Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Journal of the South African Veterinary Association
On-line version ISSN 2224-9435
Print version ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.82 n.4 Pretoria Dec. 2011
ARTICLE ARTIKEL
A literature review and investigation of staphylococcal necrotic dermatitis in sheep
G F BathI,*; A Janse van RensburgI; K P PetteyI; M van VuurenII; A KidanemariamII
IDepartment of Production Animal Studies, Faculty of Veterinary Science, University of Pretoria, Onderstepoort, 0110 South Africa
IIDepartment of Veterinary Tropical Diseases, Faculty of Veterinary Science, University of Pretoria, Onderstepoort, 0110 South Africa
ABSTRACT
An outbreak of necrotic dermatitis in sheep was intensively investigated. Initially 19 of 147 Letelle (Merino-type) ewes were identified but closer inspection revealed 57 sheep that had skin lesions, some very slight, and that the majority (46 or 80 %) had lesions only above the lips. A small number of them had multiple lesions on the legs or vulvae apart from lip lesions. Seven had only vulvar lesions and 2 only leg lesions. Among the sheep with lip lesions, twice as many had lesions on the right as on the left. Electron micrographs did not reveal any virus particles from the lesions, but all bacterial swabs yielded pure cultures of beta-haemolytic, Gram-positive cocci that were catalase, coagulase and DNase positive. The organism was identified as Staphylococcus aureus. Histopathology was consistent with a dermotoxic insult. A review of available literature indicated that this outbreak was consistent with a diagnosis of ovine necrotic (staphylococcal) dermatitis, supported by data on signalment, lesions, distribution, size, number, epidemiology as well as specific tests. The range of differential diagnoses and possible confusers are discussed.
Keywords: dermatitis, necrotic, ovine, sheep, Staphylococcus aureus.
INTRODUCTION
Skin lesions, particularly those that occur on the face of sheep present certain diagnostic challenges because of some macroscopic similarities between conditions that have widely divergent aetiologies. Incomplete descriptions, or limited investigations, may lead to further confusion.
The earliest reference to the disease of sheep now known as staphylococcal dermatitis (SD) was apparently made in Scotland in 196620,42. It was recorded as 'periorbital eczema' and even 'facial eczema', although the latter term is now regarded as inappropriate as it is used for photosensitisation caused by Pithomyces chartarum16,43. In 1951 a condition was described in America that bore several similarities to the disease in Britain12. Another disease in the USA with some similarities to SD became known as lip and leg ulceration, and was later known as ulcerative dermatosis (UD). This disease was 1st described in 1910 and subsequently in 194137,39. It was later ascribed to infection by a virus similar to the orf virus37,38 but because of the macroscopic similarities of lesions compared with those of SD, some confusion may have occurred. SD has also been reported in Ireland24, New Zealand43 and France31, and staphylococcal folliculitis, not associated with necrotic dermatitis, in several other countries14,43. Most of the investigations that led to the elucidation of the cause, epidemiology and control measures for SD were conducted in Britain, and the identification of dermonecrotic strains of Staphylococcus aureus led them to change the name from the more restrictive and inaccurate periorbital eczema to SD.
An intensively investigated outbreak of a disease of sheep in South Africa is described that corresponds with the essential features of the condition termed staphylococcal dermatitis in sheep21.
LITERATURE REVIEW
From published descriptions and investigations, the following information emerges:
Signalment
The disease does not appear to be related to any breed or type, and has been recorded in Texels6, Suffolks12,32, Scottish Blackface and Greyface24, Dorsets26, Cheviots33 and crosses10,11,34. SD has been recorded mainly in adults6,10-12,20,21,24,32,33,35 but also in unweaned lambs26. Most outbreaks have been described in ewes6,10,11,24,33 mainly in lactation6,10,11,24,33, but also in late pregnancy24,42 and once in rams12. Lesions may be associated with mastitis8,10,11 and poor growth or emaciation6,12.
Lesion descriptions
Several investigators describe an initial lump or swelling11,20 especially when the disease was induced experimentally8,32,34. Papules14,20,32 and pustules8,14,26,33,34 as well as pus8,11,20,21,24,33,34 are frequently mentioned while most authors describe scabs6,8,10-12,14,20,21,24,26,32-34,42 and/or necrotic eschars8,20,21,33, and many authors describe ulcers6,10,11,14,20,33,34. Some record alopecia20,21,24,34 and others report a foul smell21,32 or scars26 and melanosis24.
Lesion distribution
Lesions may occur on pigmented or non-pigmented skin24,32. They are most commonly recorded on the face11,20,21,24,26,32,33,42, around the eyes 20,21,32,33,42, the base of the ears12,21,24,32, the lips20,26, legs21,34, teats6,10,21,26,33 and also sites like the neck, withers, shoulders, back and rump11,12.
Lesion size and number
Most reports indicate a size up to 4 cm across11,20,21,32 and some larger6,12,32,up to 15 cm, but in many reports size is not recorded. The number of lesions per sheep appears to be variable and although this is not specified in the reports, it can be inferred by the references to multiple sites that multiple lesions were often seen.
Epidemiology
Morbidity is reported to vary between 'low'12, around 10 %6,11,up to59%26,or 'most' of 7434, with the majority of estimates in the 20-50 % range10,20,21,32. Mortalities associated with SD, when reported or estimated, are given as low or rare20,21,33. Spread is reported to be 'rapid'21,24,32,42. The course of the natural disease in an outbreak appears to be quite protracted, with reports indicating anything between 2 and 9 weeks, mostly 3-4 weeks20,21,24,26,32, but in experimental cases the course in an individual animal may be much shorter, around 4-8 days4,8,12. Where recurrence is reported, all authors indicate that there is no apparent immunity6,8,20,21,26,32. Cases of SD appear to respond to several antibiotics6,8,11,20,24,32,34. Most authors report a history of feeding mainly concentrates or silage6,10,11,20,24,32 and some particularly note an association with trough feeding20,21,24. In 2 articles an association between leg lesions and thistles is reported21,34. Some authors note that outbreaks were associated with poor weather or late winter or early spring6,10,11,21,24.
Specific tests
None of the authors who reported testing for viruses found any indication of viral involvement6,10,11,20,21,24,26,32-34,42. Although most reported initial mixed bacterial cultures from lesions6,10,11,20,24 the only consistent and aetiologically important organism isolated was Staphylococcus aureus6,11,20,21,26,34, although in 1 case S. albus was recorded12 and in another Mannheimia (Pasteurella) haemolytica10. The S. aureus isolates were recorded as being haemolytic8,11,20,21,24,32-34, mainly (α) or (β) haemolytic but also (γ) and (δ). Nearly all the isolates were coagulase-positive8,11,20,21,32-34 and 2 articles reported the isolates as catalase-positive26,32. There was 1 report of micro-abscessation6 on histopathological examination.
Differential diagnosis
There are many diseases that could complicate making a definitive diagnosis of SD, particularly in a clinical situation where diagnosis is based on macroscopic lesions, signalment and epidemiology. These diseases comprise at least the following: ulcerative dermatosis19,23,36-38, photosensitisation2,16,orf2,18,19,29, lumpy wool or dermatophilosis2,19,27, sheep pox17, papillomatosis, peste des petits ruminants30 , abscesses caused by Corynebacterium pseudotuberculosis1,2, Arcanobacterium pyogenes2,5, Pseudomonas aeruginosa43, or Actinobacillus lignieresii,2,15 Mannheimia (Pasteurella) haemolytica10 and foot-andmouth disease35. Ectoparasites include blackfly lesions (Simulium sp.)2, ringworm2,27, trombiculid mites25, leishmaniosis40and sheep scab2. Irritant substances like the sap of Euphorbia species and plant awns16 may also produce lesions that cause confusion. With lesions on the lower leg, diseases like foot rot and foot abscess2,7,43 as well as strawberry foot rot and foot scald would have to be considered. The reported association of UD with genitals may require consideration of diseases like posthitis2,9, ulcerative balanoposthitis and vulvitis, and urolithiasis2. It is only by careful examination of the lesions, the history and epidemiology that a satisfactory clinical diagnosis can be made. It may be necessary to confirm or rule out SD by bacterial culture in cases of doubt.
MATERIALS AND METHODS
History
The outbreak occurred on the farm Border Brook, southwest of Pretoria, Gauteng. The farmer had bought about 150 Letelle (Merino-type) ewes from a farmer in the Smithfield district, Free State Province between 6 and 15 months prior to the outbreak. On enquiry, that farmer reported that he had never experienced any similar cases or outbreaks. The current owner, however, believed that a single case had occurred the previous year in his own flock. The sheep were sent out to graze on Eragrostis curvula hay stubble during the day. Within some of the windrows not completely harvested during baling, a number of thistle plants were observed which, together with a few weeds, comprised the only green plants available at that time of year. At night the sheep were housed in close confinement as a precaution against theft. Here they were given access to commercial sheep pellets in feed troughs, as well as a block lick and hay bales. Normal vaccinations and anthelmintic treatments had been given but not immediately prior to the outbreak. The outbreak was reported to the private practitioner on 12 May 2002 (autumn-early winter) and referred to the authors on 21 May for further investigation.
Examination and recording
All 147 sheep present on the farm were examined for skin lesions on the wool-free areas of the face, udder, legs and perineum. These were classified according to the size, location, severity and stage. Photographs were taken and sketches of lesions made. Age was recorded according to the number of incisor teeth erupted. All data recorded were subsequently analysed for patterns of disease occurrence according to age, lesion distribution, lesion size and lesion type.
Histopathology
Histopathology was undertaken by a veterinary pathologist on 3 typical lesions collected in formalin for examination.
Specimen collection
Swab samples were taken from the edges of active lesions of 7 sheep using commercially available sterile cotton wool swabs. The swabs were cut and placed into cryovials containing 0.5-1 ml brain heart infusion broth as transport medium for bacteria, and Hayflick's broth for mycoplasma isolation. Wound scabs and crusts were collected for virus particle detection by means of electron microscopy from 7 sheep. All specimens were transported to the laboratory in a coolbox containing frozen ice packs.
Microbiological examination
For bacterial culture, specimens were inoculated onto Columbia blood agar (Oxoid) with 5-6 % horse blood and MacConkey agar (Oxoid). Two blood agar plates were inoculated with each specimen and incubated respectively in an atmosphere of 5%CO2 and anaerobically at 37 ºC. All cultures were incubated for 48-72 hours. The bacterial isolates were identified using standard methods28.
Specimens were inoculated onto Hayflick's agar and into Hayflick's broth13 and incubated in a moist chamber with 5%CO2 at 37 ºC to detect mycoplasms. The cultures were incubated for a minimum of 7 days and considered negative if there was no growth after 10 days of incubation.
No attempt was made to isolate viruses in cell cultures. Standard procedures for transmission electron microscopy were used to detect viral particles in the wound scabs.
The in vitro antimicrobial susceptibility of the bacterial isolates was established according to the method described by Bauer and Kirby3.
RESULTS
Lesions
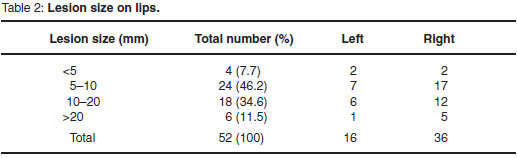
Initially the farmer reported 19 of the 147 sheep to be affected (13 %). Of these, the lesions of 4 were judged to be severe (>20 mm), 9 moderate (10-20 mm) and 6 mild (5-10 mm). Closer examination of all sheep showed that lesions were much more widespread in the flock (57 sheep or 39 %), although most of the additional cases were minor. The size, distribution, type and age group occurrence of the lesions are summarised in Tables 1-3.
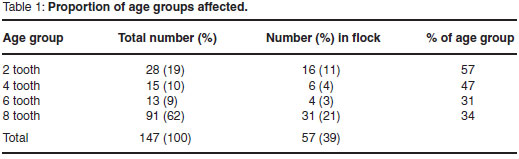
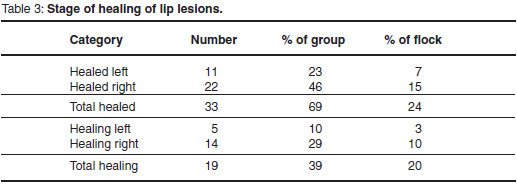
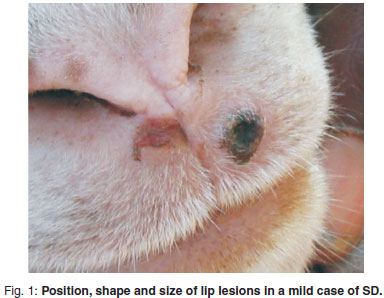
Figures 1-4 illustrate the position, shape and size of the lesions found on the upper lip, where the great majority of lesions occurred, from mild to severe. The early cases showed full-depth necrosis of the affected skin, which was sharply demarcated from the surrounding healthy skin. At the junction, the healthy skin was inflamed for a few millimetres. Removal of the necrotic, scabby layer revealed the presence of pale yellow thick pus and a rough, bleeding, granular ulcer. Within a week of initial examination most lesions showed significant improvement with scarring but no regrowth of hair. Nearly all the sheep subsequently recovered completely, except for the worst cases, which had permanent scarring and disfigurement. The mildest cases were of very short duration and within 2 weeks only the most careful examination could reveal a previous infection. Two of the worst cases appeared to extend by millimetres into the nares. Most lesions appeared to be only on the right side of the upper lip. Foot lesions recorded at the coronary junction appeared to be traumatic rather than infectious but are included for the sake of completeness. The vulvar lesions recorded did not appear to be contemporaneous with the facial lesions in that they were all chronic, scarred lesions that may have been caused by ticks in previous seasons. From the time that the 1st signs were noted by the farmer the disease appeared to have spread rapidly, but between the 1st and 2nd examinations 1 week apart, only 1 possible new case was recorded. After this, no further problems were encountered, including during the subsequent 2 seasons
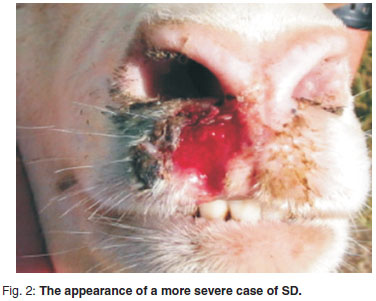
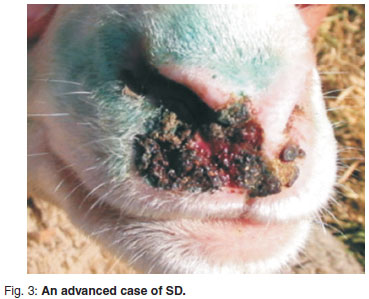
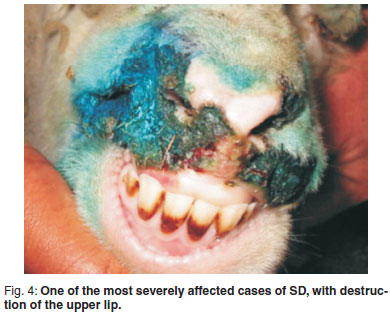
The farmer had initially treated the affected cases with an antiseptic chlorhexidine (Hibitane, Dismed) spray and later a tetracycline (Terramycin, Pfizer) spray, but after 3 days the animals appeared worse. The private practitioner recommended injectable florfenicol (Nuflor, Schering Plough) for all cases and this was associated with sustained improvement. There were no rams present on the farm and none of the ewes were pregnant or lactating during the outbreak.
Altogether 57 sheep (39 %) had lesions, of which 43 (75 %) had only unilateral lip lesions and 3 (5 %) had lesions on both sides. Six (11 %) had multiple lesions, and all of these had lip lesions. Two sheep had only bilateral lip lesions with vulvar lesions, 1 had a unilateral lip lesion with a leg lesion, 1 had a unilateral lip lesion with a vulvar lesion and 1 a unilateral lip lesion with both leg and vulvar lesions. There were 7 sheep with only vulvar lesions and 2 with only leg lesions. Altogether there were 10 vulvar lesions (6 %) and 4 leg lesions (3 %) compared with 46 with only lip lesions (80 %). A surprising total of 36 (55 %) of the 57 sheep with lesions had lip lesions on the right side, but just 16 (24 %) on the left. The total number of leg lesions in the flock (4) represented just 3 % of the total flock but 7 % of the affected group. Similarly, the total number of vulvar lesions (10) represented 6% of the flock but 16 % of affected sheep.
Microbiological examination
Electron microscopic investigation did not reveal any viral particles.
All specimens yielded pure cultures of a beta-haemolytic, greyish, smooth colony that microscopically appeared as Gram-positive cocci. The organisms were catalase-positive, coagulase-positive and DNase-positive. Further confirmation of these isolates as Staphylococcus aureus was achieved using the biochemical tests described by Quinn et al.28. No mycoplasmas were isolated. The growth inhibition zone sizes and their interpretation for each drug tested on 3 selected isolates are shown in Table 4.
Histopathology
Skin biopsies revealed full-depth necrosis of the epithelium with severe ulceration and acute tissue necrosis. There was prominent oedema, rhabdomyolysis and acute neutrophilic infiltration in some parts of the subcutis. A pathological anatomical diagnosis of severe multiflocal necrotic dermatitis accompanied by exudation and ulceration was made. The lesions were found to be consistent with a dermotoxic insult.
DISCUSSION
This investigation into an outbreak of necrotic dermatitis in a sheep flock revealed key features consistent in all material aspects with previous reports of the disease now generally referred to as ovine staphylococcal dermatitis. In fact, the great majority of findings confirm those documented by previous investigators. It is therefore strange, given the ubiquity of S. aureus22,41 and the frequency of encountering the predisposing factors, that this disease has not been properly documented before in South Africa. Informal discussions with veterinarians revealed that it has probably occurred over a long time, but infrequently or only affecting a few sheep, or in a mild form. The great range of potential confusers that are listed under the differential diagnoses clearly indicates that misdiagnoses may easily occur and are excusable unless all factors, lesions and tests are carefully considered. Since the outcome of SD is usually complete healing and no mortality, an incorrect diagnosis can easily go unnoticed and unchallenged. Superficial examination in this outbreak only showed 19 clearly affected sheep, while a detailed and careful inspection uncovered another 38 sheep with milder lesions that in all probability would have gone undetected had there been no severe cases that triggered the full investigation. One factor that should not be overlooked is that S. aureus isolates can vary markedly in their dermotoxicity and therefore only particular isolates will cause significant disease. Although SD on the basis of current knowledge cannot be regarded as a significant threat to the local sheep industry, it is important that it is clearly differentiated from other skin conditions so that appropriate measures can be taken to control an outbreak.
Recommendations to the farmer comprised twice-weekly examinations, separation of affected and healthy sheep, wound treatment, antibiotic injections, removal of old feed pellets, disinfection of hands and feed troughs and checking and repair of any sharp edges in the troughs.
One intriguing and possibly instructive aspect of this investigation was the marked preponderance of lip lesions on the right side of the face, compared with the left. It is unlikely, given the numbers involved, that this 2:1 ratio was mere chance. Rather, it points to a causative factor that created more skin damage on one side of the muzzle than the other. This could be associated with the thistles in the paddock, if the sheep were consistently grazing more from 1 side. A more likely explanation is that sheep were feeding at the troughs more from 1 side than the other and that skin damage and contamination, when present, was more frequently on the right. Unfortunately this could not be confirmed by examination of the troughs or observation of the sheep.
The range of size and severity of the lesions and the large number of unaffected sheep may point to differences in susceptibility between the animals. Since the same range of predisposing factors previously reported were found in this outbreak, eliminating or ameliorating them may assist with preventing or containing outbreaks.
ACKNOWLEDGEMENTS
We thank Hester van der Schyff and Elize Orsmond for typing, Lana Botha and Reinette van Reenen for technical assistance, and Dr W Botha for histopathological evaluation.
REFERENCES
1. Baird G J 2007 Caseous lymphadenitis In Aitkin I D (ed.) Diseases of sheep (4th edn). Blackwell Science, Oxford: 306-311 [ Links ]
2. BathGF,deWetJAL2000 Sheep and goat diseases. Tafelberg, Cape Town [ Links ]
3. Bauer A W, Kirby M M 1966 Antibiotic susceptibility testing by standard single disk method. American Journal of Clinical Pathology 45:493-496 [ Links ]
4. Brodie T A, Holmes P H, Urquhart G M 1986 Some aspects of tick-borne disease of British sheep. Veterinary Record 118:415-418 [ Links ]
5. Collett M G, Bath G F 2004 Arcanobacterium pyogenes infections. In Coetzer J A W, Tustin R C (eds) Infectious diseases of livestock (2nd edn), Vol. 3. Oxford University Press, Cape Town: 1946-1957 [ Links ]
6. Doherty M L, Bassett H F 1989 Staphylococcal dermatitis in sheep. Veterinary Record 124:470 [ Links ]
7. Egerton J R 2007 Diseases of the feet In Aitkin I D (ed.) Diseases of sheep (4th edn). Blackwell Science, Oxford: 273-281 [ Links ]
8. Fraser J, ScottFMM, Angus K W, Martin W B 1982 Experimental re-infection of the skin of sheep with Staphylococcus aureus. Veterinary Record 111:485-486 [ Links ]
9. Greig A 2007 Ulcerative balanitis and vulvitis In Aitkin I D (ed.) Diseases of sheep (4th edn). Blackwell Science, Oxford: 143-145 [ Links ]
10. Gunning R F, DaviesCHV1989 Isolation of Pasteurella haemolytica from teat lesions in ewes. Veterinary Record 125:490 [ Links ]
11. Gunning R F, Bosworth P A 1989 Staphylococcal dermatitis involving the teats of lactating ewes. Veterinary Record 124:146- 147 [ Links ]
12. Hardy W T, Price D A 1951 Staphylococcic dermatitis of sheep. Journal of the American Veterinary Medical Association 119:445-446 [ Links ]
13. Hayflick L 1965 Tissue cultures and mycoplasmas. Texas Reports on Biology and Medicine 23: 285-303 [ Links ]
14. Henton M M 2004 Staphylococcus aureus infections. In CoetzerJAW,Tustin R C (eds) Infectious diseases of livestock (2nd edn), Vol.3. Oxford University Press, Cape Town: 1754-1758 [ Links ]
15. Henton M M, van der Lugt J J 2004 Actinobacillus lignieresii infections. In Coetzer J A W, Tustin R C (eds) Infectious diseases of livestock (2nd edn), Vol. 3. Oxford University Press, Cape Town: 1648-1651 [ Links ]
16. Kellerman T S, Coetzer J A W, Naude T W 1990 Plant poisonings and mycotoxicoses of livestock in southern Africa. Oxford University Press, Cape Town [ Links ]
17. Kitching R P 2004 Sheeppox and goatpox. In Coetzer J A W, Tustin R C (eds) Infectious diseases of livestock (2nd edn), Vol. 2. Oxford University Press, Cape Town: 1277-1281 [ Links ]
18. Kitching R P 2004 Orf. In Coetzer J A W, Tustin R C (eds) Infectious diseases of livestock (2nd edn), Vol. 2. Oxford University Press, Cape Town: 1282-1286 [ Links ]
19. Kimberling C V 1988 Jensen and Swift's diseases of sheep (3rd edn). Lea & Febiger, Philadelphia: 274-276 [ Links ]
20. Martin W B 1983 Staphylococcal dermatitis in sheep. Veterinary Annual 23:104-108 [ Links ]
21. McNeil P E 2007 Staphylococcal skin infections In Aitkin I D (ed.) Diseases of sheep (4th edn). Blackwell Science, Oxford: 312-315 [ Links ]
22. Mitchell G B B, Brodie T A, Webster K A 1988 Tick pyraemia (enzootic staphylococcal infection): recent developments. Veterinary Annual 28:69-73 [ Links ]
23. Munz E, Dumbell K 2004 Ulcerative dermatosis. In Coetzer J A W, Tustin R C (eds) Infectious diseases of livestock (2nd edn), Vol. 2. Oxford University Press, Cape Town: 1287-1288 [ Links ]
24. O'Brien J J, McCracken R M 1971 Periorbital eczema in sheep. Irish Veterinary Journal 25:69-71 [ Links ]
25. Otto Q T, Jordaan L C 1992 An orf-like condition caused by trombiculid mites on sheep in South Africa. Onderstepoort Journal of Veterinary Research 59:335-336 [ Links ]
26. ParkerBNJ, Bonson M D, Carroll, P J 1983 Staphylococcal dermatitis in unweaned lambs. Veterinary Record 113:570-571 [ Links ]
27. Plant J 2007 Bacterial and fungal infections of the skin and wool. In Aitkin I D (ed.) Diseases of sheep (4th edn). Blackwell Science, Oxford: 315-320 [ Links ]
28. Quinn P J, Carter M E, Markey B K, Carter G R 1994 Clinical veterinary microbiology. Wolfe Publishing, London: 118-126 [ Links ]
29. Reid H W, Rodger S M 2007 Orf. In Aitkin I D (ed.) Diseases of sheep (4th edn). Blackwell Science, Oxford: 297-302 [ Links ]
30. Rossiter P B 2004 Peste de petits ruminants. In Coetzer J A W, Tustin R C (eds) Infectious diseases of livestock (2nd edn), Vol. 2. Oxford University Press, Cape Town: 660-672 [ Links ]
31. Savey M, Person J M, Chermette R, Espinasse J, Delvac R 1983 Identification de la dermatite staphylococcique du mouton. Recueil de Médicine Vétérinaire 159:701-705 [ Links ]
32. Scott P R, Murphy S 1997 Outbreak of staphylococcal dermatitis in housed lactating Suffolk ewes. Veterinary Record 140:631-632 [ Links ]
33. ScottFMM,Fraser J, Martin W B 1980 Staphylococcal dermatitis in sheep. Veterinary Record 107:572-574 [ Links ]
34. Synge B A, ScottFMM, MacDougall D C 1985 Dermatitis of the legs of sheep associated with Staphylococcus aureus. Veterinary Record 116:459-460 [ Links ]
35. Thomson G R, BastosADS 2004 Foot-andmouth disease. In CoetzerJAW,Tustin R C (eds) Infectious diseases of livestock (2nd edn), Vol. 2. Oxford University Press, Cape Town: 1324-1365 [ Links ]
36. Trueblood M S, Chow T L, Griner L A 1963 An immunologic study of ulcerative dermatosis and contagious ecthyma. American Journal of Veterinary Research 24:42-46 [ Links ]
37. Trueblood M S, Chow T L 1963 Characterization of the agents of ulcerative dermatosis and contagious ecthyma. American Journal of Veterinary Research 24:47-51 [ Links ]
38. Trueblood M S 1966 Relationship of ovine contagious ecthyma and ulcerative dermatitis. Cornell Veterinarian 56:521-526 [ Links ]
39. Tunnicliff E A, Matisheck P H 1941 A filterable virus demonstrated to be the infective agent in ovine balano-posthitis. Science 94:283-284 [ Links ]
40. Van der Lugt J J, Stewart C G 2004 Leishmaniosis. In CoetzerJAW,Tustin R C (eds) Infectious diseases of livestock (2nd edn), Vol. 1. Oxford University Press, Cape Town: 378-381 [ Links ]
41. Watson W A 1965 The carriage of pathogenic staphylococci by sheep. Veterinary Record 77:477-479 [ Links ]
42. Watt J A 1966 Indoor sheep husbandry: associated conditions. Veterinary Record 69:750-757 [ Links ]
43. West D M, Bruère A N, Ridler A L 2002 The sheep: health, disease and production (2nd edn) Veterinary Continuing Education, Palmerston North [ Links ]
Received: May 2011
Accepted: August 2011
* Author for correspondence. E-mail: gareth.bath@up.ac.za














