Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Journal of the South African Veterinary Association
On-line version ISSN 2224-9435
Print version ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.82 n.4 Pretoria Dec. 2011
CLINICAL REVIEW KLINIESE OORSIG
Treatment rationale for dogs poisoned with aldicarb (carbamate pesticide)
L F ArnotI,II; D J H VealeIII; J C A SteylII; J G MyburghII,*
IOutpatients Clinic, Onderstepoort Veterinary Academic Hospital, Department of Companion Animal Clinical Studies, Faculty of Veterinary Science, University of Pretoria, Private Bag X04, Onderstepoort, 0110 South Africa
IIDepartment of Paraclinical Sciences, Faculty of Veterinary Science, University of Pretoria, Private Bag X04, Onderstepoort, 0110 South Africa
IIITygerberg Poison Information Centre, Division of Pharmacology, Department of Medicine, Stellenbosch University, PO Box 19063, Tygerberg, 7505 South Africa
ABSTRACT
The treatment rationale for dogs poisoned by aldicarb is reviewed from a pharmacological perspective. The illegal use of aldicarb to maliciously poison dogs is a major problem in some parts of the world. In South Africa, it is probably the most common canine poisoning treated by companion animal veterinarians. Aldicarb poisoning is an emergency and veterinarians need to be able to diagnose it and start with effective treatment immediately to ensure a reasonable prognosis. Successful treatment depends on the timely use of an anti-muscarinic drug (e.g. atropine). Additional supportive treatment options, including fluid therapy, diphenhydramine, benzodiazepines and the prevention of further absorption (activated charcoal) should also be considered. Possible complications after treatment are also briefly discussed.
Keywords: aldicarb, carbamate, malicious poisoning, supportive treatment, Temik®.
INTRODUCTION
Malicious poisoning of dogs, especially with aldicarb (carbamate pesticide), has become an increasingly common emergency faced by companion animal veterinarians in some parts of the world20,43,64.To date, there have been very few published articles discussing the treatment of aldicarb poisoning in dogs2,64, with most published reports focusing on human cases9,31,66. Aldicarb poisoning is an emergency 2,51,64 and patients may die within minutes after ingestion due to respiratory failure22,31,51. Therefore, veterinarians need to be able to promptly diagnose aldicarb toxicity and start with effective treatment immediately to offer a reasonable prognosis. The main focus of this article is to review the treatment options for veterinarians dealing with accidental or malicious aldicarb poisonings.
BACKGROUND INFORMATION TO THE PROBLEM IN SOUTH AFRICA
The malicious use of aldicarb in South Africa to poison dogs is of major concern and it is arguably the most common intoxication treated by veterinarians64. The Onderstepoort Veterinary Academic Hospital (OVAH), Faculty of Veterinary Science, University of Pretoria, South Africa, treats between 50 and 100 clinical aldicarb cases per annum (L F Arnot, OVAH, unpubl. data.) giving some indication of the magnitude of the problem. The number of cases seen depends on criminal activity within the area64. The use of aldicarb to poison animals is, however, not only restricted to South Africa, with reports indicating large-scale intentional malicious poisoning of dogs and other species from the United Sates of America2,20 and Spain43.
The extensive use of aldicarb in agriculture, and resultant easy accessibility, has been suggested as 1 of the reasons why the malicious use of aldicarb in recent years has increased, in preference to the more traditional poisons such as strychnine43,57,59,62. Criminals use aldicarb to eliminate dogs within residential areas with the intention of gaining access to these properties for malicious activities. Cats are generally not intentionally targeted, but are assumed to be incidentally affected64.
Bayer CropScience (Bayer CropScience, Isando, South Africa) is the sole distributor of aldicarb in South Africa. Aldicarb is marketed world wide under the trade name of Temik®7. It is an agricultural insecticide and nematicide7,24,26, containing 15 % aldicarb and is usually formulated as small black granules (Fig. 1). Temik® is registered for agricultural use in South Africa under the Fertilizers, Farm Feeds, Agricultural and Stock Remedies Act (Act 36 of 1947) and the sale of it to farmers within South Africa is tightly controlled. Only certified farmers are legally able to purchase the product and strict control is enforced regarding the use and storage of the product on farms (Act 36 of 1947) (P Fourie, Bayer CropScience, pers. comm.).
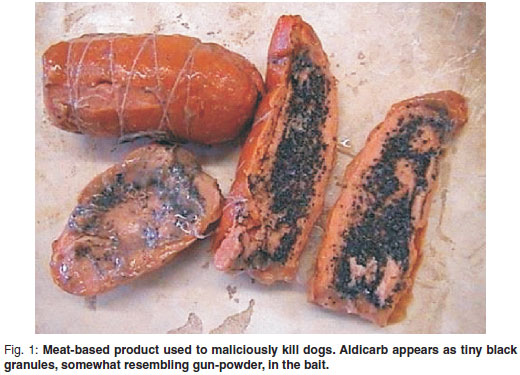
In 2001, Bayer CropScience began adding an extremely bitter substance (denatonium benzoate) to Temik® in order to prevent its use for suicides or homicides in humans (P Fourie, Bayer CropScience, pers. comm.). Unfortunately most animal species do not possess the specific taste receptors found in humans that detect the bitter taste sensation and this protective measure therefore does not absolutely guarantee that animals would not ingest aldicarb treated with denatonium benzoate19.
It is estimated that at least 60 % of the aldicarb-containing pesticides used in South Africa for the malicious poisoning of dogs are illegally smuggled across the border into South Africa from neighbouring countries such as Mozambique and Zimbabwe, where strict agricultural product control is lacking (G H Verdoorn, Griffon Poison Information Centre, pers. comm.). In South Africa Temik® is sold illegally by informal street traders as a rodenticide called 'Two Step'56 and it is often the cause of accidental poisoning in children and intentional poisoning in adults63. Similar problems are experienced in the United States of America with aldicarb sold illegally as a rodenticide under the name Tres Pasitos (3 little steps), and is sometimes used in human suicide attempts9,45,66. It has also been reported that aldicarb is illegally used as a household rodenticide in Brazil and the Caribbean Islands and that humans are sometimes poisoned51.
Criminals in South Africa wishing to gain access to a property where dogs are present typically insert aldicarb granules into cheap meat baits, such as viennas, sausages or polony, to kill the resident dog(s) (Fig. 1). Pieces of bait are clandestinely thrown over property fences for the dogs to consume, with criminals gaining access to the property once the dogs have been eliminated.
ALDICARB (CARBAMATE) POISONING: DESCRIPTION AND DIAGNOSIS
Chemistry of aldicarb
Aldicarb (2-methyl-2-(methylthio)proprioaldehyde-o-methylcarbamoyloxime) is an extremely toxic substance7,20,24,45 and is classified as an 'extremely hazardous pesticide' (class 1a) according to the World Health Organization classification of pesticides67. The rat oral LD50 varies between 0.46 mg/kg21 and 0.93 mg/kg62.In humans it is 0.80 mg/kg66. The canine oral LD50 for Temik® is about 6.5 mg/kg and it contains 15 % aldicarb (G H Verdoorn, Griffon Poison Information Centre, pers. comm.).
Mode of action of aldicarb in dogs
Both the carbamate and organophosphate pesticides are classified as cholinesterase inhibitors24,60,61. Carbamates, such as aldicarb, cause inhibition of the activity of acetylcholinerase (AChE), an enzyme responsible for the hydrolysis of the neurotransmitter acetylcholine (ACh) into choline and acetic acid21,30,33. The ingestion of a carbamate results in the inactivation of AChE activity by carbamylation of the serine hydroxyl group located at the active site of AChE31,52,59. The carbamylation of the active site on the AChE is temporary and rapidly reversible, and AChE activity is restored when spontaneous hydrolysis of the carbamylated enzyme occurs (often within an hour), resulting in the release of AChE26,31,34,59.
Exposure to organophosphates results in the phosphorylation of the same active site of AChE, but a stable and irreversible bond is formed after 24-48 hours. This process is referred to as 'aging'15,21,25,31,34,60.
Once AChE has been inactivated, ACh accumulates at muscarinic and nicotinic cholinergic receptors in the sympathetic and parasympathetic nervous systems, as well as at neuromuscular junctions resulting in continuous stimulation of these receptors10,15,31,33. Central nervous system (CNS) toxicity is less common in carbamate poisoning because many carbamates do not readily cross the blood-brain barrier (BBB)35,55 .
Clinical signs
The clinical presentations of carbamate and organophosphate poisonings are clinically indistinguishable from each other14,57,61. If a dog presents with clinical signs indicating possible organophosphate or carbamate toxicity, unless there is a clear history that an organophosphate was used (for example, history that the dog was recently dipped by the owner with an organophosphate ectoparasicide), the clinician should assume carbamate poisoning, due to the very high prevalence of malicious misuse of aldicarb in South Africa.
The clinical signs of aldicarb poisoning are associated with muscarinic, nicotinic and central effects33,57,60. Muscarinic receptors are located in the smooth muscle of glands, intestine, cardiac muscle, CNS and the iris. The nicotinic cholinergic receptors are located at the neuromuscular junctions of striated muscle and the ganglionic synapses of the autonomic ganglia25.
Muscarinic signs are often very pronounced and include bradycardia, miosis, bronchospasm, bronchorrhea, hypersalivation, lachrymation, urination and diarrhoea. Nicotinic signs include muscle tremors, fasciculations, muscle stiffness, weakness and paralysis8,16. Prolonged muscle activity finally results in exertional rhabdomyolysis41. Central effects include apprehension and seizures, followed by CNS depression and coma31.
Hyper-stimulation of the nicotinic receptors may cause tachycardia, mydriasis and hypertension, instead of the bradycardia, miosis and hypotension that are seen when muscarinic stimulation predominates66. Tachycardia may also be due to hypoxia. This is an important consideration when a patient is initially examined and subsequently treated. When using atropine as an antidote, it is important to remember that the endpoint for atropinisation is when secretions have dried up, not the presence of tachycardia or dilated pupils66.
In dogs, the clinical signs most often reported by veterinarians include muscle tremors and hypersalivation, followed by emesis, miosis, bradycardia, seizures and dyspnoea64. Excessive urination, paresis and paralysis have only been occasionally recorded. Death is caused by respiratory failure, due to bronchospasm, paralysis of the diaphragm and intercostal muscles, and depression of the respiratory centre16,22,31. In a study undertaken in humans the most common muscarinic effect was diarrhoea, the main nicotinic sign was muscle fasciculations and CNS depression occurred in about 50 % of patients51.
Diagnosis
At the Outpatients clinic of the OVAH, most aldicarb emergencies are diagnosed on the basis of the presenting clinical signs and a history of a very acute onset. Owing to the fact that in a clinical emergencies there is little time to confirm a definite diagnosis of aldicarb poisoning before treatment commences, a positive response to initial anti-muscarinic treatment can assist in confirming a preliminary diagnosis.
Some owners may report that the dog vomited up unfamiliar food in which the aldicarb granules were concealed or that they found pieces of meat products containing small black granules in the vicinity of the animal.
Measurement of decreased cholinesterase concentrations in the blood may be considered to confirm the diagnosis of aldicarb toxicity1,5,24. However, due to the fact that the temporary AChE-carbamate bond is rapidly reversed, this test must preferably be performed immediately, seeing that the diagnostic test results could become inconclusive if the test is not performed within a few hours27.A blood sample may be collected prior to initiating the treatment and the AChE concentration determined as soon as possible.
THERAPEUTIC OPTIONS
Due to the highly toxic nature of aldicarb, dogs presented to a veterinary clinic with the 'classic' clinical signs indicative of aldicarb toxicity, or a relevant history, should always be handled as an emergency. At the OVAH it is generally accepted that without prompt treatment most malicious cases will die within 20-30 min of ingestion, leaving little time for the attending veterinarian to perform additional tests to confirm the diagnosis.
A proposed treatment protocol for dogs poisoned with aldicarb is summarised in Table 1. The sequence of the different recommended procedures detailed in the table is only a guide and the treatment protocol may need to be adjusted on a case by case basis depending on the severity of the poisoning. However, it is essential to administer atropine as soon as possible.
The mortality of clinical cases is high, with a mortality rate of 25-50 % recorded even after prompt treatment64. Treatment is directed towards reversing or preventing over-stimulation of the muscarinic receptors by the accumulation of ACh in the junctions31,33,66. Effective therapy can be expensive, often requiring intensive care and monitoring with a guarded prognosis, which may cause some owners to elect for humane euthanasia instead of treatment64.
Immediate action and fluid therapy
Initially, an intravenous catheter should be placed, as intravenous access is crucial for the administration of drugs and fluids33. At the OVAH, if the patient is convulsing, a naso-oesophageal tube is inserted to reduce the risk of aspiration of medication requiring administration per os.
Intravenous crystalloid fluids should be administered to all patients55. If the patient is vomiting or exhibiting severe diarrhoea, the fluid rate should be increased to accommodate these losses. Otherwise, fluid therapy should be sufficient to maintain hydration and prevent hypovolaemia33,55.
Muscarinic receptor antagonists
The administration of an antimuscarinic drug as soon as possible is critically important in all cases. Constant over-stimulation of the muscarinic receptors results in severe and fatal consequences. Severe bradycardia and bronchospasm are often life-threatening and need to be treated immediately55. Muscarinic receptor antagonists compete with ACh for a common binding site on the muscarinic receptors33. Two specific muscarinic receptor antagonists that may be considered are atropine sulphate and glycopyrrolate50.
Atropine sulphate
Atropine is the drug of choice in the treatment of acute aldicarb toxicity. It is a competitive muscarinic receptor antagonist at postganglionic parasympathetic neuroeffector sites50. Atropine administration reverses the severe bronchospasm, bronchorrhea, bradycardia and circulatory depression associated with over-stimulation of the muscarinic receptors31,33,61. Atropine activity is, however, specific to muscarinic receptors and has no effect on the nicotinic receptors or the AChE-carbamate complex50,55. Atropine administration will therefore not counteract the muscle tremors, weakness and paralysis associated with aldicarb toxicity8,16,31. Atropine may lower the cerebral glucose threshold and thereby reduce the likelihood of brain damage during seizures40,48.
The dose of atropine required to counteract the effects of aldicarb toxicity is extremely high, more than 10 times the recommended pre-anaesthetic dose. The dose is between 0.2-0.5 mg/kg with a quarter of the dose given IV and the balance administered subcutaneously (SC)18. This dose can be repeated every 15-30 minutes as needed, until the bronchospasm, excessive bronchial secretions and bradycardia are alleviated and mydriasis is seen31,66. A dose of up to 2 mg/kg has also been cited to counteract carbamate toxicity50, but clinical experience has indicated that a dose of up to 0.5 mg/kg appears to be adequate in most cases. If IV access is problematic, the atropine can be administered intramuscularly (IM) or SC.
Atropine is well absorbed from all routes of administration, reaching peak effects 3-4 min post IV administration. It is widely distributed and crosses the BBB. It is metabolised in the liver and excreted in the urine, with up to 50 % being excreted in the unchanged form50.
Atropine can cause a range of dose-related adverse effects50. It crosses the BBB easily and central signs of atropine toxicity are common complications in humans treated with high doses3. The very high doses necessary to treat aldicarb poisoning effectively in dogs may result in neurological complications such as drowsiness, ataxia, seizures and respiratory depression. Gastro-intestinal side effects include xerostomia, dysphagia, constipation and vomition. Ocular signs include blurred vision, mydriasis, cycloplegia and photo-phobia. Cardiovascular signs include tachycardia, hypertension, arrhythmias and cardiovascular failure50.
Glycopyrrolate
Glycopyrrolate could be considered as an alternative drug to atropine for the treatment of aldicarb toxicity6,33, or it could also be used in combination with atropine3. It is a quaternary ammonium anti-muscarinic agent33 and is registered for use in dogs and cats as a pre-anaesthetic agent to treat sinus bradycardia, sino-atrial arrest and incomplete atrioventricular (AV) block50. Glycopyrrolate is more effective in controlling excessive bronchial secretions and bradycardia in rats and rabbits than atropine6. However, similar references could not be found for dogs.
In contrast to atropine, glycopyrrolate is completely ionised after administration, and is therefore poorly lipid soluble. Glycopyrrolate does not enter the CNS in any appreciable amounts and as a result, does not cause the CNS adverse effects seen with atropine after high dose administration50. This may be used to advantage in those patients showing signs of atropine toxicity. A combination of atropine and glycopyrrolate has been used in the treatment of human cases of organophosphate toxicity3.
The canine LD50 of glycopyrrolate is 25 mg/kg after IV administration50. A dose of 0.5 mg/kg has been suggested for the treatment of organophosphate and carbamate poisoning cases46. After IV administration the onset of action of glycopyrrolate is 1 min and 30-45 min following IM or SC administration respectively. It is rapidly eliminated with only minimal serum levels detectable 3 hours after IV administration50.
Diphenhydramine
Although diphenhydramine is not used routinely for the treatment of organophosphate and carbamate poisonings in humans (not registered for this use in humans), it is often used 'extra label' by some veterinary clinicians to counteract the nicotinic signs10.
Diphenhydramine is a 1st generation antihistamine that is a competitive antagonist at the H1 receptors50. It is nonselective and therefore also an antagonist at muscarinic receptors with some sedative, anticholinergic, antitussive and antiemetic effects23,50. Diphenhydramine is used as an adjunctive treatment in aldicarb poisoning in dogs to prevent over exertion of muscles by preventing excess stimulation of the nicotinic receptors at the neuromuscular junctions10,23,50.
Diphenhydramine should be administered orally at presentation10. Dosing via a naso-oesophageal tube is essential to prevent aspiration if the patient is convulsing or has a weak swallowing reflex. A dose of between 1 and 4 mg/kg, per os, every 8 hours has been advised for dogs and can be administered for up to 3 weeks if necessary10,23. It is well absorbed from the gastrointestinal tract, with a 1st pass effect of between 40-60 %. It may cause severe sedation in dogs and further treatment should be reconsidered if unacceptable sedation is observed50.
Oximes
The use of oximes in the treatment of organophosphate poisoning is commonly referred to in human medical literature31,39,61,65. Oximes, such as pralidoxime and obidoxime, are phosphorylated AChE enzyme reactivators, indicated for the treatment of organophosphate toxicity before the process of 'aging' has occurred31,39,65.
In contrast to organophosphate toxicity, the carbamate-induced inhibition of AChE is rapidly reversible with hydrolysis of the carbamylated complex often occurring within an hour31. The use of oximes in carbamate poisoning is controversial5,12,29,55 or even contraindicated27,50 and is therefore not recommended for the treatment of aldicarb poisoning in dogs. Many studies have indicated that the treatment of carbamate poisonings in animals with oximes has resulted in a protective ratio of less than 1 (i.e. worse than no treatment at all)55. Potential toxicity of oximes when used in the treatment of carbaryl (carbamate) poisoning has been reported55. However, recent data suggest that this concern may be unwarranted27,55.
If uncertainty exists as to whether the poisoning is due to an organophosphate or carbamate, the veterinarian may choose to include an oxime in the treatment protocol. In such a case, the dosage of pralidoxime in dogs is 20 mg/kg, 2-3 times a day, with slow IV administration of the initial dose and subsequent doses given IM or SC17. The dosage of obidoxime (10 % solution) is 40 mg/kg in dogs, by slow IV administration, followed by IM or SC administration, 2-3 times a day50.
Benzodiazepines
Some patients may present in a state of seizure. Diazepam (benzodiazepine) is the drug of choice to control seizure activity33,54, reduce anxiety and to induce muscle relaxation31. Benzodiazepines cause hyperpolarisation of neurons, reducing cholinergically induced depolarisation, resulting in cessation of propagation of convulsions39. Benzodiazepines are gamma-aminobutyric acid (GABA) receptor agonists and do not activate the receptor directly, but alter GABA binding at the GABAA receptors in an allosteric fashion39.
It has also been reported that diazepam acts synergistically with atropine, potentiating the efficacy of a reduced dose of atropine, improving survival and preventing CNS complications14,31,54.Ithas been shown that diazepam appears to decrease synaptic release of ACh in humans31. A dose of 0.5-1 mg/kg may be administered IV to dogs that are seizuring49.
Prevention of systemic absorption
Emesis
Stimulation of emesis shortly after intake would be beneficial31. The use of apomorphine is indicated and dogs may readily be induced to vomit by placing 0.25 mg/kg apomorphine, diluted in 3-5 ml water, on the conjunctival mucosa28. It can also be administered IV at 0.04 mg/kg53, or by the IM or SC routes at 0.08 mg/kg44. The induction of emesis is contraindicated in dogs that are already vomiting, convulsing or losing consciousness, because of the high risk of ingesta aspiration in these cases50.
Adsorption
The use of activated charcoal powder as an adsorbent is recommended in all cases to bind the aldicarb within the gastrointestinal tract, thus limiting further systemic absorption31,50. Activated charcoal powder is a highly porous form of carbon with a large surface area capable of adsorbing ingested toxins50. At the OVAH, the powder formulation of activated charcoal is preferred, as it provides a much larger adsorptive surface area than the tablet formulation.
Administration of activated charcoal per os is contraindicated in a patient that is convulsing or has a weak swallowing reflex because of the high risk of aspiration and resultant severe pneumonia in these cases50. A naso-oesophageal tube should be considered in these situations, particularly if the clinician plans to administer multiple doses of activated charcoal over time.
At the OVAH activated charcoal is administered for the initial adsorption of any aldicarb present in the gastrointestinal tract31,50. Thereafter the administration of multiple doses of activated charcoal (chronic treatment) is primarily to adsorb any aldicarb that re-circulates back into the gastrointestinal tract via enterohepatic circulation50.
Activated charcoal powder can initially be administered at a dose of 0.5-2.0 g/kg per os or via a naso-oesophageal tube. At the OVAH activated charcoal is mixed with water to form a slurry (1 g charcoal with 5 ml water) and lactulose is added to reduce the risk of constipation50. Activated charcoal treatment can be administered every 3-6 hours. Once the dog is eating, the activated charcoal can be mixed with palatable food, as it has no taste or smell50.
In contrast to veterinary practice, the use of activated charcoal in human aldicarb poisoning is not recommended, especially not repeated administrations, as it has been observed to be associated with constipation, impaction and obstruction by 1 of the authors (DJHV). In addition, the effectiveness of activated charcoal therapy in poisoned human patients is also not conclusive31.
Increase in the movement of ingesta
Lactulose is a disaccharide laxative and by drawing water into the colon, thus increasing the osmotic pressure, has a laxative effect. The chronic use of activated charcoal in patients can predispose to constipation, which is alleviated by the lactulose. In addition, the laxative effect of lactulose will increase the speed of gastrointestinal tract emptying, which will further assist in the rapid elimination of the ingested aldicarb. The dose of lactulose in dogs is 1 ml per 4.5 kg body mass 3 times daily50.
POTENTIAL COMPLICATIONS OF ALDICARB TOXICITY
In view of the fact that the aldicarb case fatality rate is so high, complications are not often seen. However, in a case that survived the initial clinical crisis, complications may be diagnosed. Although the focus of this review is the treatment rationale, veterinarians need to be aware of potential complications.
Pancreatitis
A serious complication seen with organophosphate or carbamate poisonings is pancreatitis4,38,42. Acute necrotic necrohaemorrhagic pancreatitis has been reported in about 12 % of human organophosphate and carbamate cases4. Pancreatitis was also experimentally reproduced in dogs that received diazinon (an organophosphate) IV32.
Observations from the OVAH confirmed that pancreatitis is a potential, significant complication in dogs. The clinical signs are typically seen within a few days after the intake of aldicarb, and they usually present with an acute abdomen. This condition is less likely in patients that received immediate and effective treatment42. Atropine prevents the over-stimulation of muscarinic receptors causing smooth muscle contractions (also the sphincter of Odi) in all gastrointestinal organs4. In addition, pancreatic secretion (cholinergic effect) is stimulated by organophosphate and carbamate pesticides13,38.
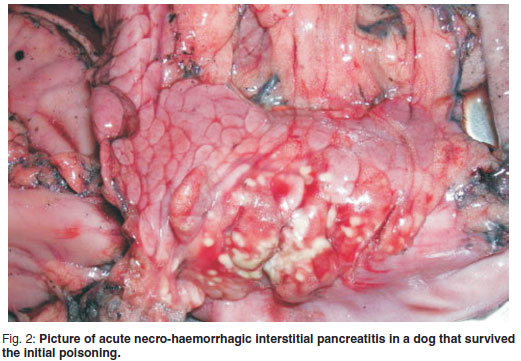
Acute necro-haemorrhagic interstitial pancreatitis (Fig. 2) was confirmed by the Pathology Section (Department of Para-clinical Sciences, Faculty of Veterinary Science, University of Pretoria) in dogs presented for necropsies after unsuccessful treatment. This is rarely seen in cases that died peracutely. A possible explanation for the development of pancreatitis after severe cholinergic stimulation is that the constrictive spasm of the sphincter of Odi and the increase in intraductal pressure increase the risk of activated pancreatic secretions escaping into the interstitial and peripancreatic tissue4,13,38.
Intermediate syndrome
The intermediate syndrome (IMS) was 1st reported in the mid-1980s, describing clinical signs, mostly of muscle paralysis, after the successful treatment (and recovery) of organophosphate poisoning cases11,15,61. It is called the IMS because it is observed after the acute cholinergic signs, but before organophosphate-induced delayed polyneuropathy (OPIDP), which is rarely seen11. It was generally accepted that it is only associated with organophosphates, until Paul and Mannathukkaran47 reported the development of IMS clinical signs in a human patient poisoned with carbofuran (carbamate).
Polyneuropathy
Organophosphate-induced delayed polyneuropathy (OPIDP) is classically associated with organophosphates36,58. However, some carbamates (e.g. methylcarbamates) also inhibit neuropathy target esterase (NTE), the target enzyme in OPIDP development. Based on mechanistic considerations, carbamates were thought to be unable to cause polyneuropathy37. Subsequently, 3 human polyneuropathy cases that occurred after poisoning by methylcarbamates were reported37.
CONCLUSION
Aldicarb poisoning is the most common poisoning in dogs seen by veterinarians in South Africa. Owing to the toxic nature of aldicarb, immediate and effective treatment is essential to obtain a positive outcome. Atropine is the drug of choice to counteract the life threatening effects such as bronchospasm, increased bronchial secretions and bradycardia. Diphenhydramine may be considered to antagonise the nicotinic effects of aldicarb, thereby reducing muscle fasciculations and tremors. Diazepam may act synergistically with atropine, allowing for a reduction in atropine dosage. Diazepam also has muscle relaxant and sedative effects that may be beneficial in some patients. The use of oximes is not recommended in carbamate poisoning. The use of activated charcoal as an adsorbent to prevent further gastrointestinal absorption is essential. Pancreatitis should be considered in a dog that presents with signs of acute abdomen after aldicarb exposure.
ACKNOWLEDGEMENTS
The authors wish to acknowledge the help and support received from the Departments of Paraclinical Sciences and Companion Animal Clinical Studies, Faculty of Veterinary Science, University of Pretoria.
REFERENCES
1. Aaron C K 2001 Organophosphate and carbamates. In Ford M D, Delaney K A (eds) Clinical toxicology (1st edn). W B Saunders, Philadelphia: 819-828 [ Links ]
2. Anastasio J D, Sharp C R 2011 Acute aldicarb toxicity in dogs: 15 cases (2001- 2009). Journal of Veterinary Emergency and Critical Care 21:253-260 [ Links ]
3. Arendse R, Irusen W 2009 An atropine and glycopyrrolate combination reduces mortality in organophosphate poisonings. Human and Experimental Toxicology 28:715-720 [ Links ]
4. Aslan S, Cakir Z, Emet M, Serinken M, Karcioglu O, Kandis H, Uzkeser M 2010 Acute abdomen associated with organophosphate poisoning. Journal of Emergency Medicine (in press) DOI: 10.1016/j.jemermed. 2010.05.072 [ Links ]
5. Aygun D, Doganay Z, Altinop L, Guven H, Onar M, Deniz T, Sunter T 2002 Serum acetylcholinerase and prognosis of acute organophosphate poisoning. Journal of Toxicology - Clinical Toxicology 40:903-910 [ Links ]
6. Bardin P G, Van Eeden S F 1990 Organophosphate poisoning: grading the severity and comparing treatment options between atropine and glycopyrrolate. Critical Care Medicine 18:956-960 [ Links ]
7. Baron R L 1994 A carbamate insecticide: a case study of aldicarb. Environmental Health Perspective 102:23-27 [ Links ]
8. Campbell A, Chapman M 2000 Carbamate insecticides. In Handbook of poisonings in dogs and cats. Blackwell Science, Oxford: 102-105 [ Links ]
9. Centers for Disease Control and Prevention (CDC) 1997 Poisonings associated with illegal use of aldicarb as a rodenticide - New York City 1994-1997. Morbidity and Mortality Weekly Report 46:961-963 [ Links ]
10. Clemmons R M, Meyers D J, Sundlof S F, Rappaport J J, Fossler M E, Hubbell J, Borsey-Lee M R 1984 Correction of organophosphate-induced neuromuscular block by diphenhydramine. American Journal of Veterinary Research 45:2167-2169 [ Links ]
11. De Bleecker J L 2006 Intermediate syndrome in organophosphate poisoning. In Gupta R C (ed.) Toxicology of organophosphate and carbamate compounds. Elsevier Academic Press, Amsterdam: 371-380 [ Links ]
12. DeSilva H J, Wijewickerema R, Senanayeke N 1992 Does pralidoxime affect the outcome of management in acute organophosphate poisoning? Lancet 339:1136-1138 [ Links ]
13. Dressel T D, Goodale R L, Hunninghake D B, Borner J W 1979 Sensitivity of the canine pancreatic intraductal pressure to subclinical reduction in cholinesterase activity. Annals of Surgery190:6-12 [ Links ]
14. Eddelston M, Buckley N A, Eyer P, Dawson A H 2008 Management of acute organophosphorus pesticide poisoning. Lancet 371:597-607 [ Links ]
15. Erdman A R 2004 Insecticides. In Dart R C (ed.) Medical toxicology (3rd edn). Lippincot Williams & Wilkins, Philadelphia: 1475- 1496 [ Links ]
16. Fikes F D 1990 Organophosphorous and carbamate insecticides. Veterinary Clinics of North America: Small Animal Practice 20:353-367 [ Links ]
17. Fikes F D 1990 Organophosphorus and carbamate insecticides. Veterinary Clinics of North America: Small Animal Practice 20:353- 367 [ Links ]
18. Firth A 2000 Treatments used in small animal toxicoses. In Kirk RW (ed.) Kirk's current veterinary therapy: XIII Small animal practice. W B Saunders, Philadelphia: 207-211 [ Links ]
19. Frank M E, Bouverat B P, MacKinnon B I, Hettinger T P 2004 The distinctiveness of ionic and nonionic bitter stimuli. Physiology and Behavior 80:421-431 [ Links ]
20. Frazier K, Hullinger G, Hines M, Liggett A, Sangster L 1999 Clinical reports: 162 cases of aldicarb intoxication in Georgia domestic animals from 1988-1998. Veterinary and Human Toxicology 41:233-235 [ Links ]
21. Garcia S J, Aschner M, Syversen T 2006 Inter-species variation in toxicity of cholinesterase inhibitors. In Gupta R C (ed.) Toxicology of organophosphate and carbamate compounds. Elsevier Academic Press, Amsterdam: 145- 158 [ Links ]
22. Goswamy R, Chaudhuri A, Mahashur A A 1994 Study of respiratory failure in organophosphate and carbamate poisoning. Heart and Lung 23:466-472 [ Links ]
23. Grauer G F, Hjelle J J 1988 Toxicology: Introduction. In Morgan R V (ed.) Handbook of small animal practice. Churchill Livingston, New York: 1083-1086 [ Links ]
24. Gupta R C 2006 Classification and uses of organophosphates and carbamates. In Gupta R C (ed.) Toxicology of organophosphate and carbamate compounds. Elsevier Academic Press, Amsterdam: 5-24 [ Links ]
25. Haddad L M 1983 The organophosphate insecticides. In Haddad L M, Winchester J F (eds) Clinical management of poisoning and drug overdose. W B Saunders, Philadelphia: 704-710 [ Links ]
26. Haddad L M 1983 The carbamate, organochloride and botanical insecticides; insect repellents. In Haddad L M, Winchester (eds) Clinical management of poisoning and drug overdose. W B Saunders, Philadelphia: 711-712 [ Links ]
27. Hoffman R S, Manini A F, Russell-Haders A L, Felberbaum M, Mercurio-Zappala M 2009 Use of pralidoxime without atropine in rivastigmine (carbamate) toxicity. Human and Experimental Toxicology 28:599-602 [ Links ]
28. Jenkins W C 1989 Drugs affecting gastrointestinal function. In Booth N M, McDonald L E (eds) Veterinary pharmacology and therapeutics (6th edn). Iowa State University Press, Ames: 657-671 [ Links ]
29. Johnson M K, Vale J A, Marrs T C, Meredith T J 1992 Pralidoxime for organophosphate poisoning. Lancet 340: 64 [ Links ]
30. Jokanović M, Maksimovic M 1997 Abnormal cholinesterase activity: understanding and interpretation. European Journal of Clinical Chemistry and Clinical Biochemistry 35:11-16 [ Links ]
31. Jokanović M 2009 Medical treatment of acute poisoning with organophosphorus and carbamate pesticides. Toxicology Letters 190:107-115 [ Links ]
32. Lee H S 1989 Acute pancreatitis and organophosphate poisoning - a case report and review. Singapore Medical Journal 30:599- 601 [ Links ]
33. Leibson T, Lifshitz M 2008 Organophosphate and carbamate poisoning: review of the current literature and summary of clinical and laboratory experience in southern Israel. Israel Medical Association Journal 10:767-770 [ Links ]
34. Li H, Schopfer L M, Nachon F 2007 Aging pathways for organophosphate-inhibited human butyrylcholinesterase, including novel pathways for isomalation, resolved by mass spectrometry. Toxicological Sciences 100:136-145 [ Links ]
35. Lima J S, Reis C A 1995 Poisoning due to illegal use of carbamates as a rodenticide in Rio de Janeiro. Journal of Toxicology - Clinical Toxicology 33:687-690 [ Links ]
36. Lotti M, Moretto A 2005 Organophosphate-induced delayed polyneuropathy. Toxicology Reviews 24:37-49 [ Links ]
37. Lotti M, Moretto A 2006 Do carbamates cause polyneuropathy? Muscle Nerve 34:499-502 [ Links ]
38. Makridges C, Koukouvas M, Achillews G, Tsikkos S, Vounou E, Symeonides M 2005 Methomyl-induced severe acute pancreatitis: possible etiological association. Journal of the Pancreas 6:166-171 [ Links ]
39. Marrs T C, Vale J A 2006 Management of organophosphorus pesticide poisoning. In Gupta R C (ed.) Toxicology of organophosphate and carbamate compounds. Elsevier Academic Press, Amsterdam: 715-733 [ Links ]
40. McDonough J H, McLeod C G, Nipwoda M D 1987 Direct micro-injection of soman or VX into amygdale produces repetitive limbic convulsions and neuropathology. Brain Research 435:123-137 [ Links ]
41. McEntee K, Poncelet L, Clercx C, Henroteaux M 1994 Acute polymyopathy after carbamate poisoning in a dog. Veterinary Record 135:88-90 [ Links ]
42. Moritz F, Droy J M, Melki J, Bonmarchand G, Leroy J 1994 Acute pancreatitis after carbamate insecticide intoxication. Intensive Care Medicine 20:49-50 [ Links ]
43. Motas-Guzman M, Maria-Mojica P, Romero D, Martinez-Lopez E, Garcia-Fernandez A J 2003 Intentional poisoning of animals in southeastern Spain. A review of veterinary toxicology service from Murcia, Spain. Veterinary and Human Toxicology 45:47-50 [ Links ]
44. Mount M E 1989. Toxicology. In Ettinger S J (ed.) Textbook of veterinary internal medicine. W B Saunders, Philadelphia: 456-483 [ Links ]
45. Nelson L S, Perrone J, DeRoos F, Stork C, Hoffman R S 2001 Aldicarb poisoning by an illicit rodenticide imported into the United States: Tres Pasitos. Journal of Toxicology - Clinical Toxicology 39:447-452 [ Links ]
46. Olson M E, Vizzutti D, Morck D W, Cox A K 1993 The parasympatholytic effects of atropine sulfate and glycopyrrolate in rats and rabbits. Canadian Journal of Veterinary Research 57: 254-258 [ Links ]
47. Paul N, Mannathukkaran T J 2005 Intermediate syndrome following carbamate poisoning. Clinical Toxicology 43:867-868 [ Links ]
48. Pazdernik T L, Nelson S R, Cross R, Churchill L, Giesler M, Samson F E 1986 Effects of antidotes on soman-induced brain damage. Archives of Toxicology 9:333-336 [ Links ]
49. Platt S R, McDonnell J J 2000 Status epilepticus: managing refractory cases and treating out-of-hospital patients. Compendium on Continuing Education for the Practicing Veterinarian 22:732-741 [ Links ]
50. Plumb D C 2008 Plumb's veterinary drug handbook (6th edn). Blackwell Publishing, Iowa [ Links ]
51. Ragoucy-Sengler C, Tracqui A, Chavonnet A, Daijardin J B, Simonetti M, Kintz P, Pileire B 2000 Aldicarb poisoning. Human and Experimental Toxicology 2000 19:657-662 [ Links ]
52. Risher J F, Mink F L, Stara J F 1987 The toxicological effects of the carbamate insecticide aldicarb in mammals: a review. Environmental Health Perspectives 72:267-281 [ Links ]
53. Riviere J E 1985 Clinical management of toxicoses and adverse drug reactions. In Davis LE (ed.) Handbook of small animal therapeutics. Churchill Livingstone, New York: 657-683 [ Links ]
54. Roberts M D, Aaron C K 2007 Managing acute organophosphorus poisoning. British Medical Journal 334:629-635 [ Links ]
55. Rosman Y, Makarovsky I, Bentur Y, Shrot S, Dushnistky T, Krivoy A 2009 Carbamate poisoning: treatment recommendations in the settling of a mass casualty event. American Journal of Emergency Medicine 27:1117-1124 [ Links ]
56. Rother H A 2010 Falling through the regulatory crack: street selling of pesticides and poisoning among urban youth in South Africa. International Journal of Occupational and Environmental Health 16:202-213 [ Links ]
57. Satoh T 2006 Global epidemiology of organophosphate and carbamate poisonings. In Gupta RC (ed.) Toxicology of organophosphate and carbamate compounds. Elsevier Academic Press, Amsterdam: 89-100 [ Links ]
58. Singh S, Ranjit A, Parthasarathy S, Sharma N, Bambery P 2005 Organo-phosphate induced delayed neuropathy: report of 2 cases. Neurology India 52:525-526 [ Links ]
59. Stevens J T, Brekenridge C B 2001 Crop protection chemicals. In Hayes A W. (ed.) Principles and methods of toxicology (4th edn). Taylor & Francis, Philadelphia: 583-591 [ Links ]
60. Sultatos L G 2006 Interactions of organophosphorus and carbamate compounds with cholinesterases. In Gupta RC (ed.) Toxicology of organophosphate and carbamate compounds. Elsevier Academic Press, Amsterdam: 209-218 [ Links ]
61. Sungur M, Güven M 2001 Intensive care management of organophosphate insecticide poisoning. Critical Care 5:211-215 [ Links ]
62. Tomlin C. 1994 The pesticide manual (10th edn). Crop Protection Publications, Farnham: 25 [ Links ]
63. VealeDJH,WiumCA, Müller G J 2010 A prospective study of the incidence and spectrum of acute poisonings in South Africa based on hospital admission and Poison Information Centre data. Clinical Toxicology 48:284 [ Links ]
64. Verster R S, Botha C J, Naidoo V, van Schalkwyk O L 2004 Aldicarb poisoning of dogs and cats in Gauteng during 2003. Journal of the South African Veterinary Association 75:177-181 [ Links ]
65. Verma S K, Ahmad S 2009 High dose pralidoxime in organophosphorus poisoning: a critical appraisal. API Medicine Update 19:448-452 [ Links ]
66. Waseem M, Perry C, Bomann S, Pai M, Gernsheimer J 2010 Cholinergic crisis after rodenticide poisoning. Western Journal of Emergency Medicine 11:524-527 [ Links ]
67. World Health Organization 2009 The WHO recommended classification of pesticides by hazard and guidelines to classification. Geneva [ Links ]
Received: August 2011
Accepted: September 2011
* Author for correspondence. E-mail: jan.myburgh@up.ac.za














