Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Journal of the South African Veterinary Association
On-line version ISSN 2224-9435
Print version ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.84 n.1 Pretoria Jan. 2013
ORIGINAL RESEARCH
Circulation of bluetongue virus in goats in the Karamoja region of Uganda
Elijah N. MulabbiI; Chrisostom AyebazibweII; Samuel MajalijaI; Carrie A. BattenIII; Christopher A.L. OuraI, III, IV
IDepartment of Bio molecular Resources and Bio laboratory Sciences, Makerere University, Uganda
IINational Animal Disease Diagnostics and Epidemiology Center, Ministry of Agriculture Animal Resources and Fisheries, Uganda
IIIInstitute for Animal Health, Pirbright, United Kingdom
IVSchool of Veterinary Medicine, University of West Indies, Trinidad and Tobago
ABSTRACT
The presence of bluetongue virus (BTV) in indigenous goats from the Karamoja region of northern Uganda was investigated. A total of 300 goats were sampled (serum and whole blood) from five districts within the Karamoja region. The samples were analysed for the presence of bluetongue (BT) antibodies using a commercial Enzyme-linked immunosorbent assay (ELISA) and for the presence of BTV viral RNA by real-time Reverse transcription polymerase chain reaction (RT-PCR), because BTV is an RNA virus. Of the 300 goats tested, 269 (90%) were positive for BTV antibodies, indicating high levels of BTV circulation within the region. Out of the 150 whole blood samples tested for the presence of the virus by realtime RT-PCR, 84 (56%) were positive for BTV RNA. This study, which is the first of its kind in Uganda, showed a high seroprevalence of BT antibodies and active circulation of BTV in a high proportion of goats in the Karamoja region.
Introduction
Bluetongue virus (BTV) is a member of the genus Orbivirus, family Reoviridae (Mertens et al. 2005) and is transmitted by biting midges (Culicoides spp.). Bluetongue (BT) disease is an infectious, arthropod-borne viral disease caused by BTV. It is found in sheep and both domestic and wild ruminants, such as goats, cattle, deer, bighorn sheep, most species of African antelopes and other artiodactyls (MacLachlan 1994). Although the frequency of infections in cattle is generally higher than in sheep, overt disease in cattle is rare and clinical signs, when they do occur, are much milder than those observed in sheep. The central role of the insect in BTV epidemiology ensures that the prevalence of disease is governed by ecological factors, such as high rainfall, temperature, high humidity and high organic matter content of the soil, which favour insect survival. Therefore in many parts of the world the disease has a seasonal occurrence (Erasmus 1990).
BTV was confined mainly to tropical and temperate areas of America, Africa and parts of Asia (Walton 2004), but recently there have been multiple incursions of BTV into Europe from Africa; the most serious caused by a strain of serotype 8 (BTV-8) (Saegerman, Berkvens & Mellor 2008). In Africa, several serotypes have been identified in Kenya (Davies, Mungai & Pini 1992) and South Africa (Gerdes 2004), where the virus has also been isolated from wild ruminants. However, in Uganda, BT disease has been suspected in three kobs (Kobus kob) in Queen Elizabeth National Park, but no laboratory confirmation has been received yet (OIE 2003), nor have there been previous reports of BTV circulation in goats in Uganda.
Animal diseases and the associated high levels of animal mortalities seen in the Karamoja region of northern Uganda pose a significant threat to the development of small ruminant farming. In addition, the region is severely affected by persistent poor harvests, drought, poor farming practices and crime (theft of animals). Goats are an important animal resource in Karamoja, with many households depending on them for livelihood and income (CPRC 2008).
The aim of this study was to improve current knowledge on the distribution, spread and clinical significance of BTV in domestic goats in Uganda.
Materials and methods
Three hundred goats that were between six and eighteen months of age and therefore free of colostral antibodies were selected randomly for the study. Blood in Ethylenediaminetetraacetic acid (EDTA), with anticogulant EDTA and without anti-coagulant was collected from goats in the five selected Karamoja districts (Figure 1). The blood samples were transported at 4 °C to the National Animal Disease Diagnostics and Epidemiology Centre (NADDEC), Ministry of Agriculture Animal Industry and Fisheries (MAAIF), Entebbe. The blood without an anticoagulant coagulated and the serum was decanted off.
Antibodies were detected in serum using a commercial competitive-Enzyme linked Immunosorbent assay (c-ELISA) test kit (ID-VET® Innovative Diagnostics, Montpellier, France) according to the manufacturer's instructions. This diagnostic kit was designed to detect antibodies directed against the BT viral protein 7 (VP7); the antibodies in the test serum competed with the monoclonal antibody against VP7 protein for binding to the antigen (VP7 protein). Whole blood samples were tested for viral ribonucleic acid (RNA) by realtime Reverse transcriptase-polymerase chain reaction (RT-PCR) (Shaw et al. 2007). RT-PCR was used because BTV is an RNA virus, and this method first synthesises complementary deoxyribonucleic acid (DNA) before PCR amplification. Real time RT-PCR was specifically preferred to conventional PCR because it is not time consuming and eliminates post PCR processing of PCR products and thus chances of carry-over contamination are reduced.
Ethical considerations
The samples used in this study were collected during animal disease surveillance surveys performed by the Ministry of Agriculture, Animal industry and Fisheries. The guidelines, as stated by the Ministry of Agriculture, Animal industry and Fisheries, which are in line with the OIE standards, were followed to ensure that animal rights were not violated.
Results
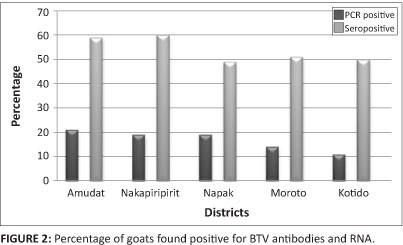
Of the 300 goats sampled, 269 (90%) were positive for BTV antibodies.The seroprevalence in goats from the five districts of Karamoja where sampling took place ranged from 85% in the Moroto district to 97% in the Nakapiripirit district (Figure 2). There was no significant difference in the seroprevalence of BTV antibodies in goats amongst the five districts of Karamoja (p = 0.9998). Out of the 150 whole blood samples that were tested for BTV RNA, 84 (56%) were positive for RNA. There was also a significant difference in the prevalence of BTV RNA in the goats, being highest in Amudat district and lowest in Kotido district (Figure 2). This study, which is the first of its kind in Uganda, showed a high seroprevalence of BT antibodies and active circulation of BTV in a high proportion of goats in the Karamoja region.
Discussion
The high seroprevalence of BT antibodies in goats in the region (90%) suggests that most of the goats have previously been exposed to the virus. The occurrence of BTV antibodies, together with the lack of reports of clinical disease, indicates that BTV infection in goats in the region may be subclinical or inapparent. This is not surprising, as local breeds of ruminants in Africa, where the virus is known to be endemic, are seen to be less clinically affected than exotic European breeds (MacLachlan 1994). Although overt clinical signs consistent with BT were not reported in the goats, further investigation is necessary in order to determine the clinical and sub-clinical significance of BTV and other pathogens in goats in Uganda.
BT can develop and spread when susceptible hosts, virus and competent insect vectors are all present at the same time. The presence of BTV antibodies and BTV RNA in the samples confirms the presence of circulating BTV in the Karamoja region.This region experiences low rainfall and has increasingly become prone to frequent droughts, leading to a lack of both pasture and water (NEMA 2006/2007). As a result there has been a large scale movement of livestock to alternative grazing areas. This increases the risk of spread of BTV and may partly explain the equally high seroprevalence in all five districts in the Karamoja region. The high seroprevalence and levels of BTV circulation seen in the goats in Karamoja, which is a semi-arid, drought-affected region of Uganda, gives an insight into what the situation could be like in more humid, rainy areas of Uganda, such as around the major lakes, where vectors are likely to be more abundant.
The presence of BTV RNA in the blood samples collected confirms the presence of circulating BTV in the goats in the region, although the study did not confirm the presence of infectious virus. The results of this study clearly indicate that BTV infection occurs in goats in the Karamoja districts, although overt clinical signs consistent with BT were not reported in the goats. It is highly likely that these goats, which do not show overt clinical signs, act as reservoirs for the virus and could pose a significant risk to naive ruminants if they are moved into this region. Therefore additional studies are needed in order to fully understand the epidemiology of BT, its clinical significance in different ruminant species and to determine the circulating BTV serotypes in the country. Understanding more about the molecular epidemiology of BTV at source in Africa may help to prevent future incursions into countries free of the disease.
Acknowledgements
The authors acknowledge greatly the technical assistance of the scientists at the Institute for Animal Health, Pirbright, UK and the laboratory staff of the National Animal Disease Diagnostics and Epidemiology Center, Ministry of Agriculture Animal Resources and Fisheries, Uganda
Competing interests
The authors declare that they have no financial or personal relationship(s) that may have inappropriately influenced them in writing this article.
Authors' contributions
C.A.L.O (University of West Indies, Trinidad and Tobago) and C.A.B. (Institute of Animal Health) were the project leaders. C.A. (National Animal Disease Diagnostics and Epidemiology Center) and S.M. (Makerere University) were responsible for the project design and E.N.M. (Makerere University) prepared samples and performed all of the experiments
References
Erasmus, B.J., 1990, 'Bluetongue virus', in Z. Dinter & B. Morein (eds.), Virus infections of ruminants, pp. 227-237, Elsevier Biomedical Press, Amsterdam. [ Links ]
CPRC, 2008, 'Understanding chronic poverty and vulnerability issues in Karamoja region', Chronic Poverty Research Centre, Kampala, Uganda. [ Links ]
Davies, F.G., Mungai, J.N. & Pini, A., 1992, 'A new Bluetongue virus serotype isolated in Kenya', Veterinary Microbiology 31, 2532. http://dx.doi.org/10.1016/0378-1135(92)90138-J [ Links ]
Gerdes, G.H., 2004, 'Bluetongue - a South African perspective', Veterinaria.italiana. 40, 39-42. PMid:20419632 [ Links ]
MacLachlan, N.J., 1994, 'The pathogenesis and immunology of bluetongue virus infection of ruminants', Comparative Immunological Microbiology and Infectious Diseases 17, 197-206. http://dx.doi.org/10.1016/0147-9571(94)90043-4 [ Links ]
Mertens, P.P.C., Maan, S., Samuel, A. & Attoui, H., 2005, 'Orbivirus', in C.M. Fauquet, M.A. Mayo, J. Maniloff, U. Desselberger & L.A. Ball (eds.), Virus taxonomy VIIIth report of the International Committee on Taxonomy of Viruses, pp. 466-483, Elsevier Academic Press, London. [ Links ]
NEMA, 2006/2007, 'State of the environment report for Uganda', National Environmental Management Authority, Kampala, Uganda. [ Links ]
OIE, 2003, 'Report of the meeting of the OIE working group on wildlife diseases', 71st General Session, 18-23 May 2003, Office International des Epizooties, Paris. [ Links ]
Saegerman, C., Berkvens, D. & Mellor, P.S., 2008., 'Bluetongue epidemiology in the European Union', Emerging Infectious Diseases 14, 539-544. [ Links ]
Shaw, A.E., Monaghan, P., Alpar, H.O., Anthony, S., Darpel, K.E., Guercio,A., et al., 2007, 'Development and initial evaluation of a real-time RT-PCR assay to detect bluetongue virus genome segment 1', Journal of Virological Methods 145, 115 126. http://dx.doi.org/10.1016/j.jviromet.2007.05.014, PMid:17586061 [ Links ]
Walton, T.E., 2004., 'The history of bluetongue and a current global overview', Veterinariaitaliana 40: 31-38. PMid:20419631 [ Links ]
Wells, E.A., 1962, 'A disease resembling bluetongue occurring in topi in Queen Elizabeth National Park', Veterinary Record 74, 1372-1373. [ Links ]
 Correspondence:
Correspondence:
Elijah Mulabbi
Postal address: PO Box 7062, Kampala, Uganda
Email: nichelijah@gmail.com
Received: 14 Aug. 2012
Accepted: 08 Mar. 2013
Published: 19 Apr. 2013














