Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
Journal of the South African Veterinary Association
versión On-line ISSN 2224-9435
versión impresa ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.83 no.1 Pretoria ene. 2012
SHORT COMMUNICATION
An outbreak of canine aflatoxicosis in Gauteng Province, South Africa
Luke F. ArnotI; Neil M. DuncanII; Heleen CoetzerIII; Christo J. BothaII, III
IOnderstepoort Veterinary Academic Hospital, University of Pretoria, South Africa
IIDepartment of Paraclinical Sciences, University of Pretoria, South Africa
IIIToxicology Laboratory, Agricultural Research Council-Onderstepoort Veterinary Institute, South Africa
ABSTRACT
Sporadic outbreaks of aflatoxicosis occur in dogs when they consume contaminated dog food. During 2011, low-cost brands of pelleted dog food were contaminated with very high concentrations of aflatoxins. Approximately 100 dogs were presented to the Onderstepoort Veterinary Academic Hospital. Clinically, the dogs were depressed to collapsed and icteric, with haematemesis, melaena and haematochezia. The most common pathological findings were icterus, gastro-enterorrhagia and hepatosis. On histopathological examination, fatty hepatosis and bile duct proliferation were observed. A consistent, very characteristic finding was the presence of a blue-grey granular material within the bile ducts. A total of 124 samples of the dog food fed to the affected dogs was analysed to determine aflatoxin concentrations. Concentrations ranged from below the limit of quantification (< 5 mg/kg) to 4946 mg/kg and six samples were submitted to determine the ratio of aflatoxins in the feed. It is estimated that well over 220 dogs died in the Gauteng Province of South Africa as a result of this aflatoxin outbreak.
Over the past few decades, there have been infrequent outbreaks of aflatoxicosis in dogs in southern Africa,1,2,3 with the previous published outbreak recorded in 1999.3 Aflatoxicosis is caused by potent hepatotoxic mycotoxins synthesised by the Aspergillus species.4 After absorption from the gastrointestinal tract, the aflatoxins are transported by the portal circulation and are filtered from the blood by the liver, which is consequently the primary organ affected.5
Aspergillus flavus and Aspergillus parasiticus often contaminate various food sources and fungal infestation can occur in the field and during storage or processing.6 These fungi are ubiquitous saprophytic moulds that grow on a variety of substrates such as groundnuts and maize, as well as feeds that contain grains.7 Certain genotypes of maize and peanuts are reportedly more susceptible to Aspergillus infection.8
Aflatoxins (AF) are difuranocoumarin derivatives and the four major metabolites are aflatoxin B1, B2 and G1 and G2.7 Aflatoxin B1 is the commonest, most toxic and most carcinogenic of the metabolites.8,9 Aflatoxicosis is dose-related, with the conversion of AFB1 into active electrophilic and alkylating metabolites that cause the hepatocellular toxicity.10 The rapid hepatocellular formation of a toxic AFB1-8,9-epoxide depletes hepatocellular glutathione (GSH), thereby overwhelming the natural detoxification pathways. The loss of GSH also compromises the detoxifying conjugation of AFB1 The accumulated AFB1 irreversibly binds to cell enzymes, proteins and DNA, causing impaired metabolism, gene transcription and protein synthesis.6
Species vary greatly in their susceptibility to AF, with dogs being highly susceptible.11,12 Animal species can be divided into three categories of susceptibility: those with a LD50 of 1 mg/kg or less, including dogs and cats, those with a LD50 of 10 mg/kg or more, such as pigs, and others that are resistant.9 Dogs appear more susceptible because of their inherently lower hepatocellular concentration of GSH compared with other species, as well as their inter-individual differences in activity of GSH-S-transferase.13,14 Furthermore, AF are reported to exert an anti-coagulation effect as a result of their coumarin-like structure.15 Exposure to lower AF concentrations in feed over a prolonged period can induce chronic liver failure, indicating a cumulative effect.7
From April to July 2011, a large number of dogs were affected during an AF outbreak in the Gauteng Province of South Africa. Over these months, it is estimated that 100 dogs were presented to the Outpatients Clinic of the Onderstepoort Veterinary Academic Hospital (OVAH), in which aflatoxicosis was strongly suspected based on presenting clinical signs and a history of being fed one of the three incriminated brands of dog pellets, namely Hi-PRO®, Buddy® and Legends®. All three brands are entry level, low-cost pelleted rations and were being sold, relatively cheaply, at petrol stations, vegetable markets, corner shops and similar outlets.
The most common complaints cited by owners of dogs presented to the OVAH included anorexia, food avoidance, vomition and lethargy. Owners often commented that the dogs had been reluctant to eat the incriminated pellets, but the dogs were enticed to consume the affected pellets by the addition of meat or gravy. Many owners reported that other dogs within the household exhibited similar signs and had subsequently died before veterinary assistance was sought.
On clinical examination of the more acute cases, dogs were depressed to collapsed and icteric, with haematemesis, melaena and haematochezia being commonly observed. In approximately 10% of cases petechiae were present on mucous membranes. The more chronic cases, presented during the second half of June and early July, were smaller breeds that had consumed some of the affected foods, but were simultaneously fed other foods such as table scraps with a resultant diluting effect on the AF-contaminated feed. These dogs presented with severe ascites, pale and icteric mucous membranes, were extremely depressed and often showed severe weight loss at the time of presentation at the OVAH.
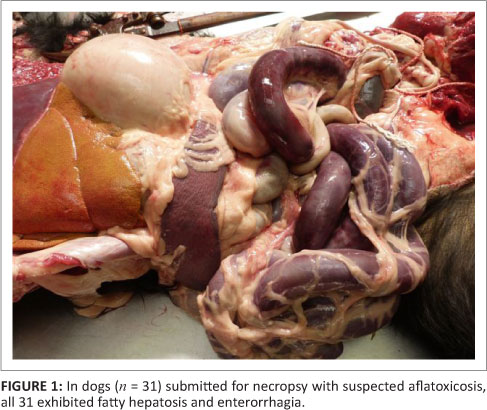
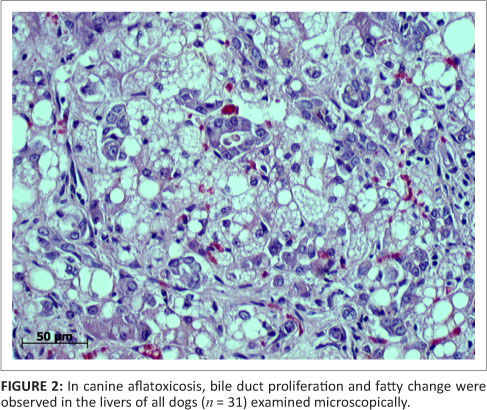
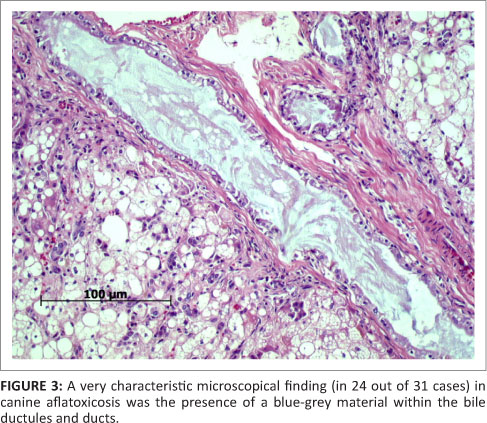
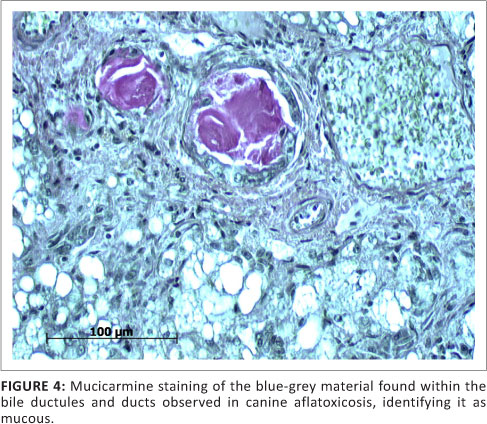
The vast majority of patients presented at the OVAH with suspected AF poisoning were euthanased because of owners' financial constraints and poor prognosis. The mortality rate of patients admitted for intensive treatment was extremely high as well, with only four patients being discharged. Following euthanasia or death, 31 post-mortem examinations were performed. The most consistent findings on macroscopic examination of the carcasses were icterus, gastro-enterorrhagia and hepatosis (31 from 31 cases) (Figure 1). Serosanguinous ascites was the next most common finding (19 from 31 cases), followed by serosal petechiation (13 from 31 cases) and pulmonary haemorrhage (12 from 31 cases). Less common findings were gall bladder oedema (9 from 31 cases), hydrothorax (6 from 31 cases), subcutaneous haemorrhage (3 from 31 cases) and meningeal haemorrhage (1 from 31 cases). On histopathological examination of the liver, fatty hepatosis and bile duct proliferation (Figure 2) were the most common findings (31 from 31 cases), with lesser numbers showing bile stasis (26 from 31 cases). The presence of a blue-grey granular material within the bile ducts was observed in 24 of the 31 cases (Figure 3), whilst 9 of the 31 cases exhibited signs of early portal fibrosis. Positive staining of the blue-grey material within the bile ducts with mucicarmine identified it as mucous (Figure 4).
Samples of the dog food eaten by affected dogs were screened by the Toxicology Laboratory, ARC-Onderstepoort Veterinary Institute using an accredited ELISA method (Veratox Aflatoxin Quantitative Test, Neogen Corporation, Lansing, USA) for total AF concentration. A total of 124 dog food samples were analysed. The concentrations ranged from below the limit of quantification (LQ < 5 mg/kg) to 4946 mg/kg and only 29 samples (23.4%) had AF levels below the LQ. The majority of positive samples were Hi-PRO®, but there were also higher than allowable AF concentrations detected in Legends® and Buddies®.
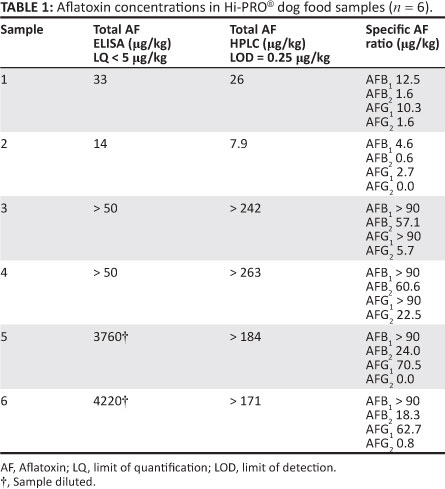
Six Hi-PRO® dog food samples were submitted for additional analysis (SANAS accredited HPLC method, Perishable Products Export Control Board Laboratory Services) to determine the specific ratio of the different AF concentrations (Table 1). Very high Aflatoxin B1 concentrations (> 90 mg/kg) were detected in four of the six samples. Regulations pertaining to the Fertilizers, Farm Feeds, Agricultural Remedies and Stock Remedies Act (Act No. 36 of 1947)16 stipulate a maximum Aflatoxin B1 level of 0.01 mg/kg (10 mg/kg) in complete feeds intended for pets. The manager of the feed company manufacturing Hi-PRO® indicated that a batch of groundnuts (peanuts) was obtained at some stage and included at a level of 5% in the feed (Van Vuuren D 2011, personal communication, April 24). It is surmised that the peanuts were the main culprit and explain the exceptionally high levels of aflatoxins determined during this outbreak when compared with previous outbreaks. The inspectorate of the abovementioned Act16 visited the premises and the registrar ordered an immediate withdrawal of the contaminated dog food and suspended manufacturing.
A questionnaire to establish the severity and extent of the aflatoxicosis outbreak was compiled and sent electronically to all South African Veterinary Association (SAVA) registered private practitioners via the office of the SAVA. Responses were received from around the country. The majority of cases were reported from Gauteng, apart from a few odd incidents in neighbouring towns. From the questionnaire, aflatoxicosis was diagnosed by private practitioners as the cause of death in an additional 120 dogs.
Owing to the sporadic and relatively rare occurrence of such outbreaks in dogs, the initial cases are often misdiagnosed by private veterinarians.3 Clinicians should always consider aflatoxicosis as a differential diagnosis when dealing with dogs presenting with the abovementioned clinical signs. A very conservative estimate of dog fatalities from the recent outbreak is in the region of 220 dogs. However, this is probably only the 'tip of the iceberg', as often only the last remaining dog in the family was presented. In addition, as these dog food brands were entry level, low-cost brands, many dog owners from the poorer socio-economic group in the townships within Gauteng may have been feeding these affected brands. They would not have presented affected dogs to a veterinarian because of a lack of access to veterinarians in these areas, as well as financial constraints, and these deaths would not have been accounted for.
Acknowledgements
The authors would like to acknowledge the SAVA for distributing the Aflatoxin survey questionnaire to all private practitioners who are members and would also like to thank the private practitioners who completed and submitted the questionnaire.
Competing interests
The authors declare that they have no financial or personal relationship(s) which may have inappropriately influenced them in writing this paper.
Authors' contributions
L.F.A. (University of Pretoria) performed most of the clinical examinations. N.M.D. (University of Pretoria) performed necropsies and microscopical examination of tissue samples. H.C. (Onderstepoort Veterinary Institute) performed the toxicological analysis. C.J.B. (University of Pretoria) interpreted the toxicological results. C.J.B. (University of Pretoria), L.F.A. (University of Pretoria) and N.M.D. (University of Pretoria) wrote the manuscript.
References
1. Bastianello SS, Nesbit JW, Williams MC, Lange AL. Pathological findings in a natural outbreak of aflatoxicosis in dogs. Onderstepoort J Vet Res. 1987;54:635-640. PMid:3444619 [ Links ]
2. Naicker D, Botha CJ. Aflatoxins in dog feed. Vet News: Newsletter of the South African Veterinary Association. 2005 Sept; 26. [ Links ]
3. Reyers F, Miller DB. Canine aflatoxicosis. Paper presented at: Congress of the South African Veterinary Association; 2000 Sept 20-22; Durban, South Africa. [ Links ]
4. Bennett JW, Klich M. Mycotoxins. Clin Microbiol Rev. 2003;16:497-516. http://dx.doi.org/10.1128/CMR.16.3.497-516.2003, PMid:12857779 [ Links ]
5. Miller DM, Wilson DM. Veterinary diseases related to aflatoxins. In: Eaton DL, Groopman JD, editors. The toxicology of aflatoxins. London: Academic Press, 1994; p. 347-364. [ Links ]
6. McClean M, Dutton MF. Cellular interaction and metabolism of aflatoxin: An update. Pharmacol Therapeut. 1995;65:163-192. http://dx.doi.org/10.1016/0163-7258(94)00054-7 [ Links ]
7. Kellerman TS, Coetzer JAW, Naude TW, Botha, CJ. Plant poisonings and mycotoxicoses of southern Africa. Cape Town: Oxford University Press; 2005. [ Links ]
8. Edds GT. Aflatoxins. In: Shimoda W, editor. Conference on mycotoxins in animal feeds and grains related to animal health. Rockville: US Department of Commerce, National Technical Information Service. PB-300 300. Food and Drug Administration Report No: FDA/BVM-79/139, 1979; p. 80-164. [ Links ]
9. Schoental R. Aflatoxins. Annu Rev Pharmacolog. 1967;7:343-353. http://dx.doi.org/10.1146/annurev.pa.07.040167.002015, PMid:5337085 [ Links ]
10. Guengerich FP. Cytochrome P450 oxidations in the generation of reactive electrophiles: Epoxidation and related reactions. Arch Biochem Biophys. 2005;409:59-71. http://dx.doi.org/10.1016/S0003-9861(02)00415-0 [ Links ]
11. Dereszynski DM, Center SA, Randolph JF, et al. Clinical and clinicopathologic features of dogs that consumed foodborne hepatotoxic aflatoxins: 72 cases (20052006). J Am Vet Med Assoc. 2008;232:329-1337. http://dx.doi.org/10.2460/javma.232.9.1329, PMid:18447777. [ Links ]
12. Raney KD, Meyer DJ, Ketterer B. Glutathione conjugation of aflatoxin B1 exo- and endo-epoxides by rat and human glutathione S-transferases. Chem Res Toxicol. 1992;5:470-478. http://dx.doi.org/10.1021/tx00028a004, PMid:1391613 [ Links ]
13. Center SA, Warner KL, Erb HN. Liver glutathione concentration in dogs and cats with naturally occurring liver disease. J Vet Res. 2002;63:1187-1197. http://dx.doi.org/10.2460/ajvr.2002.63.1187, PMid:12171175 [ Links ]
14. Watanabe T, Sugiura T, Manabe S. Low glutathione S-transferase dogs. Arch Toxicol. 2004;78:218-225. http://dx.doi.org/10.1007/s00204-003-0536-x, PMid:14685647 [ Links ]
15. Center SA. Acute hepatic injury: Hepatic necrosis and fulminant hepatic failure. In: Guilford WG, Center SA, Strombeck DR, Williams DA, Meyer DJ, editors. Strombeck's Small Animal Gastroenterology. 3rd ed. Philadelphia: WB Saunders Company, 1996; p. 654-704. [ Links ]
16. Fertilizers, Farm Feeds, Agricultural Remedies and Stock Remedies Act (Act No. 36 of 1947), South Africa. Available from: Government Gazette No. R 227, 2009 March 6. [ Links ]
 Correspondence to:
Correspondence to:
Christo Botha
Private Bag X04
Onderstepoort 0110
South Africa
christo.botha@up.ac.za
Received: 18 Nov. 2011
Accepted: 16 Jan. 2012
Published: 29 Mar. 2012














