmesis at the entry of the bony tunnel, suggesting that perhaps a structural weakness of the right facial nerve was present.
In any nerve repair, several principles must be followed, one of the most important being prevention of tension at the repair site. If any tension is present, alternative repair methods such as nerve graft should be considered (Matsuyama et al. 2000).
In the repair of severed peripheral nerves, the epineurial suture repair remains the gold standard with which other techniques must be compared. Despite many attempts being made to define the most efficacious method of nerve re-approximation, no technique has been shown to be overwhelmingly superior to others (Terris & Fee 1993). In fact, in 1977, Levinthal performed a direct comparison between epineurial and fascicular repair in a canine anterior tibial nerve model and showed that both techniques produced comparable distal axonal regeneration. Epineurial repair was chosen because it is technically easier and it is associated with less scarring and less injury to the nerve blood supply (Levinthal 1977).
Minimal numbers of epineurial sutures should be applied in order to ensure accurate anatomical coaptation of the nerve stumps whilst minimising the foreign material and scar reaction (Matsuyama et al. 2000). Surface features such as longitudinal vessels in the epineurium can confirm the rotational alignment of the nerve. This is of paramount importance to ensure that the fascicular arrangement is reoriented and matched properly and therefore to allow axon crossing (Matsuyama et al. 2000).
After nerve transection, the nerve undergoes Wallerian degeneration. This process starts immediately after injury. Schwann cells and macrophages phagocytose all of the myelin and cellular debris distal to the injury site, leaving empty endoneurial tubes (Flores, Lavernia & Owens 2000). Wallerian degeneration is well underway within a week of injury. The activated macrophages produce factors that facilitate Schwann cell migration and axon regeneration. After a lag period (at least 1 week), injured axons form a growth cone and begin to regenerate (Gaudet, Popovich & Ramer 2011). The role of surgical nerve repair is to provide an appropriate anatomical environment, so that axons from the proximal stump can regenerate into the endoneurial tubes within the distal nerve stump (Flores et al. 2000). Axons re-grow at 0.1 cm - 0.4 cm per day (Dewey & Cerda-Gonzalez 2008). This growth rate, taking into consideration the initial inflammation and debridement lag period, is consistent with the recovery time observed in this case (distance between nerve repair site and orbicularis oculi muscle was estimated to be 6.0 cm).
Historically, it has been suggested that the ability of a nerve cell to regenerate is maximal at around 3 weeks after surgery; this is the reason behind the belief that neurorraphy should be delayed for 3 weeks (McCabe 1970). Recent data suggests that early repair of transected nerves with primary neurorraphy results in an improved long-term outcome (Millesi 1990).
Electromyography (EMG) and/or nerve conduction testing would have been ideal to document the initial denervation and subsequent reinnervation. A normal relaxed muscle is electrically silent. Five to ten days after the development of a peripheral motor nerve lesion, denervated muscle fibres show spontaneous depolarisation, which is more often recorded in the form of fibrillation potentials and positive sharp waves. Motor nerve conduction studies are used to assess nerve function; the nerve in question is electrically stimulated in two sites and this should elicit evoked muscle action potential in the target muscle (Poncelet & Poma 2013). If reinnervation occurs, EMGs and nerve conduction should become normal.
EMG changes of the orbicularis oculi muscle in the form of denervation fibrillation potentials and lack of evoked muscle action potential after stimulation of the facial nerve external to the stylomastoid foramen have been previously reported in dogs following facial nerve paralysis (Braund et al. 1979). Although the lack of EMG or nerve conduction testing in this case is acknowledged as a limitation, the authors believe that the sequential nature and time to full neurologic function recovery could only be explained by the occurrence of reinnervation.
Conclusion
Bilateral TECA-LBO in a dog with concurrent cranio-mandibular osteopathy has not been previously reported to the authors' knowledge. Despite being technically challenging, it should still remain a therapeutic option for end-stage ear disease. However, owners should be informed of the potential increased risk of iatrogenic facial nerve injury. Surgical repair of a transected facial nerve is feasible but requires specialist training and equipment to be performed successfully.
Acknowledgements
Competing interests
The authors declare that they have no financial or personal relationships which may have inappropriately influenced them in writing this article.
Authors' contributions
I.C. (University of Glasgow) was the primary clinician for this case and was responsible for writing the manuscript and the final proofreading of it. I.E. (University of Glasgow) provided intellectual input to the manuscript and created Figure 3. G.H. (University of Glasgow) is responsible for interpretation of the CT. K.P. (University of Glasgow) assisted in the clinical management of this case, provided valuable input to the manuscript and assisted in the final proofreading of the manuscript.
References
Braund, K.G., Luttgen, P.J., Sorjonen, D.C. & Redding, R.W., 1979, 'Idiopathic facial paralysis in the dog', Veterinary Record 105(13), 297-299. http://dx.doi.org/10.1136/vr.105.13.297 [ Links ]
Dewey, C.W. & Cerda-Gonzalez, S., 2008, 'Disorders of the peripheral nervous system: Mononeuropathies and polineuropathies', in C.W. Dewey (ed.), A practical guide to canine and feline neurology, 2nd edn., pp. 373 & 400, Wiley-Blackwell, Ames. [ Links ]
Flores, A.J., Lavernia, C.J. & Owens, P.W., 2000, 'Anatomy and physiology of peripheral nerve injury and repair', American Journal of Orthopaedics 29, 167-173. [ Links ]
Garosi, L.S., Lowrie, M.L. & Swinbourne, N.F., 2012, 'Neurological manifestations of ear disease in dogs and cats', Veterinary Clinics of North America Small Animal Practice 42(6), 1143-1160. http://dx.doi.org/10.1016/j~.cvsm.2012.08.006 [ Links ]
Gaudet, A.D., Popovich, P.G. & Ramer, M.S., 2011, 'Wallerian degeneration: Gaining perspective on inflammatory events after peripheral nerve injury', Journal of Neuroinflammation 8, 110. http://dx.doi.org/10.1186/1742-2094-8-110 [ Links ]
Jeffery, N. & Granger N., 2013, 'Neurological abnormalities of the head and face', in S.R. Platt & N.J. Olby (eds.), BSAVA manual of canine and feline neurology, 4th edn., pp. 229-230, British Small Animal Veterinary Association, Gloucester. [ Links ]
Levine, S.M. & Esrig, R.P., 2000, 'The facial nerve in animals', in M. May (ed.), The facial nerve, 2nd edn., pp. 112, Thieme New York, New York. [ Links ]
Levinthal, R., 1977, 'Comparison of fascicular, interfascicular, and epineural suture techniques in the repair of simple nerve lacerartions', Journal of Neurosurgery 47, 744-750. http://dx.doi.org/10.3171/jns.1977.47.5.0744 [ Links ]
Mason, L., Harvey, C. & Orsher, R., 1988, 'Total ear canal ablation combined with lateral bulla osteotomy for end stage otitis in dogs. Results in thirty dogs', Veterinary Surgery 17(5), 263-268. http://dx.doi.org/10.1111/j.1532-950X.1988.tb01012.x [ Links ]
Matsuyama, T., Mackay, M. & Midha, R., 2000, 'Peripheral nerve repair and grafting techniques: A review', Neurologia Medico-Chirurgica (Tokyo) 40, 187-199. http://dx.doi.org/10.2176/nmc.40.187 [ Links ]
McCabe, B.F., 1970, 'Facial nerve grafting', Plastic and Reconstructive Surgery 45, 70-75. http://dx.doi.org/10.1097/00006534-197001000-00010 [ Links ]
Millesi, H., 1990, 'Progress in peripheral nerve reconstruction', World Journal of Surgery 14, 733-747. http://dx.doi.org/10.1007/BF01670520 [ Links ]
Poncelet, L. & Poma, R., 2013, 'Electrophysiology', in S.R. Platt & N.J. Olby (eds.), BSAVA manual of canine and feline neurology, 4th edn., pp. 61-62, British Small Animal Veterinary Association, Gloucester. [ Links ]
Salerno, G.M., Bleicher, J.N. & Stromberg, B.V., 1990, 'Blink reflex recovery after electrical stimulation of the reinervated orbicularis oculi muscle in dogs', Annals of Plastic Surgery 25, 360-370. http://dx.doi.org/10.1097/00000637-199011000-00004 [ Links ]
Smeak, D.D., 2011, 'Management of complications associated with total ear canal ablation and bulla osteotomy in dogs and cats', Veterinary Clinics of North America Small Animal Practice 41(5), 981-994. http://dx.doi.org/10.1016/j.cvsm.2011.05.011 [ Links ]
Sundine, M.J., Quan, E.E., Saglam, O., Dhawan, V., Quesada, P.M., Ogden, L. et al., 2003, 'The use of end-to-side nerve grafts to reinnervate the paralyzed orbicularis oculi muscle', Plastic and Reconstructive Surgery 111(7), 2255-2264. http://dx.doi.org/10.1097/01.PRS.0000060089.45869.59 [ Links ]
Terris, D.J. & Fee, W.E., 1993, 'Current issues in nerve repair', Archives of Otolaryngology - Head and Neck Surgery 119, 725-731. http://dx.doi.org/10.1001/archotol.1993.01880190021004 [ Links ]
Thompson, D.J., Rogers, W., Owen, M.C. & Thompson, K.G., 2011, 'Idiopathic canine juvenile cranial hyperostosis in a Pit Bull terrier', New Zealand Veterinary Journal 59(4), 201-205. http://dx.doi.org/10.1080/00480169.2011.579556 [ Links ]
 Correspondence:
Correspondence:
Ignacio Calvo
464 Bearsden Road
Bearsden
Scotland G61 1QH
United Kingdom
Email: ignacio.calvo@glasgow.ac.uk
Received: 06 May 2013
Accepted: 07 Feb. 2014
Published: 22 Aug. 2014














