Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Journal of the South African Veterinary Association
On-line version ISSN 2224-9435
Print version ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.85 n.1 Pretoria Jan. 2014
ORIGINAL RESEARCH
Reverse saphenous conduit flap in small animals: Clinical applications and outcomes
Ross C. Elliott
Department of Small Animal Surgery, Bryanston Veterinary Hospital, South Africa
ABSTRACT
Due to the lack of skin elasticity defects of the distal hind limb can be a challenge to close. This article assesses a well-described, but completely under-used technique for closure of wounds on the distal tarsus. The technique was used with good success in six cases presenting to the Bryanston Veterinary Hospital with a wide range of underlying pathology ranging from trauma to neoplastic disease of the tarsus. All six cases were treated with a reverse saphenous conduit flap and two of them underwent radiation therapy with no adverse side effects. All cases showed excellent results with a very low degree of flap necrosis that never exceeded 15% of the total flap area. This skin flap provides an excellent treatment method that is reliable in closure of defects of the distal tarsus with few adverse effects. To the author's knowledge there has been only one previously published report on the clinical use of this type of skin flap, even though the flap is well described in most texts.
Introduction
The reverse saphenous conduit flap is a type of axial pattern flap. This type of skin flap is used for one-stage reconstruction of wounds. It uses skin from surrounding areas where there is abundance, unlike the recipient site (Cornell et al. 1995). Axial pattern flaps are durable, full-thickness pedicle flaps that contain a direct cutaneous artery and vein to supply and drain the entire length of the flap. A pedicle flap without a known direct cutaneous artery is known as a sub-dermal plexus flap (Cornell et al. 1995; Degner, Bauer & Cozen 1993). The assurance of a blood supply allows a longer flap to be harvested, and an axial pattern flap shows a 50% increased graft survival area compared with a routine sub-dermal plexus flap (Moores 2009). Axial pattern flaps can be used in areas of poor vascularity, as they are not dependent on local blood supply from the wound bed. Axial pattern flaps are more resilient to movement because they do not rely completely on vascularisation from the wound bed, thus making them an excellent choice when wounds extend over, or are close to, a high-motion joint. The scar formation seen in these animals is considered to be acceptable by most of the pet owners (Degner et al. 1993).
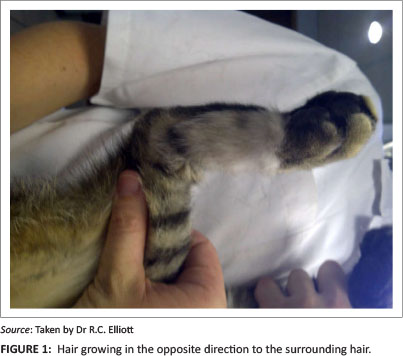
There are, however, some disadvantages to the axial pattern flaps. Their use is restricted to the anatomical area in which the direct cutaneous vessels are situated. Therefore, there are areas in which they may not be suitable for the closure of wounds. Even in the face of these limitations most wounds in the body can be treated, as there are described direct cutaneous arteries and veins throughout the body. Hair growth is reported to be normal to near normal, but given the rotational nature of the flaps it may be in the opposite direction (Figure 1) to normal hair growth (Degner et al. 1993).
There is little information published on the clinical use of the reverse saphenous flap, however, it remains a versatile and robust method of closure for wounds over the tarsus and metatarsus (Pavletic 1991). Flaps of 12 cm - 20 cm have been harvested in experimental canine patients (Pavletic 1999) and 8 cm long by 4 cm wide in feline experimental patients, both with 100% survival rates (Pavletic et al. 1983). These authors recommended the use of this flap in the feline patient on account of the well-developed medial saphenous vein, but it can also be very useful in the canine patient, as is shown in this article and indicated by Pavletic et al. (1983).
The purpose of this article is to demonstrate the clinical versatility of the reverse saphenous conduit flap, the outcome of the flap, and associated complications.
Methods
Animal selection
Animals were selected from patients that presented at the Bryanston Veterinary Hospital for pathology of the hind limb. This pathology ranged from trauma, non-healing wounds and oncological surgery. All patients had a serum biochemistry analysis performed, which included albumin, total solids, urea, creatinine and electrolytes, as well as a full haematological examination prior to surgery. The feline patients were tested for factors that may potentially affect wound healing, such as feline immunodeficiency or AIDS virus and feline leukaemia virus. All patients presenting for oncological intervention had a fine needle aspirate taken from the regional lymph nodes, ultrasonographic examination of the abdomen and thoracic radiographs. A biopsy of the neoplasm to be removed was performed prior to surgery in order to determine the type of neoplasm by histological examination.
Anaesthesia
All patients were pre-medicated with diazapam 0.2 mg/kg (Valium 5 mg/mL, Roche, New Jersey, USA) and intravenous buprenorphine 0.02 mg/kg (Temgesic® 0.3 mg/mL, Schering-Plough, Woodmead, South Africa). Induction was performed with intravenous diprivan 6.6 mg/kg (Propofol 1% 10 mg/mL, Fresenius Kabi, Midrand, South Africa). All patients were intubated and placed on isoflorane (Isofor 250 mL, Safeline, Pharmaceuticals, Johannesburg, South Africa). They were also all placed on a Ringers lactate drip (Sabax) at 10 mL/kg/h for the duration of the surgery.
A dose of intravenous amoxicillin clavulanic acid 20 mg/kg (Augmentin 600 mg, SmithKline, Wynberg Ext 6, Johannesburg) was given prior to surgery and one hour later, which coincided with the end of the surgery. No further antibiotic cover was given. Buprenorphine was used at 0.01 mg/kg every eight hours through the night after the surgery. The Ringers lactate drip was continued at maintenance dose rates according to the weight of the patient.
Anatomical background to surgical procedure
The saphenous artery is not a true, direct cutaneous artery but it supplies the skin with multiple, small direct cutaneous arteries; it therefore acts as a conduit. This flap is interesting in that it actually relies on reversing the blood flow in the saphenous artery to enable it to function and survive. The medial saphenous vein is responsible for the vascular drainage of the flap (Pavletic 1999).
These vessels are ligated just distal to the femoral artery and vein, where they originate from the femoral artery and vein. The blood supply in the saphenous artery is maintained and reversed as a result of the connections between:
• the cranial branch of the saphenous artery and the perforating metatarsal artery by way of the medial and lateral plantar arteries
• the cranial branch of the medial saphenous vein and the cranial branch of the lateral saphenous vein
• other connections with the cranial and caudal branches of the medial saphenous vein distal to the tibio-tarsal joint (Degner et al. 1993; Pavletic 1999).
Surgical procedure
All surgical procedures were performed as described below (Degner et al. 1993). The size of the defect to be covered was measured because of the need to ligate the flap just proximal to the blood vessels coming off the femoral artery. There is a maximum length to all reverse saphenous conduit flaps. A skin incision was made across the central third of the inner thigh perpendicular to the long axis of the tibia. Another useful landmark described is the patella; this incision can be made just proximal to the patella. In cats, it was possible to visualise the vasculature on the medial aspect of the tibia once clipping, shaving and cleaning was completed. To harvest the skin flap two incisions were made parallel to the long axis of the tibia on either side of the transverse incision. These two incisions extended distally just proximal to the medial malleolus. They tended to converge distally according to the availability of skin in the distal limb.
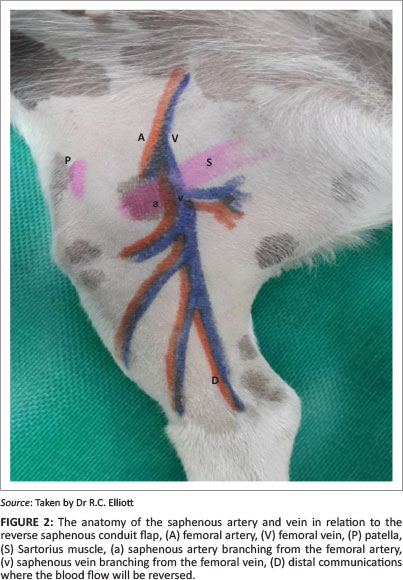
The saphenous artery and medial saphenous vein were exposed at the transverse incision where they branch off the femoral artery and vein. Both these vessels were double ligated and the flap was then undermined deep to the vasculature. A portion of the medial gastrocnemius fascia was included in the pedicle of the flap. This has been recommended in order to prevent damage to the caudal branch of the saphenous artery and medial saphenous vein (Pavletic 1999).
The epithelialised wound edges were surgically debrided and all the skin flaps were joined to their recipient beds by a bridging incision. No tubed pedicles were performed. As all of the flaps were rotated nearly 180° in order to cover the skin defects distal to the tarsal-crural joint, it was essential to make sure that there was no kinking of the vasculature in the pedicle of the flap. It is always best to allow a gradual curve of the pedicle over the bridging incision.
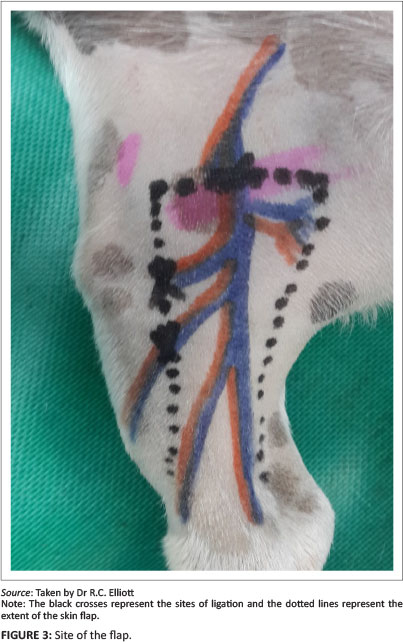
The donor site was closed with a subcutaneous continuous suture of polydioxanone 4-0 and the skin was closed with simple interrupted nylon 4-0. The flap was sutured onto the wound edges with nylon 4-0 simple interrupted sutures.
Post-operative care
A light dressing and an Elizabethan collar were placed on all patients to prevent patient interference and/or damage to the skin flap. The dressing was changed after 48 h and the flap was clinically evaluated. A large number of the flaps took on a dark purple colour at 48 h. This was thought to be due to mild post-operative venous congestion of the flap post surgery, which had disappeared completely at four days post surgery, when the flap took on a normal pink colouration. This venous congestion did not affect flap survival or increase its dehiscence. The animals were sent home with strict instructions to maintain rest and pain control for four days. The skin flaps were checked at five days post surgery and the skin sutures removed at 14 days post surgery. Radiation therapy was started at day 10 after the operation for those who required it.
Results
For ease of comparison, the results are presented in Table 1. All of the patients who were presented for radiation therapy had a full course of radiation and there were no adverse effects detected on flap survival. All patients tolerated the flap well and showed no signs of pain or discomfort during the recovery period. Some patients showed mild erythema and pruritus of the flap after radiation, but this was managed with a short course of an anti-inflammatory medication.
Discussion
The cases demonstrated that the reverse saphenous conduit flap achieved excellent results in two different species under different sets of circumstances. More studies are needed to evaluate the effects of radiation therapy on this type of skin flap, as there is great potential to use this skin flap in cats and dogs to close skin defects on the tarsus and metatarsus when removing malignant or large benign neoplasms. In the author's experience, wounds resulting from attempts to remove masses (mainly benign) in the tarsal area, which have been surgically closed by primary closure of the wound edges, have usually shown a high rate of dehiscence. A more severe complication in the metatarsal area is vascular compromise of the distal limb. This is due to swelling caused by the surgical procedure to remove these masses, the high degree of skin tension on the wound edges and the practice of placing a bandage post-operatively. This excessive skin tension produces a natural tourniquet due to the minimal soft tissue surrounding this area and subsequently causes constriction of the vascular supply, leading to severe swelling of the distal pes and possible avascular necrosis of the skin of the paw. This complication can be avoided by planning the surgery to include a reverse saphenous conduit flap to cover the skin defect. This technique prevents high tension across the wound and all of the above complications.
Deep abrasive wounds of the tarsus occur commonly after road traffic accidents due to a shearing force being applied to the skin. They may result in loss of relatively vast amounts of skin, connective tissue and even bone. Once granulated, providing the distal vascular connections are not damaged by the initial trauma, they become excellent candidates for the use of a reverse saphenous conduit flap. All ligament damage and other orthopaedic damage should also be accounted for and managed prior to closure of the skin defect.
Chronic non-healing wounds may present a different problem in that they can have an underlying cause affecting wound healing. All of these wounds should first be biopsied to rule out any neoplastic condition as being a cause of the non-healing wound. A culture should also be done to treat any underlying bacterial infection. All animals should be assessed for systemic diseases that can affect wound healing.
As the reverse saphenous conduit flap brings its own blood supply with it, its use in wounds with a poor blood supply allows for excellent healing, as it is not dependent on a health granulation bed. This factor makes it ideal for use in cases where post-operative radiation is planned and in this study was used with no adverse complications arising from the radiation.
This technique has been proven in cats, but it has only been reported in dogs as an anatomical study and not in any clinical setting. The cases described above indicate how this axial pattern skin flap was used in canine and feline patients in a clinical setting for closure of distal pelvic limb wounds. A limitation of the study was that the sample size was small, but given the high success rate of this type of skin flap, this report may provide important clinical information about using the flap.
Acknowledgement
Competing interests
The author declares that he has no financial or personal relationship(s) which may have inappropriately influenced him in writing this article.
References
Cornell, K., Salisbury, K., Jacovljevic, S., Bauer, M. & Petryk, D., 1995, 'Reverse saphenous conduit flap in cats: An anatomic study', Veterinary Surgery 24, 202-206. http://dx.doi.org/10.1111/j.1532-950X.1995.tb01319.x [ Links ]
Degner, D.A., Bauer, M.S. & Cozen, S.M., 1993, 'Reverse saphenous conduit flap: A case report in a cat', Veterinary Comparative Orthopaedics and Traumatology 6, 175. [ Links ]
Moores, A., 2009, 'Axial pattern flaps', in J. Willams & A. Moores (eds.), BSAVA manual of canine and feline wound management and reconstruction, pp. 100-143, BSAVA, Gloucester. [ Links ]
Pavletic, M.M., 1991, 'Anatomy and circulation of the canine skin', Microsurgery 12, 103-112. http://dx.doi.org/10.1002/micr.1920120210 [ Links ]
Pavletic, M.M., 1999 'Axial pattern flaps', in M.M. Palvetic (ed.), Atlas of small animal reconstructive surgery, n.p., Saunders, Philadelphia. [ Links ]
Pavletic, M.M., Watters, J., Henry, R.W. & Nafe, LA., 1983, 'Reverse saphenous conduit flap in the dog', Journal of the American Veterinary Medical Association 182, 380-389. [ Links ]
 Correspondence:
Correspondence:
Ross Elliott
PO Box 130905
Bryanston 2021
South Africa
Email: rosselliott_2@hotmail.com
Received: 19 Apr. 2013
Accepted: 14 Sept. 2013
Published: 20 Aug. 2014














